Abstract
Objective
To determine the effect of Normosol™-R as compared to normal saline on the outcomes of acute kidney injury and the need for renal replacement therapy in the resuscitation phase of sepsis.
Design
Our study is a retrospective before-and-after cohort study.
Setting
The study occurred at a 700-bed tertiary academic level 1-trauma center.
Patients
A total of 1218 patients were enrolled through emergency department admissions. The normal saline (before) cohort was defined as the dates between 1 March and 30 September 2014 and the Normosol™-R (after) cohort was assessed from 1 March to 30 September 2015.
Interventions
None.
Measurements and main results
Intravenous fluid volumes received during the first 24 h, 72 h, and total hospital stays were compared. Sodium, chloride, potassium, and bicarbonate levels at 72 h were also compared. The medical coded diagnosis of acute kidney failure, need for renal replacement therapy, hospital LOS, ICU admission, ICU LOS, in-hospital mortality, and need for mechanical ventilation were all compared. There was no significant difference in intravenous fluid volumes between groups. Regression modelling controlling for baseline characteristics and 24-h fluid intake volume found no differences between groups for the primary outcomes of acute kidney injury (P = 0.99) and renal replacement therapy (P = 0.88). Patients in the Normosol™-R cohort were found to have a lower rate of hyperchloremia at 72 h post-admission (28% vs. 13%, P < 0.0001). There was a trend toward a decrease in the hospital and ICU LOS in the Normosol™-R cohort; however, the data were not statistically significant.
Conclusions
This study was unable to detect any difference in outcomes between sepsis patients who received intravenous fluid resuscitation with either a balanced crystalloid (Normosol™-R) or normal saline, except for a decreased rate of hyperchloremia.
Introduction
Sepsis is a dysregulated, life-threatening inflammatory response to infection that is a leading cause of healthcare costs and mortality worldwide.1,2 Although the definitions and diagnostic criteria of sepsis and septic shock have evolved over time, the identification of a successful treatment strategy has remained elusive. The backbone of sepsis treatment consists of intravenous (IV) fluid resuscitation and early antimicrobial therapy. 3 No consensus currently exists regarding which type of crystalloid fluid is optimal for septic resuscitation. 3
Normal saline (0.9% sodium chloride) is currently the most commonly used crystalloid fluid worldwide. 4 Composed of 154 mmol/L each of sodium and chloride, the chemistry of “normal” saline does not reflect the characteristics of human plasma. Saline has a supraphysiologic chloride load and a strong ion difference (SID; difference between strong cations and anions) of zero, 5 much lower than plasma’s 42 mEq/L. In contrast, “balanced” crystalloid fluids are available that have a much lower chloride load, more physiologic SIDs, and contain other electrolytes that more closely mimic human plasma. Normosol™-R is manufactured by Hospira, Inc., Lake Forest, IL and has a pH of 7.4 and the following electrolytes per 1000 mL (not including pH adjustment): sodium 140 mmol, potassium 5 mmol, magnesium 3 mmol, chloride 98 mmol, acetate 27 mmol, and gluconate 23 mmol. This calcium-free balanced crystalloid has a fluid composition identical to fluids of the PlasmaLyte family of balanced crystalloids, and is the subject of our study. PlasmaLyte has been associated with reduced rates of hyperchloremic metabolic acidosis,6,7 acute kidney injury (AKI),8–10 mechanical ventilation, 11 and in-hospital mortality11,12 when compared to normal saline. To date, there are no published data available comparing the performance of normal saline and Normosol™-R in the volume resuscitation of sepsis.
Our study is a retrospective before-and-after cohort that aims to assess whether resuscitation with Normosol™-R reduces adverse events in septic patients when compared to normal saline. The primary outcomes measured were rates of AKI and renal replacement therapy (RRT). Additionally, we compared hospital LOS, ICU admissions, ICU LOS, mortality, and ventilator usage.
Materials and methods
Study design and oversight
Our study is a retrospective before-and-after cohort designed to compare clinical outcomes regarding septic patients who received normal saline versus a balanced crystalloid solution, Normosol™-R. No funding was pursued or obtained to conduct this study. The study protocol was approved by our Institutional Review Board (IRB), at the Carilion Clinic-Virginia Tech Carilion School of Medicine, with a waiver of informed consent (IRB protocol #1996).
Patient population and materials
The study occurred in a 700-bed tertiary academic level 1 trauma facility that receives over 80,000 emergency department (ED) visits annually. The normal saline arm was defined as the period of 1 March to 30 September 2014 and the Normosol™-R cohort was assessed from 1 March to 30 September 2015. In response to the newer data emerging regarding balanced IV solutions,4,8 the ED and Critical Care Departments exchanged the default resuscitation fluid from normal saline to Normosol™-R in early 2015. To assess the impact of this change, patient admissions through the ED before and after this change were examined. Inclusion criteria were age 18 years or older and a diagnosis of sepsis, severe sepsis, or septic shock (International Classification of Diseases, Ninth Edition, Clinical Modification (ICD-9-CM) codes: 038, 995.91, 995.92, or 785.52). Patients who received RRT (IC9 V45.11; ICD10 Z99.2), within one year prior to the date of admission, as well as those who had a pre-existing diagnosis of any type of heart failure (ICD-9-CM 428; ICD-10-CM I50), cardiomyopathy (ICD-9-CM 425); ICD-10-CM I42), or end-stage renal disease (ICD-9-CM 585.6; ICD10 N18.6) were excluded from the study due to potential alterations in the volume management of these patient populations. After applying the inclusion and exclusion criteria to the electronic medical record database, an initial cohort of 1233 patients was collected. Patients who were missing, necessary data points were excluded, leaving 1218 patients remaining (538 from the normal saline group, 680 from the Normosol™-R group). An additional analysis was performed which included only patients who received at least 2 L of crystalloid fluid by day 3 of hospitalization (474 from the normal saline group, 576 from the Normosol™ group) (Figure 1).
Cohort acquisition. Creation of the study cohort by application of inclusion and exclusion criteria. Renal failure is defined as pre-existing diagnosis of end stage renal disease or renal replacement therapy within one year prior to admission. Heart failure is defined as a pre-existing diagnosis of heart failure (any type) or cardiomyopathy. After excluding these populations, as well as patients missing necessary data points, the analytical cohort was acquired which consists of 1218 patients, all admitted to our hospital through our ED with an admission diagnosis of sepsis. Larger volume subgroup analysis excluded patients who received less than 2 L of IV fluid by hospital day 3. BMI: body mass index; IV: intravenous.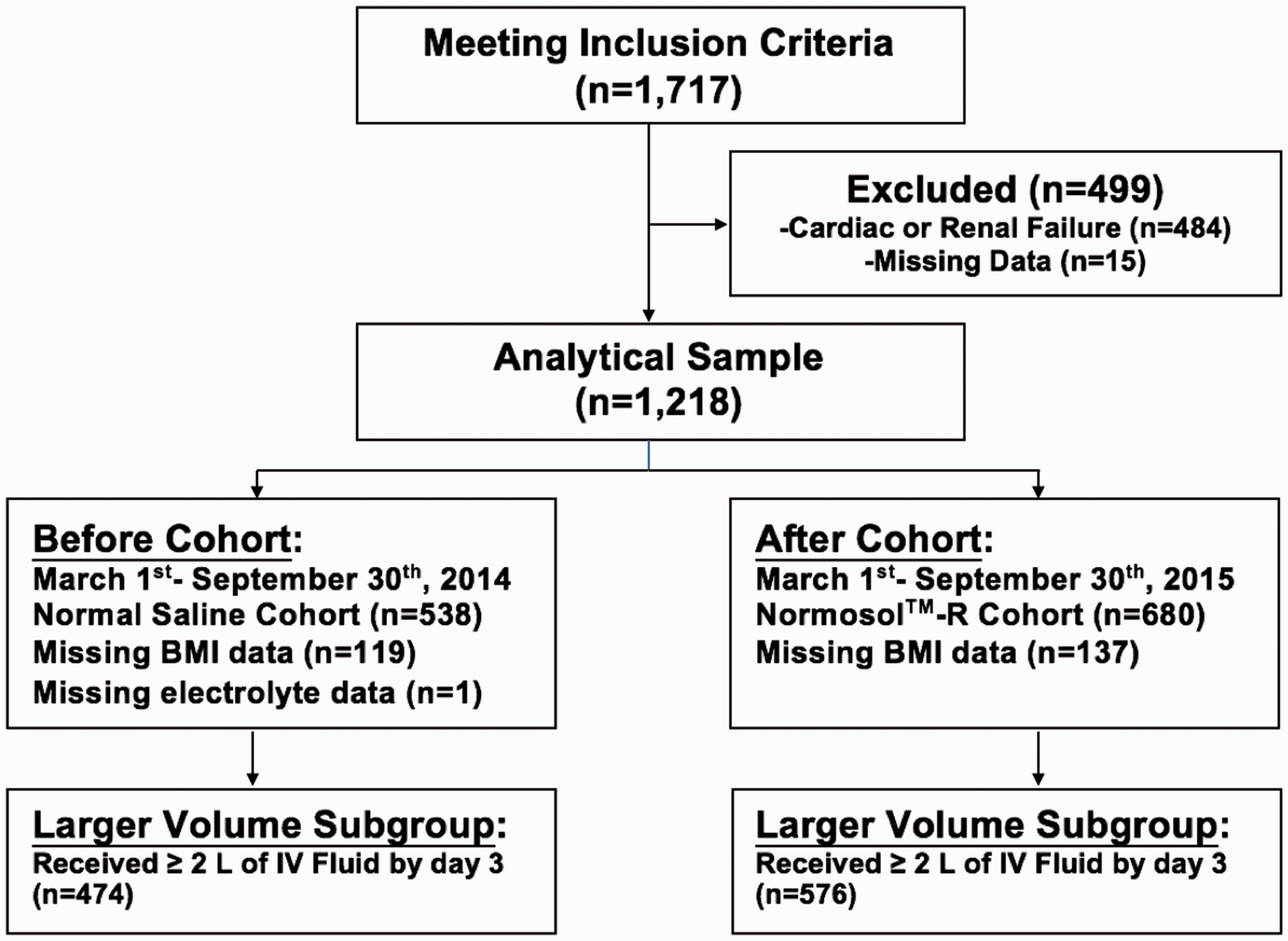
Data collection
Patient age, sex, race, body mass index (BMI), history of chronic kidney disease (CKD) (ICD-9-CM 585.x, ICD10 I12.9), IV fluid volume received during the first 24 h, 72 h of hospital admission, during total hospital stay, hospital and ICU, admission and discharge times were recorded. Vital signs on admission, and electrolyte levels (sodium, chloride, potassium, and CO2) at 72 h post-admission were also collected for each patient. The medical coded diagnosis of kidney failure as defined by the RIFLE criteria, hospital LOS, ICU admission, ICU LOS, in-hospital mortality, need for mechanical ventilation were all recorded. To control for baseline differences in severity of illness between groups, admission qSOFA score was calculated for each patient.
Study outcomes
Primary outcome measures included diagnoses of acute kidney failure (ICD-9-code 584.x/ICD-10-CM N17.9) and orders for the initiation of RRT (ICD9 V45.11/ICD10 Z99.2). Secondary outcomes included length of stay (LOS), ICU admission during hospitalization, ICU LOS, in-hospital mortality, and need for mechanical ventilation. Patients that received at least 2 L of IV fluids within the first 72 h were examined as a subgroup as we postulated that differences between fluid types would be amplified in this group.
Statistical analysis
All statistical analyses were performed using RStudio (Version 1.0.136). Baseline characteristics between groups were compared using univariate analyses. Mean values of continuous independent variables (age, BMI) were compared using two-tailed t tests. Categorical independent variables (gender, race, history of CKD) were compared using Pearson’s Chi-squared test. Wilcoxon rank sum test was used to compare each group’s admission qSOFA scores.
Chloride, potassium, sodium, bicarbonate, as well as serum creatinine levels on day 3 of admission were compared between groups for the patients for which these data were available (537 in the saline group and 680 in Normosol™-R group). The mean lab values were compared using multivariate linear regression. Hyperchloremia in this study is defined, using local reference ranges, as a chloride concentration greater than 107 mmol/L. Comparisons of clinical outcomes between fluid types were performed using logistic regressions for nominal outcome measures and multiple linear regressions for continuous outcomes. Input variables for regression models were chosen based on known clinical predictors of AKI, available patient data, and similar studies. 11 Inputs for regression modelling were patient demographics (age, gender, race), qSOFA score on admission, pre-existing CKD, and volume of IV fluid in first 24 h post-admission. BMI was not used as a covariate due to missing data points for some patients. All data, where applicable, were reported as mean values. Outcome comparisons were reported as effect estimates, defined as relative risk or absolute difference, with 95% confidence intervals. Based on Bonferroni correction for multiple analyses, a P value of 0.007 was considered significant for statistical analyses of primary and secondary outcomes. A P-value of 0.05 was used for comparison of baseline characteristics.
Results
Enrolment and baseline characteristics
Patient characteristics.
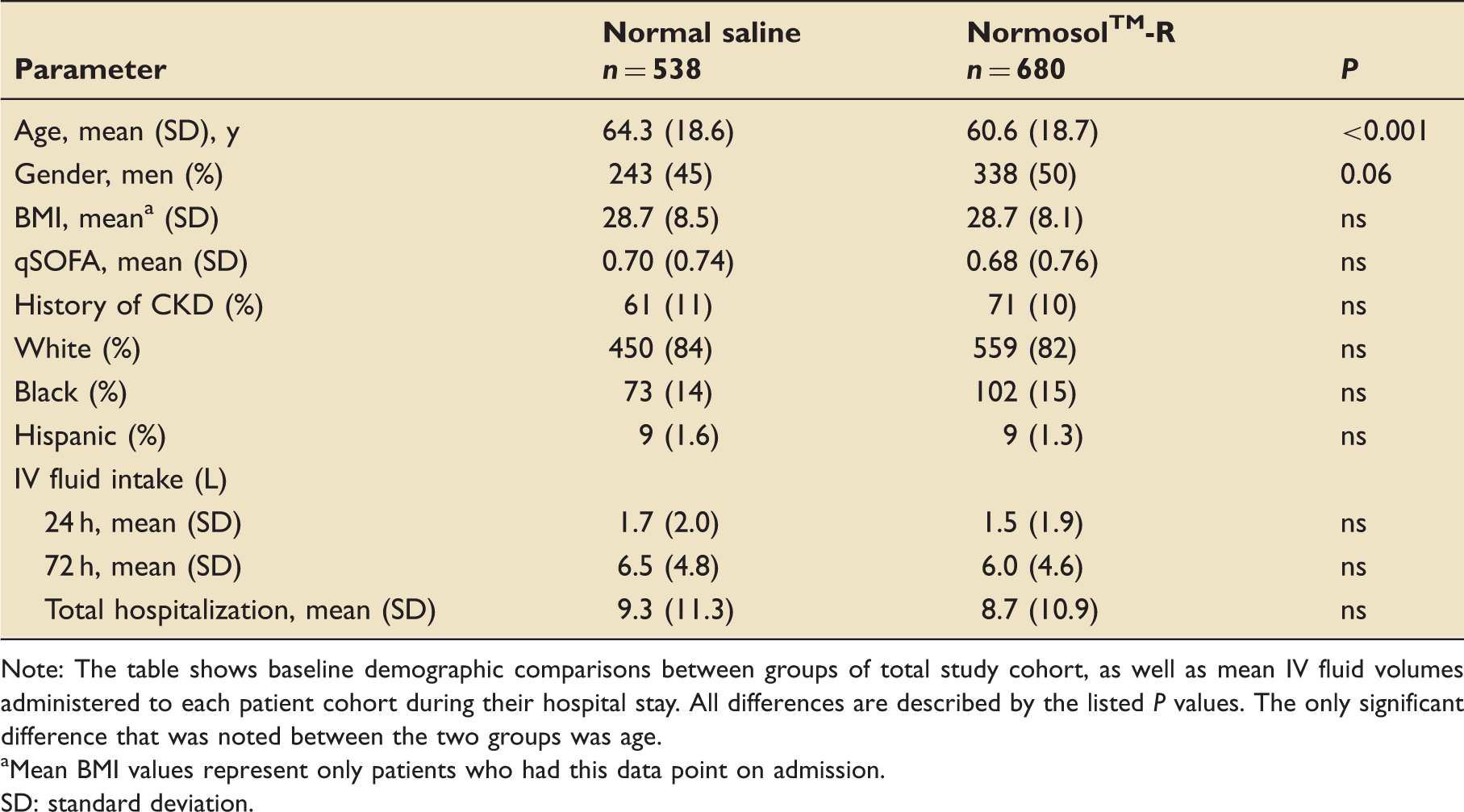
Note: The table shows baseline demographic comparisons between groups of total study cohort, as well as mean IV fluid volumes administered to each patient cohort during their hospital stay. All differences are described by the listed P values. The only significant difference that was noted between the two groups was age.
Mean BMI values represent only patients who had this data point on admission.
SD: standard deviation.
Fluid therapy
No significant differences in IV fluid volumes were observed at either 24 or 72 h post-admission, or over the entire hospital stay (Figure 2). This finding remained consistent after adjusting for age, gender, race, admission qSOFA, and history of CKD with linear regression modelling (Table 1).
Fluid volume comparison. Intravenous fluid totals following hospital admission. Fluid volume was recorded as a sum of bolus and maintenance fluids. Displayed volumes are cumulative so that the height of each bar corresponds to the average fluid intake at that time point. Volume received was similar between groups at all three time points in both univariate analyses, and after controlling for baseline characteristics.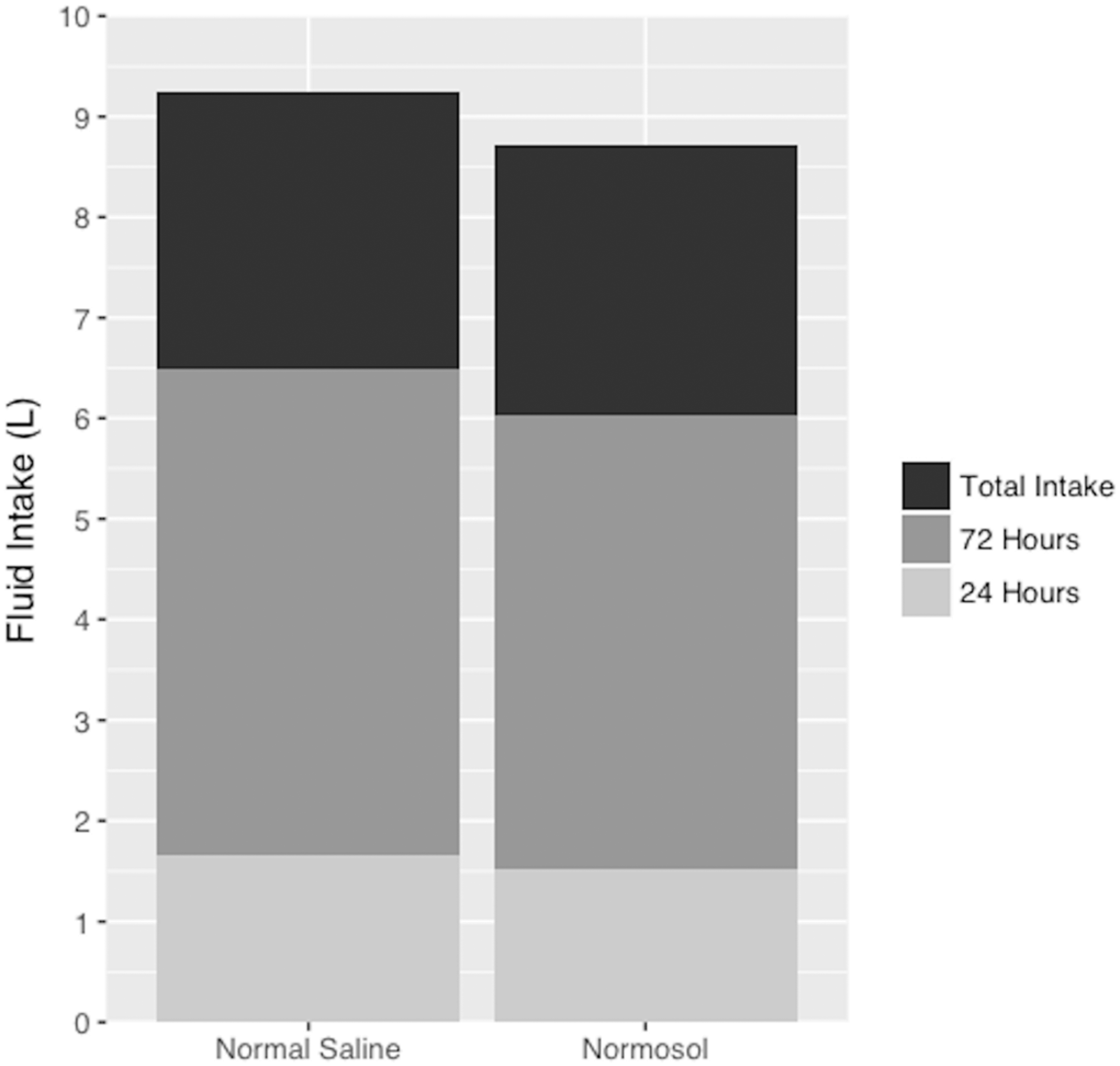
Primary and secondary outcomes
Association between resuscitation fluid type and primary and secondary outcomes among patients in total cohort and the larger volume subgroup.
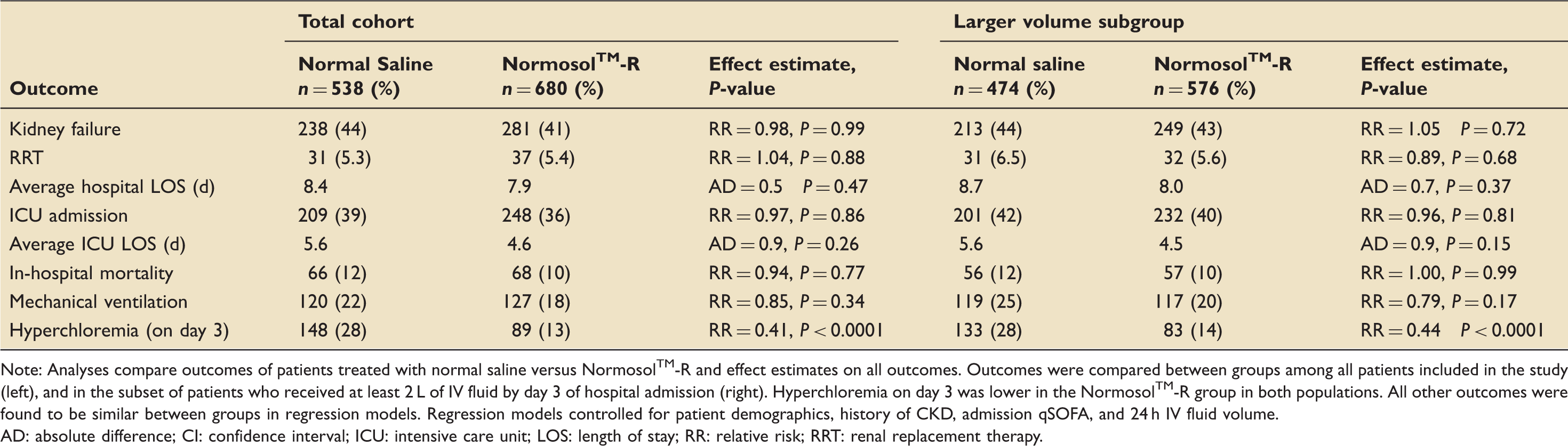
Note: Analyses compare outcomes of patients treated with normal saline versus Normosol™-R and effect estimates on all outcomes. Outcomes were compared between groups among all patients included in the study (left), and in the subset of patients who received at least 2 L of IV fluid by day 3 of hospital admission (right). Hyperchloremia on day 3 was lower in the Normosol™-R group in both populations. All other outcomes were found to be similar between groups in regression models. Regression models controlled for patient demographics, history of CKD, admission qSOFA, and 24 h IV fluid volume.
AD: absolute difference; CI: confidence interval; ICU: intensive care unit; LOS: length of stay; RR: relative risk; RRT: renal replacement therapy.
Larger volume subgroup
The large volume subgroup was defined as patients that received ≥2 L of IV fluids within the first 72 h, which included a total of 1050 patients, 474 from the normal saline group and 576 from the Normosol™-R group. After controlling for covariates, no significant differences were found between groups in the primary outcomes of AKI or RRT. Consistent with the original cohort, in this group, normal saline use was associated with an increased rate of hyperchloremia (28% vs. 14%, P < 0.0001, Table 2).
Electrolyte and metabolic disturbances
Comparison of lab values at 72 h by resuscitation fluid type.
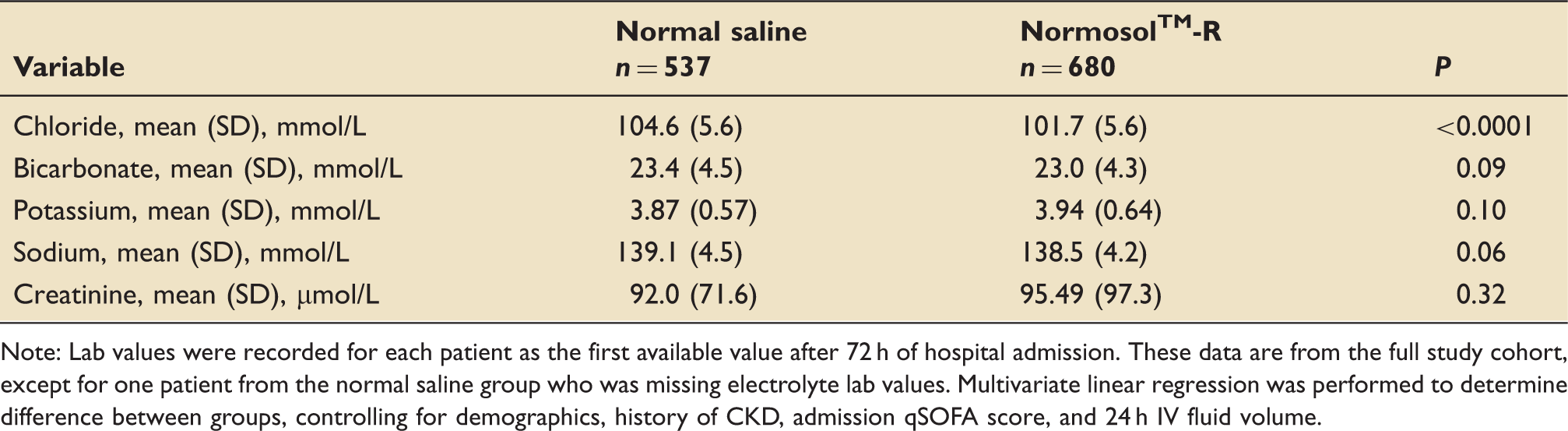
Note: Lab values were recorded for each patient as the first available value after 72 h of hospital admission. These data are from the full study cohort, except for one patient from the normal saline group who was missing electrolyte lab values. Multivariate linear regression was performed to determine difference between groups, controlling for demographics, history of CKD, admission qSOFA score, and 24 h IV fluid volume.
Discussion
In this retrospective before-and-after cohort study of septic patients resuscitated with either Normosol™-R or normal saline, no differences were observed in our primary outcomes including the rates of AKI, or RRT. Existing data comparing Normosol™-R and normal saline are diverse, but there is evidence in the literature to suggest that Normosol™-R has favourable effects on renal function when compared to normal saline. 12 Our findings suggest that although the use of Normosol™-R resulted in lower rates of hyperchloremia, this did not translate to improved renal outcomes.
Normosol™-R has been available commercially in the United States for over 25 years. 13 It is used as both a maintenance and resuscitation fluid, and because it does not contain calcium, it can be co-infused with citrate preserved blood.11,13 The rationale for the use of Normosol™-R instead of normal saline for resuscitation is that its lower chloride load and less induced acidosis may mitigate the harmful adverse effects that normal saline is known to cause.13,14 Animal models as well as studies in human volunteers have shown that the hyperchloremia induced from saline infusion leads to a reduction in renal perfusion and glomerular filtration rate.7,15 Additionally, it is theorized that the high chloride load from saline decreases the SID of the extracellular fluid, causing dissociation of water molecules to generate protons to preserve electro-neutrality, thereby decreasing the pH. This proposed mechanism of saline-induced acidosis relies on an understanding of the Stewart approach to acid-base chemistry.16,17 Although studies consistently have found that Normosol™-R tends to reduce metabolic disturbances compared to saline,14,16 it still remains unclear whether this translates into a clinically relevant improvement in patient outcomes.
Because many guidelines for initial septic resuscitation recommend that patients receive 30 mL/kg of IV crystalloid, we felt it was important to perform an analysis excluding patients who received less than 2 L of fluid (the volume an average sized adult should receive). Additionally, previous studies have suggested the renal-protective effects of balanced fluids may be more pronounced in patient populations receiving larger IV fluid volumes. 18 Patients in this larger volume subset (≥2 L by 72 h) received a median of 6.3 L over the course of their hospital stay (compared to 5.6 L in the total cohort) (Table 1). Consistent with the full cohort, patients receiving Normosol™-R had less hyperchloremia, but no differences were observed in the primary outcomes or secondary outcomes. This finding does not support previous findings which suggest saline’s detrimental effects are more pronounced at higher fluid volumes. 18
Our findings are largely consistent with those of the 2015 SPLIT (Saline versus Normosol™-R Fluid Therapy) and the 2016 SALT (isotonic Solution Administration Logistical Testing) prospective trials in which no differences were observed in outcomes between critically ill patients receiving saline or balanced IV crystalloids.18,19 Notably, while there were major differences between rates of AKI between studies, these were due to different definitions of such within the study. The need for RRT was nearly identical. The pooled results likewise reflect the similar renal outcomes between normal saline and balanced crystalloid groups (online supplementary digital content 1: table summarizing renal results of the current, SPLIT, and SALT studies).
The lack of renal-protective effects observed in the Normosol™-R group contrasts the findings from a large 2012 observational study which found that balanced crystalloid solution reduced kidney injury requiring RRT five-fold compared to saline in patients undergoing abdominal surgery. 12 Although there is no clear explanation for the discrepancy between our studies, the patient populations were substantially different. Most patients in that study were undergoing elective abdominal surgery (64.5–67.6%), while our population consisted of patients admitted through the ED with sepsis.
To address the inherent weaknesses of a before-and-after cohort study, we attempted to minimize the differences between our two cohorts by analysing data from the same months of the calendar year (March through September) for each group. Additionally, this method allowed for a six-month period in which the ED and critical care departments transitioned default fluid types. Department chairs in our hospital confirmed that no substantial structural or procedural changes occurred during this transition period. Our regression models adjusted for IV fluid volume received at 24 h, which helped control for shifting trends in the volume management of sepsis, as well as adverse events associated with aggressive fluid resuscitation. It should also be mentioned that there was a statistically significant difference between groups in patient age, as the Normosol group was on average younger (64.3 vs. 60.6 years, P < 0.001). To control for this baseline difference between groups, age was included as an independent variable in regression models.
Data collection in our study relied largely on discharge ICD9CM and ICD10CM coding, which could lead to the under or overestimation of the true incidence of various diagnoses and outcomes. However, our finding that 45.6% of patients overall were diagnosed with AKI is consistent with prior prospective studies who have observed AKI in septic patient.20,21 During the study period, implementation of ICD-10 was adopted at the study site, how this impacted the data is unknown.
A notable limitation of this study is the exclusion of patients with a history of cardiac or renal failure, as these patients comprised a large proportion of the screened population (484 out of 1717). These patients were excluded from the study because these patients are often treated less aggressively than other patients regarding the timing and volume of IV fluid resuscitation, due to fears of volume overload. This pattern of differential treatment is supported by recent evidence showing that among patients receiving crystalloid resuscitation for sepsis or septic shock, patients with comorbid cardiac and renal failure received delayed crystalloid initiation.22–25 However, it is reasonable to consider that these patients may be the very individuals who are at increased risk of the adverse effects associated with normal saline. Therefore, it should be acknowledged that because our study excluded these “high-risk” populations, our findings might not be generalizable to the entire septic population.
This study contributes to the growing body of data comparing balanced crystalloids to 0.9% normal saline for IV fluid resuscitation. Our study suggests that either balanced crystalloid or normal saline are reasonable choices in initial septic resuscitation. Further data are needed to evaluate the potential harmful effects of large volume normal saline resuscitation.
Conclusions
Among patients admitted through our ED with a diagnosis of sepsis, initial resuscitation with a balanced crystalloid (Normosol™-R) did not result in reduced rates of in-hospital mortality, total hospital LOS, ICU admission rates, AKI or RRT. The same findings were observed among the subset of patients who received at least 2 L of IV crystalloid fluid in the first 72 h following hospital admission. Further prospective studies to evaluate these findings are needed.
Supplemental Material
Supplemental material for Normal saline versus Normosol™-R in sepsis resuscitation: A retrospective cohort study
Supplemental material for Normal saline versus Normosol™-R in sepsis resuscitation: A retrospective cohort study by Ryan A Duffy, Mathab B Foroozesh, Robert D Loflin, Susanti R Ie, Bradley L Icard, Allison N Tegge, Jonathan R Nogueira, Damon R Kuehl, Dan C Smith and Anthony L Loschner in Journal of the Intensive Care Society
Footnotes
Acknowledgements
The authors thank Carilion Healthcare Analytics Team for their help with data acquisition. This study was performed at the Virginia Tech Carilion School of Medicine, Roanoke, VA.
Authors’ contributions
MF, SI, DK, DS, AL designed the study. RD analysed data. AT consulted for statistical analysis. RL, BI, JN reviewed and interpreted data. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study was approved and a waiver of informed consent was obtained by our institutional review board (IRB protocol #1996).
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
