Abstract
Background
Acute kidney injury is common in critically ill patients with detrimental effects on mortality, length of stay and post-discharge outcomes. The Acute Kidney Injury Network developed guidelines based on urine output and serum creatinine to classify patients into stages of acute kidney injury.
Methods
In this analysis we utilize the Acute Kidney Injury Network guidelines to evaluate the acute kidney injury stage in patients admitted to general and cardiac intensive care units over a period of 18 months. Acute kidney injury stage was calculated in real time hourly based on the guidelines and using these temporal stage scores calculated for the population; the prevalence and progression of acute kidney injury stage was compared between the two units. We hypothesized that the prevalence and progression of acute kidney injury stage between the two units may be different.
Results
More cardiac intensive care unit patients had no acute kidney injury (stage <1) during their intensive care unit stay but more cardiac intensive care unit patients developed acute kidney injury (stage >1), compared to the General Intensive Care Unit. Both at intensive care unit admission and discharge, more General Intensive Care Unit patients had acute kidney injury; however, the number of cardiac intensive care unit patients with acute kidney injury was three times higher at discharge than admission. Acute kidney injury developed in a different pattern in the two intensive care units over five days of intensive care unit stay. In the General Intensive Care Unit, acute kidney injury was most prevalent on second day of intensive care unit stay and in cardiac intensive care unit acute kidney injury was most prevalent on the third day of intensive care unit stay. We observed the biggest increase in new acute kidney injury in the first day of General Intensive Care Unit and second day of the cardiac intensive care unit stay.
Conclusions
The study demonstrates the different trends of acute kidney injury pattern in general and cardiac intensive care unit patient populations highlighting the earlier development of acute kidney injury on General Intensive Care Unit and more prevalence of acute kidney injury on discharge from cardiac intensive care unit.
Introduction
Acute kidney injury (AKI) is prevalent in hospitalized patients and particularly in the intensive care unit (ICU) due to sepsis, major surgery and iatrogenic complications.1–7 AKI can be defined and classified according to the Acute Kidney Injury Network (AKIN) classification that utilizes serum creatinine and urine output measurements. 8 It is a common phenomenon, with large retrospective studies quoting incidence of AKI between 22 and 67% and mortality between 14 and 36%.1–3,9 AKI is an independent risk factor for mortality in critically ill patients and the short- and long-term complications of AKI extend beyond the acute phase.5,6,10 ICU management of AKI consists of supportive therapy and minimizing further kidney injury by treating the underlying cause, avoiding nephrotoxic drugs, optimizing fluid status and perfusion pressure and the use of renal replacement therapy. However, there is no single remedy for established AKI and early identification of patients with AKI has the potential to improve outcomes. 7
In this analysis we deployed an algorithm on routinely collected electronic data in real time within a clinical information system to determine the differences in stages of AKI, AKI incidence and progression in general (GICU) and cardiac intensive care unit (CICU) patients. To our knowledge this is the first study to investigate AKI stage using electronically collected data including both serum creatinine and hourly urine output. In our institution GICU looks after a mixed medical and surgical population whereas CICU admits mainly elective post-cardiac surgery patients. We describe the incidence and progression pattern of AKI in the two units utilising the high fidelity electronic health records (EHRs).
Methods
Setting
This retrospective observational study was undertaken in two ICUs at University Hospitals Bristol, UK, a closed format tertiary medical and surgical ICU (GICU) and closed format tertiary cardiac ICU (CICU). GICU is a tertiary ICU with over 1200 admissions per year and CICU is a regional cardiac surgical centre with over 1800 admissions a year. Both units have used the Philips ICIP EHR system since 2015. This EHR system automatically collects all information relating to patient care including laboratory data and hourly vitals and observations.
Patients
Consecutive adult patients admitted to GICU and CICU over 18 months for more than 24 h were included in the study. Encounters were excluded from analysis if (1) age, race or gender information was not available; (2) age at admission >90 years; (3) had AKI on admission defined as oligoanuria in the first 6 h of admission. Additionally, encounters where no sufficient data were available to calculate AKI score (urine output, body weight, serum creatinine) were excluded.
Data collection and preprocessing
We collected anonymized data from 4571 patients (5140 encounters). After applying the exclusions, the final cohort consisted of 4000 encounters (3780 patients). This group was split into GICU 1676 encounters (1560 patients) and CICU 2324 encounters (2236 patients). Urine output data are recorded hourly when patients are catheterized and serum creatinine levels are measured daily in all patients. The study was deemed not to require individual patient consent by the institutional review board.
Information on hourly urine output, serum creatinine, body weight and demographic information were extracted for the cohort from the Philips ICIP EHR. In addition, information on primary diagnosis and nature of admission were extracted from Intensive Care National Audit and Research Centre (ICNARC) database.
AKI computation
An algorithm was developed to calculate AKI score electronically as previously described 11 using AKI staging guidelines from AKIN.
We calculated the AKI stage for the entire duration of ICU stay for all encounters. Using hourly urine output data we calculated AKI urine output stage every 15 min. Serum creatinine data were used to calculate AKI serum creatinine stage. We used an empirical formula based on age, race and gender to estimate baseline creatinine. AKI serum creatinine staging was done by comparing serum creatinine values to this baseline. The overall AKI stage was the maximum of the AKI urine output stage and AKI serum creatinine stage. The algorithm computed a new AKI stage every 15 min.
Descriptive statistical parameters
AKI stage was assessed on a population level for the patient’s duration of ICU stay. The differences in AKI prevalence between the two ICUs were evaluated using the following metrics computed from the estimated AKI stage time series values.
Maximum AKI stage over 24-hour periods for first 5 days of ICU stay in (a) GICU and (b) CICU. Each bar represents the distribution of patients in the 4 AKI stages during a 24-hour period shown as a percentage. The vertical axis is limited to 70% to better display the proportion of patients with AKI stage 1 or greater. The numbers on the bar indicate the number of patients in each stage.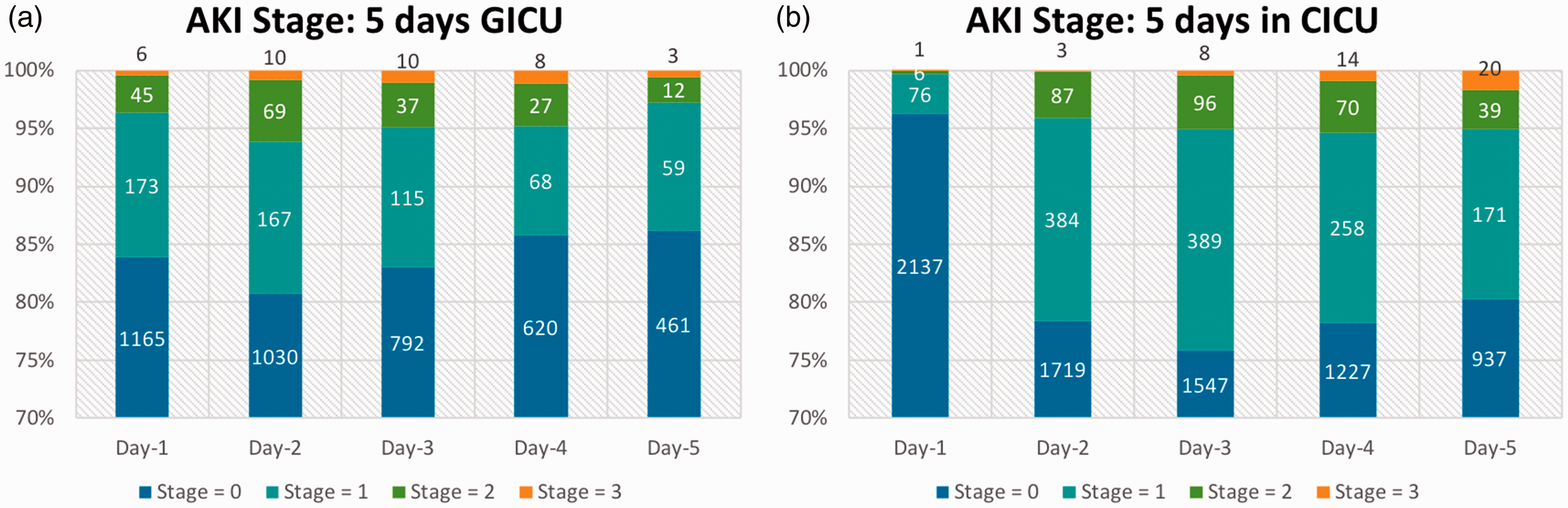
Number of patients with no change (grey), deterioration (red) and improvement (green) in AKI stage in (a) GICU and (b) CICU. The vertical axis is limited to 70% to better display the proportion of patients with AKI stage 1 or greater. The numbers on the bar indicate the number of patients in each group.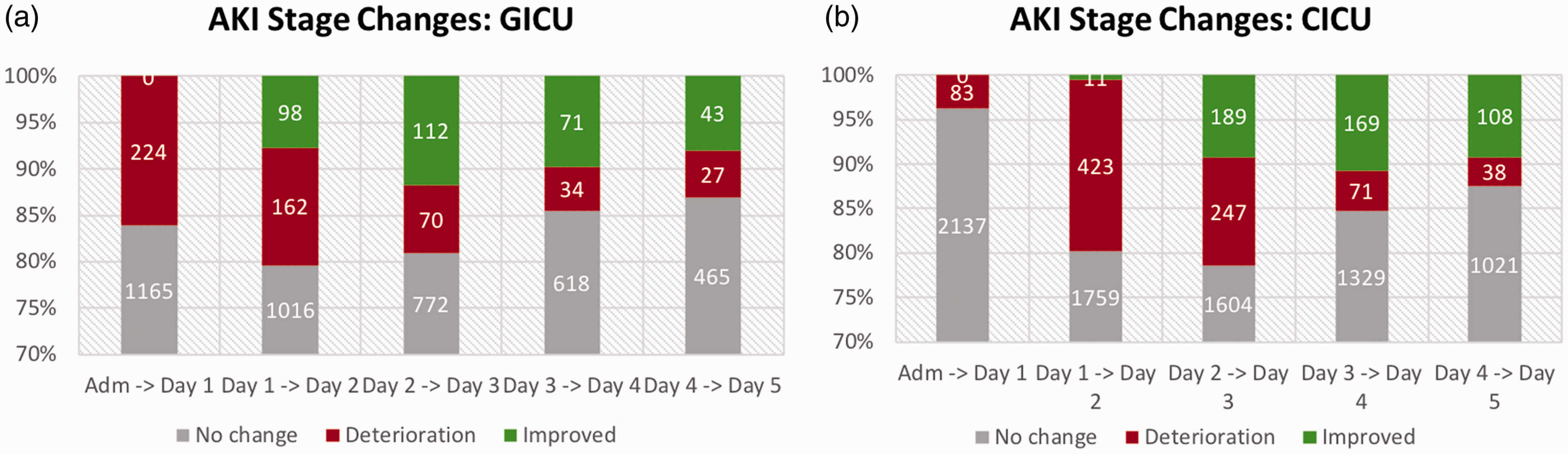
New AKI stages developing per day of ICU stay in (a) GICU and (b) in CICU. The number of patients developing new AKI is further divided into groups by magnitude of stage increase represented by different colors.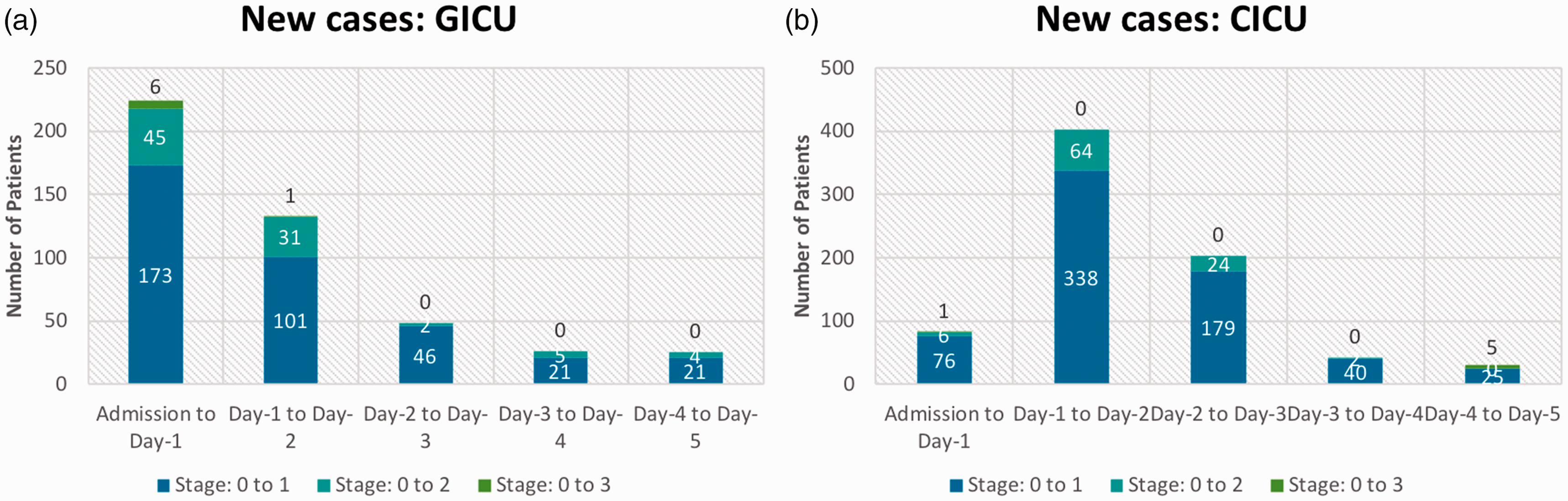
Results
Study population characteristics.
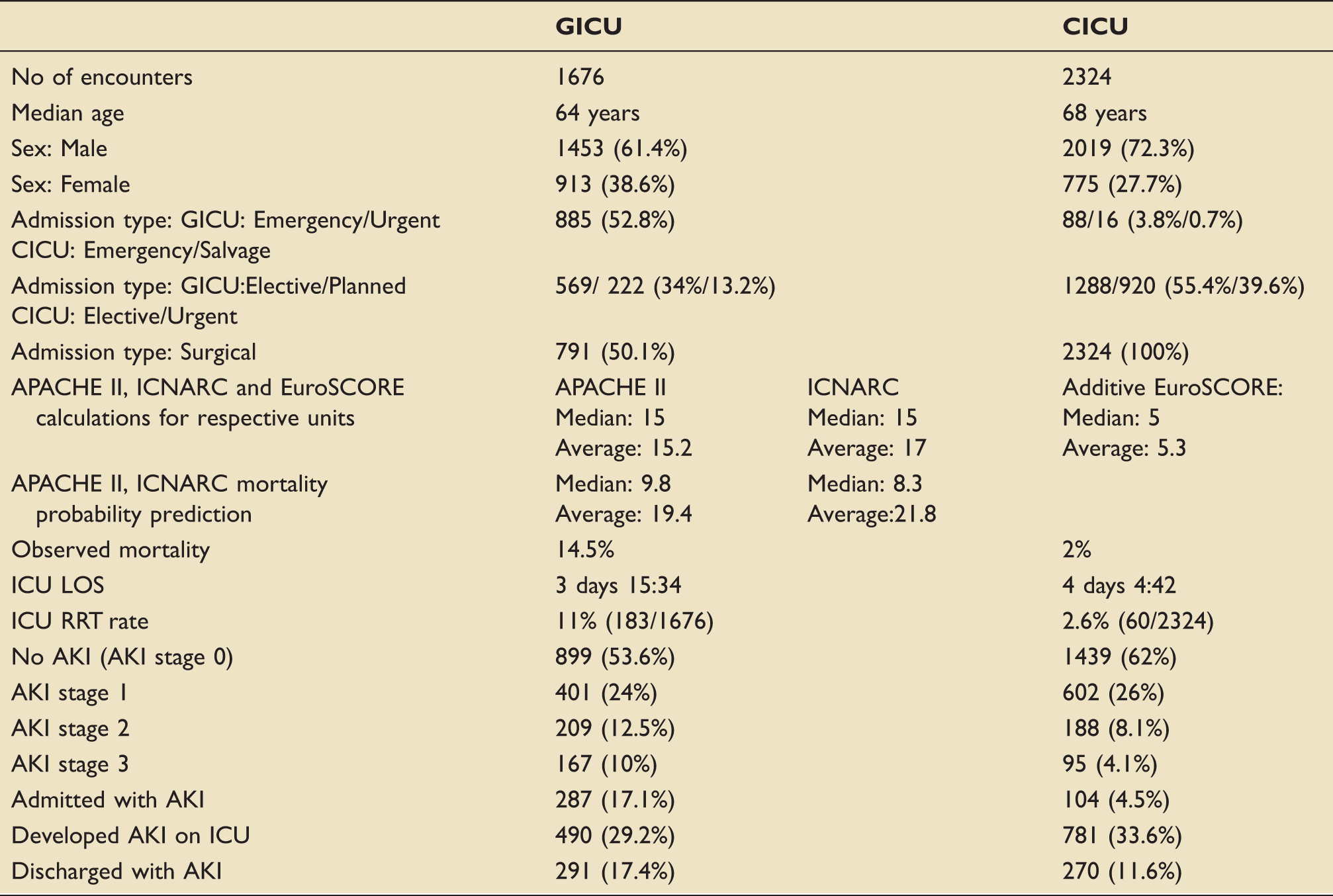
AKI: acute kidney injury; APACHE II: Acute Physiology And Chronic Health Evaluation II; CICU: cardiac intensive care unit; GICU: General Intensive Care Unit; ICNARC: Intensive Care National Audit and Research Centre; ICU: intensive care unit; LOS: length of stay; RRT: renal replacement therapy.
GICU admissions come from medical (49.9%) and general surgical (50.1%) background (see Table 1 for further details). The majority of GICU elective surgical patients have undergone cancer surgery (thoracic, head and neck, colorectal, upper GI and hepatobiliary) and medical GICU patients come from General Medicine, Hepatology, Haematooncology, Respiratory and Cardiology wards (including 10% of all patients who have suffered an out-of-hospital cardiac arrest). The median APACHE score for GICU patients admitted during the study period was 15 with average APACHE and ICNARC predicted mortality of 19.4 and 21.8%, respectively. Observed mortality for GICU was 14.5%.
CICU predominantly admits elective/urgent cardiac surgical patients. More than 3/4 of operations are performed utilizing cardiopulmonary bypass (76.3%). The main cardiac surgical procedures undertaken are as follows: CABG (54.3%), valves (38.5%), major aortic (7.2%). The median and average additive EuroSCORE for CICU patients admitted during study period was 5 and 5.3, respectively, and CICU mortality was 2.03%.
AKI incidence
Encounters were grouped by their maximum AKI stage into four groups. CICU had a higher proportion of encounters with no AKI (62%) compared to general ICU (54%). In addition, more CICU patients developed up to AKI stage 1 than GICU patients. Conversely more encounters in the GICU developed higher stages of AKI (stage 2 and 3) compared to CICU (Table 1).
At admission fewer GICU patients had no AKI (83%) than CICU (96%). Ten per cent of GICU population had AKI stage 1. AKI stage 2 and 3 prevalence was 3 and 4%, respectively. In comparison, in CICU only 3% of the encounters had AKI stage 1 and less than 2% of the encounters had AKI stage 2 and 3 combined.
At discharge in the GICU, the distribution of encounters in the four stages is similar to admission – no AKI: 83%, AKI stage 1 reduces to 9%. In the CICU, at discharge the distribution of encounters in four stages changes. The proportion of encounters discharged with no AKI decreases to 88%. The biggest increase is in AKI stage 1 group (9%) and the number of encounters discharged with AKI stage 2 and 3 is also higher.
Furthermore, the number of new AKI cases developing per day of ICU stay indicates a very different pattern of AKI development in the two ICUs. Excluding encounters with AKI at admission, we had 1389 encounters in GICU and 2220 encounters in CICU. In GICU most new AKI cases occurred on the first day and the number of new AKI cases decreased from day 1 onwards. In CICU, most new cases develop on the second day and the number of new cases decreases after that. In CICU a larger proportion of encounters develop new AKI from day 1 to day 2 (18.3%) while in GICU the maximum proportion of new cases is 16.1% (admission to day 1) (see Figure 1(a) and (b)).
These results are borne out in our analysis of AKI incidence by day of ICU stay. We found differences in patient distribution across AKI stages. In the GICU 16.1% of patients had some AKI risk on first day and this increased till day 2 after which it decreased (Figure 2(a)). The proportion of patients with any AKI risk per day varied from 19.3% (day 2) to 13.8% (day 5). Fewer patients in GICU had any risk on day 5 compared to day 1. In contrast, in the CICU, fewer patients had any AKI risk at admission (3.7%) (Figure 2(b)). This increased till day 3 to 24.2%. Thus, the variation in AKI risk between days in CICU was much larger and more patients had some AKI risk on day 5 (19.7%) than day 1.
These results are also reflected in the daily increase (deterioration) and decrease (improvement) in AKI stage. In GICU the largest deterioration of AKI risk (16.1%) is seen on day 1 and the number of patients with deteriorating AKI risk decreases (Figure 3(a)). The proportion of patients with improvement in AKI risk increases from day 2, reaching a maximum on day 3 (11.7%). In the CICU, the proportion of patients with deteriorating AKI risk increases till day 2 (19.3%) after which it decreases (Figure 3(b)). The proportion of patients with improvement in AKI risk increases from day 2, reaching a maximum on day 4 (10.8%).
Dialysis rates by ICU type
One hundred and eighty-three (10.9%) encounters in GICU and 60 (2.6%) encounters in CICU received dialysis in ICU. The median dialysis duration in GICU was three days 15 h and in CICU was four days 13 h. The median ICU stay duration for dialysis encounters was eight days 20 h in GICU and 15 days 9 h in CICU.
Discussion
In this single centre, retrospective observational study we have utilized a previously described and validated electronic surveillance tool (electronic algorithm) among a large cohort of critically ill surgical and medical patients (over 4500 consecutive intensive care unit admissions) in order to assess the incidence and progression of AKI. To our knowledge this is a first report of utilizing this electronic algorithm to describe the incidence, progression and effect of AKI on LOS and mortality among large number of consecutive ICU admissions using routinely collected data. The observed incidence of AKI and mortality associated with it in our cohort of critically ill patients is similar to the one reported in other studies. 12
Unlike previous studies2,3 we found that large proportion of patients develop AKI during ICU stay rather than present with it (especially in CICU where the highest progression to AKI stage 1 was observed between day 1 and 2).
This study demonstrated a difference in dialysis rate between the two units – 11% in GICU (183/1676) and 2.6% in CICU (60/2324). A larger proportion of GICU patients receive dialysis but do so for a median duration of eight days compared to 15 days on CICU.
More patients with higher AKI stages in GICU compared to CICU can be explained by the fact that GICU caters for emergency medical and surgical patient populations compared to CICU where the overwhelming majority of admissions are from elective cardiac surgical workload. Medical GICU patients have higher risk of AKI and account for half of admissions. This would explain why overall a larger proportion of GICU patients have AKI at admission and discharge. Emergency and urgent surgical patients are also more likely to develop AKI compared to elective and scheduled surgical population. Many risk factors for developing AKI are not modifiable (increasing age, peripheral vascular disease, hyperlipidaemia, hypertension, chronic kidney disease, previous stroke) but intensivists should be aware of these risk factors in order to potentially reduce their burden (e.g. by choosing a higher MAP target for a hypertensive patient).
Cardiac surgery 13 (on or off pump) together with aortic cannulation and cross clamping combined with the use of vasopressors and inotropes presents an unavoidable but well-defined ischaemic insult including systemic and renal inflammation, microvasculature injury and tissue oedema, increase in oxidative stress, reperfusion injury, cardiopulmonary bypass-induced haemolysis and cholesterol emboli. These mechanisms would explain why a larger proportion of patients develop AKI on CICU (34% versus 29% GICU) and are discharged with resolving but still present AKI. This very prescribed insult explains the highest incidence of AKI between days 1 and 2.
In general ICU the maximum deterioration occurs between admission and day 1 which usually represents the fact that the kidney ‘insult’ happened 24–48 h before ICU admission and is a marker of disease severity causing ICU admission in the first place.
It is not at all surprising that patients are discharged from both ICUs with AKI still present as in clinical practice factors such as cause of AKI, trends in serum creatinine, evolution of critical illness and comorbidity influence the decision of the intensivist to discharge a patient from ICU.
Strengths and limitations
The main strengths of our study include its large size, pragmatic, well-defined study population, comprehensive laboratory and urine output measurement data utilizing the time series of hourly collected weight adjusted urine output measurements, and complete follow-up and mortality data derived from the routinely collected dataset (ICNARC). We demonstrated utilising real time, daily calculation of AKI stages and progression daily.
However, the following should be considered when interpreting our results.
First, we used routine laboratory data to assess AKI at ICU admission but only for patients staying on ICU for longer than 24 h. It is possible that some patients could have developed AKI after ICU discharge but given the short ICU stay was not included in our analysis. Second, urine output criteria are affected by diuretics, which are commonly used in ICU patients. However, we looked at daily creatinine measurements as well as the time series of hourly weight adjusted urine output to determine if patients developed and/or progressed to higher stages of AKI.
Conclusion
AKI is common in general and cardiac ICUs although most often mild (stage 1 AKI according to AKIN classification). Effective treatment and prevention strategies for AKI may be possible in the future but for now the efforts should concentrate on early identification of patients with worsening AKI and initiation of care bundles designed to mitigate the modifiable risk factors and providing attentive supportive ICU care. Early identification of worsening AKI stage allows the clinicians to increase monitoring, initiate preventative and therapeutic interventions to optimize high-risk patients or even to enrol patients into clinical trials. This is particularly true in the context of cardiac surgery, which provides a well-defined and described standardized insult to patients (majority of which are elective) who are monitored in ICU environment after surgery. Ability to diagnose and spot AKI early may reduce the negative outcome it has on critical care patients and AKI sniffers (electronic algorithms designed to identify AKI based on routinely collected data in real time) have been developed to help with early detection of worsening AKI. Future studies should concentrate on deployment of interventions utilizing AKI care bundles based on active surveillance and workflow interventions comprising of interruptive checklists with clinician decision support to aid healthcare professionals in managing this syndrome more effectively.
Footnotes
Ethics approval and consent to participate
Ethics approval waived by the institution as this was a retrospective data analysis.
Consent for publication
Not applicable.
Availability of data and materials
The data that support the findings of this study are available from University Hospitals Bristol NHS Foundation Trust but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of University Hospitals Bristol NHS Foundation Trust.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EG and LE are employees of Philips Research North America. KP is employee of Philips Innovation Campus, India.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors’ contributions
Conception of idea: CPB, EG Study design: MAP, CPB, EG, LE, KP Acquisition of data: CPB, EG, LE, KP Performance of the study and data analysis: MAP, CPB, EG, LE, KP Analysis of the results: MAP, EG, LE, KP, TG, MT, CPB Writing and revising the manuscript: MP, EG, LE, KP, TG, MT, CB All authors approved the manuscript and agreed to be accountable for all aspects of the work.
