Abstract
Clostridium perfringens sepsis has been ascribed a dismal prognosis when associated with massive intravascular haemolysis. We present a 71-year-old woman’s fatal case which was compounded by isolated right ventricular cardiogenic shock. In this context, combined use of transthoracic echocardiography and pulmonary artery catheter monitoring is able to yield an individualized hemodynamic resuscitation. We discuss key aspects related to right and left heart mechanical efficiency, hypothesize as to the pulmonary hypertension mechanism of our case and set to emphasize a physiologically based framework for right ventricular failure hemodynamic management.
Introduction
The traditional description of septic shock is a vasoplegic and distributive state with profound microcirculatory and cardiac alterations. Traditional resuscitative strategies (Rivers’s early goal-directed therapy (EGDT)) focus on fluid administration but the actual incidence of fluid responsiveness is often lower than expected, close to 50% 1 in a heterogeneous group of critically ill patients and even lower (23%) in a subgroup of previously resuscitated ARDS patients. 2 Recently, a trio of trials (ProMISe, ARISE and ProCESS) failed to show a benefit of the original EGDT approach when compared to usual care.3–5 Indeed, aside for the ‘early’ concept relating to antibiotherapy and fluid resuscitation which still remains valid, the EGDT hemodynamic framework lacks a solid physiological background and may even be deleterious. 6 Nonetheless, all three trials share a substantial amount of accrued fluid at 72 h with a commensurate increase in CVP at 6 h.7,8 To further improve outcome, these findings demand a haemodynamically guided, flow-oriented, individualized and fluid restrictive approach.1,7,9 Whether it is echocardiographically driven or not, one such approach is able to minimize the untoward effects of liberal fluid administration 10 and acknowledge the following three complementary principles: fluid therapy has the potential to counterintuitively augment the hyperdynamic state, 11 diastolic dysfunction may further alter ventricular–arterial coupling and shift the fluid-pressure sensitivity 12 and the right ventricle is not merely a bypassable flow-setter as in a ‘Fontan circulation’.13,14 The latter reflects an important shift in the paradigm and underlines the permissive role of the ‘forgotten’ ventricle. 15
Herein, we describe the first reported case of Clostridium perfringens sepsis complicated by isolated right ventricular (RV) cardiogenic shock. We then characterize the importance of echocardiography in the assessment and resuscitation of septic shock.
Case presentation
A 71-year-old woman with no prior medical history or comorbidities presented to a district general hospital with a 12-h history of fever, jaundice and right upper quadrant abdominal pain. An abdominal ultrasound did not reveal any pathology and she was initially treated with intravenous Co-amoxiclav, Clindamycin and Metronidazole. Over the next 4 h, she deteriorated rapidly requiring intubation, cardiovascular support with noradrenaline and urgent transfer to our intensive care unit. On arrival, her physical examination revealed a shock state with diffuse petechiae and dark red urine. A second abdominal ultrasound revealed gallstones but a normal gall bladder and no biliary or common bile duct dilation. The patient’s clinical status precluded further imaging.
Laboratory results.
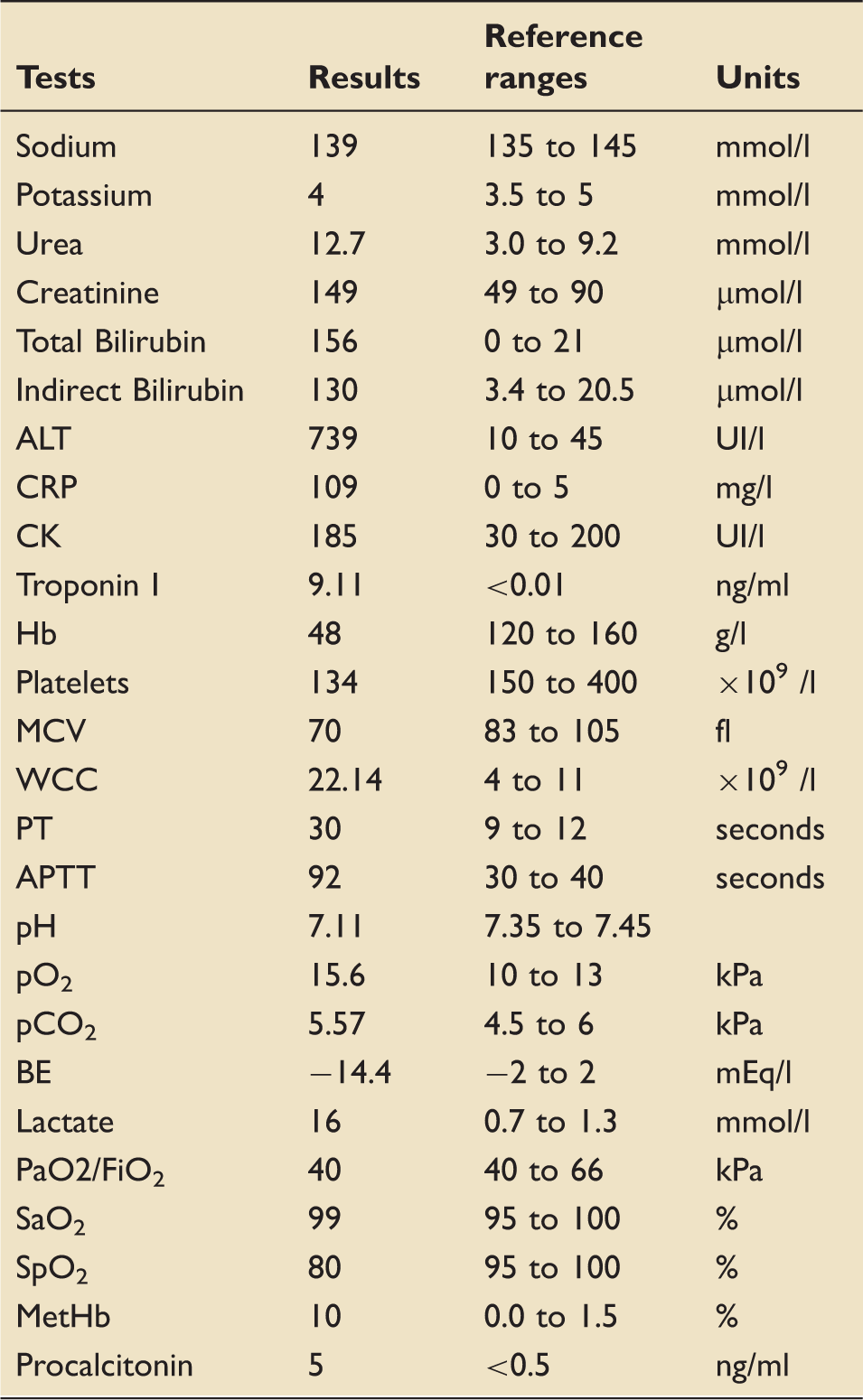
Over the course of the following 24 h, the patient received haemodynamic-directed cardiovascular resuscitation guided by repeated transthoracic echocardiography (TTE). The initial TTE showed severe dilatation of the right ventricle (RVEDA/LVEDA = 1.3) (see Figure 1) with impaired right ventricle contractility (see Table 2), raised pulmonary vascular resistance (3.16 WU or 253 dynes s/cm
5
calculated according to Abaas et al.
16
) and a flattened, D-shaped interventricular septum with paradoxical motion consistent with right-sided pressure overload and consequent acute cor pulmonale. In this regard, the pulmonary flow profile showed midsystolic deceleration and the pulmonary acceleration time was decreased (81 ms) (see Table 2). The inferior vena cava (IVC) was dilated (23 mm) and did not change with ventilation. The left ventricle (LV) was underfilled and hyperdynamic. The stroke volume was measured at 19.5 ml using the Doppler velocity time integral (VTI) method in the LV outflow tract (LVOT), in keeping with RV cardiogenic shock. Also, an arterial to ventricular elastance (Ea/Es) ratio of 1.31 (<1.36) calculated according to Chen’s method
17
revealed an adequately coupled LV to the systemic circulation whereas the ratio of the TAPSE (10 mm) to the echocardiographically derived pulmonary artery systolic pressure (PASP) (50 mmHg) of 0.20 supported the view of a severely uncoupled RV to pulmonary circulation.
18
The respiratory variation in the systolic blood pressure (SBP) and the LVOT VTI was suggestive of fluid responsiveness but given the context of severe RV impairment, it was deemed a false positive.
19
A modified mini-fluid challenge
20
(200 ml albumin 20% resulting in ΔLVOT VTI of 6%) confirmed the lack of fluid responsiveness.
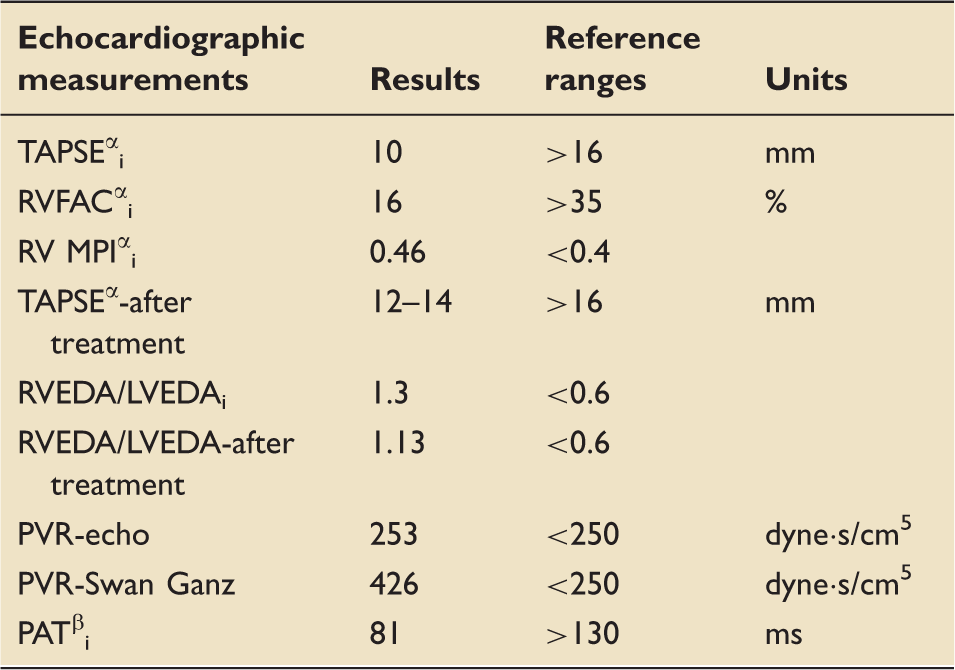
Apical four-chamber view, RVEDA to LVEDA ratio of 1.3 diagnostic for severe RV dilatation. RV function assessment. i: initial; α: RV contractility index; TAPSE: tricuspid annular plane systolic excursion; RVFAC: RV fractional area change; RV MPI: right ventricular myocardial performance index; RVEDA: right ventricular end-diastolic area; LVEDA: left ventricular end-diastolic area; PVR: pulmonary vascular resistance; PAT: pulmonary acceleration time; β: mean PAP = 79–0.45 × PAT.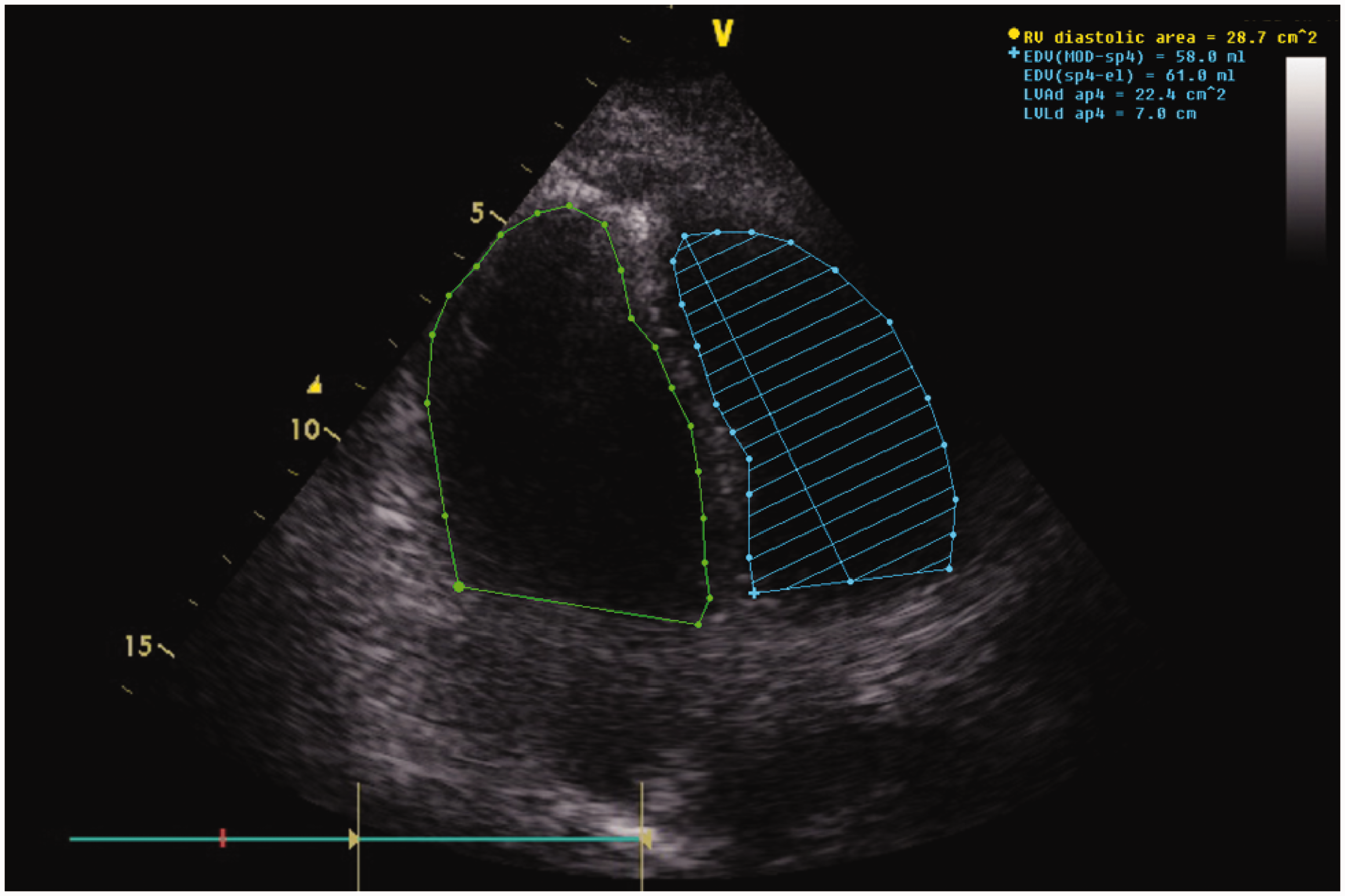
The aforementioned measurements were done on norepinephrine infusion under which the arterial lactate continued to worsen.
In the context of a dilated and impaired RV, a hyperdynamic but empty LV and lack of fluid responsiveness, a Swann Ganz catheter was inserted and confirmed a raised pulmonary artery pressure (PAP 46/31 mmHg; meanPAP 36 mmHg) with increased PVR (426 dynes s/cm5), a severely depressed cardiac output (CO) of 3 l/min but with a mixed venous saturation (SvO2) of 72%. We then initiated support with a phospho-diesterase inhibitor (milrinone) and inhaled nitric oxide (NO). This strategy resulted in a transient improvement in right-sided cardiac function and cardiac output (TAPSE increased to 12–14 mm and the RVEDA/LVEDA ratio receded to 1.13) (see Figure 2).
Apical four-chamber view, RVEDA/LVEDA receded to 1.13 consequently to an improved LVEDA reflecting an overall improvement of the RV-LV mismatch after adding NO inhalation and a PDE-3 inhibitor to norepinephrine. Not shown, LVOT-VTI increased to 11.9 cm from 9.7 cm reflecting a 22% increase in stroke volume.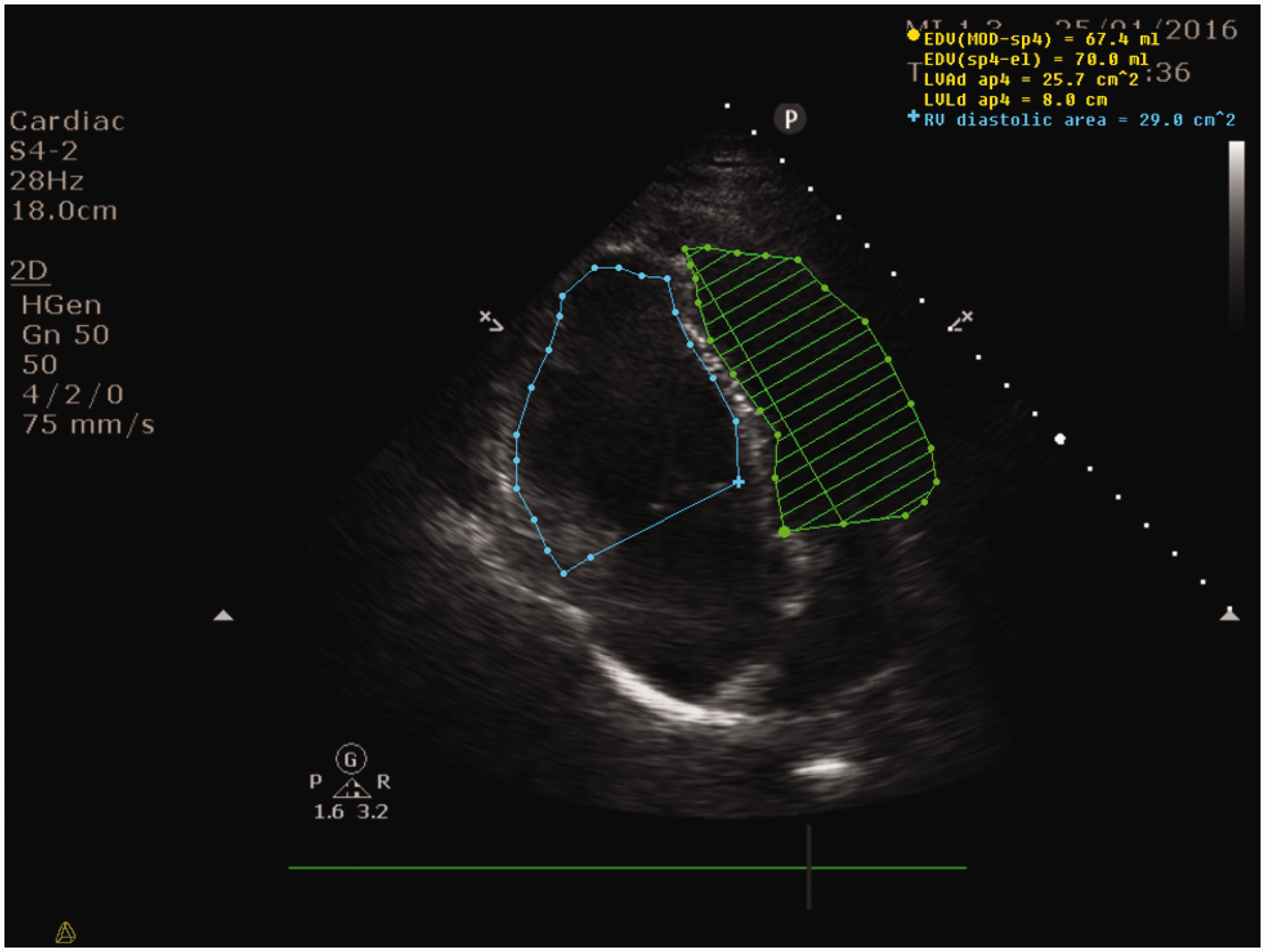
Related to a possible ventilation-induced RV strain, we report an acute cor pulmonale risk score of 0 in consonance with a driving pressure of 16 mbar, PaO2/FiO2 ratio of 40 kPa, PaCO2 of 5.57 kPa and lack of any evidence of pneumonia-induced ARDS. 21 Our ventilatory strategy included a moderate PEEP of 8 mbar and was in full concert with a lung protective approach. 22
Despite target-based resuscitation including renal replacement therapy, the lactataemia continued to worsen and death ensued within 24 h.
A hospital post-mortem was performed that revealed no evidence of pulmonary emboli, patent coronary arteries, isolated and marked dilatation of the right ventricle with normal histology of the myocytes. The liver was normal and no surgical source for the Clostridium was found.
Discussion
C. perfringens, formerly known as welchii (Micula 1900), is a Gram-positive rod, normally found in human gastrointestinal and genitourinary microbiota. It is an anaerobe but still aerotolerant, capable of doubling in only 7 min and of secreting at least a dozen of enzymes, of which the most well known is the alpha toxin. A calcium-dependent enzyme, it has the potential to hydrolyse phosphatidylcholine, lecithin and sphingomyelin. This accounts for red blood cell membrane phospholipids hydrolysis, leading to spherocytosis and subsequent haemolysis and other effects such as tissue necrosis and platelet aggregation.
Bacteraemia with C. perfringens is usually hepatobiliary or gastrointestinal and can be asymptomatic or can result in full-blown septic shock.
Mainstay of therapy for Clostridium sepsis includes organ support, antimicrobials and, when necessary, surgical/percutaneous debridement. A surgical diagnosis was not found in this case and the post-mortem did not reveal a surgical source for the sepsis.
Of particular interest to our case is the aetiology of the patient’s RV failure resulting in cardiovascular shock which to the best of our knowledge is the first description of this in C. perfringens sepsis. We have already reported similar findings in three other patients 23 with septic shock.
This case underlines the crucial role that TTE plays in septic shock, independently assessing the ventricular function and reliably guiding resuscitation. In this case, it revealed an isolated RV septic cardiomyopathy with severe systemic vasoplegia. This allowed us to focus on a restrictive fluid strategy, inotropy and pulmonary vasodilatation as well as systemic vasoconstriction along with optimization of physiological parameters (CO2, pH) aiming at further reducing the PVR. Our case report aims not only to underline the poor prognosis of a subgroup of patients with C. perfringens sepsis but also to revive a ‘forgotten’ ventricle. 24
Myocardial depression has been shown to affect both ventricles in the setting of sepsis but there is lack of agreement with regards to incidence, prognosis and therapeutic management. A recent paper aims at providing a unifying view. 25 An early paper from 1990 showed a similar pattern of ventricular dysfunction in 39 septic shock patients using radionuclide angiographic and hemodynamic studies. 26 Left ventricle dysfunction is common (60%) and is often unmasked by concurrent norepinephrine treatment. It is usually reversible and is not associated with a worse prognosis, 27 whereas RV dysfunction appears to carry a poorer prognosis according to most papers.28–31 It may have a lower incidence (41%) and may potentially resolve with norepinephrine treatment. 32 Another paper coming from the Mayo group led by Pulido 33 did not find a difference in mortality at 30 days or 1 year between patients with any myocardial dysfunction and patients with normal echocardiography but unveiled an increased severity of illness pertaining to patients with RV dysfunction.
Although the RV lags historically behind the left ventricle, it is not only a mere bystander. 34 It shows particularities with regards to embryological development, 35 underlying molecular and cellular mechanisms describing disease states, mechanical properties and/or efficiency.
Context will create misbeliefs. Such a context was facilitated by Starr et al. 13 and later by Fontan and Baudet. 14 Hence, the RV was construed as a mere flow generator which could even get bypassed if Guyton’s mean systemic filling pressure 36 would be appropriately increased. Nevertheless, this hypothesis remains valid only as long as the PVR stays normal. Should the PVR increase, just like in our case, then the RV will soon rely on its Starling heterometric adaptation as its Anrep homeometric adaptation reserve is narrow and short lived. 37
Both the RV and the LV are characterized by serial and parallel functional linkages. A failing and dilated RV will encroach on LV function mainly through diastolic interaction whereas the converse is characterized by systolic interaction and right-sided afterload mismatch. 38
Sepsis has the potential to concurrently disrupt both aspects of RV mechanical efficiency and simultaneously optimizing RV contractility and afterload becomes mandatory, 39 whereas fluid administration should be administered cautiously as it has been shown to potentially elicit harm. Hoffman et al. 29 reported RV ischemia due to fluid-driven dilatation causing right heart subendocardial compression but in reality, ischaemia itself could account for just a minority of cases as highlighted by a study looking at 20 septic shock patients who underwent forensic autopsy. 40
The reader will also appreciate that maximizing the systemic arterial blood pressure (MAP) is paramount as it is another way of augmenting the RV myocardial blood flow with a consequent improvement in RV function.41,42 In this regard, norepinephrine, alone or combined with non-sympathomimetic pressors (vasopressin), pulmonary vasodilators (inhaled NO, prostanoids, PDE V inhibitors) and/or inodilators (PDE III inhibitors, levosimendan), 43 is capable of restoring the aortic root pressure whilst preserving LV mechanical efficiency as reflected in the preload recruitable stroke work. 44
Mechanisms and aetiology of right sided heart failure.

RV: right ventricular; LVAD: left ventricular assist device; ARDS: acute respiratory distress syndrome.
Two other interesting and interconnected features of our case are the methaemoglobin level and the intravascular haemolysis. Massive intravascular haemolysis is uncommon. Only 50 cases were reported between 1990 and 2014 according to one review. 52 It was associated with a poor outcome with described death rates of 74%–80% and a median time to death of 8–9.7 h (range 0–96 h), reflecting 7%–15% of all C. perfringens bacteremias. 53 We may only speculate that the association of RV failure, far from being a mere epiphenomenon, portends an even poorer prognosis. Regrettably, in our case, it proved fatal.
There were no known hereditary or newly acquired causes of methaemoglobinemia or/and haemolysis aside from the episode of sepsis. In accordance with a well-described physiological rationale, Ohashi et al. 54 already showed that methaemoglobin levels might actually increase in sepsis or/and septic shock patients. Shamiyeh et al. 55 described a case of clostridial sepsis with methaemoglobin levels of 5%–9% similar to ours 7%–12.2%. These numbers are truly impressive given the context of massive free haemoglobin level which is known to be an avid NO scavenger. Also, haemolysis is known to release arginase which depletes the substrate for NO synthesis. 56 In line with these data, we cannot refute the possibility of at least a regional pulmonary NO dysregulation (a relative deficiency) eliciting increased PVR. 57 Hence, in our case, haemolysis might be regarded as the putative link between the RV failure and the septic injury. Putting the severe systemic vasoplegia, the profound myocardial dysfunction and a probable intense systemic NO dysregulation (a relative excess, in contrast to the pulmonary NO status) into the same frame, methylene blue, retrospectively, might have been a useful adjuvant based on a strong physiological plausibility. Nevertheless, cautious administration would have been mandatory given the already established RV strain. 58
Conclusion
We have described a case of RV failure which, by its septic nature, should stress the importance of rapid diagnosis, urgent antimicrobial therapy and source control when appropriate. We foreground an individualized echocardiography-driven approach bearing the potential to circumvent the pitfalls associated with blind cardiovascular support. Of equal importance, this case report should reinforce the importance of a physiologically guided therapy, at times being our single most important armamentarium that we can at least use to optimize and tailor therapy.
Author’s contributions
Cosmin Balan wrote the paper. David Garry and Graham Barker revised the paper. All authors read and approved the final paper.
Footnotes
Consent
This case report is published with the written consent of the next of kin.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
