Abstract
Introduction
Severe infection is one of the most common causes of critical illness. Healthcare-associated infections complicating critical illness bring the additional challenge of multidrug resistance. However, England lacks a national surveillance system for infections in intensive care units. Prior experience with surveillance systems suggests that they are most effective when placed within a collaborative quality improvement framework.
Method
A national survey of adult, paediatric and neonatal intensive care doctors, nurses, microbiologists and infection control practitioners was undertaken throughout the UK to determine stakeholder engagement.
Results
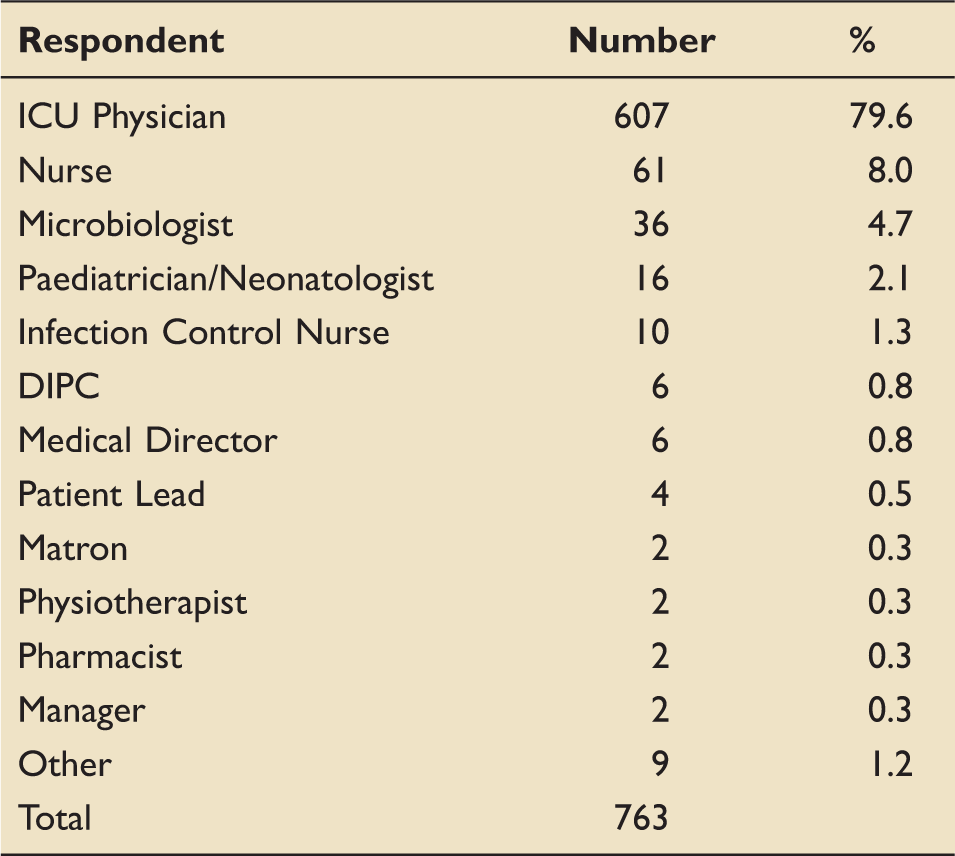
Of 763 respondents (80% ICU physicians; 8% nurses) from 158 hospital Trusts across the UK, 721 (94.4%) supported establishing a surveillance system; 63.5% preferred that data collection be mandatory; 47.5% considered that the work should be undertaken within existing resources. Respondents prioritised catheter-associated and multidrug resistant infections. Free-text responses demonstrated strong support for using the data for epidemiological information and benchmarking for quality improvement.
Discussion
The survey provides a satisfactory foundation for establishing a national surveillance system for infection prevention and control in critical care in England.
Introduction
Infections account for around 22% of all deaths worldwide, 1 6% of all deaths in the USA, 2 and 5% of all deaths in England. 3 Intensive care units (ICUs) are the focus of the most lethal forms of infection, severe sepsis and septic shock, which are responsible for 27% of admissions to UK ICUs. 4 Despite a reduction in recent years in case mix-adjusted mortality, the incidence of sepsis has increased annually (from 46 to 66 per 100,000 population between 1996 and 2003), resulting in around 14,000 deaths in the UK each year. 4 The incidence and lethality of severe sepsis is most marked at the extremes of life: Gram-negative infection in neonates has a case fatality rate of 27%, 5 and in adults mortality doubles in those over the age of 85 years. It is estimated that the cost of treating sepsis in the USA in 1995 was in the region of $16.7 billion nationally; for a comparable period in the UK the median cost of treating septic patients in intensive care was estimated to be six times that of non-septic patients.6,7 The UK's Parliamentary and Health Service Ombudsman has recently drawn attention to the importance of timely recognition and treatment of sepsis in saving patients' lives. 8 The absence of a national surveillance system for England which captures data from ICUs therefore represents an important gap in knowledge, performance feedback, and quality improvement.
Healthcare-associated infections (HAIs) add to this burden and are estimated to affect between 5 and 10% of hospitalised patients worldwide. 9 HAIs are associated with a substantially increased risk of death: for ICU patients with pneumonia the hazard ratio ranges from 1.7 to 3.5, and for blood stream infections from 2.1 to 4.0. 10 HAIs caused by multidrug-resistant organisms are an increasing problem, contributing to substantially increased length of hospital stay and costs, with the strong probability that some infections will shortly become untreatable. 11
HAI rates vary widely internationally, and between institutions in the same country, 12 suggesting that variations in local practices may contribute. Infection rates can be reduced with a sustained focus on disseminating and implementing preventative best practice. Approximately 15% of UK neonatal ICUs have been investigated for a ‘recent’ infection prevention and control issue, and 12% per year temporarily close for this reason; neonatal surgical units are particularly vulnerable. 13
In 2009 the Department of Health funded an England-wide study to reduce blood stream infections from central venous catheters blood stream infection (CVC-BSIs) in critically ill patients. The ‘Matching Michigan’ project was performed in 196 adult and 19 paediatric ICUs across England, 14 requiring them to implement technical measures to reduce infections with non-technical interventions to improve patient safety derived from a study in the USA. 15 Matching Michigan reported a reduction in CVC-BSI rates overall from 4.4 to 1.7 per 1000 CVC patient days with greater impact in adult than paediatric ICUs. However, self-reported infection control practices varied widely as did the frequency of blood culture sampling. Importantly, the novel study design showed that the reduction in infection rates was as much a feature of improvements throughout the health system as it was a specific effect of the technical and non-technical interventions located in the ICUs.
Parallel ethnographic studies in 17 of the Matching Michigan ICUs demonstrated that although infection control practices and staff focus were largely good, use of the non-technical interventions was limited, and there were concerns that the definitions of CVC-BSIs were subjective and might not fairly represent local circumstances and case mix.16,17 In units which trusted their data, performance feedback had the capacity to boost infection control efforts. However, there was wide variation within and between ICUs in the detection, diagnosis, and reporting of CVC-BSIs, a finding that has also been shown in the USA.18,19 The experience of the National Neonatal Audit Programme (England and Wales) also indicates opportunities to improve standardised reporting of CVC-BSIs. 20 Both local context and perceptions of top-down imposition of quality improvement programmes influence clinician engagement and compliance. 21
A standardised approach to detection and reporting infections in intensive care is therefore needed, with clinicians harmonising practice and organisations investing in data collection, and with the entire process having strong professional ownership and leadership. If performance measures are to have professional support, the numerator (the variable being measured) must have clinical validity, be consistently applied, and minimise opportunities for bias; and the denominator (the population being studied) must capture case mix fairly, and every effort must be made to reduce the burden of data recording placed upon clinical teams. If these conditions are satisfied, a national surveillance and feedback system would be expected to have a significant impact on infection rates.
To develop this national surveillance system, during 2011 and 2012, with the approval of the Department of Health and its standing committee on antimicrobial resistance, we formed a national collaboration of organisations representing adult, paediatric and neonatal intensive care medicine, and microbiology and infection control, under the aegis of Public Health England (PHE) (Appendix 2). We have called this collaboration the Infection in Critical Care Quality Improvement Programme (ICCQIP). ICCQIP will develop a voluntary web-based system for data capture from adult and paediatric ICU in England and will utilise existing data sources for neonatal units. Each ICU will own the data it submits, with governance of the aggregated dataset derived from PHE through the partner organisations in the Oversight Board. Although the initiative is limited to England, to maximise opportunities for learning and collaboration ICCQIP includes organisations from across the UK.
To launch the collaboration and to determine clinician engagement and priorities we conducted a survey of adult and paediatric ICUs, and report here the results of this survey as the first step informing this national surveillance and improvement system.
Methods
Members of the ICCQIP Oversight Board developed the survey questions with input from their professional organisations. It was prepared in web format using SurveyMonkey 22 and in December 2012 it was disseminated to fellows and members of the Faculty of Intensive Care Medicine, the Paediatric Intensive Care Society, the British Association of Perinatal Medicine, the UK Neonatal Collaborative, the British Infection Association, the Infection Prevention Society, and the Healthcare Infection Society. Neonatal clinical leads were also sent a letter notifying them that the survey was taking place, as they already record surveillance data for the National Neonatal Audit Programme. Although the remit for ICCQIP is England, the membership of participating organisations covers the whole of the UK. Aggregated responses and free text were analysed by frequency with thematic analysis undertaken by two physicians.
Results
Respondent roles or speciality.
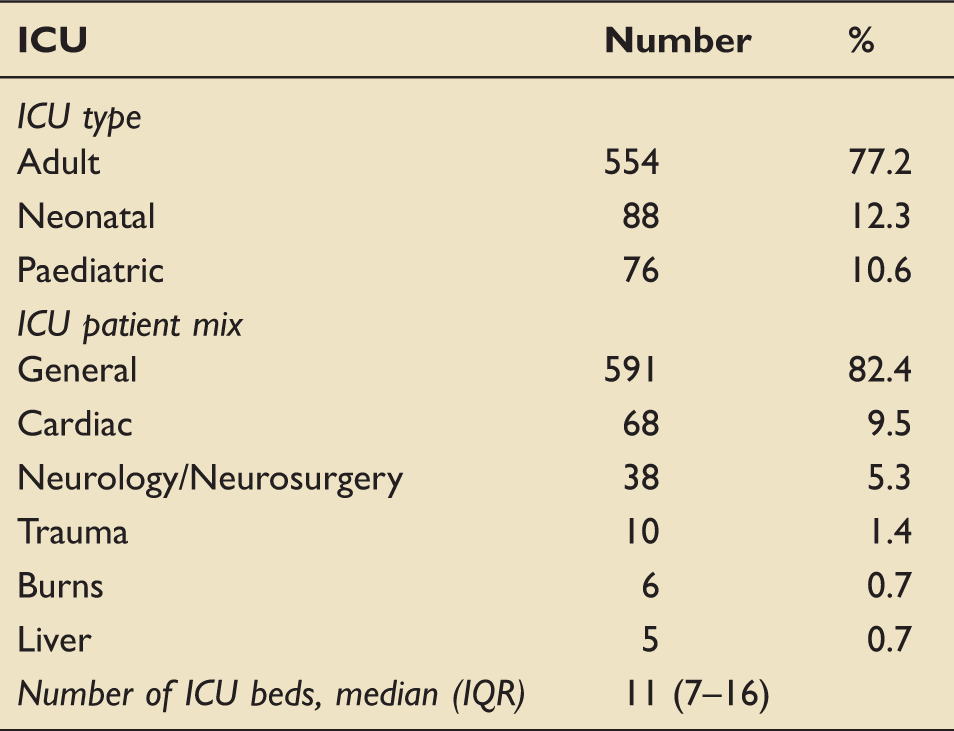
Respondents' ICU type.
Respondents' professional organisations' affiliations.
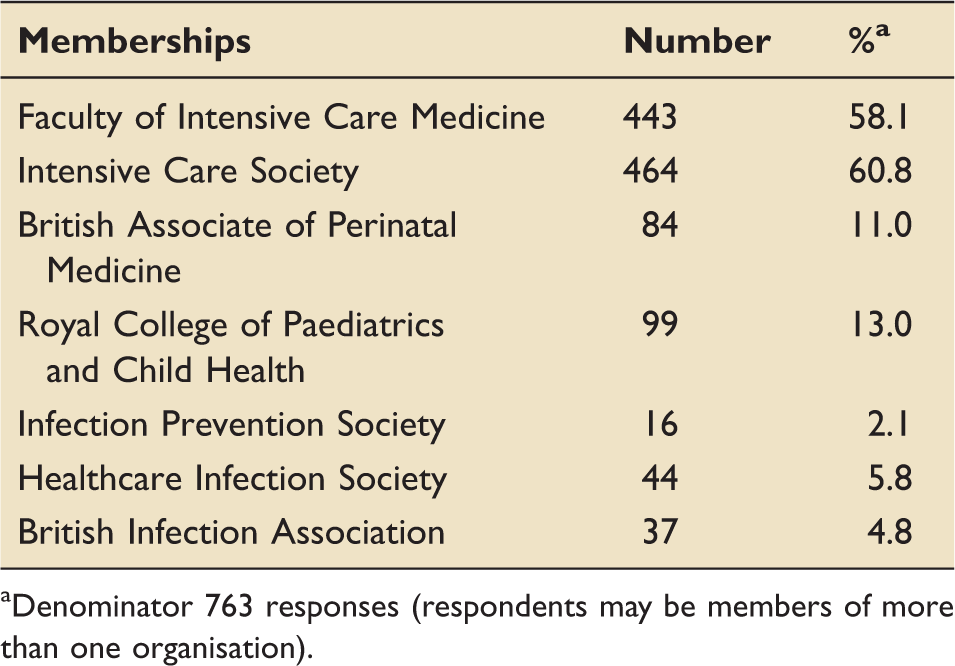
Denominator 763 responses (respondents may be members of more than one organisation).
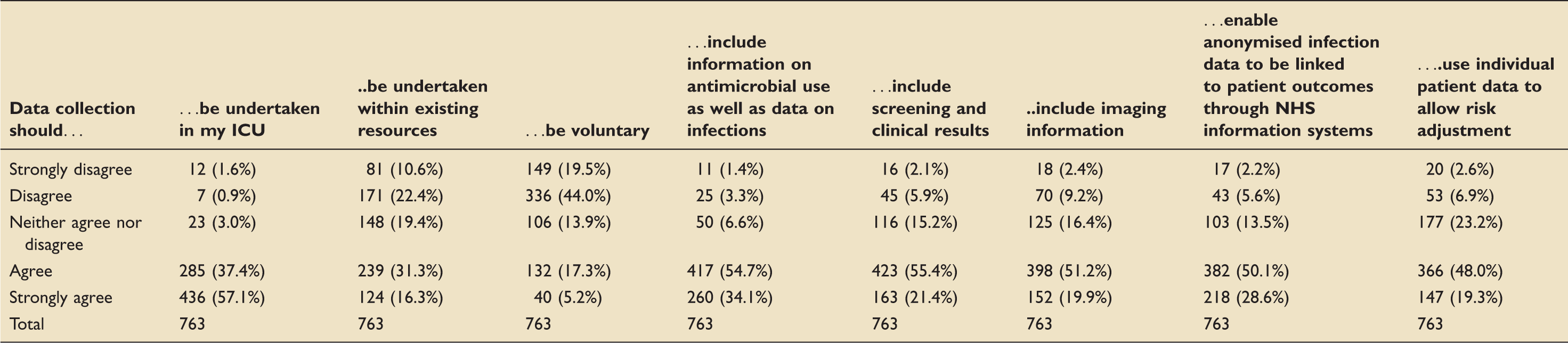
Statements on data collection (763 responses).
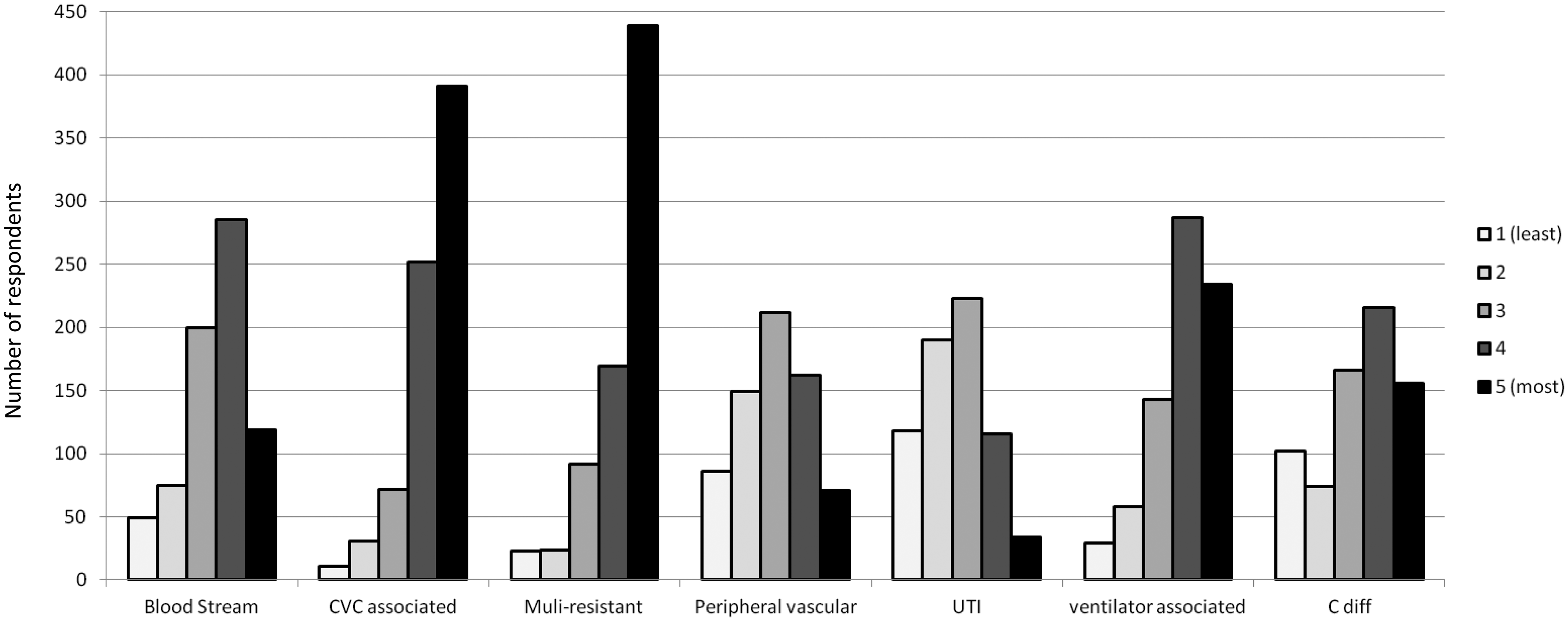
Ranking in order of importance of ICU healthcare-associated infections for priority in surveillance.
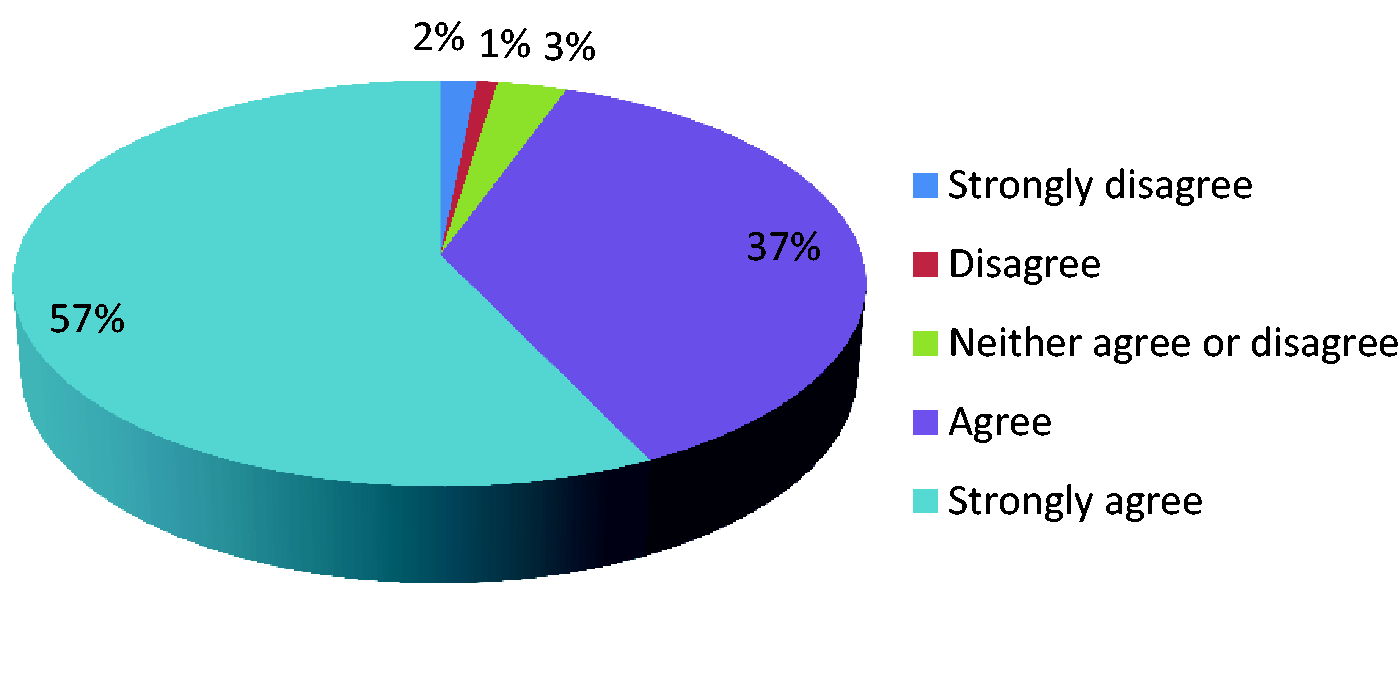
Data collection should be undertaken in my ICU.
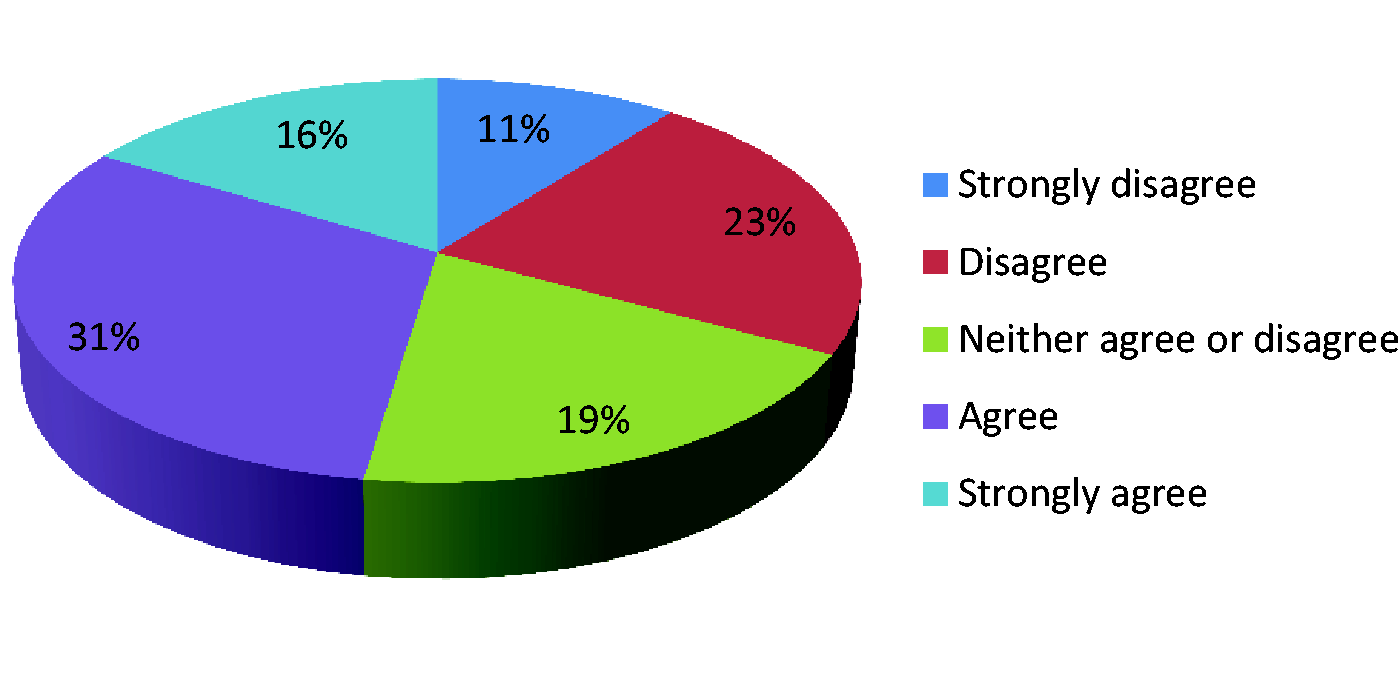
Data collection should be undertaken within existing resources.
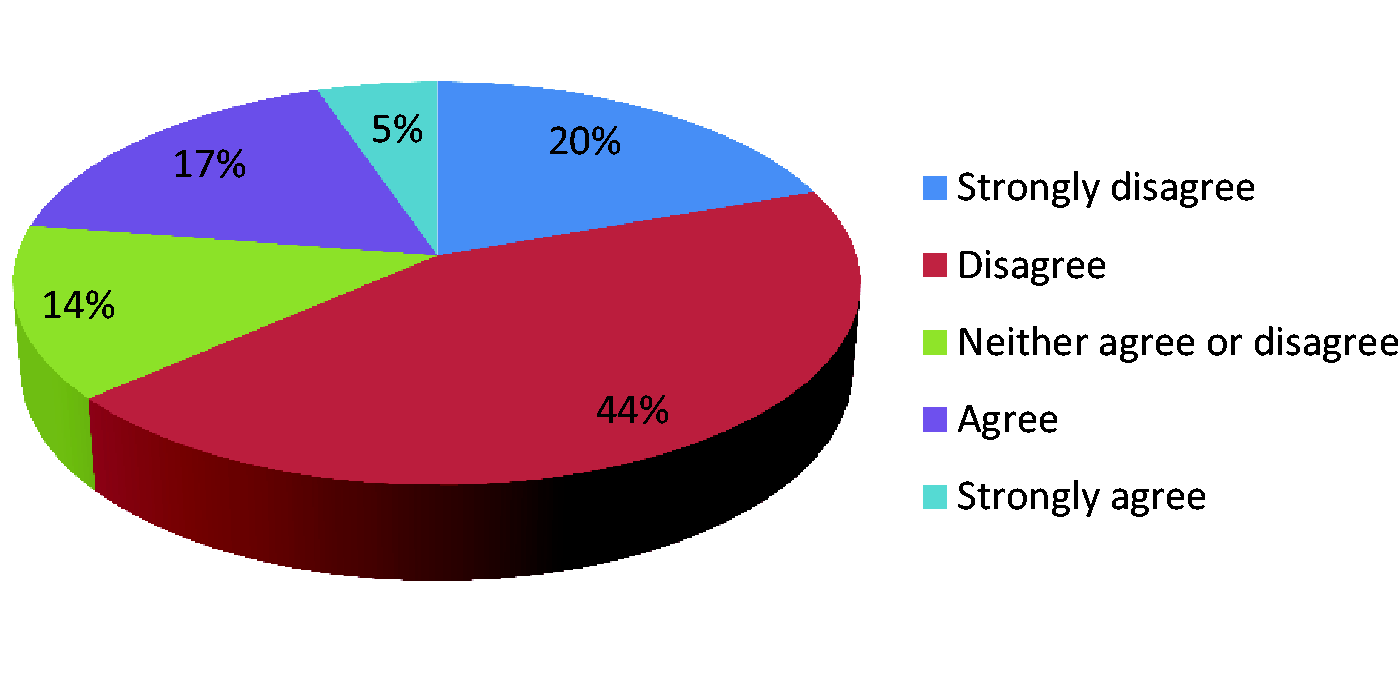
Data collection should be voluntary.
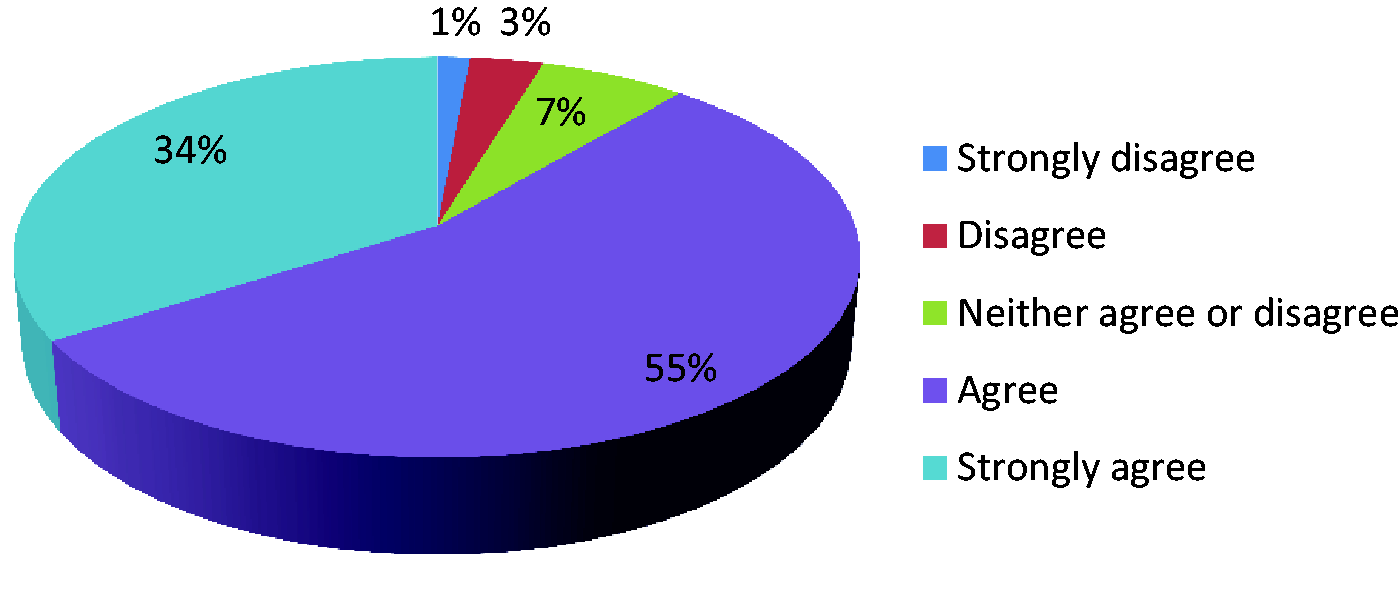
Data collection should include information on antimicrobial use as well as data on infections.
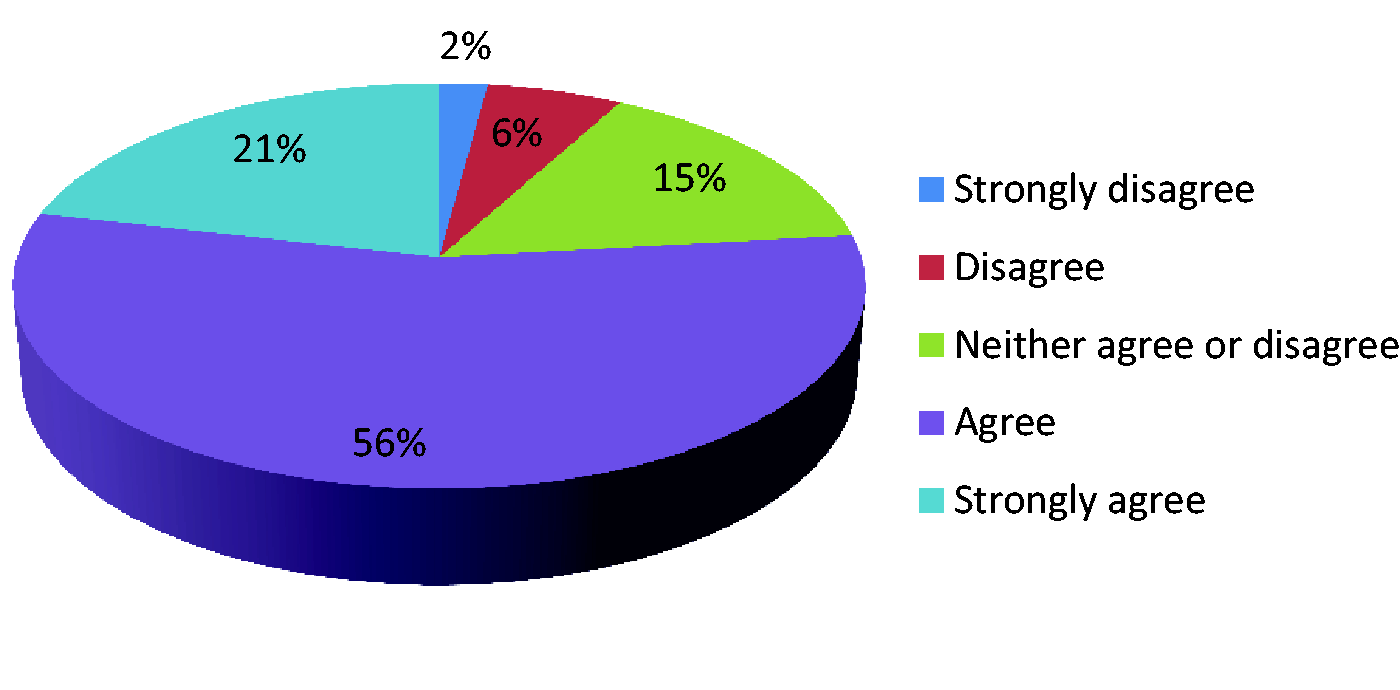
Data collection should include screening and clinical results.
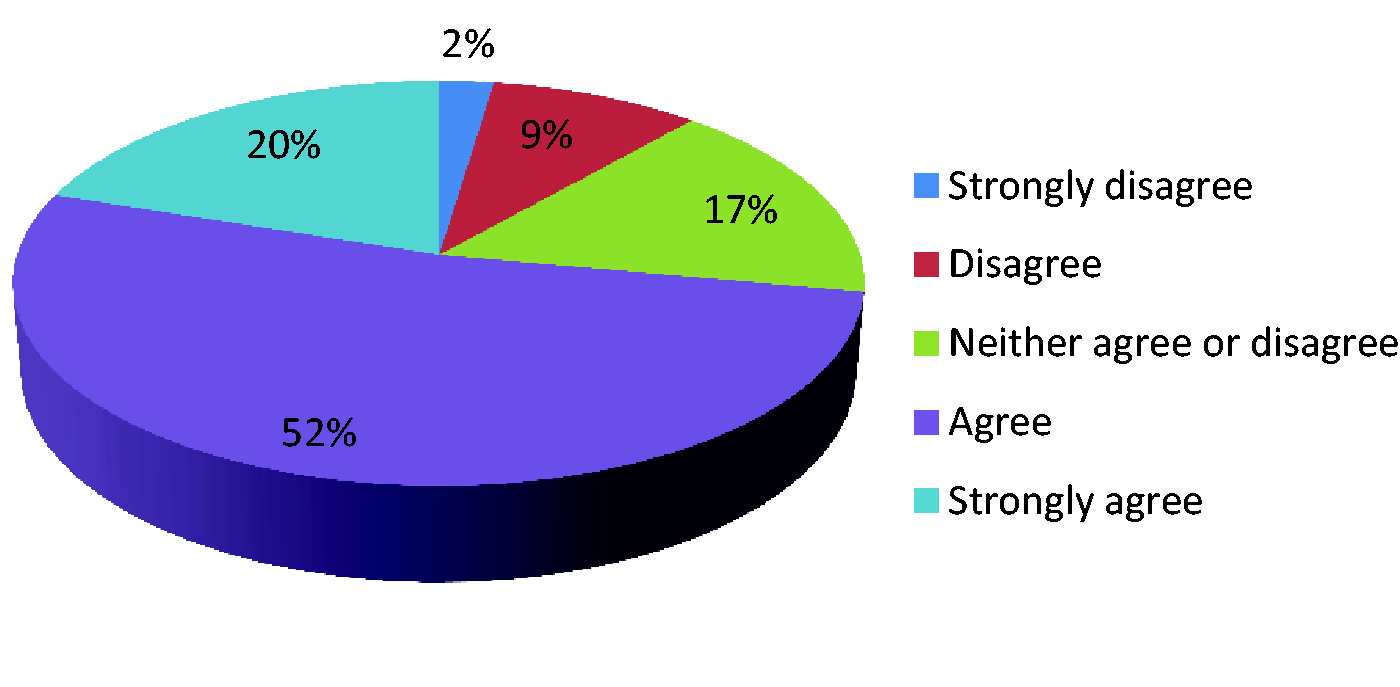
Data collection should include imaging information.
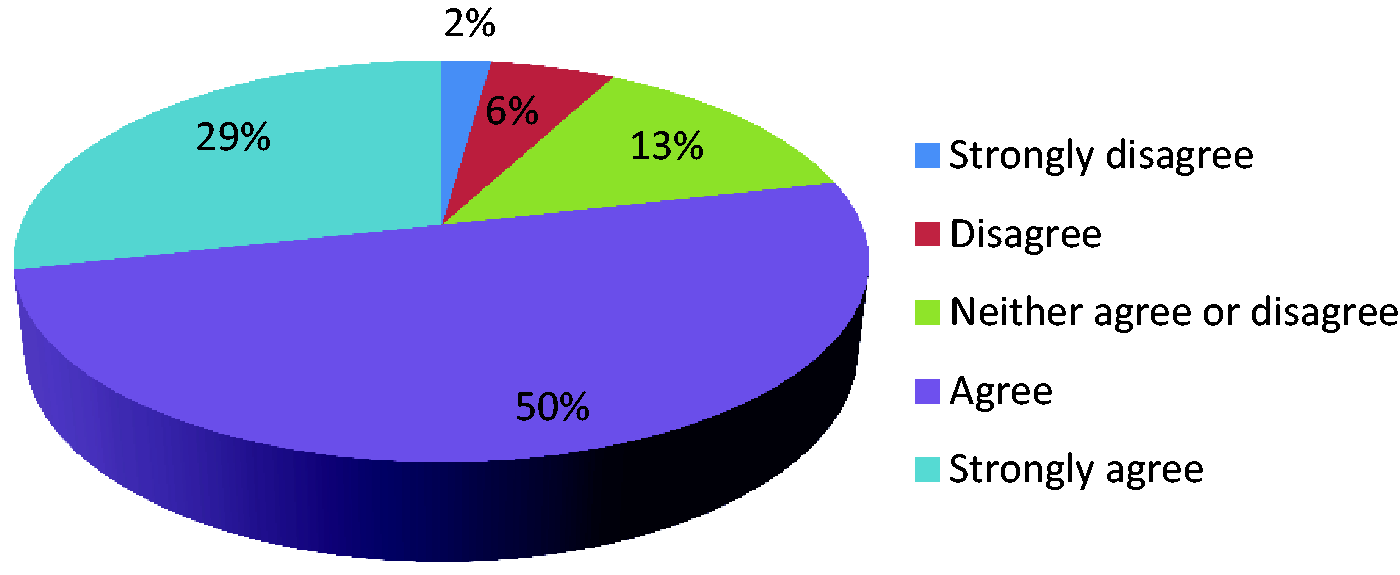
Data collection should enable anonymised infection data to be linked to patient outcomes through NHS information systems.
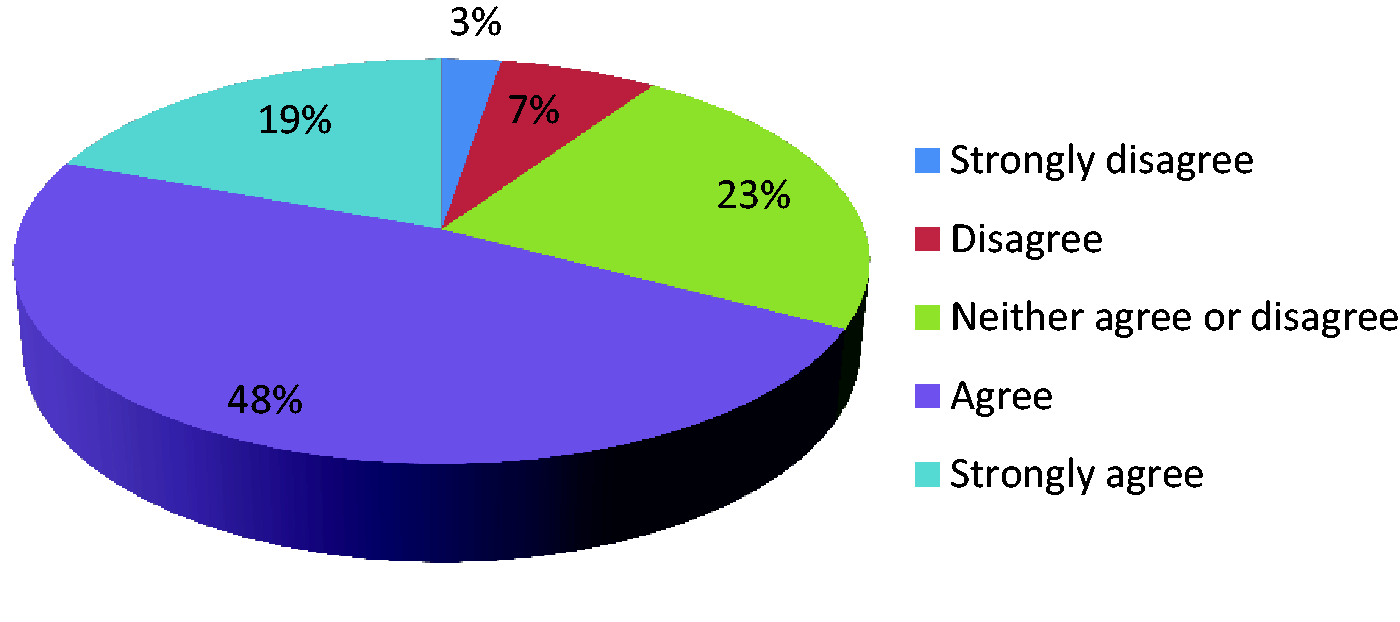
Data collection should use individual patient data to allow risk adjustment.
Ranking HCAIs by importance, CVC-associated infections, and multiresistant infections were accorded the highest priorities (Figure 1). Ventilator-associated pneumonia and blood stream infections received a lower priority.
Free-text response themes (n=869).
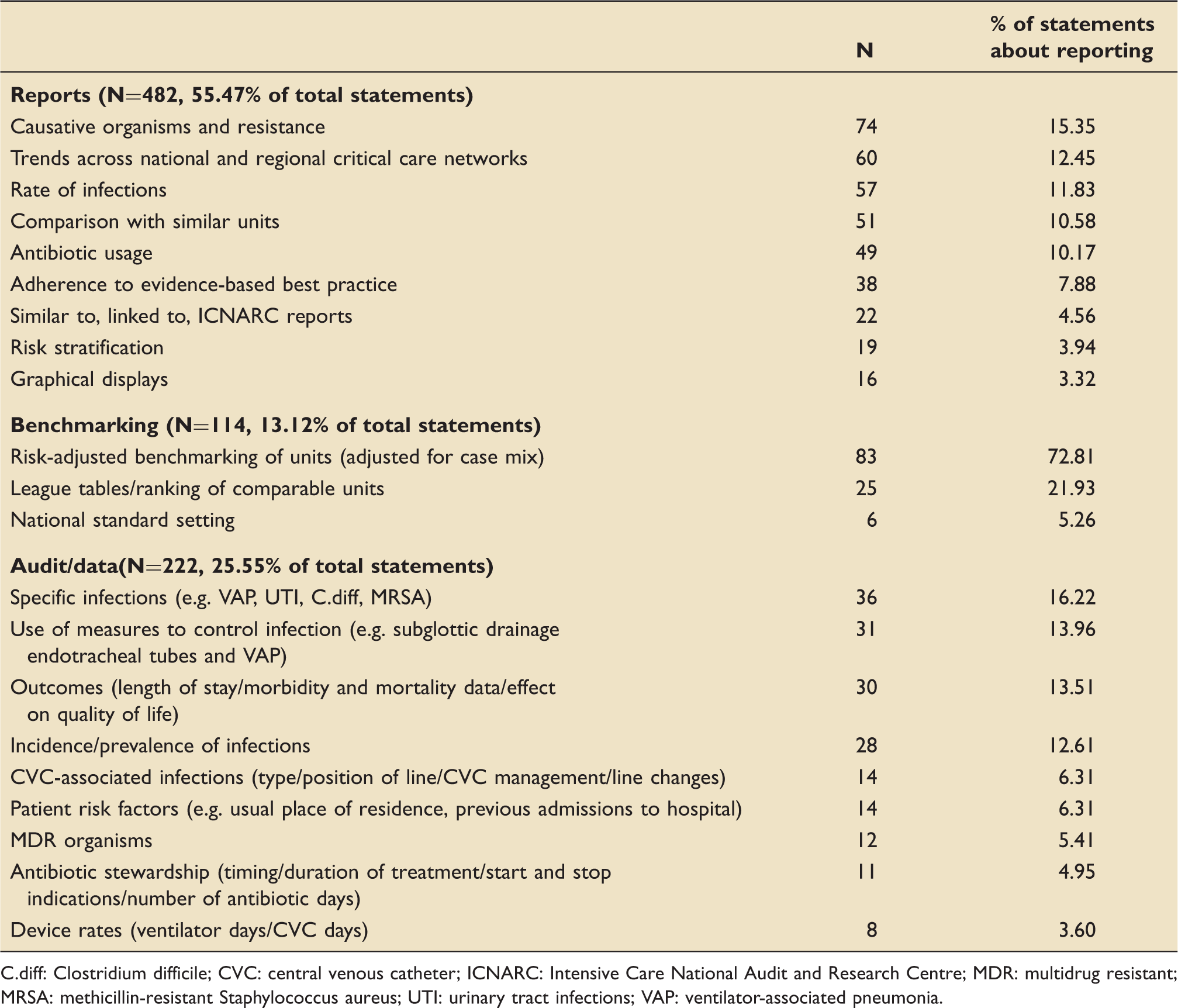
C.diff: Clostridium difficile; CVC: central venous catheter; ICNARC: Intensive Care National Audit and Research Centre; MDR: multidrug resistant; MRSA: methicillin-resistant Staphylococcus aureus; UTI: urinary tract infections; VAP: ventilator-associated pneumonia.
There were seven free-text criticisms of the proposed collaboration. These included concerns that it would duplicate work, be a bureaucratic exercise with no useful outcomes, be unachievable without electronic patient records, or that it was designed to enhance the academic status of the project team rather than those collecting the data.
Discussion
This survey has acted as a prior diagnostic phase to help determine priorities, barriers to, and facilitators of change before investing in the technology required to establish a national quality improvement programme. 23 The survey has shown substantial support amongst responding intensive care clinicians and infection control staff for a professionally owned national surveillance and reporting system for intensive care infections, focussing initially on blood stream infections. The great majority stated a desire for quite detailed information which will take time to evolve and which will certainly require the parallel development of local electronic patient records to minimise the burden of data collection as is already the case for neonatal units.
An intensive care infection surveillance system cannot stand in isolation. Many countries have national infection surveillance systems, 24 but despite the central importance of the ICU in infection epidemiology, not all include adult critical care. The European Centre for Disease Prevention and Control (ECDC) incorporates ICU infections using standardised definitions,25,26 as does the USA's National Healthcare Safety Network (previously the Centre for Disease Control and Prevention (CDC) National Nosocomial Infections Surveillance system), 27 Germany's Krankenhaus Infektions Surveillance System, 28 the French National Program for Prevention of Healthcare-Associated Infections and Antimicrobial Resistance, 29 and Australia's state of Victoria Hospital Acquired Infection Surveillance System. 30
In the UK, only Scotland specifically includes data from ICUs through collaboration between Health Protection Scotland (HPS) and the Scottish Intensive Care Society Audit Group. Surgical site infection (SSI) surveillance in Scotland started in 2002 following the publication of ‘A Framework for National Surveillance of Hospital Acquired Infection in Scotland’ by the Scottish Government. 31 This required NHS Boards to implement SSI surveillance, using standardised definitions which have been adopted from the CDC in the USA, permitting international comparisons. HPS also contributes selective SSI data to the ECDC for inclusion in European reports.
In England, PHE – formerly the Health Protection Agency – has responsibility for collecting data on a wide range of infections in primary and secondary care and has the capacity to link existing data with new surveillance systems such as ICCQIP. The national case mix programmes for intensive care in England have varying capacity to capture infection data:
For adult intensive care the Intensive Care National Audit and Research Centre (ICNARC) manages case mix information for 220/240 (95%) of adult ICUs in England. The dataset permits collection of blood stream infections only, excluding Staph epidermidis. The paediatric intensive care case mix programme in England is PICAnet, covering all 30 PICUs. This does not currently include any infection data. In neonatal practice, all 173 neonatal units in the UK contribute data to the National Neonatal Research Database (NNRD). Data are recorded onto a real-time patient management system (primarily the Badger.net system). A pre-specified data extract, the variables comprising the NHS Information Standards Board approved national Neonatal Dataset (ISB1595) is transmitted to the Neonatal Data Analysis Unit at Imperial College London and held in the NNRD on a secure NHS server. The electronic patient management system permits the recording of daily data including an ‘ad hoc’ field completed when any cultures are drawn from a sterile site >72 h of age. The NNRD is utilised for analyses for the National Neonatal Audit Programme. In addition, 26 neonatal units contribute to a separate stand-alone system ‘NEONin’, which has provided valuable data to inform antibiotic choice.
5
Currently, neonatal units are also required to enter catheter-associated infection data into a national neonatal dashboard. However, this is difficult to use, completeness is poor, and the approach has been criticised for requiring repetitive entry of data that are already available in the NNRD.
A key question is whether feedback of data through surveillance systems will in fact improve patient outcomes. French and German experience suggests that this is the case.27,32 To do so the data must be contemporaneous, reliable, representative of the population from which it was drawn, credible to clinicians, comparable across different centres, and be derived from activities which are susceptible to modification of care processes. This is particularly important if the data are to be used for performance management including commissioning of services.
Sustaining improvements in performance is challenging once the initial interest and novelty have worn off. 33 Participation and outcomes are highly context dependent.16,17,34 Mandating participation has the desired effect on data returns, 35 but accurate data still requires active engagement by clinicians, particularly senior doctors; it must be seen as a professional obligation to our patients. 36 This is particularly important when datasets are used for benchmarking, since although there is general support amongst clinicians for the public presentation of performance data it is important that these data have the confidence of the clinicians and are accompanied by adequate explanatory variables including case-mix adjustment to avoid misclassification and misinterpretation.37,38
The ICCQIP web-based data entry system has been designed to take into account the responses we have received from the survey and is currently being piloted. In later phases, we intend to link ICCQIP to adult (ICNARC' case mix programme), paediatric (PICAnet), and neonatal databases to adjust results for severity of illness and to associate infection data with patient diagnoses and outcomes.5,39–41
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Appendix 1. Authorship and Writing Committee
Appendix 2. ICCQIP Participating Organisations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
