Abstract
Thyroid storm is a rare and serious complication of pregnancy which can lead to spontaneous abortion, preterm delivery, preeclampsia and cardiac failure. It is also associated with high maternal and foetal mortality if not diagnosed and managed promptly. The diagnosis of thyroid storm in pregnancy can pose significant challenges due to its presentation being similar to other pregnancy-related complications. We present a patient who developed thyroid storm at 29 weeks of pregnancy, which resulted in pre-term delivery, cardiac failure and thyroidectomy. We discuss the treatment of thyroid storm in pregnancy, the decision making involved in proceeding to thyroidectomy or to use radio-iodine, and foetal thyroid status in thyrotoxicosis.
Case report
A 29-year-old pregnant woman presented with symptoms of lower back pain and vaginal bleeding. She was 29 weeks pregnant (gravida 2, para 2) with no previous antenatal history. Her past medical history included iron-deficiency anaemia, hypertension and a familial history of type II diabetes mellitus. On examination, the patient’s vital signs were as follows: temperature 37.4℃; heart rate 140 beats/min; blood pressure 140/80 mmHg; respiratory rate 40 breaths/min; SpO2 94% while breathing air. Vaginal examination revealed blood-stained mucus in the vagina and a dilated cervix of 2 cm. Her initial chest radiograph (CXR) showed some opacification at both the left and right base (Figure 1); however, in view of her high body mass index (>30), and sudden increase in her tachypnoea, a computed tomography pulmonary angiogram (CTPA) was arranged to investigate for potential pulmonary emboli. While the CTPA was being arranged, her cardiotocography (CTG) monitoring became ‘non-reassuring’ and emergency caesarean section was undertaken. This took place under general anaesthesia given her respiratory distress.
Initial chest radiograph on admission to hospital.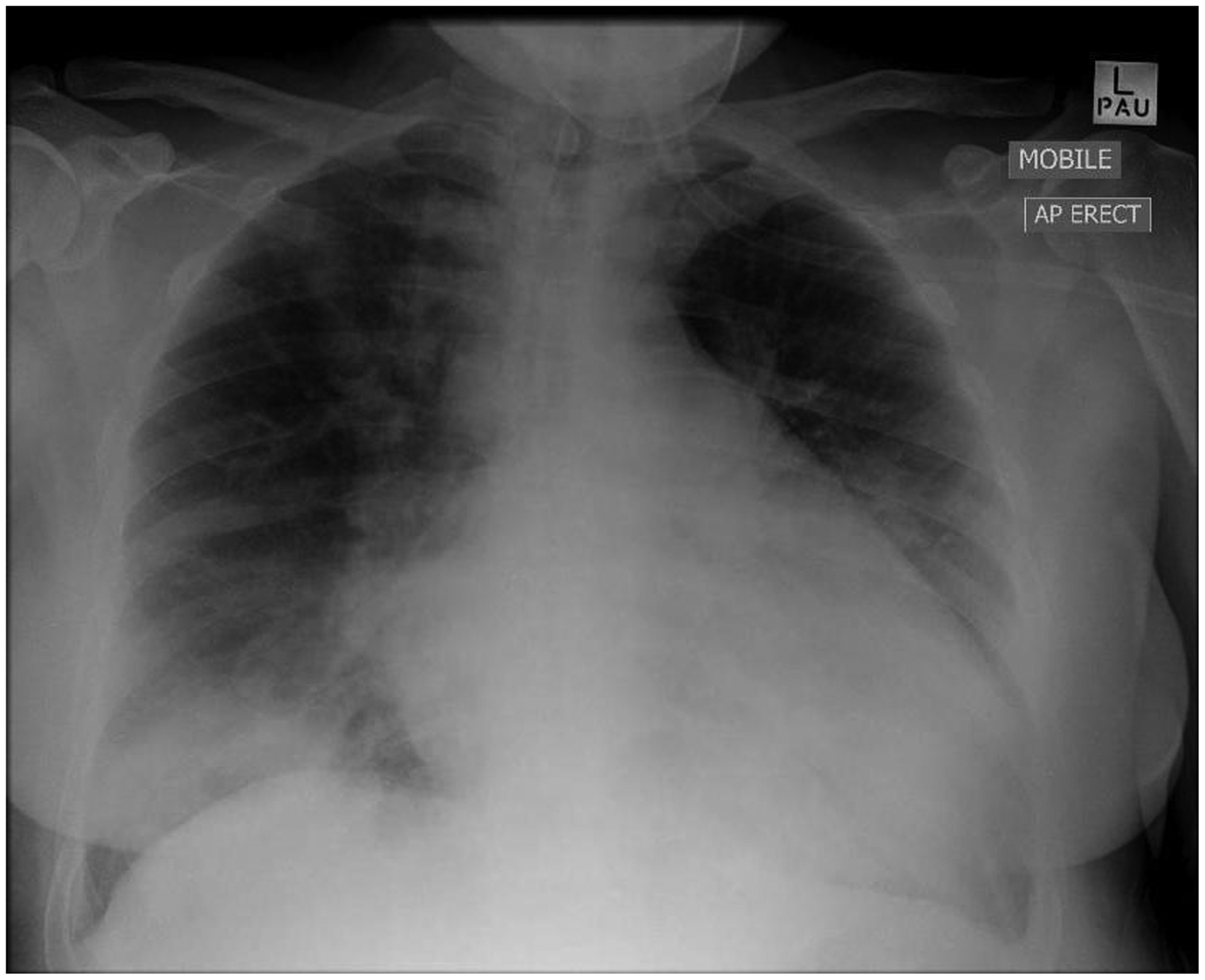
On induction of anaesthesia with sodium thiopental and suxamethonium, she developed hypertension of 220/120 mmHg and atrial fibrillation (AF) with a rapid ventricular response of 180 beats/min. This was treated by increasing the depth of anaesthesia, intravenous amiodarone 300 mg and magnesium sulphate 20 mmol, following which she reverted to sinus rhythm at a rate of 130 beats/min. She required an FiO2 of 0.55 and Positive end-expiratory pressure (PEEP) 10 cm H2O to maintain adequate oxygenation, and her compliance appeared reasonable with manual ventilation not being difficult. She was found to have a placental abruption, though the operative blood loss was estimated at 1.5 L which was less than expected. Thick meconium was noted at the time of delivery. She received transfusion of six units of packed red blood cells, four units of fresh-frozen plasma, and two units of cryoprecipitate guided by her estimated blood loss and the pre-operative full blood count and coagulation studies: Hb 100 g/L; white cell count (WCC) 28.7 × 109/L; platelets 165 × 109/L; prothrombin time (PT) 20 s; activated partial thromboplastin time (APTT) 53 s;Fibrinogen 2.5 g/L.
An intraoperative transoesophageal echocardiogram (TOE) was performed specifically to assess right ventricle (RV) systolic function, left ventricle (LV) systolic function, visibility of clots in the pulmonary arteries and superior vena cava distensibility to assess resuscitation and fluid status. The TOE revealed a mildly dilated and mildly hypokinetic LV, a normal size and contractile RV and moderate mitral regurgitation. The appearance, pulse, grimace, activity, respiration (APGAR) score of the baby was 9 post-delivery.
On arrival to intensive care unit (ICU), she had a temperature of 37.9℃, heart rate of 140 beats/min and blood pressure of 145/105 mmHg; she was intubated and ventilated with an SpO2 94% (FiO2 0.75). She was initially ventilated with synchronised intermittent mandatory ventilation (SIMV), but her compliance began to worsen requiring lung protective strategies (low tidal volume/high frequency); her oxygenation also worsened requiring increasing levels of positive end-expiratory pressure (PEEP) and FiO2. She developed atrial flutter with no haemodynamic compromise requiring direct current (DC) cardioversion of 50 J which successfully restored sinus rhythm. Over the next 24 h, her oxygenation worsened further requiring FiO2 of 1.0 and PEEP of 15 cm H2O, and her CXR developed bilateral infiltrates along with a fever of 38.7℃ (Figure 2). It was felt she was either developing acute respiratory distress syndrome (ARDS) due to sepsis or transfusion related acute lung injury (TRALI) from the intraoperative blood products. This was managed with continuing lung protective ventilation, diuresis aiming for a negative fluid balance, and a broad spectrum antibiotic regimen of ceftriaxone, gentamicin, and metronidazole.
The patient’s chest radiograph on her first day in ICU with worsening of her respiratory status.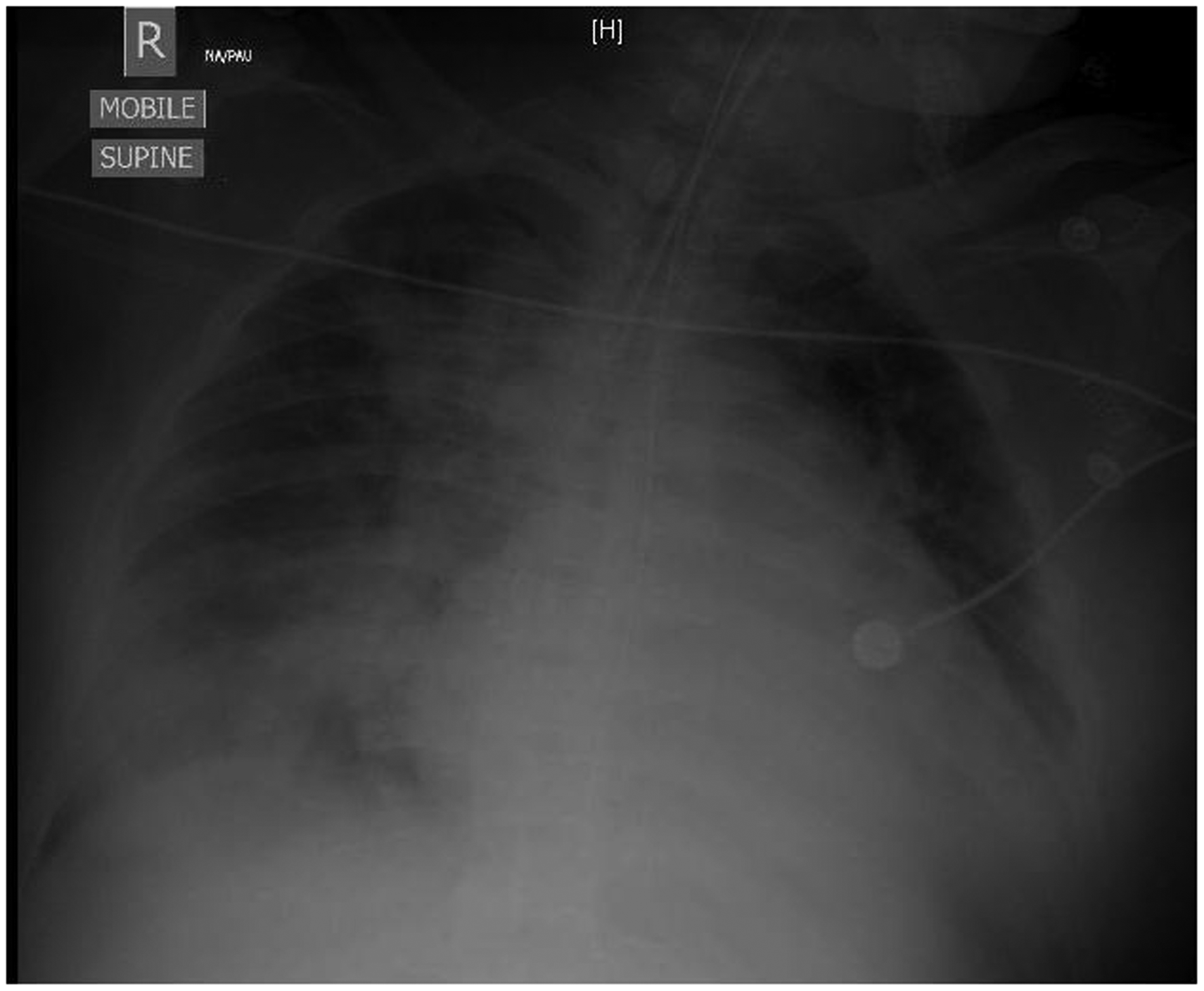
It was felt that she was too unstable to be transferred for a CTPA, and since her clinical picture appeared more consistent with sepsis, the CTPA was deferred. In the event of her having suffered a pulmonary embolus (PE), it was considered that anticoagulation was not an option at this moment in time, and therefore, consideration of an inferior vena cava filter (IVC) filter would be required. However, venous Doppler scans of the upper and lower limbs were negative which furthermore reinforced the plan to continue her current management. At this time, she was still mildly coagulopathic and required ongoing fresh-frozen plasma. Other investigations showed a normal urine protein to creatinine ratio, and a mild troponin rise which was consistent with arrhythmias and cardioversion. In the setting of no electrocardiogram (ECG) changes and no regional wall motion abnormalities on TOE, the troponin rise was not investigated further. She also developed an acute kidney injury (creatinine 163 µmol/L), and acute hepatic impairment (AST-aspartate transaminase 790 U/L, ALT-alanine transaminase 253 U/L) which was felt at the time to be related to presumed sepsis, and therefore, current management was continued.
Invasive haemodynamic monitoring was established with PiCCO, which demonstrated a cardiac index of 3.65 L/min/m2, pulse pressure variation of 13% and intrathoracic blood volume index of 980 mL/min/m2. Transthoracic echocardiography (TTE) once again confirmed reasonable LV and RV systolic function. It was therefore felt that fluid status was adequate, and in the absence of catecholamine requirements, on-going diuresis was appropriate.
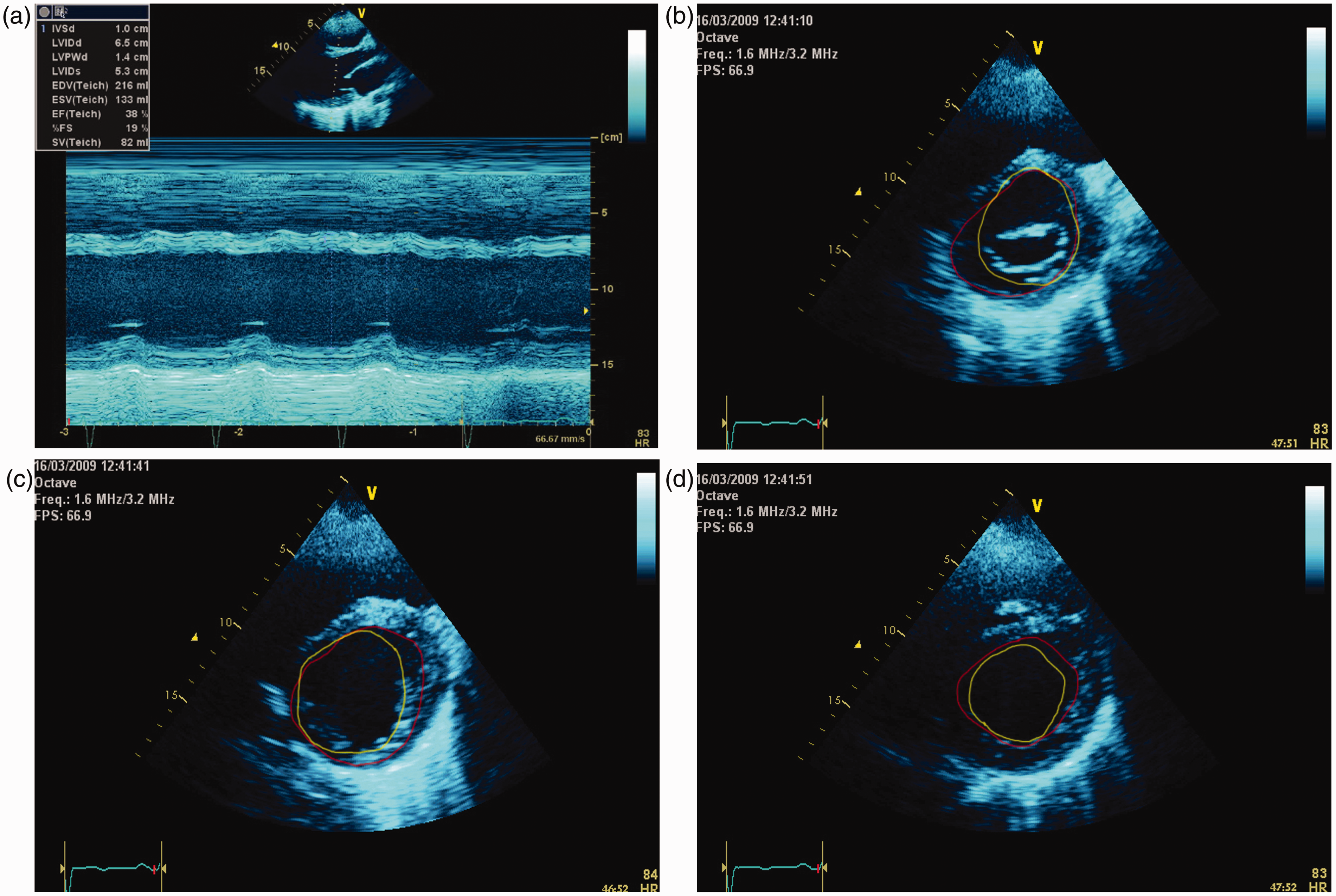
On day 2 in ICU, she remained tachycardic with a rate of 135 beats/min but became increasingly febrile with a temperature of 40.7℃ despite her WCC falling to 19 × 109/L, although her procalcitonin was raised at 68 µg/L. Her oxygenation and compliance improved and her ventilation was weaned to an FiO2 of 0.6, and PEEP 10 cmH2O. Her CXR also started to clear (Figure 3) having achieved a negative fluid balance (−1500 ml) through i.v. frusemide therapy (10 mg 6 hourly). However, she also had multiple episodes of AF with a rapid ventricular response between 160 and 175 beats/min requiring a total of four DC cardioversions. The final DC cardioversion was successful, and she was commenced on an amiodarone infusion. During this period, her cardiac index as measured by PiCCO was 3.8–4.3 L/min/m2; however, her TTE now showed global left and right ventricular hypokinesia, with akinesia of the anterolateral wall and anterior septum in the mid-zones and apical section of the LV (Figure 4(a)–(d)). Her renal function continued to worsen which seemed out of keeping with her good diuresis and the lack of need for catacholamine support. Towards the end of day 2, she suddenly spiked a fever to 41℃, which coincided with worsening AF (rapid ventricular response of 190 beats/min) and hypotension with a mean arterial pressure of 45 mmHg. She was managed with DC cardioversion which was unsuccessful, a noradrenaline infusion, on-going amiodarone infusion and active cooling measures.
The patient’s chest radiograph from her second day in ICU; the radiograph corresponds with resolution of respiratory but worsening of cardiac symptoms. Inner border of the LV during systole (green line) and diastole (red line). What is evident from the area tracings is that there are regional wall motion abnormalities. The basal sections of the LV are akinetic, except the inferior segment that is hypokinetic. In the mid-sections, the LV is akinetic except for the lateral section which is hypokinetic. The apical sections are akinetic. Measurements of the change in area at the mid-zone and apex show a difference of 22%, which is significantly less than the calculated EF of 38% using only the basal sections in M-Mode. (a) M-Mode through the basal sections of the left ventricle. (b) Parasternal short axis of the LV at the level of the mitral valve. (c) Parasternal short axis of the LV at the level of the mid-section. (d) Parasternal short axis of the LV at the level of the apex.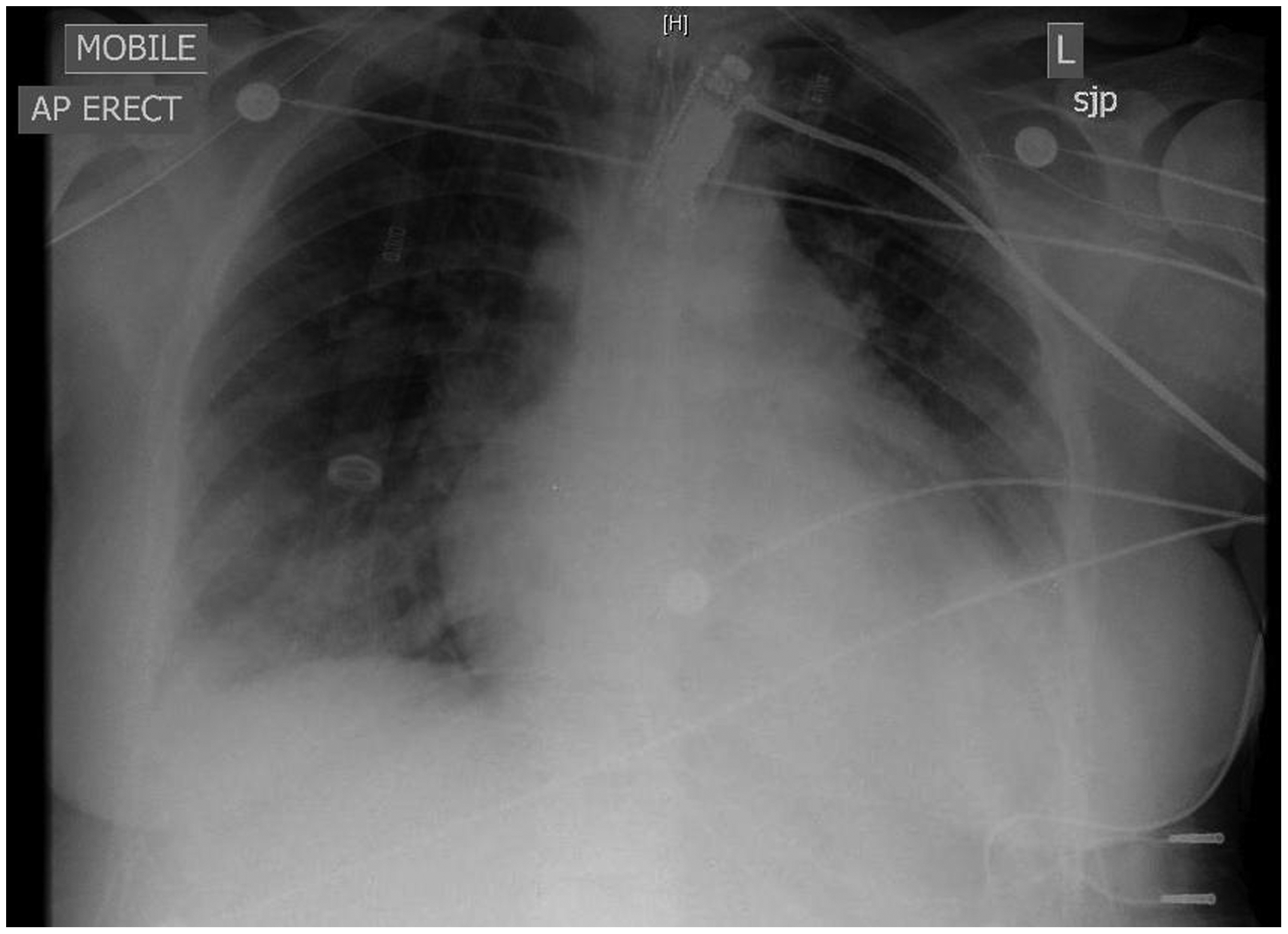
Due to the unusual presentation, fevers, ongoing tachycardia and arrhythmias, her thyroid status was investigated. This revealed thyroid-stimulating hormone (TSH) levels of 0 U/mL, free T4 of 74.3 pmol/L, and free T3 of 12.1 nmol/L. A diagnosis of thyroid storm was made, and she was treated with propranolol, Lugols Iodine, propylthiouracil and dexamethasone.
Renal function from day 1 to day 8, showing the rise in urea and creatinine associated with her worsening clinical condition.

The table also shows a faster return towards baseline of the creatinine rather than the urea due to her ongoing steroid therapy as part of her anti-thyroid regimen. ICU: intensive care unit.
Trend in thyroid function tests after commencement of carbimazole and Lugol’s solution.
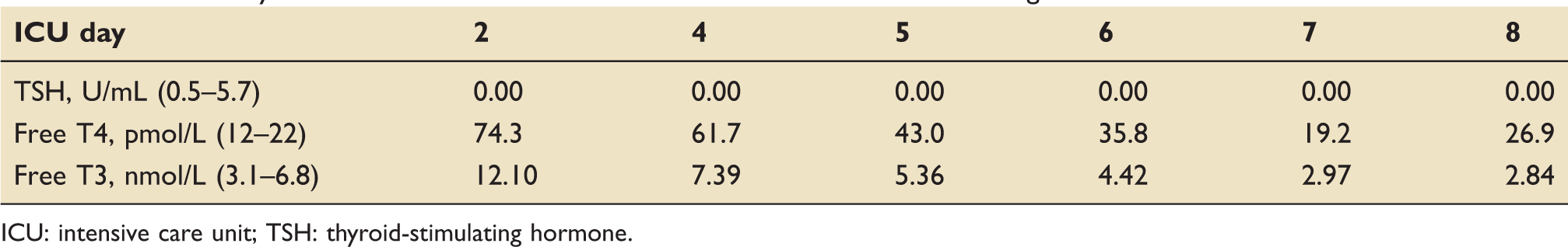
ICU: intensive care unit; TSH: thyroid-stimulating hormone.
Discussion
Hyperthyroidism occurs in about 0.1–0.4% of pregnant females and approximately 85% of cases are due to Graves’ disease. 1 Transient hyperthyroidism due to inappropriate secretion or action of human chorionic gonadotropin hormone (hCG) and hyperemesis gravidarum are the other common cause. 2 Thyrotoxicosis in pregnancy is associated with both maternal and foetal complications. Maternal complications include pre-eclampsia, congestive heart failure, placental abruption and caesarean delivery. 3 Foetal risks of maternal thyrotoxicosis include spontaneous abortion, premature labour and low birth weight, stillbirth and congenital abnormalities. 3
Cardiac dysfunction in thyrotoxicosis
In patients with uncontrolled thyrotoxicosis, cardiac dysfunction is frequent and may be precipitated by coexistent complications of pregnancy. 4 Thyrotoxicosis is usually associated with high output heart failure; however, low output heart failure can also occur.
The pathophysiology of low output heart failure is unclear but may include chronic tachycardia-induced cardiomyopathy (CTIC). 5 Dilated thyrotoxic cardiomyopathy (DTC) is less common with an incidence of 1%. The exact mechanism for DTC is unknown, but a hypothesis is that prolonged ventricular hypertrophy can transform into dilation which may develop in prolonged untreated hyperthyroidism. Many studies suggest that DTC is reversible; however, a recent longitudinal six-month follow-up study has shown that there is high incidence of irreversibility. Reversibility status was associated with time since diagnosis of thyrotoxicosis, with complete reversibility being associated with a disease diagnosis of less than 13 months. 6 The patient we report was undiagnosed, although the family reported that her symptoms may have commenced more than a year previously. Although her initial TOE showed mild left ventricular dilatation, she did not have left ventricular hypertrophy.
As our understanding of cardiac dysfunction in the critically ill improves, it is becoming more evident that terms such as CTIC, DTC and even takotsubo cardiomyopathy are all variants of stress-induced cardiomyopathy (SIC). The numerous acronyms and syndromes manifest since the clinical findings happen to occur in different forms of critical illness with different echocardiographic findings. 7 Our patient demonstrated an echocardiographic pattern of dysfunction more commonly seen with regional ischaemia but has also been described as part of the spectrum of SIC. 8 It can also be seen that the M-Mode 2D section through the basal sections of the LV significantly underestimated the extent of her myocardial dysfunction (Figure 4(a)–(d)) which remained abnormal for the duration of hospital stay and subsequent discharge.
Diagnosis and management of thyrotoxicosis in pregnancy
Diagnostic criteria for a thyroid storm.
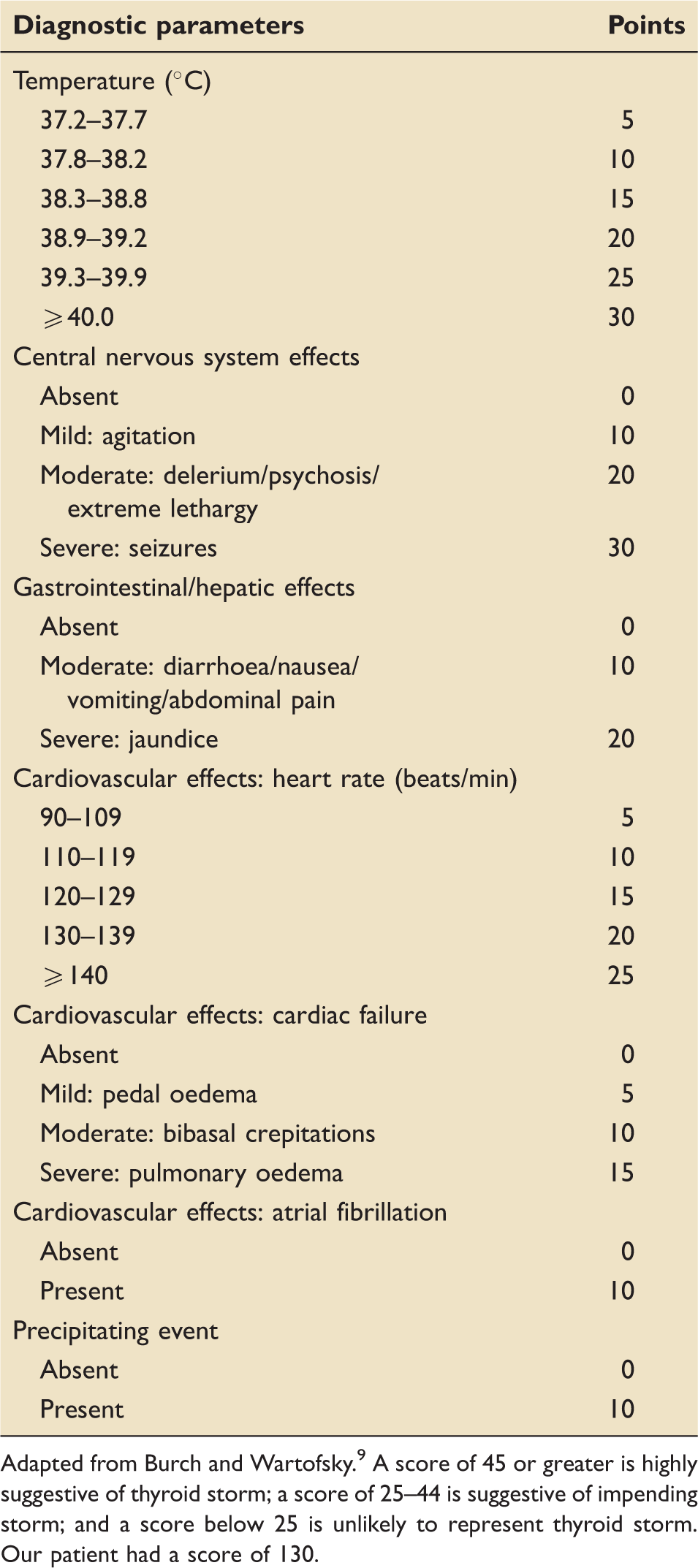
Adapted from Burch and Wartofsky. 9 A score of 45 or greater is highly suggestive of thyroid storm; a score of 25–44 is suggestive of impending storm; and a score below 25 is unlikely to represent thyroid storm. Our patient had a score of 130.
Management of thyroid storm in pregnancy remains standard with anti-thyroid drugs such as carbimazole and propylthiouracil, iodides in the form of Lugol’s iodine or saturated solution of potassium iodide, beta-blockade, steroids to prevent peripheral T4 to T3 conversion, and antipyretics and intravenous fluids to prevent dehydration. 10 The use of amiodarone as treatment for AF is controversial since the effect of amiodarone on the thyroid gland ranges from abnormal thyroid function test findings to overt thyroid dysfunction, which may be either amiodarone-induced thyrotoxicosis or amiodarone-induced hypothyroidism. 11 However, it is generally accepted in the acute situation that maintaining optimal haemodynamic status is more important, and beta-blockade should be avoided. 12
Iodine therapy is complementary to the anti-thyroid drugs and reduces the release of pre-stored hormone and transport and oxidation of iodide in the follicular cells. This decrease in organification due to large doses of inorganic iodide such as Lugol’s iodine is termed the ‘Wolff-Chaikoff’ effect. 13 The thyroid gland escapes this inhibition in approximately 48 h, and most patients return to hyperthyroidism within two to three weeks if no other treatment is given. In the acute setting if the Lugol’s iodine is given before the anti-thyroid drugs, new hormone synthesis can be stimulated (the ‘Jod-Basedow’ effect 14 ); hence, Lugol’s iodine should be given 30–60 min following the dose of anti-thyroid drugs. Lugol’s iodine saturates the thyroid gland with iodine and hence definitive treatment with radioiodine ablation cannot be carried out till the iodine load is adequately cleared. 13 However, definitive therapy in the form of thyroidectomy can be carried out within a period of two weeks before there is escape from the ‘Wolff-Chaikoff’ effect. For these reasons, our patient underwent thyroidectomy on the 10th day of ICU admission.
Definitive treatment: Thyroidectomy vs. radioiodine?
The decision of whether to use radioiodine versus surgery for definitive treatment of thyrotoxicosis is controversial. Radioiodine is safe, effective and cheap and is suitable for the elderly and patients with heart disease. It is the preferred treatment option in the United States, whereas in Europe there has been greater preference for anti-thyroid medications and/or surgery. 13 Contraindications to radioiodine include pregnancy, lactation, coexisting thyroid cancer or suspicion of thyroid cancer, females planning for pregnancy in four to six weeks and patients’ inability to comply with radiation safety guidelines. 13 The indications for surgery include situations where radioiodine is contraindicated along with patient preference, inadequate uptake of radioactive iodine, age younger than five years and patients where there is a need for immediate control of the disease. 15
Foetal thyroid status
The foetal thyroid gland starts secreting T4 by 10–12 weeks and starts responding to the TSH secreted by the foetal pituitary gland by 20 weeks. However, the overall function of the thyroid gland remains low. TRAB in pregnant females with Graves’ disease can cross the placenta and affect the foetal thyroid gland inducing foetal thyrotoxicosis. However, aggressive therapy with anti-thyroid drugs during pregnancy can induce a state of foetal hypothyroidism and related complications including neurocognitive developmental deficits. Hence, routine blood screening and appropriate management of the newborn baby is essential. The baby born to our patient had hyperthyroidism and was managed accordingly.
Conclusion
Thyroid storm is rare in pregnancy but can cause significant maternal and foetal mortality. It is often a difficult diagnosis due its unusual presentation and similarities to other pregnancy related disorders; hence, prompt diagnosis is essential for early treatment and improving outcomes. It is associated with cardiac dysfunction that may not resolve in tandem with the clinical thyroid status. Definitive treatment for thyroid storm involves stabilisation with anti-thyroid medications and iodine; however, the decision for thyroidectomy versus radioactive iodine as definitive therapy is still debated.
Footnotes
Consent
Written consent for publication of this report was obtained from the patient.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
