Abstract
An extensive caval thrombus was incidentally detected in a neurosurgical patient by clinician-delivered critical care echocardiography. Recent intracranial haemorrhage prevented therapeutic anticoagulation; the very proximal nature of the thrombus precluded standard deployment of an inferior vena cava filter. We describe the novel radiological technique employed to manage the thrombus, and examine whether a thermoregulatory central venous catheter inserted as part of standard neuro-critical care may have contributed to the risk and extent of the caval thrombus.
Case report
An 18-year-old man was transferred to the regional neuroscience centre with a 10-day history of headache and features suggestive of raised intracranial pressure. Imaging revealed a cystic lesion of the fourth ventricle with associated hydrocephalus.
Endoscopic third ventriculostomy was undertaken. The procedure was complicated by haemorrhage. Ventriculostomy was abandoned and extraventricular drain (EVD) inserted. Subsequent imaging demonstrated diffuse subarachnoid blood but no apparent vascular injury.
The patient was admitted to intensive care unit (ICU) for neuro-protective management including insertion of a left femoral thermoregulatory central venous catheter (CVC) to assist in maintenance of normothermia. He was deemed high risk of venous thrombus. In keeping with the National Institute for Health and Care Excellence (NICE) guidelines, 1 mechanical thromboprophylaxis (graded compression stockings and sequential pneumatic compression device (Flowtron®)) was instituted. Anticoagulation was felt to pose an unacceptable risk of bleeding and was omitted but reviewed daily.
On day 11 of his ICU admission, the EVD dislodged and required reinsertion. The procedure was uneventful. Routine post-operative ECG demonstrated new deep T wave inversion on the anteroseptal leads. An echocardiogram, performed by the ICU registrar in search of underlying cardiac pathology, revealed a structurally unremarkable heart with normal function and no regional wall motion abnormality. Examination of the inferior vena cava (IVC) as part of the study led to the incidental finding of a large echogenic mass (6 cm × 2 cm) consistent with thrombus. Subsequent computed tomography images demonstrated the presence of right common iliac thrombus with extension of free floating thrombus into the distal IVC. Further thrombus was seen more proximally in a juxtarenal location (Figure 1(a)).
(a) Cavagram demonstrating thrombus within the juxtarenal cava and within the distal cava (arrows). (b) Cavagram demonstrating the position of the filter cranial to the juxtrenal thrombus (curved arrow) and to the distal IVC thrombus (arrow).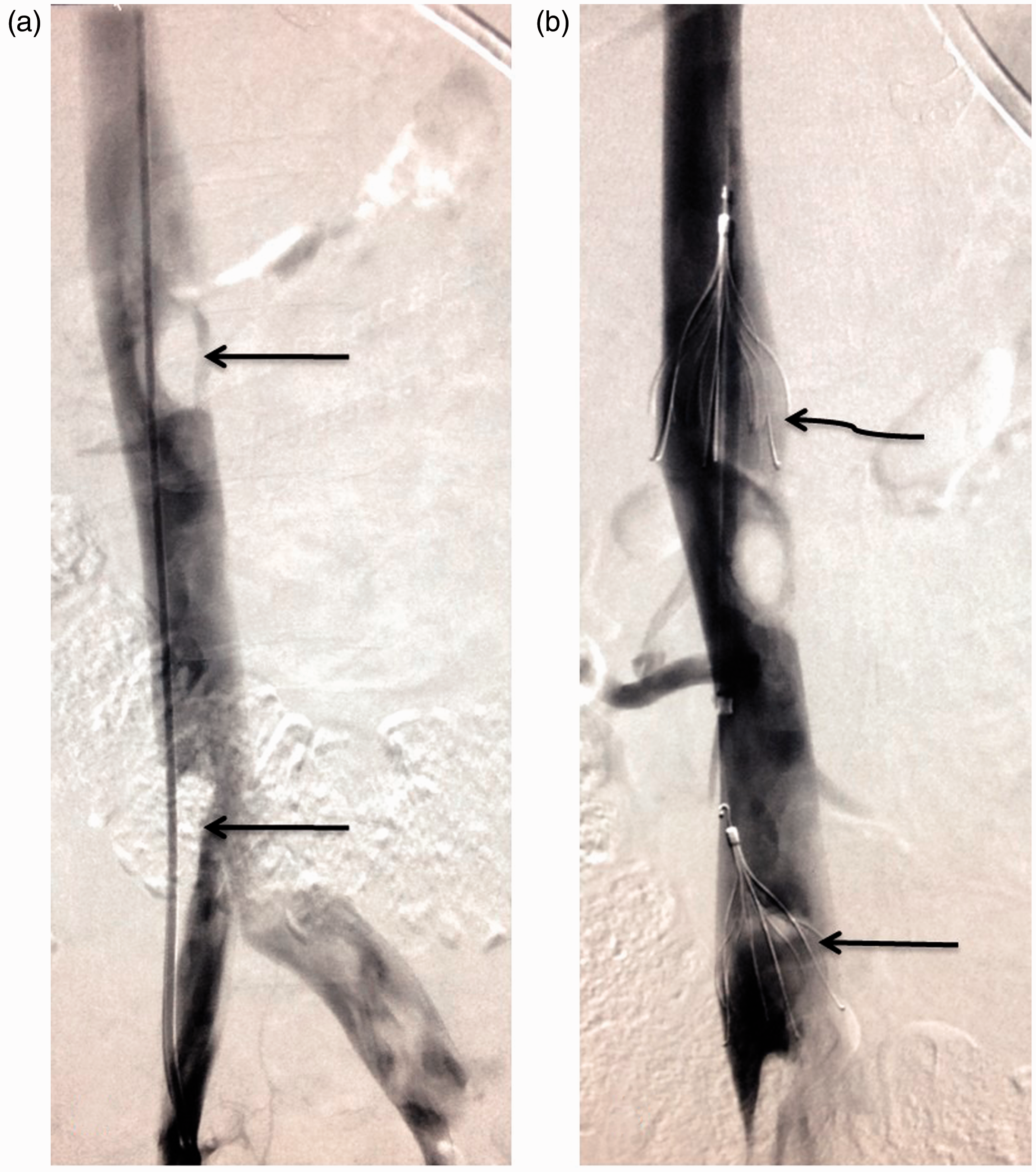
This case was of a large caval thrombus, at risk of embolisation, in a patient with contraindication to anticoagulation. In keeping with the NICE guidelines, 1 an IVC filter would be indicated. The juxta-renal nature of the proximal thrombus, however, raised concern that filter placement may result in renal vein obstruction and renal infarction. Following discussion with both interventional radiology and vascular surgery, radiological clot evacuation and filter placement was felt to be a viable option and the most appropriate course of action.
Two IVC filters (Cook Celect®) were utilised. One filter was placed superior to the renal veins in order to protect against embolisation to the lungs. This was done with access through the right internal jugular vein. A further filter was placed within the distal IVC in order to prevent further thrombus embolising to a juxtarenal location worsening the local clot burden and potentially causing renal vein thrombosis (Figure 1(b)). With both filters in situ, mechanical aspiration of clot using a 6F guide catheter to reduce the juxtarenal clot burden was undertaken. Although a significant amount of clot was retrieved, there remained a large amount of thrombus within the IVC. Both filters were therefore kept in situ with a view to removing when the patient was fully anticoagulated and dissolution of thrombus achieved.
Discussion
This case raises two important points. First, while this patient had multiple risk factors for venous thrombus, the extent of the clot was somewhat surprising. We examine whether the use of a femoral thermoregulatory CVC may have contributed to the risk and size of the clot. Second, contraindications to anticoagulation and the very proximal nature of the clot posed management issues: there was concern that placement of an IVC filter proximal to the clot would risk occlusion of the renal veins and resultant renal infarction.
Thermoregulatory CVCs and venous thromboembolism
Critically ill patients universally have risk factors for venous thromboembolism (VTE); this risk is frequently compounded by contraindication to anticoagulation. CVCs (particularly those in the femoral vein) are a recognised risk factor for VTE. 2 The presence of a catheter within the venous system causes stasis of flow and inflammation: two of the conditions described by Virchow in his pro-thrombotic triad. 3 The incidence of VTE following standard femoral CVC insertion in the critically ill or injured patient has been reported to lie between 9.6% and 26%. 3
In recent years, CVCs have been adapted to provide a means of temperature control. 4 Our institution utilises the Coolgard 3000® (Alsius) system with ICY® catheters (ZOLL Medical Corp., Chelmsford, MA, USA). These triple lumen catheters are of 9.3F diameter and thus wider than standard CVCs. They have along their length a varying number of reservoirs (dependent upon the model) connected to a source of temperature-controlled water. A thermistor-tipped Foley catheter provides feedback to the thermoregulatory device regarding core temperature. The system has been demonstrated to achieve a more rapid rate of cooling, and more reliable maintenance of core temperature at target, than other forms of cooling. 5 The system is indicated for ‘fever reduction, as an adjunct to other antipyretic therapy, in patients with cerebral infarction and intracerebral hemorrhage <sic> who require access to the central venous circulation and who are intubated and sedated’. 6 A range of three catheters, of varying length and with two, three or four water reservoirs are available. The smallest catheter may be inserted into the subclavian, jugular or femoral vein; the length of the other catheters limits their insertion site to the femoral vein.
The larger diameter of thermoregulatory catheters, in addition to the reservoirs, impedes venous flow to a greater extent than standard CVCs. 7 Furthermore, hypothermia is associated with a local procoagulatory state. 8 Combined, the associated risk of VTE is theoretically greater with the thermoregulatory CVC than its standard counterpart. 4 This association has been discussed in a number of case reports and series.
Two case reports relating to three patients9,10 associate caval thrombus with femoral thermoregulatory CVCs: one after cardiac arrest management, one after traumatic brain injury management, and the other in a patient with extensive burns; the latter two patients had contraindications to pharmacological thromboprophylaxis. These cases were reported on the basis of discovery of a potential complication of a new technology.
Pichon et al. 11 report a series of 40 patients with thermoregulatory CVC placed to assist in therapeutic hypothermia post cardiac arrest. Duplex ultrasound of the lower limbs was conducted routinely following CVC removal: no thrombi were identified. All patients underwent coronary angiography and therefore received heparin as part of routine care.
Gierman et al. 12 retrospectively reviewed the incidence of VTE in a total of 83 major trauma patients who underwent thermoregulatory CVC insertion as part of their routine initial trauma care. Forty-seven patients survived their initial injury, none of whom were suitable for pharmacological thromboprophylaxis. Ten were referred for prophylactic IVC filter of whom five (50%) were incidentally found to have VTE at the time of procedure. A further five patients had clinical evidence of VTE, the presence of which was confirmed on subsequent Doppler ultrasound. Therefore, in 10 of the 47 (21%) patients who survived initial injury VTE was detected on imaging. However, the true incidence might have been significantly higher because routine screening was not used. Of particular concern, six of the 10 confirmed cases of VTE (60%) extended into the vena cava and were therefore at greater risk of PE. 13
In a series of 11 patients with thermoregulatory CVCs inserted to assist management of traumatic brain injury, Simosa et al. 14 routinely conducted Doppler ultrasound following catheter removal or if there was clinical suspicion of VTE. An incidence of 50% is reported with two cases extending into the IVC. A non-statistically significant relationship between duration of catheter use and incidence of VTE is described.
It should be noted that in the case we describe, the iliac thrombus lay on the right whilst the thermoregulatory CVC had been inserted in the left femoral vein. It could be argued that this reduces the likelihood that catheter and clot were related. In the 20 cm femorally inserted thermoregulatory CVC, however, the reservoirs – the point where flow stasis and local procoagulation are likely to be greatest – lie within the IVC. It would seem plausible therefore that thrombus initially formed in the IVC and spread distally down veins free of catheter.
In summary, thermoregulatory CVCs carry a theoretically increased risk of deep vein thrombosis (DVT) formation. In a limited number of case studies and series, the incidence of DVT appears higher than for standard CVC; there is a high incidence of extensive caval thrombus. Any thrombotic risk associated with thermoregulatory CVC is compounded by the near universal contraindication to anticoagulation within the intended, neurologically injured, patient population. Whilst there is insufficient evidence to reach a definitive conclusion regarding thermoregulatory CVC usage in this group, the growing body of evidence suggests the need for further, robust investigation. In the interim, the thermoregulatory benefits of these devices should be carefully weighed against the theoretical increased thrombotic risk.
Radiological approach
The IVC filter is a mechanical device which reduces the risk of large thrombi in the deep venous system migrating to the pulmonary vessels. IVC filters are indicated for management of proven VTE in patients in whom anticoagulation is contraindicated or are at high risk of embolisation despite anticoagulation. 1 In keeping with the Society of Interventional Radiologists and the Eastern Association for the Surgery of Trauma guidelines, their role may be extended to prophylaxis for those patients at high risk of VTE, with no proven thrombus and a risk of bleeding too great to institute anticoagulation. 15
In this case, the identified thrombus extended to a position proximal to the renal veins. While the presence of an IVC filter would have provided protection against embolisation, an increase in clot density distal to the filter carried a theoretical risk of renal vein occlusion with subsequent renal infarction. Previous reports of cases of this nature describe management by surgical thrombectomy.9,10 The radiological approach described was deemed by our departments to provide an alternative, minimally invasive approach which could potentially reduce clot burden within the IVC and protect against embolisation of more distal thrombi.
Initially, the deployment of two filters – proximal and distal to the renal vein – was planned only as a peri-procedural measure; it was proposed that the proximal filter be removed once juxta-renal thrombus had been radiologically aspirated. Complete aspiration of the thrombus was not, however, possible and therefore both filters were left in situ: the proximal to protect against embolisation of juxtarenal thrombus to the pulmonary circulation; the distal to prevent an increase in juxtarenal thrombus burden and associated risk of renal infarction.
Conclusion
This is a case of an extensive caval thrombus, incidentally detected by clinician-delivered echocardiography on the critical care unit. There are theoretical reasons to believe this may have been associated with the thermoregulatory CVC inserted in the femoral vein, a concern which has been raised in other case reports and series from Europe and North America. Contraindication to anticoagulation precluded pharmacological management of the thrombus, and a novel radiological technique utilising two IVC filters was employed to reduce clot burden and minimise the risk of embolic complications.
Careful consideration of the risk to benefit ratio of thermoregulatory CVC use in the neuro-intensive care population is advised.
Footnotes
Consent
This article is published with the written assent of the patient’s parents.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
