Abstract
Polypharmacy is becoming more prevalent due to an ageing population. As more patients are undergoing surgical procedures, it is important to determine which group of patients are at higher risk of poorer outcomes. This review aimed to provide a summary of existing literature and to determine if polypharmacy is associated with poorer perioperative outcomes and to identify any gaps in the literature. This systematic review was conducted using electronic databases PubMed, Embase and Web of Science from their inception to December 2024. Statistical analysis was performed using generic inverse variance method. We identified 45 eligible studies from different countries and different surgical populations. Thirty-two studies (71.11%) defined polypharmacy as the use of five or more medications. Polypharmacy is significantly associated with postoperative delirium (odds ratio = 1.62, 95% confidence interval = 1.32–1.98, I2 = 0%). Although polypharmacy is found to be significantly associated with postoperative delirium, the relationship between polypharmacy and postoperative delirium remains complex.
Introduction
Polypharmacy is the use of multiple medications, more commonly in older patients with multiple comorbidities (Krustev et al 2022). Although different definitions exist in the literature, polypharmacy is usually defined as the use of five or more medications (Masnoon et al 2017). Polypharmacy is associated with poorer health outcomes such as drug interactions and adverse drug events, but is becoming more common due to the burden of chronic diseases in an ageing population (Kim et al 2024, Maher et al 2014). Moreover, more than 300 million major surgical procedures are performed yearly, and as the demand for surgical procedures continues to increase, many of these patients with polypharmacy will undergo surgical procedures (Etzioni et al 2003, Weiser et al 2015).
Preoperative screening tools can be useful to aid preoperative optimisation and risk counselling as they help to identify patients who are at high risk of postoperative complications (Aitken et al 2021). Polypharmacy is included in some preoperative screening tools such as the comprehensive geriatric assessment (CGA). CGA evaluates other domains such as function, nutrition, cognitive, mood, comorbidity and frailty (Aitken et al 2021). Perioperative outcomes that are measured include length of stay (LOS), intensive care unit (ICU) admission, patient-centred measures such as disability-free survival and pain score have been evaluated as well (Myles 2016). A meta-analysis of six studies on gastrointestinal cancer patients evaluated the effectiveness of CGA in predicting postoperative complication (Xue et al 2018). Polypharmacy was found to be one of the predictive factors for postoperative complications.
To our knowledge, there is no systematic review that has assessed the association of polypharmacy on perioperative outcomes. The rationale for this review is to provide a summary of existing literature that evaluates the association of polypharmacy and perioperative outcomes. The primary objective of this review is to determine whether polypharmacy is associated with poorer perioperative outcomes and the secondary objective to identify any gaps in the literature with regard to this topic.
Methods
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Page et al 2021). The inclusion criteria were English peer-reviewed journals which studied adults 18 years old and above and sought to determine the effect of polypharmacy on perioperative outcomes. Exclusion criteria were studies that involve subjects below 18 years old and non-English studies. In addition, guidelines, systematic reviews, meta-analyses, case reports and case series were excluded. We did not register a protocol for this review.
The literature search was conducted using electronic databases PubMed, Embase and Web of Science. The databases were searched from their inception to December 2024. The keywords used were (‘polypharmacy’ OR ‘multiple drugs’ OR ‘multiple medications’ OR ‘drug interaction’ OR ‘many drugs’ OR ‘many medications’) AND (‘outcome’ OR ‘outcomes’) AND (‘perioperative’ OR ‘postoperative’ OR ‘preoperative’ OR ‘operative’ OR ‘surgical’ OR ‘surgery’).
Two independent reviewers were involved in the data extraction and article selection process based on inclusion and exclusion criteria. Data such as type of study, country of study, number of patients, type of surgical procedures, definition of polypharmacy and perioperative outcomes assessed were extracted and presented accordingly.
The perioperative outcomes explored include those identified by the International Standardised Endpoints in Perioperative Medicine (StEP) initiative such as LOS, death within 30 days of surgery, death within 1 year of surgery, postoperative morbidity, admission to ICU within 14 days of surgery, readmission to hospital within 30 days of surgery, postoperative delirium and discharge disposition (Evered et al 2018, Haller et al 2019, Jackson et al 2023, Moonesinghe et al 2019). Postoperative delirium was commonly assessed using Confusion Assessment Method (Ho et al 2021). Postoperative morbidity was commonly measured using Clavien–Dindo classification (Jackson et al 2023). Grade I is defined as any complications without the need of intervention, and Grade II is complications requiring pharmacological treatment or blood transfusion. Grade III is complications requiring surgical, endoscopic or radiological intervention. Grade IV is life-threatening complications requiring ICU management. Grade V is patient death.
Patient-reported outcomes assessed include health-related quality of life (HRQOL), pain, disability and functional decline. HRQOL was measured using questionnaires such as Medical Outcomes Study Short Form 36 (SF-36) and Patient-Reported Outcomes Measurement Information System (PROMIS) (Cella et al 2010, Lins & Carvalho 2016). Disability was measured using World Health Organization Disability Assessment Schedule (WHODAS2.0) and Oswestry disability index (Ustün et al 2010). Functional decline was deemed to be present if there is decrease in WHODAS2.0. Pain was assessed using visual analogue scale (VAS) and was also included as a component of SF-36.
The quality of studies was assessed using the Newcastle-Ottawa Scale (NOS). The score ranges from 0 to 9 points, with a higher score indicating better quality. Studies with NOS scores of at least 7 are considered as high quality. For cohort studies, NOS addresses three areas, namely selection, comparability and outcome (Wells et al 2014).
Statistical analysis was performed using Review Manager Web (RevMan Web) Version 8.4.0. Studies that define polypharmacy as five or more medications were included in the meta-analysis. Outcomes from studies were pooled using unadjusted odds ratio with 95% confidence intervals (CIs), calculated using the generic inverse variance method. If odds ratio was not available, absolute numbers were used to calculate the unadjusted odds ratio. Due to heterogeneity across selected studies, random effect models were used. A p value of <0.05 was considered to be statistically significant. The I2 value measured consistency between included trials in the meta-analysis. According to the Cochrane guidelines, I2 value <50% was considered to be low, 50%–74% considered to be moderate and ⩾75% is considered to be high (Higgins et al 2003). Sensitivity analysis was performed using the leave-one-out method, and each study was omitted sequentially from the analysis to test the stability of the result.
Results
A total of 3459 articles were retrieved from the three databases, consisting of 800 from PubMed, 2141 from Embase and 518 from Web of Science. Figure 1 shows the PRISMA 2020 flow diagram (Page et al 2021). Out of the 3459 articles, only 45 articles were included in this review as shown in Table 1, 44 were cohort studies and one study was a case–control study (Marušič et al 2017).
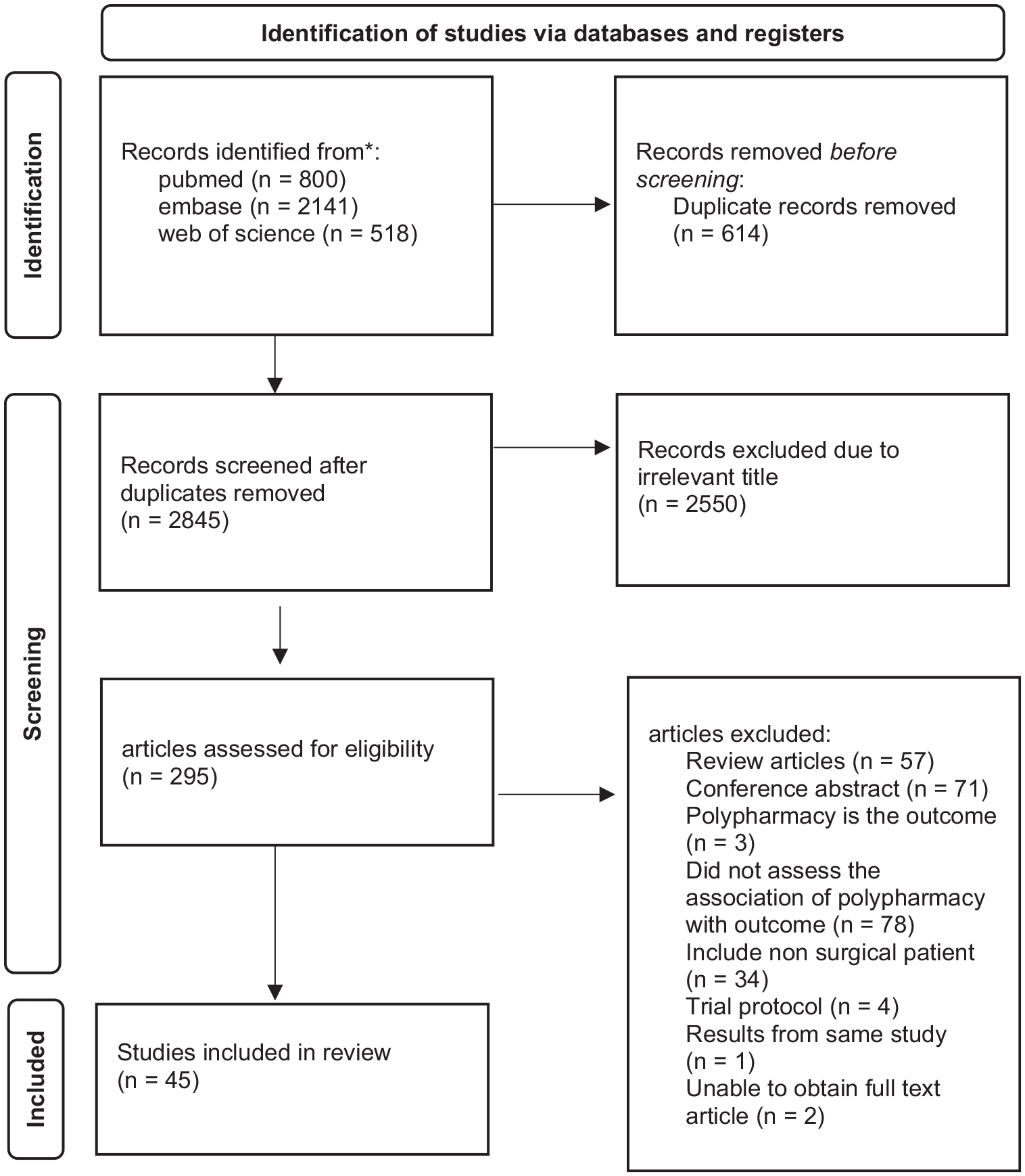
PRISMA flow diagram
Characteristics of included studies
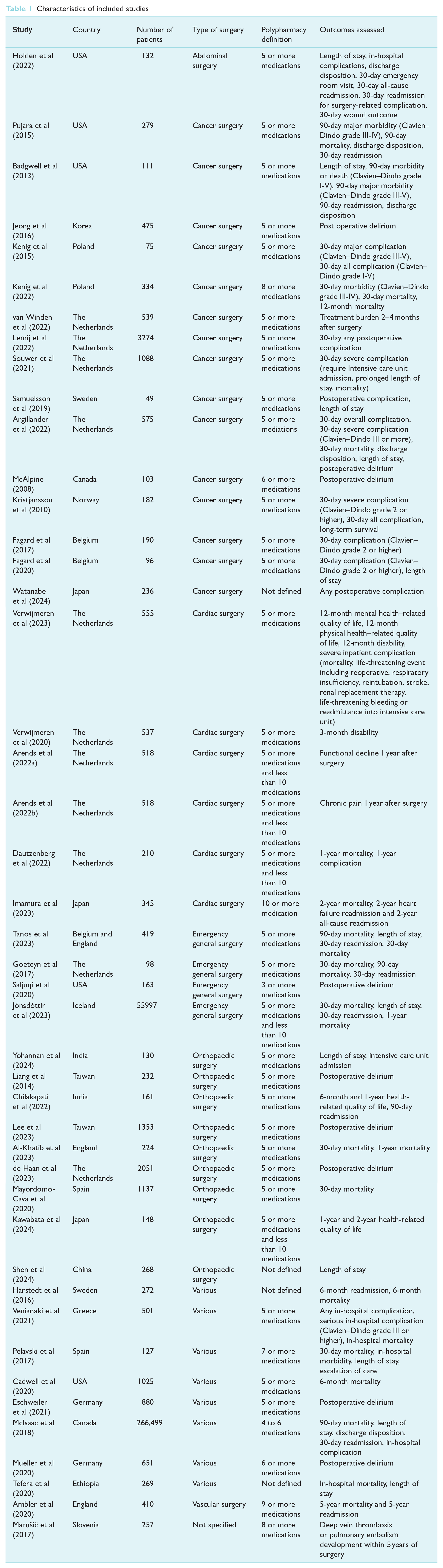
As shown in Table 1, studies were conducted in different countries. Of 45 studies, 28 studies (62.22%) were conducted in Europe, nine studies (20%) were conducted in Asia, five studies (11.11%) were conducted in the United States, two studies (4.44%) were conducted in Canada and one study (2.22%) was conducted in Africa. Studies were conducted on different surgical populations, including abdominal, cancer, cardiac, emergency, orthopaedic and vascular surgical procedures.
Of 45 studies, 32 studies (71.11%) defined polypharmacy as the use of five or more medications as shown in Table 1. Other reported cut-offs for polypharmacy ranged from three or more medications to ten or more medications. For patients on ten or more medications, two studies defined that as excessive polypharmacy and two studies defined that as hyper polypharmacy. For perioperative outcomes, nine studies (20%) evaluated postoperative delirium and 12 studies (26.67%) have evaluated on prolonged LOS. Eight studies (17.78%) evaluated 30-day complication. Eight studies (17.78%) evaluated 30-day mortality.
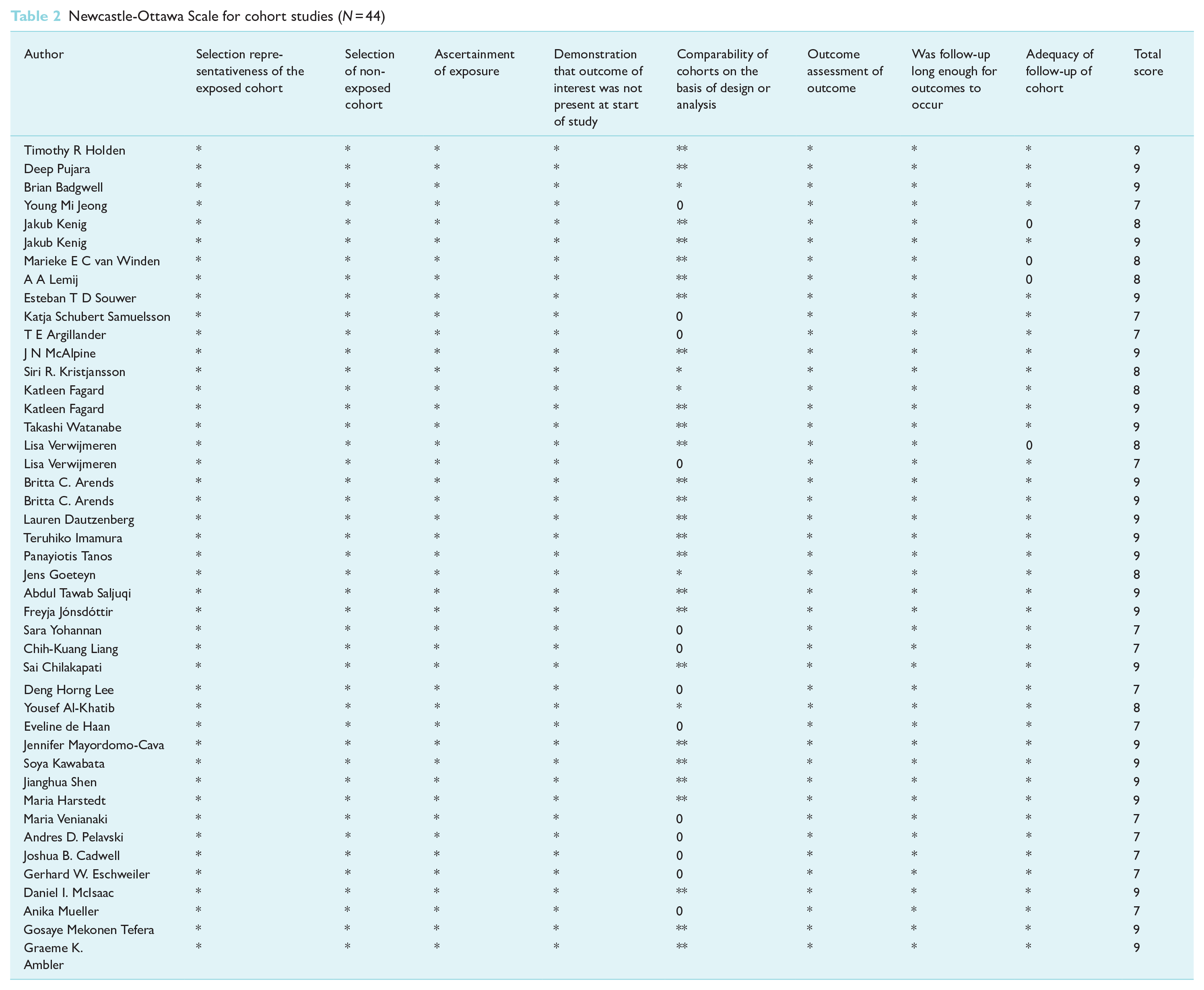
The studies included are of good quality, with at least seven stars in the NOS rating scale as shown in Table 2. Twenty-three studies (52.27%) had 9 stars, eight studies (18.18%) had 8 stars and 13 studies (29.55%) had 7 stars.
Newcastle-Ottawa Scale for cohort studies (N = 44)
Two studies were included in the forest plot of ICU admission in patients with polypharmacy compared to non-polypharmacy as shown in Figure 2. Polypharmacy is not significantly associated with ICU admission (odds ratio (OR) = 1.73, 95% CI = 0.96–3.10, I2 = 0%). Two studies were included in the forest plot of discharge to another care facility in patients with polypharmacy compared to non-polypharmacy as shown in Figure 3. Polypharmacy is significantly associated with discharge to another care facility (OR = 1.68, 95% CI = 1.08–2.61, I2 = 0%). Three studies were included in the forest plot of 30-day mortality in patients with polypharmacy compared to non-polypharmacy as shown in Figure 4. Polypharmacy is significantly associated with 30-day mortality (OR = 5.28, 95% CI = 2.50–11.17, I2 = 14%). Five studies were included in the forest plot of postoperative delirium in patients with polypharmacy compared to non-polypharmacy as shown in Figure 5. Polypharmacy is significantly associated with postoperative delirium (OR = 1.62, 95% CI = 1.32–1.98, I2 = 0%).
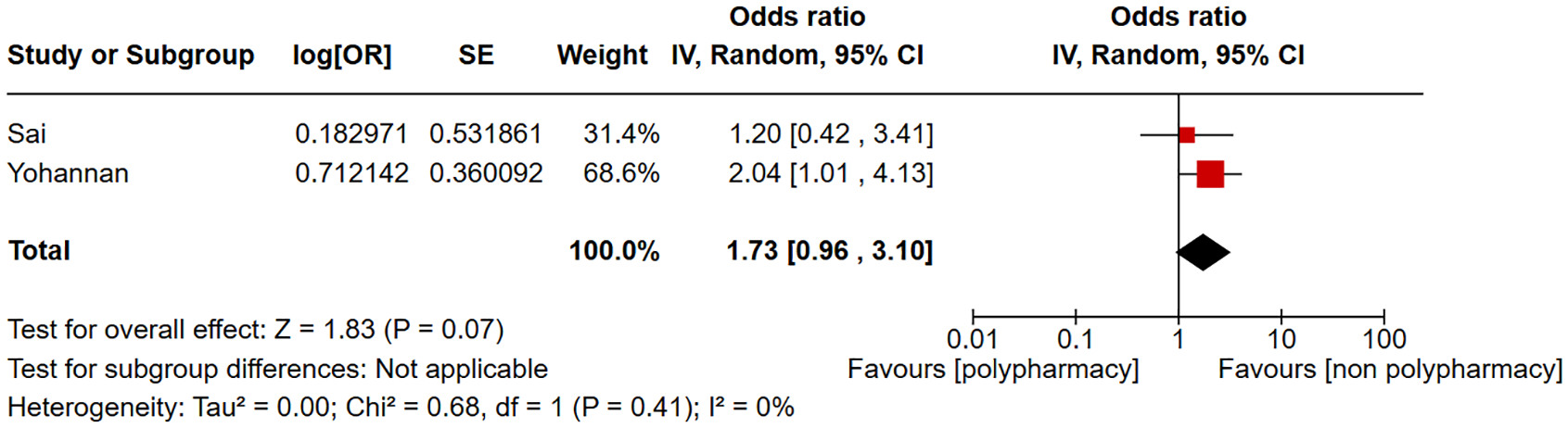
Forest plot of ICU admission in patients with polypharmacy compared to non-polypharmacy
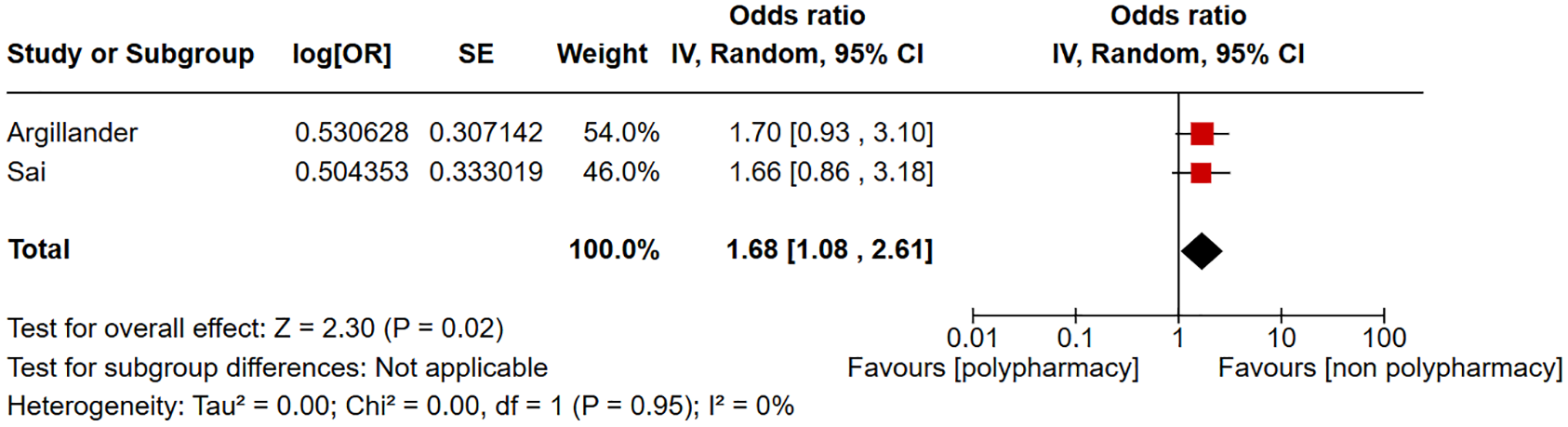
Forest plot of discharge to another care facility in patients with polypharmacy compared to non-polypharmacy
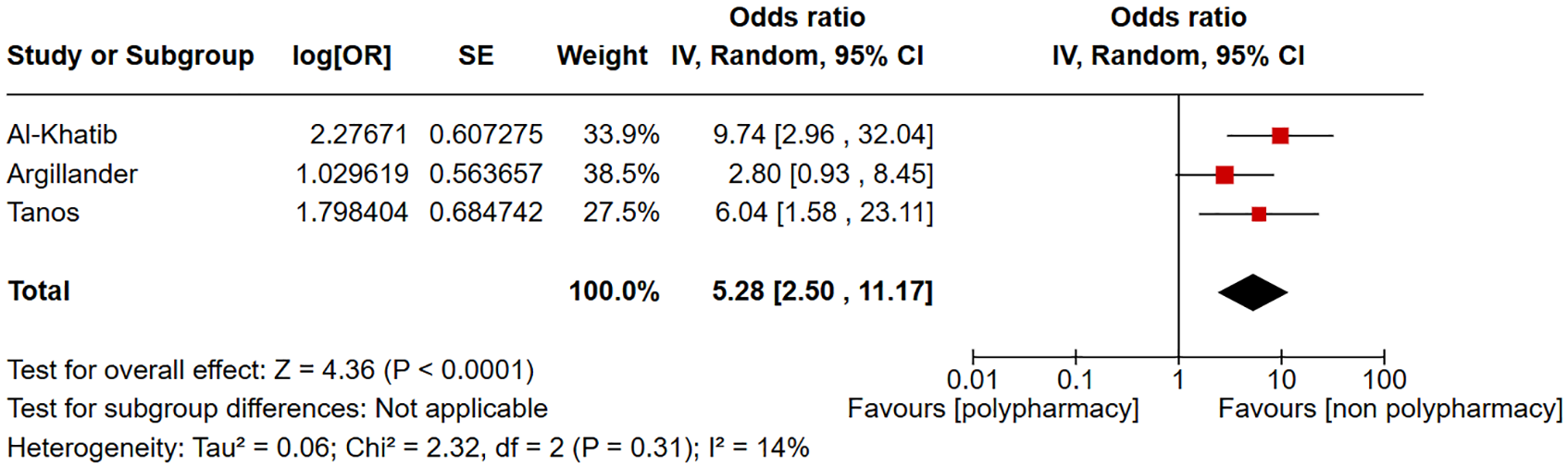
Forest plot of 30-day mortality in patients with polypharmacy compared to non-polypharmacy
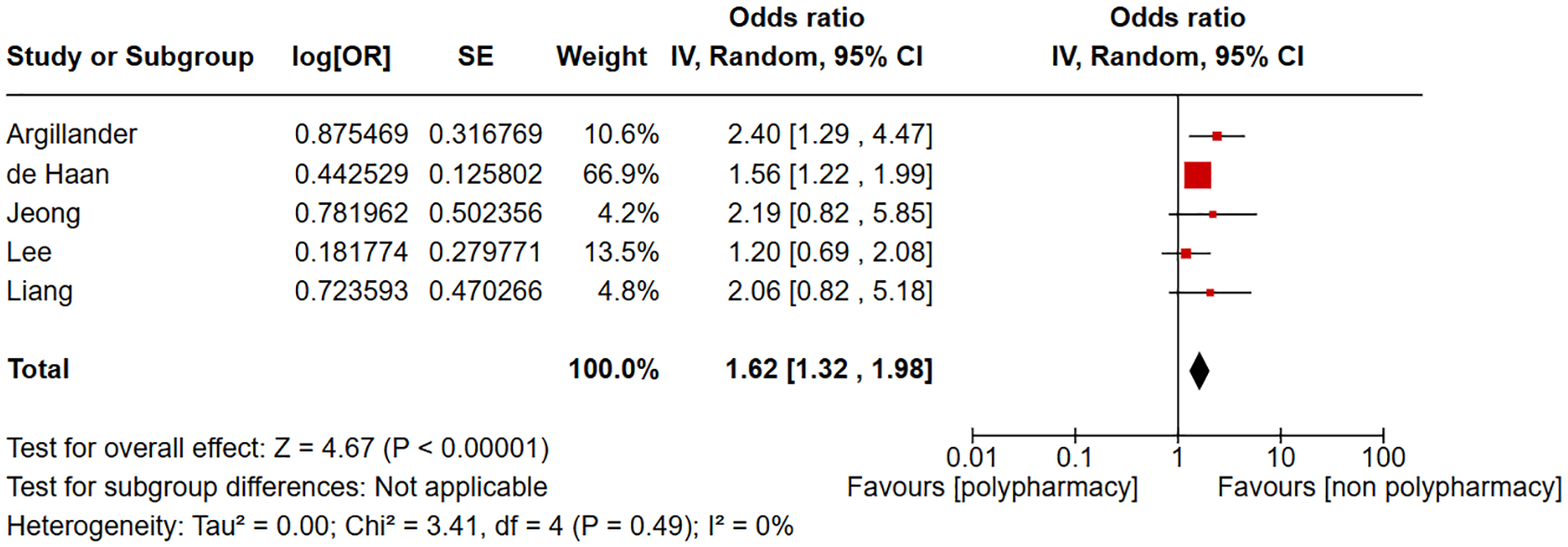
Forest plot of postoperative delirium in patients with polypharmacy compared to non-polypharmacy
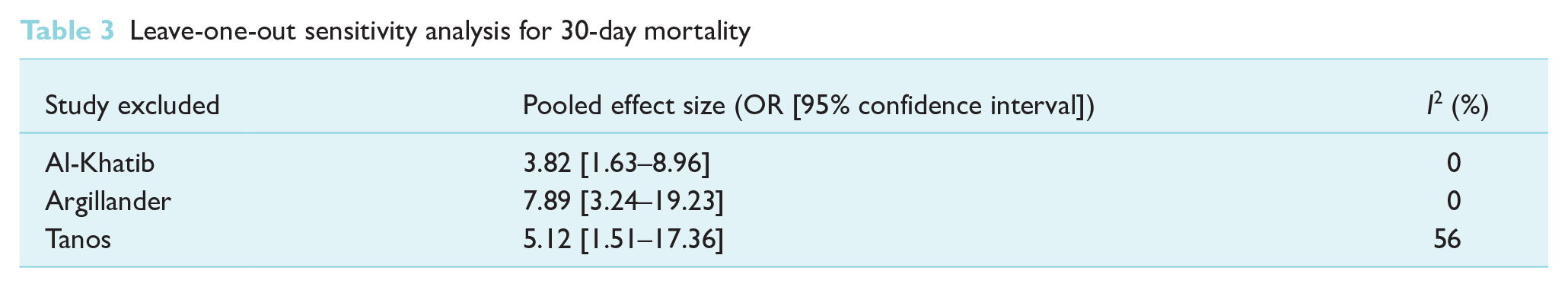
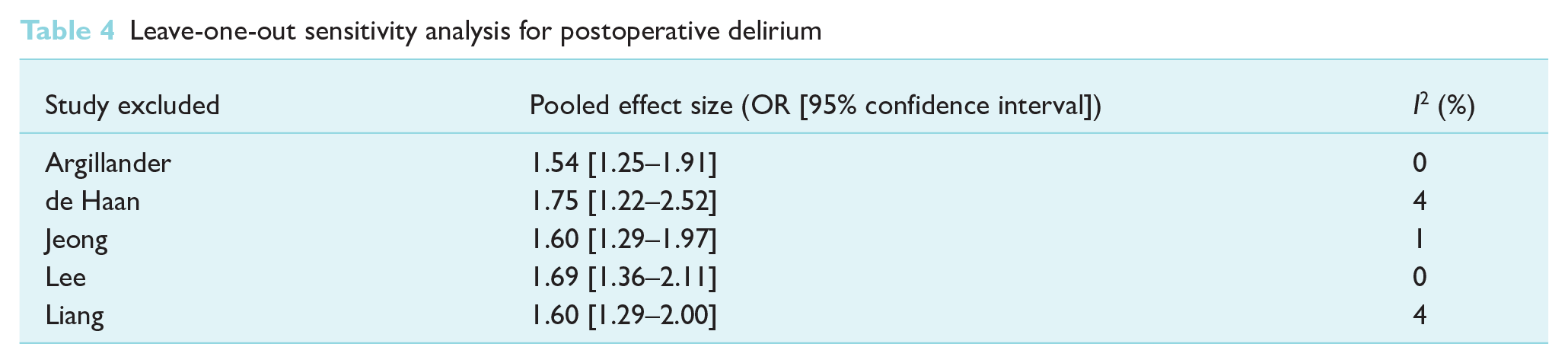
Leave-one-out sensitivity analysis for 30-day mortality in Table 3 showed that with the removal of study by Tanos et al, heterogeneity I2 increased from 14% to 56%. In Table 4, the leave-one-out sensitivity analysis for postoperative delirium showed no significant change in pooled effect size and heterogeneity was caused by excluding any one study.
Leave-one-out sensitivity analysis for 30-day mortality
Leave-one-out sensitivity analysis for postoperative delirium
Discussion
In this systematic review, we have identified 45 studies that explored the association of polypharmacy with various perioperative outcomes. Polypharmacy is most commonly defined as the use of five or more medications. Apart from comparing perioperative outcomes between polypharmacy and non-polypharmacy groups, there were some studies that investigated the association between the number of medications and perioperative outcomes. For perioperative outcomes, there were variations in the definitions of postoperative complications and different time points were selected for evaluation of complications. Different definitions were also used to define prolonged LOS. These differences made comparison challenging.
Our meta-analysis of five studies found that polypharmacy is significantly associated with postoperative delirium (Argillander et al 2022, de Haan et al 2023, Jeong et al 2016, Lee et al 2023, Liang et al 2014). The sensitivity analysis showed stability of results as no significant change in pooled effect size and heterogeneity was caused by excluding any one study. This finding presents a different perspective compared to the existing literature. A prior systematic review by Kassie et al, evaluating preoperative medication use and postsurgical delirium, found that there were limited number of high-quality studies that quantify the direct association between preoperative medication use and postsurgical delirium (Kassie et al 2017). Polypharmacy is known to be associated with delirium, but explanation of the link between polypharmacy and delirium remains complex (Hein et al 2014). Classes of medications that may induce delirium include anticholinergics, benzodiazepines and narcotics. These are commonly prescribed in the healthcare systems and some may be used as anaesthetic agents (Alagiakrishnan & Wiens 2004, Ghossein et al 2024, Smith et al 2024). However, it is also important to take note that delirium assessment tools only reveal the patient’s state at the time of assessment, and the frequency of postoperative delirium may increase with increased charting by healthcare professionals (Young et al 2022).
Our meta-analysis also found that polypharmacy is significantly associated with 30-day mortality and discharge to another care facility. However, a small number of studies were included in these meta-analyses. Furthermore, the sensitivity analysis for 30-day mortality showed that the removal of the study by Tanos et al led to increase in heterogeneity from 14% to 56%. This suggests that more studies are needed to investigate the association between polypharmacy and 30-day mortality as well as polypharmacy with discharge to another care facility. There is complex interplay between polypharmacy, frailty and chronic comorbidity, which may be associated with higher mortality. Poorer recovery after an operation may result in higher care needs, requiring transfer to care facilities such as a nursing home, instead of discharging patients back home (van Dam et al 2022).
Preoperative deprescribing is a possible consideration for patients with polypharmacy. A systematic review and meta-analysis identified 16 articles that studied deprescribing intervention (Lee et al 2022). It was found that deprescribing interventions were associated with positive or neutral outcomes. However, some studies included patients without polypharmacy. Furthermore, the authors highlighted that the evidence is weak due to heterogeneity of interventions and outcomes. In addition, polypharmacy is closely associated with comorbidities and frailty (van Dam et al 2022). This needs to be taken into consideration when deprescribing. Potential harms of deprescribing include adverse drug withdrawal reactions, pharmacokinetic and pharmacodynamic changes and return of medical condition (Reeve et al 2014).
Apart from clinical outcomes, this systematic review identified only six articles that evaluated patient-reported outcomes. Four of the six studies were derived from the Anaesthesia Geriatric Evaluation study cohort that included patients 70 years and above who underwent elective open-heart surgical procedure, and patient-reported outcomes were physical and mental HRQOL, disability and functional decline by using SF-36 and WHODAS2.0 questionnaires (Arends et al 2022a, 2022b, Verwijmeren et al 2020, Verwijmeren et al 2023). Another study was conducted on a cohort of spinal deformity patients aged 65 years and above, and patient-reported outcomes were disability, pain, and physical, mental and social wellbeing, measured by Oswestry Disability Index, VAS and PROMIS, respectively (Chilakapati et al 2022). The last study was conducted on patients with lumbar spine stenosis, and the HRQOL was assessed using questionnaires commonly used in patients with lumbar spinal stenosis, namely, Zurich Claudication Questionnaire (ZCQ), the Roland Morris Disability Questionnaire (RDQ) and the Japanese Orthopaedic Association Back Pain Evaluation Questionnaire (JOABPEQ) (Kawabata et al 2024). Different questionnaires were utilised in different studies with different time points and study populations. Thus, it may be difficult to compare outcomes between these studies.
This systematic review provides a comprehensive review of the articles that evaluate association of polypharmacy with various perioperative outcomes in different surgical populations across different countries. Our study identified key perioperative outcomes associated with polypharmacy. Given the increasing prevalence of polypharmacy, especially among the elderly population, it is important for perioperative practitioners to identify patients at higher risk of developing perioperative complications and, where possible, implement additional precautionary measures to mitigate these risks. Furthermore, our study highlighted areas for future research in perioperative practice, particularly in exploring more patient-reported outcomes during the perioperative period. Some limitations of our systematic review include significant heterogeneity as populations are drawn from different countries and surgical specialities. Furthermore, our analysis is limited by risk of bias from missing data and reporting from individual studies.
In conclusion, polypharmacy is found to be significantly associated with postoperative delirium. However, the relationship between polypharmacy and postoperative delirium is complex. Reduction of polypharmacy through deprescribing may potentially be explored for future research. More studies are needed to determine whether polypharmacy is associated with other perioperative outcomes. Patient-reported outcomes are not commonly evaluated as perioperative outcomes, and this may be an area for future research.
Footnotes
Acknowledgements
This work was carried out solely by the listed authors.
Author contributions
Y.L. and Y.L. prepared the manuscript draft. All authors revised the draft critically for important intellectual content and agreed to the final submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
