Abstract
Objective
In this survey, we assessed the current clinical management of postoperative delirium (POD) among Chinese anesthesiologists, after publishing the European POD guideline.
Methods
We administered an electronic survey, designed according to the European POD guideline. The survey was completed using mobile devices.
Results
In total, 1,514 respondents from China participated in the survey. Overall, 74.4% of participants reported that delirium is very important. More than 95% of participants stated that they routinely assessed POD. In total, 61.4% screened for POD using clinical observation and 37.6% used a delirium screening tool. Although the depth of anesthesia (a POD risk factor) was monitored, electroencephalogram monitoring was unavailable to 30.6% of respondents. Regarding treatment, only 24.1% of respondents used a standard algorithm; 58.5% used individualized treatment.
Conclusion
Our survey showed that there are high awareness levels among Chinese anesthesiologists regarding the importance of POD. However, routine assessment and monitoring of all patients, including perioperative anesthesia depth monitoring, and a treatment algorithm need to be implemented on a larger scale. According to the results, efforts should be made to improve the knowledge of POD among Chinese anesthesiologists.
Introduction
Postoperative delirium (POD) is a typical complication after surgery, and interventional procedures can affect patients of any age. 1 According to the Fifth Edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), delirium can be defined as a disturbance in attention and awareness that develops over a short period of time and represents a change from baseline attention and awareness. Delirium tends to fluctuate in severity during the course of a day and can be accompanied by additional disturbance in cognition. 2 Delirium can influence perception, thinking, memory, psychomotor behavior (e.g., hypoactive vs. hyperactive), emotion, and the sleep–wake cycle. 3 Generally, hypoactive delirium has a worse prognosis and poorer outcome because it is more common and can easily be overlooked without a screening instrument, thereby delaying early treatment.1,4
To address this topic, recommendations for the prevention and treatment of POD were released in May 2017 by the European Society of Anaesthesiology (ESA) in its evidence- and consensus-based guideline. One group most vulnerable to POD is older patients, who experience cognitive decline after surgery that is associated with POD two to three times more often than in younger patients.5–7
China has a rapidly growing older population, which could reach over 200 million people aged ≥60 years by the end of 2020. China has also had increasing cases of geriatric anesthesia. 8 Therefore, implementation of POD prevention and treatment strategies in upcoming years will have an important role in the country with the largest aged population, to avoid long-term cognitive decline. The purpose of this survey conducted in China was to evaluate the current importance of POD and clinical practice among Chinese anesthesiologists with respect to the ESA guideline, as well as to advance perspectives regarding global approaches to POD.
Methods
Survey design and target population
The ESA designed the present cross-sectional survey. The institutional ethical review board of Huazhong University of Science and Technology, Wuhan approved this survey targeting Chinese anesthesiologists (TJ-IRB20180601).
The original questionnaire was developed in English by members of the taskforce and the advisory board, who were responsible for development of the ESA guideline. The final survey was translated into Chinese and consisted of 21 questions that were subdivided into five sections: basic demographic data of respondents, importance of delirium, assessment of pain and delirium, monitoring the depth of anesthesia (DOA), and treatment for delirium.
The survey was proofread and then launched via WeChat©, a mobile software application developed by Tencent Mobile International Limited. The survey was accessible using any web-enabled mobile device able to run the WeChat© application.
Data sampling
The survey was conducted from 10 to 24 September 2017, during and after the 25th Annual Meeting of the Chinese Society of Anesthesiology (CSA 2017) in Zhengzhou, China. We invited all congress attendees to participate in the survey. All attendees received daily reminder messages about the questionnaire survey, sent through the broadcasting system during the meeting. The questionnaire survey was delivered using WeChat© and could be forwarded with a Quickmark scanner application.
All participants in the study provided electronic informed consent. Informed consent was presented in an introductory paragraph at the beginning of the survey.
Statistical analysis
The analysis encompassed frequency distributions of responses, generated using IBM SPSS version 25 (IBM Corp., Armonk, NY, USA). Responses to questions that involved written input from respondents were translated from Chinese into English, categorized, analyzed for redundancy, and then counted. Figures were created using Microsoft Excel.
Results
General data of respondents
Altogether 1,514 Chinese anesthesiologists completed the survey. Of the total, 987 (65.2%) were not members of the Chinese Society of Anesthesiology. According to the congress committee, 7,368 attendees participated in the congress. It is unknown how many attendees actually received or noticed a message about or the questionnaire survey; however, we estimated that the response rate was at least 20.5% (1514/7368).
In terms of their current position, 943 respondents reported being either the department head and/or a full or assistant professor (n = 313, 20.7%) or a consultant/specialist in anesthesiology (n = 630, 41.6%). A total 170 (11.2%) respondents were anesthesiologists in training. Medical students and nurses as well as other health care personnel also completed the survey. Table 1 shows the distribution of survey respondents.
General data and characteristics of respondents.
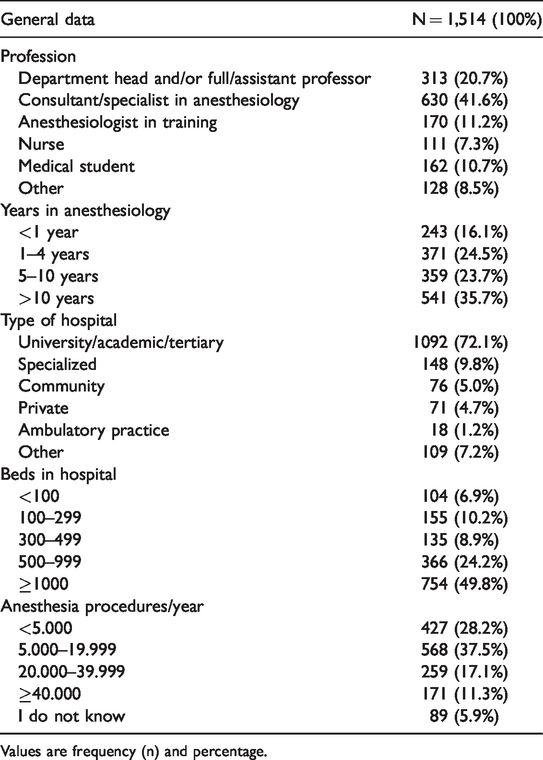
Values are frequency (n) and percentage.
Regarding work experience, 35.7% (n = 541) of respondents had more than 10 years of experience in their field, 359 (23.7%) had been working for 5 to 10 years and 371 (24.5%) for 1 to 4 years.
Most participants (n = 1,092, 72.1%) were working in a university, academic, or tertiary care hospital. Nearly half of respondents (n = 754, 49.8%) worked in a hospital with more than 1,000 beds. The number of anesthetic procedures performed per year varied from more than 40,000 (n = 171, 11.3%) to fewer than 5,000 (n = 427, 28.2%); most respondents (n =568, 37.5%) performed between 5,000 and 19,999 procedures per year. Table 1 presents a complete summary of the general data of respondents.
Indicated importance of delirium
POD was considered to be “very important” by 1,127 participants (74.4%), and “important” by 353 participants (23.3%). Only six respondents answered that POD was “not important” (0.4%), followed by 28 (1.9%) who responded that POD was “not very important” (Figure 1).

Reported importance of delirium.
Assessment of delirium and pain
Regarding the question “Do you routinely assess POD?”, 665 participants (43.9%) specified that they assessed POD only in patients presenting symptoms; 562 participants (37.1%) indicated that they checked for POD only in patients with risk factors. Of the remaining respondents, a more general answer was common; 103 (6.8%) participants assessed POD in more than half of their patients and 124 (8.2%) monitored for POD in fewer than half of their patients. Only 60 respondents (4.0%) reported that they never assessed POD (Figure 2).
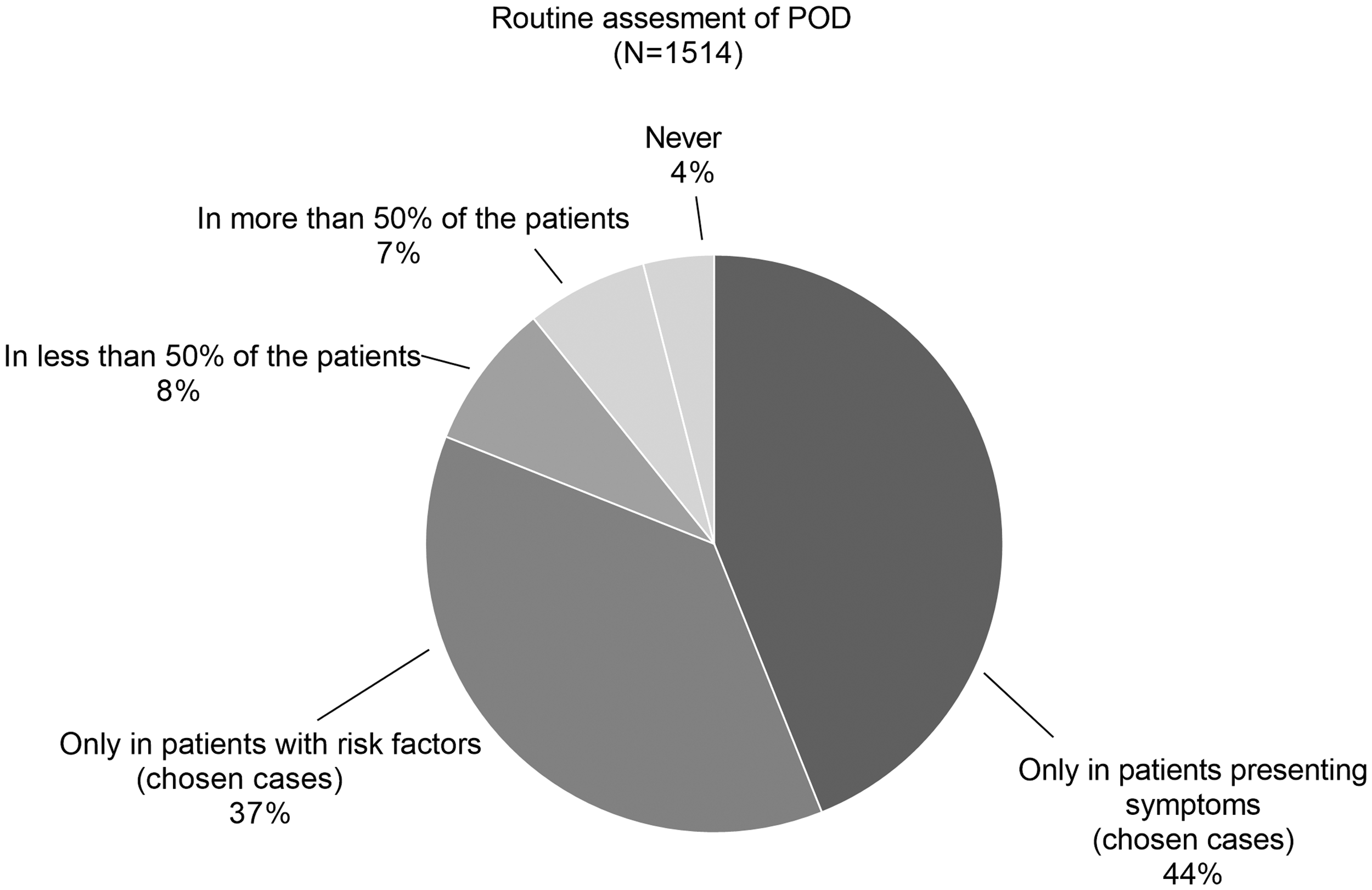
Routine assessment of postoperative delirium (POD).
We analyzed responses regarding the time factor of POD measurement; multiple answers were possible. Most respondents checked for delirium either “before OR recovery room discharge” (n = 854, 56.4%) or during “the first post-op day” (n = 815, 53.8%). “Up to 3 post-operative days” and “up to 5 post-operative days” was selected by 511 (33.8%) and 143 (9.5%) participants, respectively (Figure 3).

Assessment time of postoperative delirium (POD).
Most respondents (n = 930, 61.4%) used clinical observation to assess POD; 37.5% (n = 567) used a quantitative score or a delirium screening tool to evaluate the patient (Figure 4). In total, 577 (64.1%) of the anesthesiologists with 5 or more years’ experience (N = 900) stated that they used clinical observation and 316 (35.1%) used a quantitative score or delirium screening tool. For anesthesiologists with fewer than 5 years of experience (N = 614), 57.5% (n = 353) used clinical observation and 40.9% (n = 251) used a quantitative score or delirium screening tool.

Assessment of postoperative delirium (POD).
Among scales and instruments used to assess POD, the five most frequently selected were the Clinical Assessment of Confusion (CAC) (n = 639, 42.2%), the Bedside Confusion Scale (BCS) (n = 526, 34.7%), the Cognitive Test for Delirium (CTD) (n = 526, 34.7%), the Clinical Global Impressions Scale Delirium (CGID) (n =444, 29.3%), and the Confusion Assessment Method (CAM) (n = 344, 22.7%) (Figure 5).

Delirium screening tools.
Regarding post-operative pain assessment, slightly more than half of participants (n = 800, 52.8%) stated that they used clinical observation; the other 714 respondents (47.2%) used a quantitative score. The five most frequently selected scales were the visual analogue scale (n = 812, 53.6%), numeric rating scale (NRS) (n = 653, 43.1%), verbal rating scale (n = 542, 35.8%), faces pain scale (n = 487, 32.2%), and the NRS visually enlarged and laminated (n = 391, 25.8%).
Monitoring depth of anesthesia (DOA)
Regarding the question “Do you monitor the depth of anesthesia (using electroencephalogram (EEG)- and/or electromyography-based monitoring)?”, 463 participants (30.6%) indicated that they had no such equipment available. This was followed by 142 (9.4%) respondents who said they never monitor the DOA, although monitoring is available. The remaining 60% of participants reported monitoring the DOA: 38.2% (n = 579) in selected cases only, 9.4% (n = 142) in more than 50% of patients, 5.5% (n = 83) in 20% to 50% of patients, and 6.9% (n = 105) in fewer than 20% of patients (Figure 6).

Depth of anesthesia monitoring.
When asked the reasons for measuring the DOA, the most frequently chosen answers were to “reduce the risk of intraoperative awareness” (n = 1,300, 85.9%), followed by “to reduce the risk of burst suppression” (n = 633, 41.8%) and “reduce turnover time (time to extubation)” (n = 455, 30.1%).
Treatment for delirium
Responses regarding the reported POD therapy regimes comprised free text as well as multiple-choice answers. In total, 885 respondents (58.5%) said that they applied individualized treatment. Only 24.1% (n = 365) treated POD according to a standard algorithm and 264 (17.4%) said they did not treat POD at all. A total 63.4% (n = 571) of the anesthesiologists with 5 or more years of experience (N = 900) stated that they applied individualized treatment and 19% (n = 171) treated delirium according to a standard algorithm. Among anesthesiologists with fewer than 5 years of experience (N = 614), a total 51.1% (n = 314) used individualized treatment and 31.6% (n = 194) treated POD according to a standard algorithm.
When asked to specify the characteristics of the treatment algorithm, the most frequently mentioned approach was “symptom based” (n = 1,001, 66.1%), followed by “specialist consultation (neurologist/geriatrician/psychiatrist)” (n = 854, 56.4%), and “cause-based” (n = 627, 41.4%).
In the final two questions of the survey, respondents were asked to enter a brief free-text description of symptom-based or cause-based treatment. Multiple answers were possible. The five most frequently used terms in the analysis of symptom-based treatment were “sedation” (n = 163, 29.6%), “analgesia” (n = 93, 16.9%), “drugs/medication” (n = 62, 11.3%), “dexmedetomidine” (n = 30, 5.5%), and “fluid” (n = 27, 4.9%; Table 2). The five most frequently entered terms regarding cause-based treatment were “analgesia” (n = 76, 20.2%), “fluid” (n = 29, 7.7%), “nothing” (n = 29, 7.7%), “don’t know” (n = 28, 7.5%), and “drugs/medication” (n = 26, 6.9%) (Table 3).
Text analysis of symptom-based treatment strategies for postoperative delirium.

Note: The underlying question was “Briefly explain your symptom-based treatment”; multiple manual text entries were allowed. Of 1,514 participants, 935 left this question blank. After the text analysis, 550 responses were included in the statistical analysis.
Text analysis of cause-based treatment strategies for postoperative delirium.

Note: The underlying question was “Briefly explain your cause-based treatment”; multiple manual text entries were allowed. Of 1,514 participants 1,016 left this question blank. After the text analysis, 376 responses were included in the statistical analysis.
Discussion
Current awareness of the importance of POD was excellent among respondents to this survey in China. 9 In total, 98% of respondents indicated delirium to be “highly important” or “important”. POD screening was also performed in many hospitals, and only 4% of respondents said they never screened for POD. However, POD was only assessed in certain patient populations at risk for POD or those with POD symptoms and not routinely in all patients. Even if assessed, a validated score was used in only one-third of patients. POD prevention tools such as EEG monitoring were unavailable to one-third of respondents and not used by another third of participants. In addition, a POD treatment standard algorithm was only available in one-fourth of hospitals.
The European guideline recommends routine screening for POD in all patients (starting in the recovery room) up to postoperative day 5, using a validated screening tool. 1 The reason is that clinical POD has low sensitivity and high specificity.1,10–12 We found that POD monitoring decreased on each day after surgery. Postoperative studies have shown that POD can be avoided by monitoring and using delirium-prevention programs like the Hospital Elder Life Program (HELP)13,14 and modified HELP.13–15
Regarding the diagnosis and treatment of POD, our survey revealed that most anesthesiologists (61.4%) still used clinical observation to diagnose POD, compared with 37.5% who used a validated screening tool. Inexperienced anesthesiologists used a screening tool more frequently (40.9%) than experienced ones (35.1%), and more experienced anesthesiologists relied on clinical observation (64.1%) more often than those with less experience (57.5%). A validated scale increases POD detection owing to fewer false negative results.1,10–12 Clinical judgement, however, can result in high failure rates and has low diagnostic validity. 16 Therefore, to diagnose POD according to the DSM-5, it is recommended to use validated instruments, in addition to clinical observation and chart review.1,17 Early diagnosis and immediate treatment of POD are extremely important in terms of reducing the duration of POD. 1
In terms of validated POD screening tools, the guideline recommends the Nursing Delirium Screening Scale (Nu-DESC) and CAM for the recovery room setting. 1 The former can be used without additional training, and the CAM requires training. 18 Whereas the CAM ranked fifth in our survey results, the Nu-DESC was only used by 106 respondents and ranked in position 17. Survey respondents most often reported using the CAC (n = 639, 42.2%), followed by the BCS (n = 526, 34.7%), CTD (n = 526, 34.7%), CGID (n = 444, 29`.3%), and CAM (n = 344, 22.7%).
The CAC was developed to quickly assess confusion using a 25-item checklist and is not based on the DSM criteria. 19 It is an instrument for nonexperts that is quick to use. 20 However, the CAC has poor criterion validity when measured against the DSM-IV criteria.21,22 The BCS has been validated against the CAM and takes about 2 minutes to complete.22–24 Like the CAC, it is designed for nonexperts. 20 The CTD was originally developed for the ICU and for patients who are intubated and cannot speak.25,26 It is based on the DSM-III-R and has been validated for use in non-ICU settings.20,21,27,28 The CGID was designed to be an easily applied tool for clinicians to assess a patient’s progress and treatment response. 29 The scale has been validated against the Delirium Rating Scale-Revised-98 27 and can be used by nonexperts as a quick screening tool to measure delirium severity.20,30
Regarding monitoring of the DOA, our survey revealed that one-third of respondents used monitors in selected cases, one-third did not use them routinely, and one-third did not have any equipment. Previous studies from other countries have shown similar results regarding the frequency of bispectral index (BIS) monitoring usage and different results in terms of availability.
In Korea, DOA monitoring is used to avoid excessively light or deep anesthesia in patients at high risk for adverse outcomes, but no surveys regarding its general usage could be found. 31 One study from Hong Kong showed that some anesthesiologists tend not to use certain anesthetics owing to the unavailability of DOA monitoring equipment; however, again no further surveys were found. 32 A study reported low availability of monitoring equipment among rural areas of India and mentioned that DOA monitoring was mainly used for the prevention of awareness. A study from Thailand regarding adverse events during anesthesia also showed very low availability of monitoring equipment.33,34
A questionnaire survey among anesthesiologists in Japan showed that 37% use DOA monitoring equipment in almost all cases, 29% in about half of cases, and 34% did not use DOA monitoring at all. However, there was no information regarding the availability of BIS monitoring. 35 A national survey among anesthetists in the United Kingdom showed that overall, 62% of the centers surveyed were equipped with DOA monitors but only 1.8% of the anesthetists used monitoring in every patient; 25% used DOA monitoring in selected cases only. 36 In Ireland, the availability of DOA monitors is 80% of centers; 6.7% of participating anesthesiologists reported using monitoring routinely and 54.8% in selected cases only. 37 A survey in Sweden revealed that 50% of clinics have monitoring equipment, which is used by 12% of anesthetists in all cases, 22% in selected cases only, and 12% rarely use monitoring. 38 In Australia, overall availability of DOA equipment as high as 98.8% was reported. DOA monitoring was used by 29% of anesthesiologists in more than one-third of patients, by 66% in fewer than one-third, and monitoring was never used by 5% of survey respondents. 39
If neuro-monitors were available, our survey revealed that these monitors were most often used to avoid awareness. However, it has not been proven that neuro-monitors can detect intraoperative awareness.40,41 As suggested by approximately 40% of respondents, the proven value of these monitors is to avoid unnecessarily deep anesthesia, which might lead to burst suppression and increase the risk of POD.42–44 However, it is crucial to mention that simple monitoring using machine-generated indices may be insufficient to prevent burst suppression, particularly older patients in whom the values underestimate the DOA. 45 Recent studies have shown that these indices should not be used without expert visual analysis of the alpha-band EEG trace.45–47
Regarding POD treatment, the guideline recommends non-pharmacological measures be used first. 1 This was also mentioned in the survey (5.9% of respondents) and included ensuring the involvement of family, rest in a comfortable and soothing environment with cognitive stimuli, as well as psychotherapy and communication with the patient, which represent the framework of self-healing capacity.1,15,48 Furthermore, our survey revealed that experienced anesthesiologists (5 years or more) tended to use individualized treatment more frequently than less experienced anesthesiologists (fewer than 5 years). If a non-pharmacological approach alone is unsuccessful, specific agents mentioned for POD treatment by respondents were dexmedetomidine, propofol, benzodiazepines, haloperidol, droperidol, antipsychotics, and antagonists in general. The guideline suggests treatment with a well-titrated dose of haloperidol (titrated to a maximum of 3.5 mg) or low-dose atypical neuroleptics. Benzodiazepines are not routinely recommended except in cases of withdrawal, and propofol and antagonists are not mentioned. 1 With respect to alpha-agonists, the guideline suggests perioperative application when undergoing cardiac or vascular surgery. 1 Clonidine can be considered preoperatively in high-risk patients, as well as perioperatively. 1 Dexmedetomidine has been investigated in several recent studies analyzing its use in POD.49,50 The findings showed that prophylactic intraoperative infusion of dexmedetomidine does not prevent delirium.51,52 However, when used after a surgical procedure, studies have revealed a positive effect on POD.8,53
HELP is a new model of care designed to prevent functional and cognitive decline in older people during hospitalization. Proposed in 2000 by Inouye, 54 HELP has been proven very effective in preventing cognitive and functional decline in at-risk older patients. Patients are screened for risk factors on admission and if they are found to be at risk, patients are visited by HELP staff who are assisted by trained volunteers. Examples of intervention protocols include orientation, sleep enhancement, oral volume repletion, feeding assistance, therapeutic activities, early mobilization, and vision and hearing improvement. The program was modified in 2014 to better suit the needs of the perioperative environment, and HELP remains an ongoing research topic. 55
Limitations of this survey are that the target population was limited in distribution and the response rate could have been higher. A low response rate might be owing to a lack of interest or time owing to work overload among anesthesiologists; therefore, efforts should be made to encourage more anesthetists to take part in future surveys. The response rate in surveys is usually unpredictable.9,56 Anesthesiologists who are interested in POD will be more likely to take part in a survey; therefore, our results may be biased based on this consideration. In addition, as this survey only addressed clinical practitioners, a discrepancy between observed practice and perceived practice must be taken into account. 57
In conclusion, the present survey revealed that most anesthesiologists in China are aware of the importance of POD. Collaborative educational approaches of the ESA and national societies might be used to further implement POD screening and POD prevention and treatment tools. Close interdisciplinary collaboration might enhance POD screening after surgery to improve postoperative cognitive outcomes, 9 especially in patients with hypoactive delirium, which accounts for most cases of POD, 58 to overcome Steiner’s so-called burden of delirium. 59
Footnotes
Acknowledgements
The authors are very grateful to Professor Li-Ze Xiong (Immediate Past President of the Chinese Society of Anesthesiology) and Professor Dong-Xin Wang (Current President of the Chinese Association of Perioperative Organ Protection, CSCTVA) for their generous help and suggestions on this work.
Authors’ contributions
CS and MW conceived performance of the survey. SD, BW, BN, and CS helped to design the questions. SD and BN analyzed the data. SD prepared the draft of the manuscript. All authors contributed to the revision of the manuscript.
Congresses
Preliminary data for this study were presented as a poster at the Capital Congress of the German Society of Anaesthesiology and Intensive Care Medicine for Anesthesiology and Intensive Care Therapy with Nursing Symposium and Rescue Service Forum on 20 September 2018 in Berlin.
Declaration of conflicting interest
The authors certify that there is no conflict of interest with respect to any financial organization regarding the material discussed in the manuscript, except for the task force, who received travel grants for guideline development. BW received personal fees from Orion Pharma Ltd. and personal fees from Dr. Franz Köhler Chemie GmbH outside the submitted work. CS received funding from the European Union-funded Seventh Framework research program [FP7/2007-2013], under grant agreement no. HEALTH-F2-2014-60246, BioCog. Dr. Spies received grants from Dr. Franz Köhler Chemie GmbH, grants from AppAdventure, grants from J&P Medical Research Ltd., grants from Pharmaceutical Research Associates GmbH, grants from Delcath Systems, Inc, grants from INC Research UK Ltd., grants from La Jolla Pharmaceutical Company, grants from Aridis Pharmaceutical Inc., grants from B. Braun Melsungen AG, grants from Drägerwerk AG & Co. KGaA, grants from Grünenthal GmbH, grants from Infectopharm GmbH, grants from Sedana Medical Ltd., grants from Arbeitsgemeinschaft industrieller Forschungsvereinigungen, “Otto von Guericke” e. V. (AiF)/German Federation of Industrial Research Associations, grants from Berufsverband Deutscher Anästhesisten e. V. (BDA)/Professional Association of German Anaesthesiologists, grants from Deutsche Forschungsgemeinschaft/German Research Society, grants from Deutsches Zentrum für Luft- und Raumfahrt e. V. (DLR)/German Aerospace Center, grants from Einstein Stiftung Berlin/Einstein Foundation Berlin, grants from the European Society of Anaesthesiology, grants from Gemeinsamer Bundesausschuss/Federal Joint Committee (G-BA), grants from Inneruniversitäre Forschungsförderung/Inner University Grants, grants from Projektträger im DLR/Project Management Agency, grants from Stifterverband/Non-Profit Society Promoting Science and Education, grants from the World Health Organization Collaborating Centre, grants from Westfälische Wilhelms-Universität Münster/DFG, personal fees from B. Braun Melsungen AG, grants from AbbVie Deutschland GmbH & Co. KG, grants from Aguettant Deutschland GmbH, grants from B. Braun Melsungen, grants from Dr. F. Köhler Chemie GmbH, grants from MAQUET Vertrieb und Service Deutschland GmbH, grants from Orion Pharma GmbH, grants from Philips Electronics Nederland B.V., grants from Sintetica GmbH, grants from Baxter Deutschland GmbH, grants from Biotest AG, grants from Cytosorbents Europe GmbH, grants from Edwards Lifesciences Germany GmbH, grants from Fresenius Medical Care, grants from Grünenthal GmbH, grants from Masimo Europe Ltd., grants from Medtronic GmbH, grants from Pfizer Pharma PFE GmbH, personal fees from Georg Thieme Verlag, grants from the European Commission, and grants from BMUB (Bundesministerium für Umwelt, Naturschutz und nukleare Sicherheit), outside the submitted work. In addition, Dr. Spies has patent 10 2014 215 211.9 pending, patent application no. PCT/EP2015/067730 pending to Graft Gesellschaft von Architekten mbH, and patent application no. PCT/EP2015/067731 pending to Graft Gesellschaft von Architekten mbH.
Funding
This work was supported by institutional grants and an ESA grant for guideline development (CS, chair of the guideline task force).
