Abstract
Guidelines for prophylactic antibiotic administration in total joint replacement vary considerably in terms of drug, dosage, route of administration and duration of cover. Despite the range of treatment options available, infection remains the most common reason for arthroplasty failure in the decades following a procedure, simultaneously increasing health care costs and lowering patient satisfaction considerably. This work aims to evaluate whether there are benefits to administering further doses of antibiotic post-arthroplasty, in addition to the recommendations of current protocols. We present a review of evidence surrounding infection rates in a variety of prophylactic regimens, and weigh this against further considerations such as cost to the patient and risks of nephrotoxicity. In summary, the available evidence does not suggest a benefit to administering additional doses post-arthroplasty in most cases. However, further doses may benefit those deemed at high risk of infection, or those in areas of high methicillin-resistant Staphylococcus aureus prevalence.
Introduction
It is widely accepted that the use of prophylactic antibiotics leads to a reduced rate of infection post-arthroplasty (Holtom 2006, Ponce et al 2014, Smith et al 2012). Despite virtually ubiquitous antibiotic use, prosthetic joint infection (PJI) remains the primary reason for arthroplasty failure in the 25 years following total joint arthroplasty (TJA; Myers et al 2020). Large retrospective studies on TJA show PJI rates between 0.9% and 2.5% (Kheir et al 2017, Koh et al 2017, Lindeque et al 2014), depending on factors including the antibiotic administered and the time of administration (Classen et al 1992).
Minimising infection rate is a pertinent issue, as PJI is the most common cause of revision surgery (Koh et al 2017). Further treatment inconveniences patients and may reduce patient satisfaction (Kay et al 1983). Additional treatments following a diagnosis of PJI may include further antibiotic courses, debridement and, in some cases, amputation (Windsor & Bono 1994). PJI also has financial implications: in UK NHS trusts between 2006 and 2009, it was estimated that the cost of inpatient and day cases alone was approximately £33,000 greater for those experiencing PJI, compared to those who did not, in the five years following treatment (Garfield et al 2020).
This review will assess the effectiveness of the current antibiotic administration regimens for arthroplasty in terms of the number of doses administered, and examine available literature to determine whether revision of these protocols should be pursued.
Discussion
Current treatment protocols
Current recommendations
Prophylactic antibiotic protocols vary between regions, even within countries, likely due to evidence for the effectiveness for a wide range of treatments, as well as regionally relevant concerns about prevalence of resistant organisms (Aujla et al 2013). However, only seven causative organisms are responsible for 89% of the infections affecting both hip and knee arthroplasties, which does not justify the widespread variation in treatment approaches (Hickson et al 2015).
Regional variation is often influenced by local policy makers. For example, there is currently no consensus on timing, dose or choice of antibiotic prophylaxis in the United Kingdom (Graham et al 2020). Therefore, protocols for administration of antibiotics for arthroplasty differ between NHS trusts.
Globally, there is some agreement on the duration of administration, with 80% of the countries sampled in a review by Parsons et al (2022), recommending no more than 24 hours of coverage in TJA. However, this study focused on high-income countries, meaning that this may not truly represent international practice.
Generally, recommendations suggest that prophylactic antibiotic administration is initiated preoperatively within an hour of skin incision and before tourniquet inflation, and continued for not more than 24 hours (Holtom 2006).
More stringently, Centers for Disease Control and Prevention guidelines from 2017 also suggest that no postoperative antibiotics should be administered for clean and clean-contaminated surgery (Berríos-Torres et al 2017) including TJA (Myers et al 2020). National Institute for Health and Care Excellence guidelines reflect this, suggesting that antibiotic prophylaxis is only administered once preoperatively (Leaper et al 2008, NICE Guidelines 2020).
One drug frequently listed in TJA protocols is the first-generation cephalosporin, cefazolin. Cefazolin use has demonstrated a significantly reduced rate of PJI following hip and knee arthroplasties, compared to several non-cefazolin antibiotics (including vancomycin and clindamycin), with increasing benefit as time increases from surgery (Wyles et al 2019). In contrast, a retrospective review found that vancomycin prophylaxis administration alone, as opposed to cefazolin, led to a significantly decreased rate of infections (Smith et al 2012). Importantly, when considering additional dosages, there was no difference in overall infection rate when vancomycin was administered in addition to cefazolin in patients who had undergone TJA (Ponce et al 2014).
Alternative choices include flucloxacillin and gentamicin. A study by Torkington et al (2017) showed that in hip replacements, flucloxacillin (2g intravenous (IV) dose) achieved femoral and tibial concentrations which met the stringent epidemiological cutoffs (ECOFFs) for resistance for the common infectious pathogens Staphylococcus epidermidis and Staphylococcus aureus in 100% of hip arthroplasties and 95% of knee arthroplasties. The single case in which this threshold was not met may have been confounded by tourniquet inflation shortly after administration. Gentamicin (3mg/kg IV) reached the ECOFF standards much less frequently, however. Finally, cefuroxime (1.5g IV) was the preferred regimen for antibiotic prophylaxis in elective hip and knee arthroplasty in 30% of NHS trusts in 2013 (Hickson et al 2015).
Efficacy of alternative administration practices
There are several routes of antibiotic administration, which offer surgeons alternative delivery options when optimising dosing regimens (Figure 1). Intraoperative administration allows for intraosseous delivery of antibiotics. This has been found to reduce periprosthetic joint infection in follow-up of knee arthroplasty by up to seven fold (0.22% versus 1.4%), when using a tibial infusion of 0.5g vancomycin + a first-generation cephalosporin, compared to IV vancomycin at 15mg/kg + a first-generation cephalosporin (Park et al 2021). However, these data were retrospective and across multiple surgeons, leaving room for variability in the administration technique. Nevertheless, this demonstrates the importance of considering route of administration when optimising dosing.
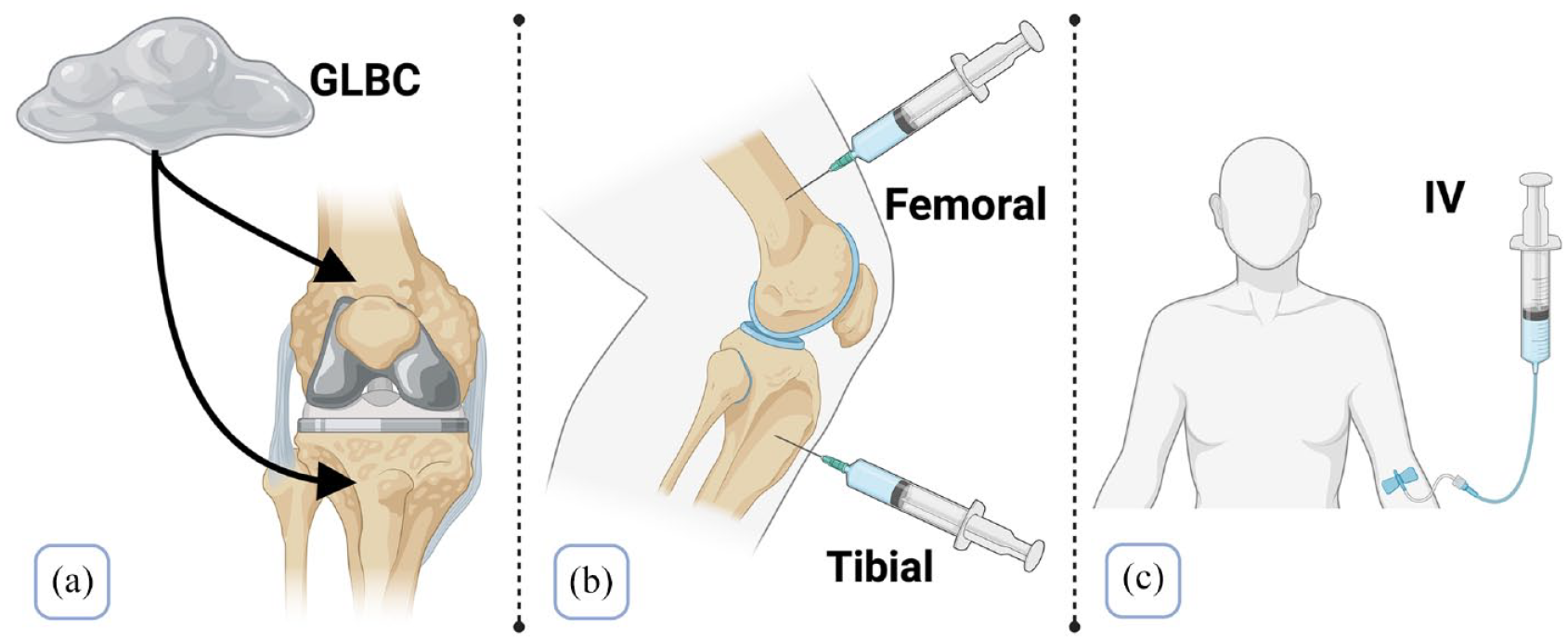
Different antibiotic delivery methods: (a) gentamicin-loaded bone cement (GLBC) – used to anneal the prosthetic implant to bone, (b) intraosseous delivery – antibiotics can be administered into the distal femur or the proximal tibia and (c) intravenous (IV) administration.
Similarly, Chang et al (2013) found that gentamicin-loaded bone cement (GLBC) led to extended durations of antibiotic release relative to other antibiotics, while providing a broad spectrum of coverage against bacterial classes. Further evidence shows a significant reduction in infections when using GLBC (0.8%), compared to systemic antibiotics (1.9%) in the five years following the operations (Josefsson et al 1990).
Timing of onset: Postoperative versus preoperative administration
In isolation, dosing postoperatively, compared to preoperatively or perioperatively (within three hours after incision), leads to higher rates of infection in general surgical procedures (3.3% versus 0.6% versus 1.4%; Classen et al 1992). This was reflected in the finding that there are no benefits of administering systemic antibiotics more than three hours post-incision, while the most effective time is pre-incision (Burke 1961).
For IV administration, current methods aim to have the greatest concentration at the time of incision. Hence, dosing should be started between 30 and 60 minutes prior to incision for optimum concentration (Galandiuk et al 1989, Gyssens 1999).
Current dosages may be inefficient
Dosing for TJA prophylaxis antibiotic regimens is often weight determined. Cefazolin is given in 2g doses for patient body masses between 60 and 120kg, and 3g for masses greater than 120kg. Alternatively, a 1500mg dose of the second-generation cephalosporin cefuroxime can be administered, while vancomycin, given in doses of 15mg/kg (up to 2g), may be used in cases of β-lactam antibiotic allergy or in instances of high risk of methicillin-resistant Staphylococcus aureus (MRSA; Myers et al 2020).
However, administering larger IV boluses in larger patients may not be more efficacious in reducing infection rates. Administration of 2g versus 1g of IV prophylactic cefazolin produced a greater concentration in the distal femur and proximal tibia, but Angthong et al (2015) found no significant correlation between concentration and inhibitory effect on bacterial growth at either of these locations. Kheir et al (2017) also found no significant difference in infection rate when patients were ‘underdosed’ or ‘overdosed’ according to weight administration guidelines; however, the confidence in this finding was limited by low study power. Surgeons should consider plateauing antibiotic effects when optimising dosing regimens.
Evidence surrounding extended dosing regimens
Evidence against the postoperative administration of extra doses
Administration of multiple antibiotic doses is common in TJA, despite there being little evidence for this approach (Dellinger et al 2015).
Procedures on American veterans found increasing duration of antibiotic administration led to no improvement in the incidence of surgical site infection rate (Branch-Elliman et al 2019). In addition, there was increased risk of acute kidney injury (AKI), as well as increased risk of Clostridium difficile infection. Looking at the data holistically, 502 out of the 38,675 patients (1.3%) who underwent joint arthroplasty experienced surgical site infection, across a range of prophylactic antibiotic administration durations. This is within the range reported by other studies where one dose was administered (Tan et al 2019, Tang et al 2003), suggesting that additional doses had little effect. However, as analysed by Myers et al (2020), this study did not detail exclusion of data in the event of an infection, which could underlie some of the prolonged antibiotic regimens, and may confound the authors’ findings.
Two randomised controlled trials studying the efficacy of additional doses of cefuroxime failed to find any significant difference when increasing the number of doses post-hip arthroplasty (Ritter et al 1989, Wymenga et al 1992). Wymenga et al (1992) compared the efficacy of one- and three-dose cefuroxime regimens; infection rates of 0.83% and 0.45% were not significantly different.
There was no significant difference in deep infection rates by two years post-operation between a single dose of cefazolin given at anaesthetic onset, compared to three doses of cefuroxime in TJA (Pulido et al 2008). These data were specific to operations less than four hours in length, and in which no intraoperative antibiotic was given. The study is limited by its follow-up period, as PJIs have been diagnosed more than four years post-operation. Despite this, the rate of PJI observed in the single-dose cefazolin group is similar to one- and three-day dosing regimens seen in other arthroplasty studies (Williams & Gustilo 1984; <1%), suggesting that further doses of cefazolin may be ineffective. Supporting this, a large prospective cohort study by Badge et al (2022) found no significant difference in surgical site infection when applying cephalosporin for greater or less than 24 hours. However, this study too was limited in its one-year follow-up period.
These comparisons demonstrate the relative scarcity of data on infection following TJA, where the number of prophylactic doses is the independent variable and the identity of the antibiotic is controlled. There has been a failure to find superiority of multiple doses in other surgical procedures however, such as the treatment of closed long-bone fractures (Slobogean et al 2008), as well as an extra dose of cefazolin in general surgical procedures compared to the more-regularly recommended single dose (Scher 1997).
Evidence supporting extra doses: Reduced rates and MRSA
Evidence for administration of extra doses may be provided by small-scale studies which show lower rates of infection but cannot reliably prove significance due to a lack of power. For example, a study on cefuroxime use conducted by Wymenga et al (1992) failed to find significance in the 0.38% reduction in infection rate difference in the 12 months following arthroplasty, possibly due to its modest patient population (n = 2651). Despite the lack of significance, the study recommended the administration of three doses until more data on the longer term effects and larger cohort sizes could be produced. Due to the already high success rate of antibiotic application intraoperatively, or preoperatively, it may be that significant reductions in PJI with additional doses only present across very large patient populations.
Certain patients are at increased risk of infection, and so surgeons could be tempted to increase dosing in an effort to improve protection. For example, end-stage renal failure leads to an increased rate of infection post-TJA, with an odds ratio of 7.54 (Erkocak et al 2016). Similarly, malignancy produced an odds ratio of 3.1 relative to controls (Berbari et al 1998).
Those afflicted by autoimmune conditions are at greater risk of infection following joint arthroplasty. Those with rheumatoid arthritis experienced a hazard ratio of 4.08 relative to those not afflicted in a matched cohort study (Bongartz et al 2008).
Diabetic and morbidly obese patients have a significantly higher risk of PJI, with approximately five times and nine times greater risk of infection, respectively: 2.6% versus 0.49% (Dowsey & Choong 2008, Iorio et al 2012). Recent analysis however failed to show a significant difference in PJI for extended antibiotic administration for morbidly obese patients compared to controls (Carender et al 2021).
A summarising study on high-risk patients led to 1% and 2.2% 90-day infection rates post-total knee arthroplasty (TKA) and post-total hip arthroplasty, respectively, in a group administered seven-day prophylactic oral antibiotics. These were 4.9 and four times lower than in control groups of high-risk patients not offered the extended treatment (Inabathula et al 2018). The patients were classified as high risk due to smoking, obesity, autoimmune disease, chronic kidney disease and positive nasal MRSA swabs, providing evidence for further antibiotic use in many cases.
There is some evidence that increasing doses could be beneficial in arthroplasty when the further doses are of a different drug. For example, a study by Tornero et al (2014) found that using teicoplanin in combination with two doses of cefuroxime, as opposed to two doses of cefuroxime alone, led to a significantly reduced rate of infection: 3.51% as opposed to 1.26%. The use of teicoplanin is indicated in the cases of MRSA or for allergy to penicillin. This study found a significantly reduced rate of staphylococcal infections in the group also administered teicoplanin (0% versus 1.6%).
Similarly, vancomycin combined with cefazolin led to a significant decrease in the number of staphylococcal infections (Sewick et al 2012). Contrastingly, a retrospective review found that in TJA, vancomycin prophylaxis alone, as opposed to cefazolin, significantly decreased the rate of both total infections and specifically MRSA infections (Smith et al 2012). These findings warrant further consideration of additional dosing in areas with a high prevalence of MRSA.
Effects of the duration of a procedure:
For long procedures, an extra dose of antibiotic is advised. This is when the procedure duration lasts one to two times the half-life of the antibiotic, or when the procedure is associated with extensive blood loss (Hanssen & Osmon 1999). This is because clearance of the antibiotic within the patient’s body post-administration, or blood loss, means that the antibiotic is at a sub-effective concentration.
Infection rates for general surgical procedures more than three hours led to 6.1% rate versus 1.3% at less than three hours, using cefazolin (short half-life). However, using a single preoperative dose of cefotetan (longer half-life) led to a similarly reduced infection rate. The suggestion is therefore that a single dose is sufficient for short procedures, but another dose is needed for longer procedures or switching to a long-half-life antibiotic (Scher 1997).
Due to the relatively short durations of hip and knee arthroplasty operations (Dicks et al 2015), the need for additional doses would be limited to a fraction of cases, perhaps due to difficult anatomy, or surgical complications.
Further considerations for extended dosing regimens
Patient compliance
A disadvantage to protracted dosing regimens is a reduction in patient compliance. In a prospective study on lumbar fusion, extended antibiotic regimens were reported to be the causative reason behind a reduction in patient compliance. Interestingly, the same study found no difference in the infection rate of the compliant and non-compliant patients, given that all had had a prophylactic dose (Rodríguez-Caravaca et al 2014). Longer dosing regimens may therefore result in an unnecessary expenditure on behalf of the patient or health care provider.
Antibiotic stewardship
Antibiotic stewardship programmes aim to balance immediate benefits to an individual patient with the long-term population goal of reducing the rate of development of antibiotic-resistant strains. It is therefore important to dose optimally, to avoid contributing to microbial resistance. When considered alongside poor patient compliance, the chances of promoting multi-drug-resistant strain development should be considered with extended dosing.
Wasting resources
Balanced against the cost of treating an infection is the cost of administering unnecessary antibiotics. Individual doses of cefazolin and vancomycin can cost $12 and $30, respectively. Although these costs will vary between institutions, the millions of TJA operations performed annually (Maradit Kremers et al 2015) could make additional doses a significant financial burden (Tan et al 2019).
Gut microbiota
The spotlight is increasingly being shone on antibiotic use due to the effects on the microbiome of the gut, which is becoming implicated in an ever-increasing range of conditions, with the overall aim to reduce the use of unnecessary oral antibiotics in aid of its protection (Iizumi et al 2017).
Kidney injury
AKI is a serious side effect of antibiotic administration following many hospital procedures, and is associated with increased mortality and duration of hospital stay (Chertow et al 2005). As mentioned in the study on the American veterans, there was an increased risk of AKI when prescribed protracted regimens of antibiotics post-TJA (Branch-Elliman et al 2019). The addition of vancomycin to a cefazolin regimen post-TJA has also been shown to significantly increase the risk of AKI in patients (13% versus 8%), without evidence of significantly reducing infection rates (Courtney et al 2015).
The rate at which AKI occurs will vary with the antibiotic of choice, so surgeons must weigh the appropriate risk against any benefits of additional doses. Antibiotics with known nephrotoxic functions such as aminoglycosides should be used with caution in extended dosing regimens: four 2g doses of flucloxacillin as well as a single dose of gentamicin (1.5mg/kg) led to an AKI rate of 13% following TKA, compared to four 2g doses of flucloxacillin alone, which led to an AKI rate of 8.5% (Graham et al 2020).
Surgeons should also be conscious of appropriate dosing if the patient has previously established renal dysfunction. Cefazolin is mainly cleared by the glomerulus and has much reduced clearance in patients with nephrotic dysfunction (Rein et al 1973).
Conclusion
The current evidence suggests that extra doses post-surgery are not needed for most patients; there is limited evidence for any benefit, and rates of post-surgical infection are generally already very low.
When the limited scope for benefits is weighed against additional costs and potential for kidney injury to the patient, extra doses seem an unlikely recommendation.
Optimising other parameters including type of antibiotic, time of initiation of antibiotic administration, route of administration and body-weight-dependent dosing formulae may allow surgeons to reduce infection rates further, while maximising convenience for patients.
There is evidence that additional doses of antibiotics are beneficial in certain patients who are at high risk of infection. There is also evidence that MRSA infections can be reduced with the addition of vancomycin or teicoplanin to standard dosing regimens. Furthermore, extra doses may also be needed for elongated procedures, for example in the occurrence of surgical complications.
This review highlights the need for larger studies of higher power which can determine significance of even slight benefits of further doses. Finally, this review highlights the need for studies for optimising current guidelines, assessing alternative administration practices such as intraosseous delivery and antibiotic-laden bone cement, as well as the optimal drug doses and durations for patients undergoing TJA.
Key points
Joint infection remains the most common reason for arthroplasty failure in the 25 years following the procedure.
Prophylactic protocols vary widely in terms of drug, dose and duration, likely due to evidence for effectiveness of a wide range of treatments.
Randomised controlled trials failed to find a significant reduction in infection rates post-hip arthroplasty when additional doses of cefuroxime were used.
A significant reduction in MRSA infection rate has been observed with the addition of teicoplanin to cefuroxime in at-risk populations.
Potential downsides to extended dosing regimens must also be considered, including AKI and further costs to patients.
