Abstract
Despite advances in management strategy, traumatic brain injury remains strongly associated with neurological impairment and mortality. Management of traumatic brain injury requires careful and targeted management of the physiological consequences which extend beyond the scope of the primary impact to the cranium. Here, we present a review of the principles of its acute management in adults. We outline the procedure which patients are assessed and the critical physiological variables which must be monitored to prevent further neurological damage. We describe current interventional strategies from the context of the underlying physiological mechanisms and recent clinical data and identify persisting challenges in traumatic brain injury management and potential avenues of future progress.
Keywords
Introduction
Traumatic brain injury (TBI) encompasses a diverse collection of intracranial injuries following impact to the cranium. This review aims to highlight the current perioperative guidelines in the United Kingdom with regard to the management of the adult patient with TBI and secondary prevention of further damage. TBI is the most prominent cause of morbidity and mortality in people aged one to 40 in the United Kingdom, and had an aggregate cost of £5.1 billion in the United Kingdom in 2010, with an average cost per case of £11,340 (Parsonage 2016). Damage sustained from TBI can be grouped into primary injuries, including haematomas and traumatic axonal injury (TAI) sustained due to the initial trauma and secondary injuries which develop some time after, ranging from minutes to days. TBI may be viewed as a progressive injury, with optimal patient recovery contingent upon reducing the magnitude of the secondary insult.
Incidence
The Global Burden of Diseases, Injuries and Risk Factors (GBD) study has estimated the age-standardised worldwide incidence of TBI to be 369 per 100,000 people in 2016 (James et al 2019). However, the true consequences of TBI extend beyond its incidence and can also be considered from the perspective of disease burden, which can be assessed using the measures of years of lost life (YLLs) and years lived with disability (YLDs). The GBD study (James et al 2019) reported that TBI was the cause of an estimated 8.1 million YLLs in 2016. Consistent with its high global incidence, TBI remains one of the commonest causes of death and disability in the United Kingdom. Data from NICE (2014) have indicated that 1.4 million people annually from England and Wales attend emergency departments with acute head injury.
A crucial part of public health prevention strategies is to reduce the risk of incurring the initial trauma. Prevention strategies identified by the Lancet Neurology Commissions on TBI (Maas et al 2022) include attention to road traffic safety, fall prevention in the elderly and implementation of measures to mitigate risks in contact sports.
However, once the injury has occurred, the clinical focus is on maintaining metabolic normality for surviving cerebral tissue, while seeking to prevent further secondary injury. Here we present the principles of acute management of TBI and the perioperative considerations.
Acute management
The acute management phase of TBI is crucial to stabilising the patient and preventing any secondary brain injury resulting from physiological insults such as ischaemia, reperfusion injury and hypoxia (Moppett 2007).
Patient assessment
Initial patient assessment is carried out in accordance with Advanced Trauma Life Support (ATLS) guidelines. An important component of the assessment is the Glasgow Coma Score (GCS) to estimate the extent of injury and provide a rough approximation of morbidity and mortality risk of the patient (Moppett 2007). Full cervical spine immobilisation should be performed for patients with any of the following risk factors: GCS less than 15 on initial assessment, neck pain or tenderness, focal neurological deficits, paraesthesia in the extremities and/or any other clinical suspicion of cervical spine injury (NICE 2019). In patients with reduced consciousness (GCS equal to or less than eight), there should be early involvement of an anaesthetist or critical care physician to establish a secure airway (eg: endotracheal intubation) (NICE 2019).
Endotracheal intubation is considered the gold standard, but it is essential to assess the benefits, risks and timing of this on a case-by-case basis. For example, patients who are unable to maintain SpO2 > 90% with supplemental oxygen, who are agitated and who are suspected of having cerebral herniation where maintaining adequate oxygenation is a concern, should also be considered for intubation (Sönmez 2022). While anaesthetic agents allow for quick control of the airway and modulation of intracranial pressure (ICP) through CO2 control, many are also potent vasodilators resulting in an undesirable drop in mean arterial pressure (MAP) (Dinsmore 2013). To maintain cerebral perfusion pressure (CPP), care must be taken to maintain the MAP during induction of anaesthesia, largely through the use of accompanying vasoconstrictors, such as metaraminol. Therefore, the need to induce anaesthesia must be balanced against the risk of decreasing blood pressure.
Effective pain management is also essential as uncontrolled pain may lead to a rise in ICP. The patient should be provided with reassurance if conscious, any fractures splinted and pain managed with small doses of intravenous opioids titrated against clinical response and baseline cardiorespiratory measurements recorded (NICE 2019).
The time elapsed before intervention is a major determinant of outcome (Trivedi et al 2022). Major Trauma Networks operate to ensure that the three overlapping phases of the patient’s journey (pre-hospital, in-hospital and post-hospital) are well-organised, so that patients are treated at the time and place which has the greatest benefit for them (Moran et al 2018).
Imaging
Computed tomography (CT) is the core imaging modality in acute TBI. Early imaging helps to detect serious life-threatening abnormalities, allowing for an appropriate treatment plan to be developed. The following guidelines are the criteria recommended by NICE for performing CT imaging in TBI (NICE 2019) (see Table 1).
This table outlines the risk factors used to determine the time interval within which a CT scan should be performed.

CT: computed tomography; TBI: traumatic brain injury; GCS: Glasgow Coma Score.
Management of raised ICP
As the brain is contained within a non-expandable case of bone, the Monro-Kellie doctrine describes the sum of the volumes of the cerebrospinal fluid (CSF), intracranial blood and CSF must be constant, such that an increase in one necessitates a decrease in either or both of the other two (Mokri 2001). An increase in ICP can therefore impair cerebral blood flow and cause secondary ischaemia (Haider et al 2018) (see Figure 1).
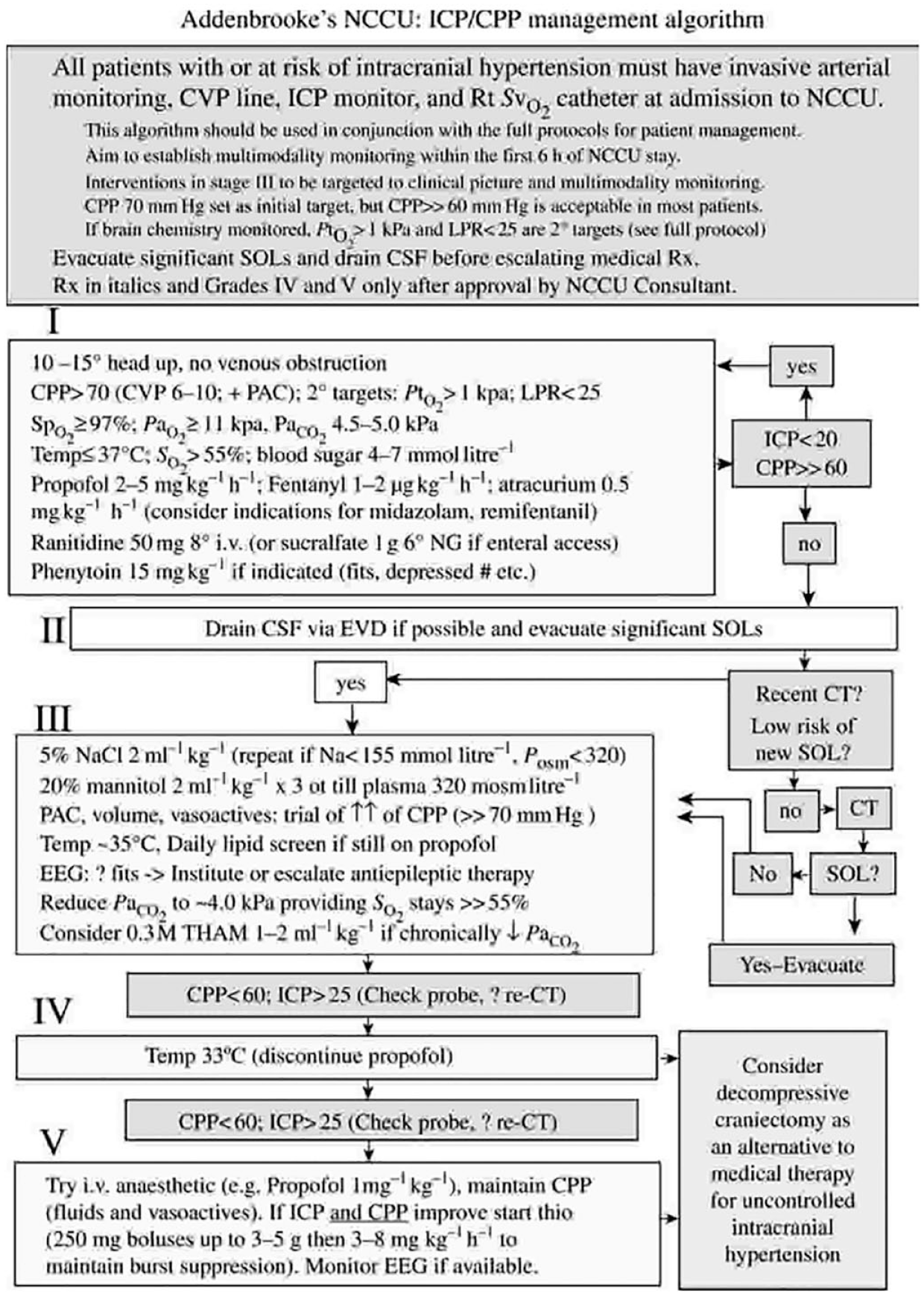
An example of an ICP/CPP management algorithm used by Addenbrooke’s Hospital, Cambridge
ICP monitoring remains the mainstay of cerebral monitoring in these patients. A range of interventions exist to manage elevated ICP, with evidence that standardised protocols improve management and outcomes.
Head elevation
Raising the patient’s head reduces ICP by displacing CSF from the intracranial department and promoting venous outflow. The head of the bed should be raised to 30°, with the patient’s head in a midline position to avoid compression of the internal jugular vein (Schizodimos et al 2020). Cerebral blood flow is unaffected (Feldman et al 1992). Head elevation above 45° should be avoided to prevent paradoxical increases in ICP in response to a fall in cranial perfusion pressure (CPP) (Moraine et al 2000). Care is taken in the polytrauma patient, particularly with spinal fracture(s), to protect other organ systems. In this instance, a reverse Trendelenberg position can be used to maintain spinal alignment, while still allowing the head to be raised.
Hyperventilation
Hyperventilation refers to an elevated minute alveolar ventilation, which decreases the intraarterial carbon dioxide partial pressure (PaCO2), resulting in cerebral vasoconstriction and a consequent decrease in ICP (Grubb et al 1974). PaCO2 levels which rise above normal (5.2kPa) result in cerebral vasodilatation and an increase in ICP. PaCO2 values which are uncontrolled may increase the risk of cerebral haemorrhage (Deng et al 2020).
However, hyperventilation is also associated with numerous adverse effects. If PaCO2 levels fall below 4.2kPa, cerebral vasoconstriction can start to limit cerebral perfusion, while hyperventilation has also been associated with electrolyte imbalances, such as hypokalaemia and hypocalcaemia (Godoy et al 2017). Furthermore, hyperventilation may cause respiratory alkalosis, increasing the oxygen affinity of haemoglobin and reducing oxygen delivery to peripheral tissues (Curley et al 2010). These explain why this intervention is not recommended prophylactically and is typically reserved for brief periods of acute neurological dysfunction in severe TBI.
Hyperosmolar therapy
Intermittent boluses of a hyperosmolar agent such as mannitol lower ICP via two distinct mechanisms (Dinsmore 2013, Peters et al 2018). First, the formation of an osmotic gradient across the blood-brain barrier causes water to move from the parenchyma into the intravascular space. Second, mannitol reduces blood viscosity, thereby promoting plasma expansion. Due to autoregulation, plasma vasoconstriction occurs, decreasing cerebral blood volume.
However, mannitol is also associated with a range of adverse effects, such as arterial hypotension and rebound of cerebral oedema (Shi et al 2020). Therefore, hypertonic saline is increasingly being used instead (Gu et al 2018).
Hypothermia
Inducing hypothermia reduces cerebral metabolic demands. Selective brain cooling can be achieved by using a cooling cap, while systemic cooling can be achieved with a cooling blanket or endovascular catheters. Treatment outcomes may be related to the time taken to reach the goal temperature, when the cooling began and the duration of cooling (Finkelstein & Alam 2010). Some studies suggest that longer periods of cooling may be associated with more favourable outcomes, as cerebral swelling and oedema are greatest three to five days after injury (Fox et al 2010). However, the adverse effects associated with hypothermia, such as coagulation disturbances, may counteract its positive effects, and thus there is lack of support for the use of therapeutic hypothermia as a first-tier treatment (Sandestig et al 2014).
Glycaemic control
Hyperglycaemia is associated with poor outcomes following TBI (Jeremitsky et al 2005). Hyperglycaemia may arise as a consequence of the catecholamine surge and may be further exacerbated by the administration of exogenous steroids and catecholamines. Monitoring is required to ensure blood sugar levels are maintained between 4mmol/L and 8mmol/L (Dash & Chavali 2018).
The importance of blood glucose monitoring was illiustrated in a seminal single-centre study by Van de Berghe (2001) which showed that intensive glycaemic control reduced mortality in the surgical intensive care unit (ICU) setting. However, subsequent randomised controlled trials (RCTs) identified that hypoglycaemia was also associated with a substantial increase in mortality (Godoy et al 2017). Therefore, patients with TBI are particularly sensitive to both hyperglycaemia as well as hypoglycaemia.
Neurosurgical interventions
Surgical intervention may be required to evacuate an epidural or subdural hematoma. The local mass effect of hematomas may cause an increase in ICP and clinical signs such as mental status changes, dilated pupils or extensor posturing consistent with brainstem herniation. Subdural hematomas which are over 10mm in size or causing more than 5mm midline shift may be considered for operative intervention (Fomchenko et al 2018).
A craniectomy is a further surgical procedure which can be used to reduce ICP. This involves the removal of part of the skull, which allows the brain to swell without increasing the ICP. Options for the repair of the cranial defect include the use of the previously removed bone flap or the use of a variety of synthetic implants. While there is still no consensus on the timing of the repair, some institutions report performing this after at least three months to allow the swelling to subside (Martin et al 2014).
Fluid intervention and blood pressure management
Secondary brain injury may arise from episodes of hypotension after TBI and are associated with worse patient outcomes. To maintain cerebral perfusion and intravascular volume, intravenous fluids are commonly administered, with the choice traditionally being 0.9% saline, as it is relatively isotonic to plasma. Isotonic solutions do not increase brain water content as they do not change plasma osmolality and distribute freely in the extracellular fluid compartment (Alvis-Miranda et al 2014). However, a systematic review and meta-analysis found that 0.9% saline and low-molecular weight hydroxyethyl starch were associated with lower mortality than other intravenous solutions, such as balanced crystalloid solutions (Tseng et al 2020). In addition, hypertonic crystalloids may be commonly used in patients with cerebral edema and raised ICP.
Vasopressor therapy is also often administered in combination with intravenous fluid administration. Although noradrenaline is most commonly used, there has been concern that noradrenaline infusion may result in hypoperfusion to other organs as a consequence of excessive vasoconstriction. A systematic review was unable to show a significant benefit associated with noradrenaline infusion in patients with TBI (Lloyd-Donald 2020).
Seizure management
Seizures typically happen where there is tissue damage that arises as a consequence of the injury (Englander et al 2014). The tissue damage results in scarring, which alters astrocyte physiology and can cause neurotransmitter imbalance (Robel 2016). Seizure risk is reduced by the use of anti-epileptic medications in the first seven days (Wiles 2022). Although phenytoin is commonly used, it is associated with several side effects which include liver enzyme induction, cardiac arrhythmias and hypotension. Due to its low incidence of adverse effects, levetiracetam has become increasingly popular, with a meta-analysis demonstrating that levetiracetam showed similar efficacy to phenytoin in patients with TBI (Fang et al 2021).
Hypoxia
With cessation of blood flow, the intracellular production of adenosine triphosphate is reduced. The subsequent reduction in primary active transporter ion channels results in the intracellular accumulation of sodium ions and cytotoxic edema. Furthermore, ischaemia also promotes the release of glutamate, which activates N-methyl-
In contrast to the well-known association between hypoxia and adverse outcomes in TBI, the influence of hyperoxia is less certain. A systematic review demonstrated that hyperoxia was not associated with an increase in mortality (Ni et al 2019). It is reasonable to tolerate a degree of hyperoxia after TBI to allow adequate oxygen reserves for unanticipated events, especially in light of the established dangers of hypoxia to the injured brain.
Recent progress and challenges in TBI management
In 2017, the Lancet Neurology published its first commission on TBI (Maas et al 2022). This drew attention to the largely unrecognised public health challenge caused by TBI and called for a concerted approach to better understand its origins and mechanisms. Since then, there have been several large-scale observational studies, such as CENTRE-TBI and TRACK-TBI, which highlighted prognostic factors such as frailty, comorbidities and alcohol misuse (Lecky et al 2021, Steyerberg et al 2019).
There is the need to develop new prediction tools to stratify TBI patients for more personalised care (Chinnery 2022). A potential new approach could be to measure novel CSF and serum biomarkers which could, in the future, become easy-to-use biochemical tests. While there has clearly been progress over the past five years, many advances are yet to achieve routine clinical implementation. Avenues of future research could involve identifying subgroups of patients which would be most likely to benefit from certain interventions and developing greater understanding of the effects of genetic variation on the biology of TBI to facilitate individualised management.
Conclusion
The management of TBI poses many challenges, as TBI does not a comprise a single disease entity, but rather a collection of discrete disease types. Despite a diverse arsenal of management strategies, TBI continues to be associated with neurological impairment and mortality, with an estimated half of the global population experiencing at least one TBI during their lifetime (Young & Hughes 2020). The wide range of treatment strategies further emphasises the complexity of treating TBI, especially as the specific context of each injury and patient variability must be taken into account. Despite improving and evolving management strategies, the global burden of TBI is increasing, so health care systems must continue to adapt (Feigin et al 2019).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
