Abstract
Therapeutic hypothermia is often used for traumatic brain injury because of its neuroprotective effect and decreased secondary brain injury. However, this procedure lacks clinical evidence supporting its efficacy, and adverse outcomes have been reported during general anesthesia. A 61-year-old man with a history of percutaneous coronary intervention (PCI) was admitted with traumatic brain injury. Immediately after admission, he underwent mild therapeutic hypothermia with a target temperature of 33.0°C for neuroprotection. During general anesthesia for emergency surgery because he developed a mass effect, hypothermic cardiac arrest occurred following an additional decrease in the core body temperature. Moreover, myocardial infarction caused by restenosis of the previous PCI lesion also contributed to the cardiac arrest. Although the patient recovered spontaneous circulation after an hour-long cardiopulmonary resuscitation with rewarming, he eventually died of subsequent repetitive cardiac arrests. When anesthetizing patients undergoing therapeutic hypothermia, caution is required to prevent adverse outcomes that can be caused by unintentional severe hypothermia and exacerbation of underlying heart disease.
Keywords
Introduction
Therapeutic hypothermia is an attractive strategy for traumatic brain injury (TBI), which demonstrates neuroprotective effects and decreases secondary brain injury. 1 Despite the lack of clinical evidence regarding efficacy, therapeutic hypothermia after TBI has been used for at least half a century because of its potential benefits, which are supported by numerous preclinical studies.2,3 However, there have been no reports on the danger of unintentional severe hypothermia during general anesthesia caused by the additional drop in temperature of patients undergoing therapeutic hypothermia. When anesthetizing patients with comorbidities, such as ischemic heart disease, caution is required to prevent adverse outcomes that can be caused by unintentional severe hypothermia and exacerbation of underlying heart disease.4,5
Case report
A 61-year-old man (height, 168 cm; weight, 70 kg) presented to our emergency room (ER) with head trauma after falling on a cement floor from a 5-meter-tall construction site. Upon arrival at the ER, the patient had a Glasgow coma scale score of 11 and the following vital signs: blood pressure (BP), 147/83 mmHg; heart rate (HR), 86 beats per minute (bpm); respiratory rate, 24 breaths per minute; body temperature from the auditory canal, 36.8°C; and percutaneous oxygen saturation (SpO2), 97%. Upon discovering signs of acute subdural hemorrhage, subarachnoid hemorrhage, and intraventricular hemorrhage from a brain computed tomography (CT) scan performed in the ER, we admitted the patient to the intensive care unit (ICU) for close monitoring. After approximately 6 hours, we performed follow-up brain CT because the patient’s mental status had deteriorated from drowsy to stupor. Upon observing a mass effect characterized by an increase in the hemorrhage volume and a left midline shift on the CT images, the patient was scheduled for immediate intubation and emergency decompressive craniectomy (Figure 1).
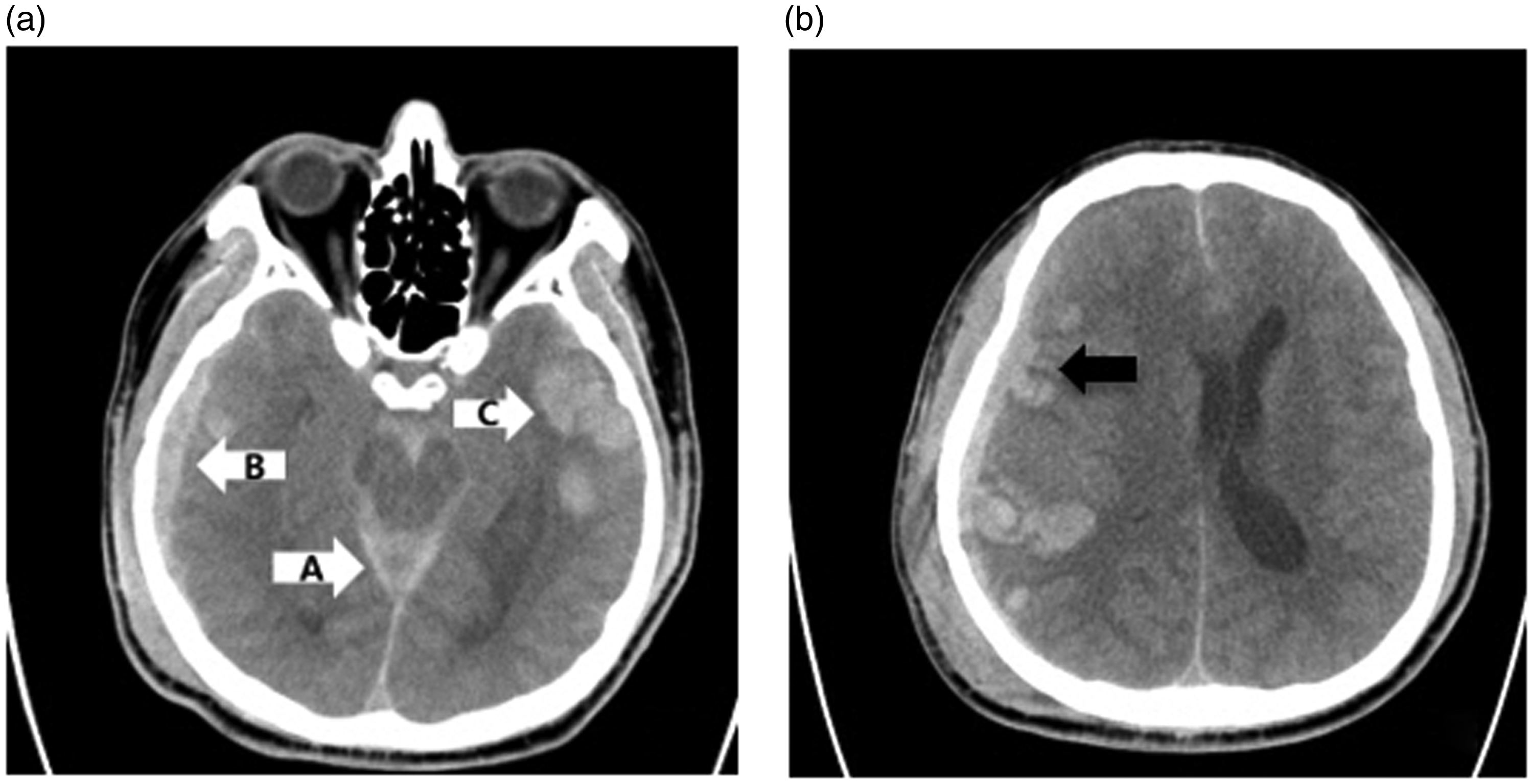
Brain computed tomography (CT) images taken 6 hours after intensive care unit admission. (a) Axial view showing acute subarachnoid hemorrhage in the peri-mesencephalic cistern (arrow A) and subdural hemorrhage in the right inferior temporal (arrow B) and right parietal (arrow C) regions. (b) Axial view showing acute subdural hemorrhage in the right temporal region (black arrow) and left midline shift.
Although the patient had a history of ischemic heart disease with percutaneous coronary intervention (PCI), it was difficult to obtain accurate information because there was no relevant data in the patient’s medical records from the authors’ hospital, and his drowsy state interfered with detailed interviews. According to the chest CT scan performed immediately after admission, coronary stents had been inserted at the bifurcation site of the left main coronary artery (Figure 2). It also appeared that the inserted stents were drug-eluting stents (DES) considering that he had received dual antiplatelet therapy (DAPT) with aspirin and clopidogrel (Plavix®; Sanofi, Bridgewater, NJ, USA) a switching platelet P2Y12 inhibitor, for approximately 4 months. However, DAPT was discontinued owing to the worsening cerebral hemorrhage after hospitalization; bridging antiplatelet therapy was also not performed. Before the surgery, the patient showed a normal electrocardiogram (ECG), and cardiac biomarker testing revealed a troponin-T concentration of 0.006 µg/L, which was within normal limits (Figure 3). The patient demonstrated satisfactory functional capacity to be able to work at a construction site just before the trauma. Therefore, an additional preoperative cardiac evaluation was not considered necessary.

Chest computed tomography (CT) images showing coronary stents in the main bifurcation of the left coronary arteries (white arrow in (a) and (b)).

Preoperative electrocardiogram of the patient showing normal sinus rhythm with a heart rate of 95 bpm.
Immediately after admission to the ICU, the patient’s bladder temperature was 38.0°C. Using a hydrogel-coated water-circulating pad (Arctic Sun®; Medivance Inc., Louisville, CO, USA), which is a surface cooling device, the patient underwent mild therapeutic hypothermia with a target temperature of 33.0°C. During therapeutic hypothermia, sedation and shivering control were performed using continuous rate infusions of midazolam at 5 mg/hour and remifentanil at 0.2 µg/kg/minute. The patient’s bladder temperature was 33.4°C just before he was transferred to the operating room. Preoperative laboratory tests showed no unusual findings except for mildly elevated aspartate aminotransferase (73 U/L) and alanine aminotransferase (51 U/L) concentrations.
The patient’s vital signs upon arrival at the operating room were a BP of 130/70 mmHg and SpO2 of 100%. Lead II of the ECG was monitored, and there were no unusual findings except sinus tachycardia with an HR of 100 bpm on the initial ECG. Arterial BP (ABP), central venous pressure (CVP), and esophageal temperature were monitored from the right radial artery, right subclavian vein, and lower third of the esophagus using a temperature sensor attached to the esophageal stethoscope, respectively. The initial ABP, CVP, and esophageal temperature were 140/85 mmHg, 6 mmHg, and 33.0°C, respectively. Arterial blood gas analysis was performed in the arterial line, with values calculated as for a body temperature of 37°C, which revealed the following: pH, 7.400; partial pressure of carbon dioxide (PCO2), 40 mmHg; partial pressure of oxygen (PO2), 174 mmHg; bicarbonate (HCO3), 24.8 mmol/L; base excess, 0.0; lactate, 0.17 mmol/L; ionized calcium, 1.04 mmol/L; and potassium, 3.3 mmol/L. Total intravenous (IV) anesthesia was performed using a target-controlled infusion of propofol and remifentanil in accordance with the Schnider 6 and Minto 7 models, respectively. Propofol and remifentanil were infused continuously with effect-site concentrations (Ce) of 1.5 µg/mL and 1.5 ng/mL, respectively. Concurrently, 50 mg of rocuronium was administered by IV bolus injection for neuromuscular blockade. Mechanical ventilation was adjusted to maintain the end-tidal carbon dioxide value at 30 to 33 mmHg.
As ABP decreased to 78/42 mmHg and CVP decreased to 4 mmHg within 10 minutes after anesthesia, the Ce of propofol and remifentanil were lowered to 0.8 µg/mL and 1.0 ng/mL, respectively, and approximately 1 L of balanced crystalloid fluid loading was performed. However, the patient’s BP did not recover, and a dopamine continuous infusion (5 µg/kg/minute) was initiated. When a surgical incision was made 10 minutes after beginning the dopamine infusion, the patient’s ABP and HR increased to 115/70 mmHg and 120 bpm, respectively, and marked ST-segment depression (−2.4 mm) was found, which was not observed on the preoperative or initial ECGs (Figure 4). Subsequently, the depth of anesthesia was increased by increasing the Ce of propofol to 1.0 to 1.2 µg/mL, followed by a continuous infusion of isosorbide dinitrate (1 µg/kg/minute). While HR was maintained at 110 to 120 bpm, ABP gradually decreased to 70/40 mmHg after approximately 5 minutes. To treat the hypotension, isosorbide dinitrate was discontinued, and a bolus injection of 100 µg phenylephrine was administered, after which, ABP increased temporarily to 95/60 mmHg. Subsequently, HR decreased suddenly and rapidly, and ventricular fibrillation occurred. The anesthesiologists immediately discontinued the surgery and performed cardiopulmonary resuscitation (CPR). The patient’s esophageal temperature at the time was 32.0°C.

Intraoperative electrocardiogram of the patient showing sinus tachycardia with significant ST depression. The ST depression on the monitor represented approximately −2.4 at the time.
Chest compressions were performed with periodic defibrillation with 200 J, and epinephrine injections were administered every 3 minutes; these measures elicited no response. During CPR, ECG monitoring showed temporary severe bradycardia (HR, 10–30 bpm). However, this was considered pulseless electrical activity because no pulse was detected in the patient’s femoral artery. Soon afterward, there was a rapid transition to ventricular fibrillation, and CPR was repeated. At the time, the patient’s esophageal temperature dropped to 30.0°C. According to the refractory state after the defibrillation and drug injections, we considered that the cardiac arrest was caused by hypothermia. To reverse the cause, a forced-air warming device and a warming blanket (Bair Hugger®; Arizant Healthcare Inc., Eden Prairie, MN, USA) were used for surface warming, the IV fluids were warmed, and surgical wound closure and dressing were performed rapidly to prevent surface heat loss. The first arterial blood gas analysis after ventricular fibrillation, calculated as for a body temperature of 37°C, showed the following: pH, 7.690; PCO2, 15 mmHg; PO2, 260 mmHg; HCO3, 18.1 mmol/L; base excess, −1.2; lactate, 0.33 mmol/L; ionized calcium, 0.88 mmol/L; and potassium, 5.0 mmol/L. Follow-up arterial blood gas analyses were repeated periodically. As we continued to perform CPR, the patient developed severe metabolic acidosis and hypocalcemia characterized by a pH of 6.840, HCO3 of 15.4 mmol/L, and ionized calcium of 0.66 mmol/L. To correct the abnormal findings, we administered appropriate amounts of sodium bicarbonate and calcium chloride. After an hour-long CPR and rewarming, the patient’s esophageal temperature recovered to 33.0°C, which was followed by observing sinus rhythm characterized by an HR of 75 bpm on an ECG monitor and detecting a pulse in the femoral artery with a characteristic ABP waveform that eventually led to the recovery of BP to 130/55 mmHg. Upon confirming recovery of spontaneous circulation (ROSC), the patient was transported to the ICU while we administered dopamine (10 µg/kg/minute), norepinephrine (0.1 µg/kg/minute), and vasopressin (2 U/hour). ABP, HR, and esophageal temperature during anesthesia are shown in Figure 5.

Changes in vital signs (blood pressure (BP), heart rate (HR), and esophageal temperature) over time (5-minute intervals).
Cardiac biomarker testing performed after the patient’s transfer to the ICU showed an increased troponin-T level of 1.432 µg/L. Portable Doppler transthoracic echocardiography (TTE) revealed total occlusion of the left main coronary artery, which had undergone PCI previously. Because emergency coronary artery bypass grafting (CABG) was impossible at the time, repeated PCI was considered an alternative treatment option. However, even after his transfer to the ICU, the patient experienced repetitive ventricular fibrillation. Four hours after the patient’s transfer to the ICU, the patient’s guardian requested that CPR should be stopped. Without resuscitation, the patient died.
Discussion
In this case, the patient, who was receiving therapeutic hypothermia, underwent emergency craniectomy owing to worsening brain hemorrhage, and cardiac arrest occurred during general anesthesia. We deduced that the cardiac arrest was owing to the additional decrease in the patient’s core body temperature and myocardial infarction caused by restenosis of the previous PCI-treated lesion.
Therapeutic hypothermia is used to improve neurological outcomes in cardiac arrest, TBI, stroke, and hypoxic–ischemic encephalopathy.1,4 Studies on the efficacy of therapeutic hypothermia for TBI have been less convincing, and they show conflicting results. However, hypothermia is consistently used as an option for TBI because it reduces intracranial pressure and the cerebral metabolic rate, thereby protecting neuronal function in the brain and decreasing the wide range of injuries or reparative processes by attenuating temperature-sensitive mechanisms, such as excitotoxicity, free radical generation, apoptosis, and inflammation.1,2
The cardiovascular effects associated with hypothermia are complex. Hypothermia affects the electrical conduction of myocardial cells, which can induce hypotension or bradycardia and even increase the risk of ventricular fibrillation by prolonging repolarization.4,8 However, mild hypothermia of 32.0°C to 35.0°C demonstrates cardioprotective effects by decreasing myocardial oxygen demand and increasing myocardial perfusion through vasodilation of coronary vessels. 9 Therefore, there has been a growing interest in the clinical use of mild therapeutic hypothermia as an adjunctive therapy during ischemic heart disease. 10 Our patient received mild therapeutic hypothermia with a target core temperature of 33.0°C, which may have yielded cardioprotective as well as neuroprotective benefits. 11
During general anesthesia, hypothermia is induced following an increase in heat loss caused by peripheral vasodilation after the injection of anesthetic drugs, heat redistribution from the core to the periphery, and impaired normal thermoregulatory responses, such as vasoconstriction or shivering. 5 In our patient, the cold operating room temperature and irrigating an uncovered surgical area in the brain with cold saline also may have contributed to the additional core body temperature drop in addition to the effects of general anesthesia. 12 However, in our case, neurosurgeons requested that anesthesiologists not perform warming during anesthesia. Instead, esophageal temperature and signs that could result from hypothermia, such as ECG changes, were carefully monitored. The esophageal temperature decreased from 33.0°C, which was measured immediately after entering the operation room, to 32.0°C at the time of cardiac arrest. The esophageal temperature further decreased to a minimum of 30.0°C during CPR. With rewarming, the temperature recovered to 33.0°C and finally, ROSC was achieved. Considering that chest compressions, drugs, and defibrillation did not induce ROSC, which was achieved with the recovery of an esophageal temperature of 33.0°C, the additional rapid fall in the patient’s core body temperature during anesthesia may have caused the cardiac arrest.
In this case study, we suspected that restenosis at the site of the previous stent insertion contributed to the cardiac arrest, given the following three findings: First, the patient had a normal preoperative ECG with no signs of ischemia but developed ST-segment depression during anesthesia. Second, repetitive cardiac arrests occurred even after achieving ROSC with rewarming. Third, an increased troponin-T concentration was observed, and suspected re-stenotic occlusion of the left main coronary artery was confirmed by Doppler TTE after the patient’s transfer to the ICU. Accordingly, myocardial infarction was considered the main cause of death.
The mechanisms of myocardial infarction owing to stent restenosis after DES insertion are rapid stent thrombosis or profuse in-stent restenosis (ISR). 13 We assume that the lesion characteristics in the left main bifurcation of the coronary artery and DAPT interruption owing to the increasing amount of cerebral hemorrhage contributed to the stent restenosis in this patient. As evidence in support of this assumption, previous studies have shown that stenting at the left main bifurcation and DAPT interruption are strong risk factors for stent thrombosis or ISR.14,15 While controversial, not interrupting aspirin perioperatively could have avoided poor outcomes. 16 However, there were no clinical signs related to expected ongoing restenosis of the coronary artery or myocardial ischemia preoperatively in our patient. We suspect that the possible ischemic signs may have been masked by the aforementioned cardioprotective benefits of therapeutic hypothermia. We also believe that the following three conditions contributed to the intraoperative myocardial ischemia and infarction caused by acute restenosis of the coronary stent: First, intraoperative hypotension may have contributed to myocardial injury. The IV anesthetics used during anesthesia, propofol and remifentanil, induced redistribution of blood volume by peripheral vasodilation, resulting in profound hypotension. 4 Profound hypotension was strongly associated with myocardial injury and especially harmful for this patient, who had preexisting ischemic heart disease. 17 Second, tachycardia, which may have been caused by the inotrope dopamine, used to compensate for the low BP or the stress response to surgical stimulation from shallow anesthetic depth, may have triggered myocardial ischemia by decreasing oxygen delivery and increasing oxygen consumption.17,18 If cardiogenic shock is suspected, as in our case, norepinephrine could be a better option than dopamine. 19 Third, the bolus injection of phenylephrine, a selective α1-adrenergic agonist, just before cardiac arrest may have temporarily and abruptly increased cardiac afterload and impaired myocardial perfusion. 20
Had we known that coronary artery occlusion would result from hypothermia-induced cardiac arrest and coronary stent restenosis, the treatment of choice would have been CABG surgery while maintaining circulation with emergency cardiopulmonary bypass (CPB) or extracorporeal membrane oxygenation (ECMO) via the femoral artery and vein. 11 However, it was difficult to determine the etiology of the cardiac arrest at the time. Even if we had known the cause, ideal treatment would have been impossible, given circumstances beyond our control; all thoracic surgeons were busy operating, and emergency CPB and ECMO were not available. As an alternative, repeat PCI after the patient achieved ROSC could have been performed. However, the patient had a prolonged CPR duration, and he did not maintain the ROSC state for a period sufficient for intervention, which led to a poor outcome.
In this case, it was not possible to accurately determine the reason for the cardiac arrest and for not achieving consistent ROSC. Instead, we conclude that cardiac arrest developed and persisted as a result of the unintentional severe hypothermia that occurred during general anesthesia and/or coronary artery restenosis at the site of the previous PCI. A thorough evaluation of heart-related history is required for TBI patients who have a history of ischemic heart diseases and who undergo therapeutic hypothermia because there may be undiscovered coronary insufficiency owing to the cardioprotective effects of therapeutic hypothermia. In addition, careful monitoring of the patient’s core body temperature, and preventive and countermeasures should be fully prepared and taken against adverse hemodynamic effects, even cardiac arrest, which can occur because of additive hypothermic responses during anesthesia.
Footnotes
Ethics statement
This case report was approved by our Institutional Review Board (EMC 2020-05-005-001), and the case report was performed in compliance with the EQUATOR Network guidelines (the CARE guidelines). All patient details were de-identified, and written consent was obtained from the patient’s guardian for treatment and publication of this report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
