Abstract
The aim of the present study was to simultaneously audit hormonal contraceptive (HC) use, menstrual cycle characteristics and training/nutrition related profiles in pre-menopausal women from varying athletic and exercise backgrounds. Elite (n = 51), sub-elite (n = 118) and amateur (n = 392) female athletes and exercisers were examined via an anonymous quantitative/qualitative survey tool. All analyses for ratio data were conducted utilising one- and two-way ANOVA/ANCOVA and odds ratio models, with ordinal data analysed via Pearson's Chi-squared tests. HC use was similar across elite, sub-elite and amateurs (34–44%). Menstrual cycle length was not different (P = 0.08) between competitive levels (28 ± 13 days), but 66% of respondents reported cycle variability (10 ± 11 days). Training profiles were not different based on contraceptive status (P > 0.05) yet were across competitive groups (P < 0.05). Daily meal/snack intakes were not different between contraceptive status (P > 0.05), though elite and sub-elite groups reported a higher daily meal consumption compared to amateurs (P < 0.01). Forty percent of all respondents skipped meals, attributed to lack of time, schedule and alterations in appetite, with reported changes in taste preferences during pre-menses (14–35%) and menses (15–25%) towards sweet foods (60%), with cravings for chocolate and other confectionary (25%). Prevalence of dietary supplement use was not influenced by contraceptive status (P = 0.31), though elites (76%) reported higher use (P = 0.04) than amateurs (63%). Data demonstrates that in female athletes and exercisers, competitive level appears to affect simple markers of training and nutrition practice, yet contraceptive status does not.
This is a visual representation of the abstract.
Introduction
Between the years of 2014 and 2020, 63% of studies published within leading sport and exercise science related journals included mixed sex cohorts, with 31% including only males and 6% solely females within their study design. 1 This obvious sex data gap across the sport and exercise sciences is often attributed to the methodological challenges of conducting carefully controlled research studies within female populations. Indeed, the need to consider variations in endocrine profiles between hormonal contraceptive (HC) and non-HC users, as well as control for fluctuations in reproductive hormones throughout the menstrual cycle, can substantially increase the cost of conducting such research and require specialist knowledge and expertise, therefore lengthening testing schedules.2–4 Although a recent meta-analysis concluded that exercise performance may be trivially reduced in the follicular phase of the menstrual cycle, 5 the authors specifically noted that our current understanding is significantly limited by the large number of low quality investigations and between study variations in methodological rigor. Additionally, a recent special edition on Nutrition for Female Athletes also highlighted the need for a much larger scope of female-specific sport nutrition research, given the existing body of evidence does not presently support nutritional recommendations that are sex specific or that appear modifiable throughout the menstrual cycle. 6 Furthermore, the uniquely individual contexts that each female may experience throughout the menstrual cycle has also been demonstrated to influence these areas.7–11 On this basis, it may therefore be premature to suggest evidence-based training and/or nutritional guidelines that can be tailored according to phases of the menstrual cycle or types of HC.12–14
HCs can be administered in numerous forms inclusive of oral contraception (OC), hormonal intrauterine devices (global terminology) or systems (United Kingdom terminology) (IUD/S), injections, implants, vaginal rings and transdermal patches. These methods deliver various doses of exogenous steroid hormones that induce a suppressed level of endogenous sex steroid hormones and therefore inhibit ovulation. 15 In 2019, the United Nations commissioned a worldwide report highlighting that across 1.9 billion women of reproductive age (15–49 years), 922 million used various forms of contraception, of which 407 million (43%) were users of differing forms of HC, including IUD/S (17%), OC (16%), injections (8%) and implants (2%). 16 The prevalence of HC use amongst athletic populations appears marginally higher than these general trends, whereby in a study by Martin and colleagues published in 2018, 50% of elite athletes reported currently using HCs. 15 Despite such prevalence, it is noteworthy that the effects of HCs on the physiological/metabolic responses of exercise performance and the associated impact of nutritional interventions are less frequently studied when compared to the research literature examining the effects of the menstrual cycle.6,17,18 Therefore, the use of HCs and their potential associated effects on reproductive hormones, further complicates the notion of prescribing a one size fits all approach to training and nutritional practices in athletic and exercising females.
Previous audits of menstrual cycle characteristics and HC use amongst female athletes have often focused on elite cohorts.8,15,19,20 As such, the applicability of this data to other athletic and exercising female groups (i.e. sub-elite, amateur and recreationally active) is unclear, particularly if elite athletes exhibit an increased prevalence of HC use in an attempt to manipulate and control their menstrual cycle around sporting schedules. 20 Additionally, it is possible that other factors intended to support daily training schedules (e.g. nutritional habits and dietary supplement use) may also vary between groups of females with differing training and/or competitive demands. In this regard, it could be postulated that HC status itself may also modulate dietary behaviours, given that fluctuations of ovarian hormones across the menstrual cycle can result in altered energy intake, taste sensitivities and cravings.21–26
To that end, the aim of the present study was to simultaneously audit HC use, menstrual cycle characteristics and training/nutrition related profiles in pre-menopausal women of varying athletic and exercise backgrounds. On this basis, we hypothesised that both contraceptive status and competitive level would affect these profiles. Therefore, the goal of the study was to identify any potential areas for future education and behaviour change, alongside ascertaining which of these factors could potentially be generalised around the menstrual cycle and HC use.
Methods
Participants
Participants were recruited via dissemination of the study information throughout the authors’ professional networks and social media platforms inclusive of Twitter, Facebook and Instagram in a random sampling approach. In order to capture individuals of post menarche and pre-menopause, inclusion criteria stated an age range of 18 to 40 years and those completing a minimum of 3 hours of exercise per week. Participants self-classified their current sport and/or exercise ranking as either professional, international, semi-professional, national, local or recreational, whilst also self-reporting current contraceptive status (both within the past 12 months). Inclusion criteria also stipulated those individuals who classified themselves as professional, international, semi-professional or national sporting levels, must be recognised and/or licensed as a member of a professional sporting organisation and/or national governing body. Participants who indicated HC use were subsequently grouped separately to non-HC users. Sport and/or exercise ranking were collated into competitive levels, with participants competing at professional and/or international standard classified as an ‘elite’ category, semi-professional/national as ‘sub-elite’ and local/recreational as ‘amateur’. A total of 607 individuals participated within the study, with 561 data sets included within the final analysis. Individual respondents who fell outside of the inclusion criteria and/or had incomplete data sets were excluded (see Figure 1). The validity of all groupings was cross-referenced against the Participant Classification Framework, 27 with the elite category being confirmed at Tier > 4, the sub-elite Tier 3 and amateur category a combination of Tier 2 (n = 266) and Tier 1 (n = 126). Finally, participants were also further sub-grouped into sport type including aesthetic, combat, endurance, multi, strength/power, team and other events, with an additional general category within the amateur group, however, some smaller group numbers did not allow further analysis. All participants provided electronic informed consent prior to starting the survey and the study was approved by the research ethics committee of Liverpool John Moores University (Protocol code: U20_SPS_1071).
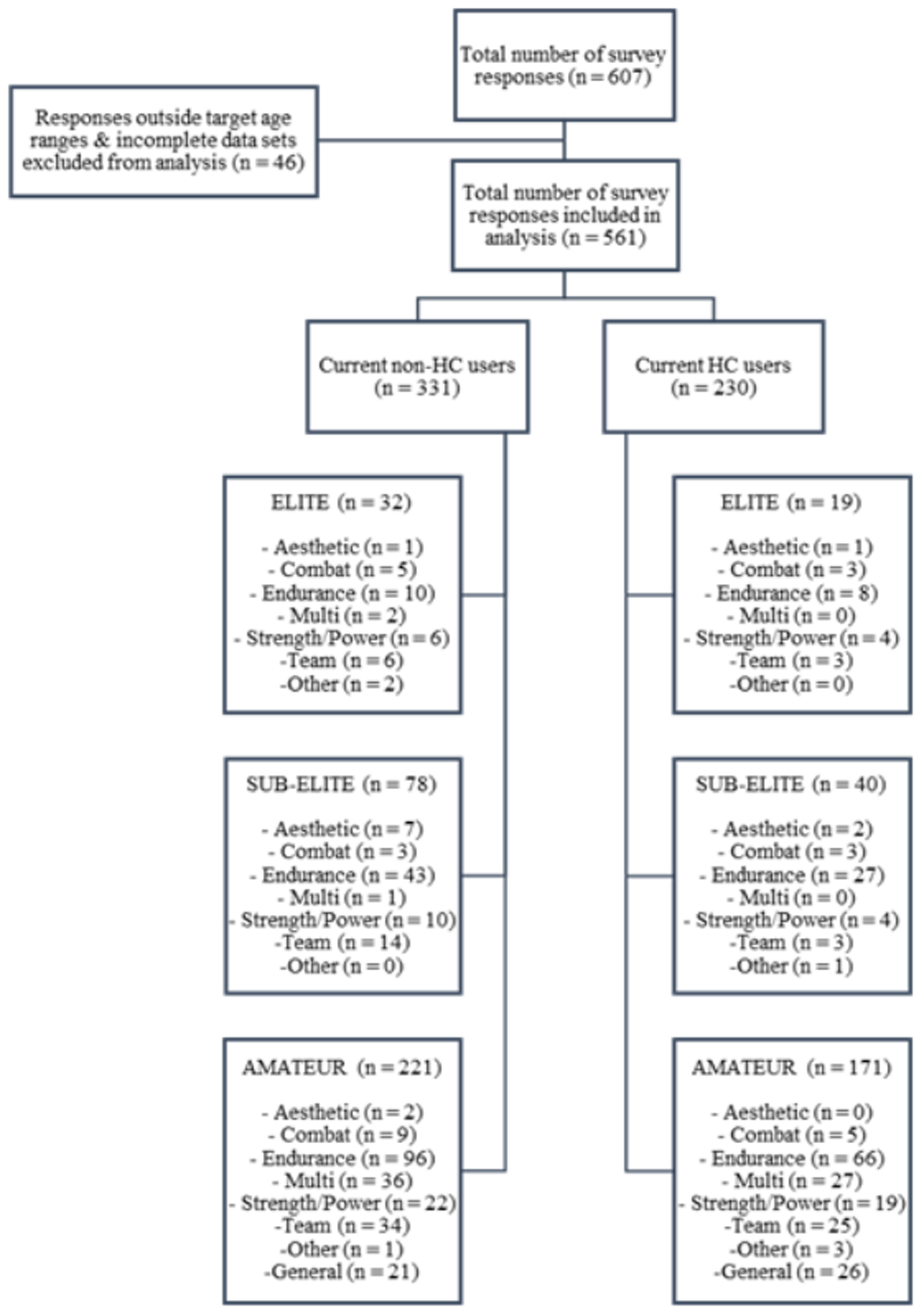
The prevalence of current non-HC and HC users, competitive status and sporting background within the survey respondents (HC: hormonal contraceptive users; non-HC: non-hormonal contraceptive users).
Survey tool
Data were collected using a specifically designed questionnaire administered via an anonymous and combined quantitative/qualitative survey tool. The survey included items from two previously published studies, which were employed on similar populations to those stipulated within the inclusion criteria.15,20 The survey was communicated in English and transposed into an electronic version hosted on the encrypted onlinesurveys.ac.uk platform (Jisc, Bristol, UK). This permitted participation on several types of electronic devices inclusive of laptops, tablets and mobile telephones, whilst also affording a world-wide geographical reach. Dependent on responses, the questionnaire took approximately 20–30 minutes to complete, and access was active between November 2020 and January 2021. The survey collated a number of sets of quantitative data including current general information (i.e. age, stature, body mass, body mass index, sport and/or competitive level), training history and profile (i.e. training years, number of training sessions per week, typical minutes per training session, weekly training volume), current (within the past 12 months) HC use/type or menstrual cycle characteristics (i.e. cycle occurrence, length, variability, days of variability and tracking, self-reported and medically diagnosed instances of secondary amenorrhea and oligomenorrhoea, cycle effect on and avoidance of training/exercise, use of treatments/medications to alleviate menstrual symptoms) and typical nutritional profile (i.e. frequency for daily occasions of meal and snack intake, skipping meals, dietary supplement usage, perceived changes in taste preferences and food cravings across the menstrual or HC cycles). Questions related to training and nutritional practices stipulated responses that were provided retrospectively, based on the specific HC or menstrual cycle phase of each individual participant. Responses were collected in open-ended numeric formats and rating scales, for example, do you use nutritional supplements (i.e. for health and/or sport performance); yes or no? Additionally, qualitative responses stipulating further open-ended worded detail on HC, menstrual cycle characteristics and nutritional profiles were included, for example, which nutritional supplement(s) do you use (i.e. type and/or brand)?
Statistical analyses
Descriptive statistics inclusive of mean ± SD, range, 95% confidence intervals (95% CI), odds ratio (OR) and frequency are provided for data where appropriate. All analyses were performed using SPSS® version 28 (IBM® United Kingdom Ltd, Hampshire, UK) with the alpha level set at P < 0.05. Initially, a univariate one-way ANOVA exploring menstrual cycle length and days of variability as the dependent variable and competitive level as the independent variable was conducted. All subsequent analyses were examined with contraceptive status and competitive level as independent variables. A univariate two-way ANOVA was employed to compare several dependent variables relating to participant characteristics (age, stature, body mass, body mass index). Furthermore, univariate two-way ANCOVA's were also utilised to compare dependent variables of participant training histories and profiles (training years, number of training sessions per week, typical minutes per training session, weekly training volume), with both age and body mass index included as covariates within the models. Data within all ANOVA and ANCOVA analyses were explored for normality via histograms, box plots were examined for outliers and a Levene's test was employed to assess homogeneity of variances. During any violations of these assumptions, data were log transformed and post hoc testing for multiple pairwise comparisons was assessed with Fisher's least significant difference. Ordinal regression models to generate OR were also used, with number of meal and snack occasions as outcomes. Pearson's Chi-squared test was employed to cross tabulate frequency percentages for HC type, menstrual cycle occurrence, length, variability and tracking, self-reported and medically diagnosed instances of secondary amenorrhea and oligomenorrhoea, cycle effect on and avoidance of training/exercise, use of treatments/medications to alleviate menstrual symptoms, daily occasions of meal and snack intake, skipping meals, dietary supplement usage, perceived changes in taste change preferences and food cravings across the menstrual or HC cycles. Finally, qualitative data were assessed via content analysis utilising data matrices, in order to identify the most common phrase responses, which were then ordered into frequencies. 28
Results
Overview of respondent demographics
An overview of respondent demographics and training history/profiles is displayed in Table 1. The survey was represented by 9% elite, 21% sub-elite and 70% amateur participants. When categorising the total sample by contraceptive status, 41% were current HC users and 59% non-HC users, with the division of HC and non-HC users being 37% to 63% in elite, 34% to 66% in sub-elite and 34% to 56% in amateur competitive levels, respectively. In relation to age and anthropometrical profile, the sub-elite group were younger (P < 0.01; 95% CI = -4 to 1 years) and greater in stature (P = 0.01; 95% CI = 0.6–3.7 cm) than the amateur group. Although body mass was not different between competitive levels (P = 0.80), the sub-elite group displayed a lower body mass index than the amateur group (P = 0.02; 95% CI = 0.2–1.3 kg·cm2).
Respondent's demographic and training profiles within the elite, sub-elite and amateur cohorts from both HC and non-HC users.
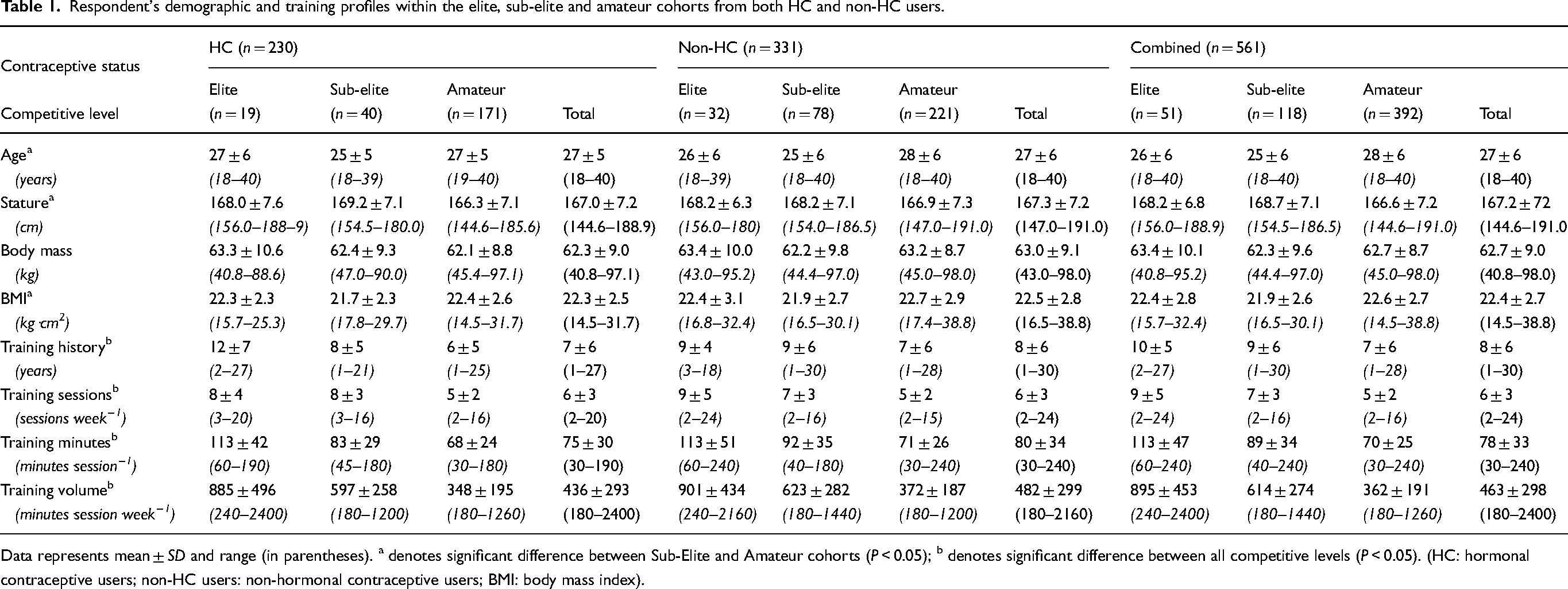
Data represents mean ± SD and range (in parentheses). a denotes significant difference between Sub-Elite and Amateur cohorts (P < 0.05); b denotes significant difference between all competitive levels (P < 0.05). (HC: hormonal contraceptive users; non-HC users: non-hormonal contraceptive users; BMI: body mass index).
Between competitive levels, distinct differences were reported in training history and profiles across cohorts, whereby the elite group reported longer training history (*P = 0.04; 95% CI = 0–4 years | ^P < 0.01; 95% CI = 1–5 years), higher weekly training frequency (*P = 0.02; 95% CI = 0–2 sessions·week−1 | ^P < 0.01; 95% CI = 2–4 sessions·week−1), longer individual training session duration (*P < 0.01; 95% CI = 16–36 minutes·session−1 | ^P < 0.01; 95% CI = 33–51 minutes·session−1) and higher weekly training volume (*P < 0.01; 95% CI = 203–371 minutes·session·week−1 | *P < 0.01; 95% CI = 457–604 minutes·session·week−1) compared to the sub-elite (*) and amateur (^) cohorts, respectively. Furthermore, the sub-elite group also reported longer training history (P < 0.01; 95% CI = 0–3 years), higher training frequency (P < 0.01; 95% CI = 1–3 sessions·week−1), longer training session duration (P < 0.01; 95% CI = 9–22 minutes·session−1) and higher weekly training volume (P < 0.01; 95% CI = 190–297 minutes·session·week−1) compared to the amateur group.
When making the same comparisons between groups based on contraceptive status, there were no significant differences in participants age (P = 0.97), stature (P = 0.91), body mass (P = 0.77), training history (P = 0.38), training frequency (P = 0.71), training session duration (P = 0.38) or weekly training volume (P = 0.34).
Menstrual cycle characteristics amongst non-HC users
Menstrual cycle characteristics of non-HC users are presented in Table 2. When compared between competitive levels, respondents reported no difference in number of cycles over a 12-month period (P = 0.23), cycle length (P = 0.08), frequency of menstrual cycle variability (P = 0.30), days of variability (P = 0.29), self-reported (P = 0.62) and medically diagnosed (P = 0.98) instances of secondary amenorrhea or self-reported (P = 0.35) and medically diagnosed (P = 0.90) instances of oligomenorrhoea. Additionally, no differences were reported between groups in relation to whether training was affected (P = 0.57) or avoided (P = 0.64) at any stage of the menstrual cycle. Menstrual cycle tracking was utilised by 91%, 83% and 84% of the elite, sub-elite, and amateur cohorts, respectively, with no differences between groups (P = 0.60). Methods used to track the cycle included mobile phone apps (83%), calendar (14%) or a diary (6%), with 7% of participants reporting the use of multiple tracking strategies.
Overview of menstrual cycle characteristics in non-HC users within the elite, sub-elite and amateur cohorts.
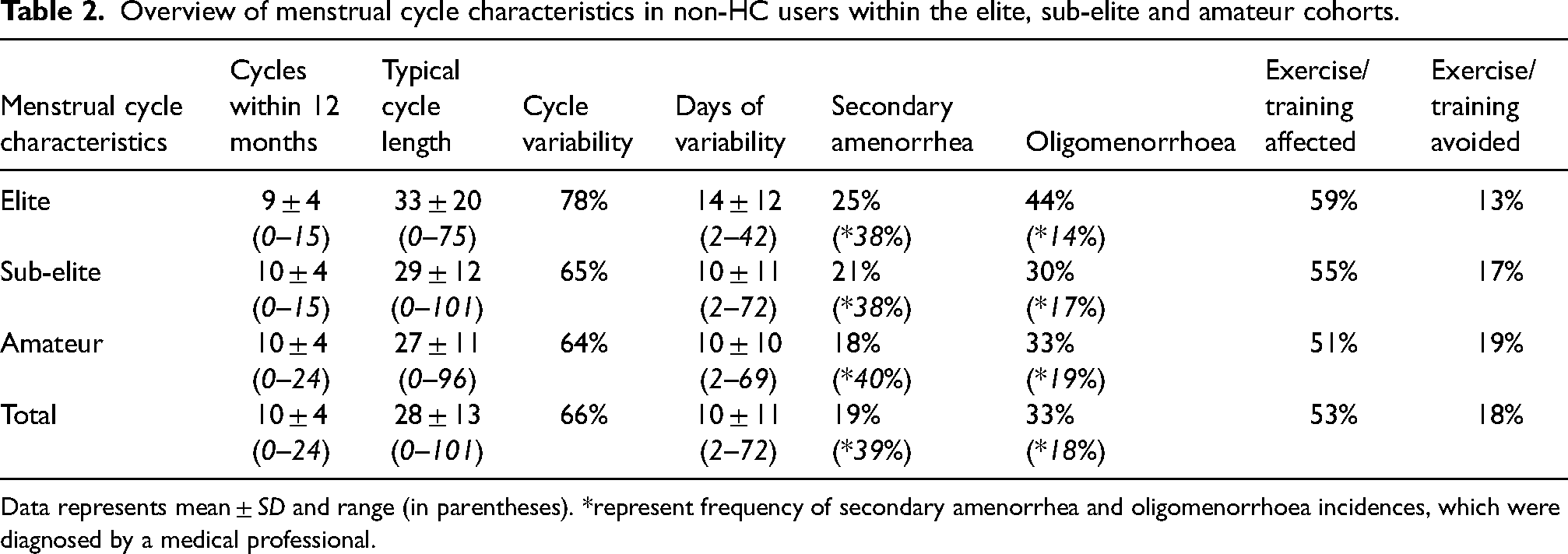
Data represents mean ± SD and range (in parentheses). *represent frequency of secondary amenorrhea and oligomenorrhoea incidences, which were diagnosed by a medical professional.
Frequency distribution of types of contraceptive use amongst HC users
The frequency of HC type within each of the competitive levels and the total participant sample is displayed in Table 3. The reported use of OC (P < 0.01) was significantly greater than all other methods. Additionally, the prevalence of IUD/S and implant use was also greater than the use of injections, vaginal rings and transdermal patches (P < 0.01). However, the prevalence of OC (P = 0.72), IUD/S (P = 0.30), implant, (P = 0.45), injection (P = 0.68), vaginal ring (P = 0.46) and transdermal patch (P = 0.84) use was not different between competitive levels.
The prevalence of hormonal contraceptive types within the elite, sub-elite and amateur cohorts.

denotes significant difference between OC and all other forms of contraception (P < 0.01). b denotes significant difference between IUD/S-Implant and all other forms of contraception (P < 0.01). (OC: oral contraception; IUD/S: intrauterine device/system; VR: vaginal ring; TP: transdermal patch). *group totals exceed 100% due to some participants using multiple contraceptive types.
Reported symptomology during the menstrual and contraceptive cycles
An overview of reported symptoms experienced by both non-HC and HC users is presented in Figure 2, with distinct differences reported between groups (see Figure inset panel). In non-HC users, 28%, 30% and 24% of the elite, sub-elite, and amateur cohorts reported using treatments and/or medication with a view to alleviate menstrual symptoms, with no differences evident between groups (P = 0.60). Treatments cited included ibuprofen (54%), paracetamol (35%), codeine (7%), naproxen (7%), heat applications (5%), cannabidiol (5%), acetaminophen (4%), tramadol (1%) and diclofenac (1%).
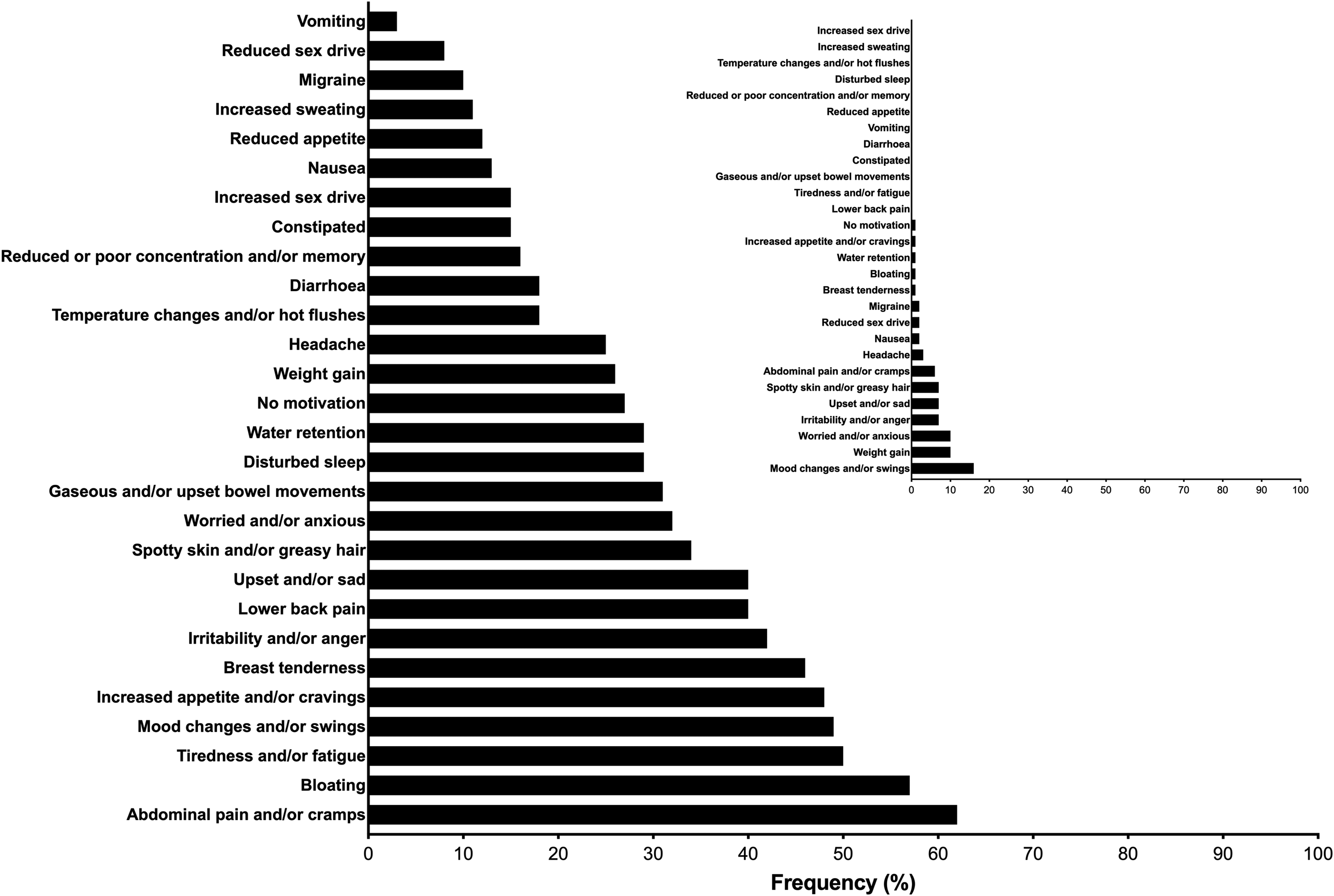
The frequency of reporting for negative symptoms amongst non-HC and HC users (panel inset).
Frequency of daily occasions of meal and snack intake
The frequency for daily occasions of meal and snack intake were 2 to 5 meals and 0 to 5 snacks across competitive levels and contraceptive statuses, respectively. There was a significant influence of competitive level on the odds of increased meal (OR = 1.74; P < 0.01; 95% CI = 1.30–2.32) and snack (OR = 1.37; P < 0.01; 95% CI = 1.09–1.72) intake, whereby the elite and sub-elite group reported a significantly higher frequency (P < 0.05 for all comparisons) of consuming 4 meals (21.6 and 13.6%) and snacks (9.8 and 11.0%) per day when compared with the amateur group (6.9% meals and 5.4% snacks). The amateur group also reported a higher frequency of consuming only one snack per day (23.7%) when compared with elite (7.8%) and sub-elite (13.6%) groups (P = 0.01). In contrast to competitive level, there was no influence of contraceptive status on frequency of either meal (OR = 1.19; P = 0.39; 95% CI: 0.80–1.76) or snack (OR = 0.97; P = 0.85; 95% CI = 0.71–1.32) intake.
Frequency of skipping meals and associated qualitative reasons
The frequency of those participants who self-reported skipping meals is presented in Figure 3. Neither contraceptive status (P = 0.68) nor competitive level (P = 0.37) affected the frequency of reporting for skipping meals. When evaluated across the whole sample, 40% of participants reported skipping meals. The frequency of reasons provided for skipping meals is presented in Figure 4. Lack of time, schedule and alterations to appetite were cited as the most frequent reasons within all cohorts.
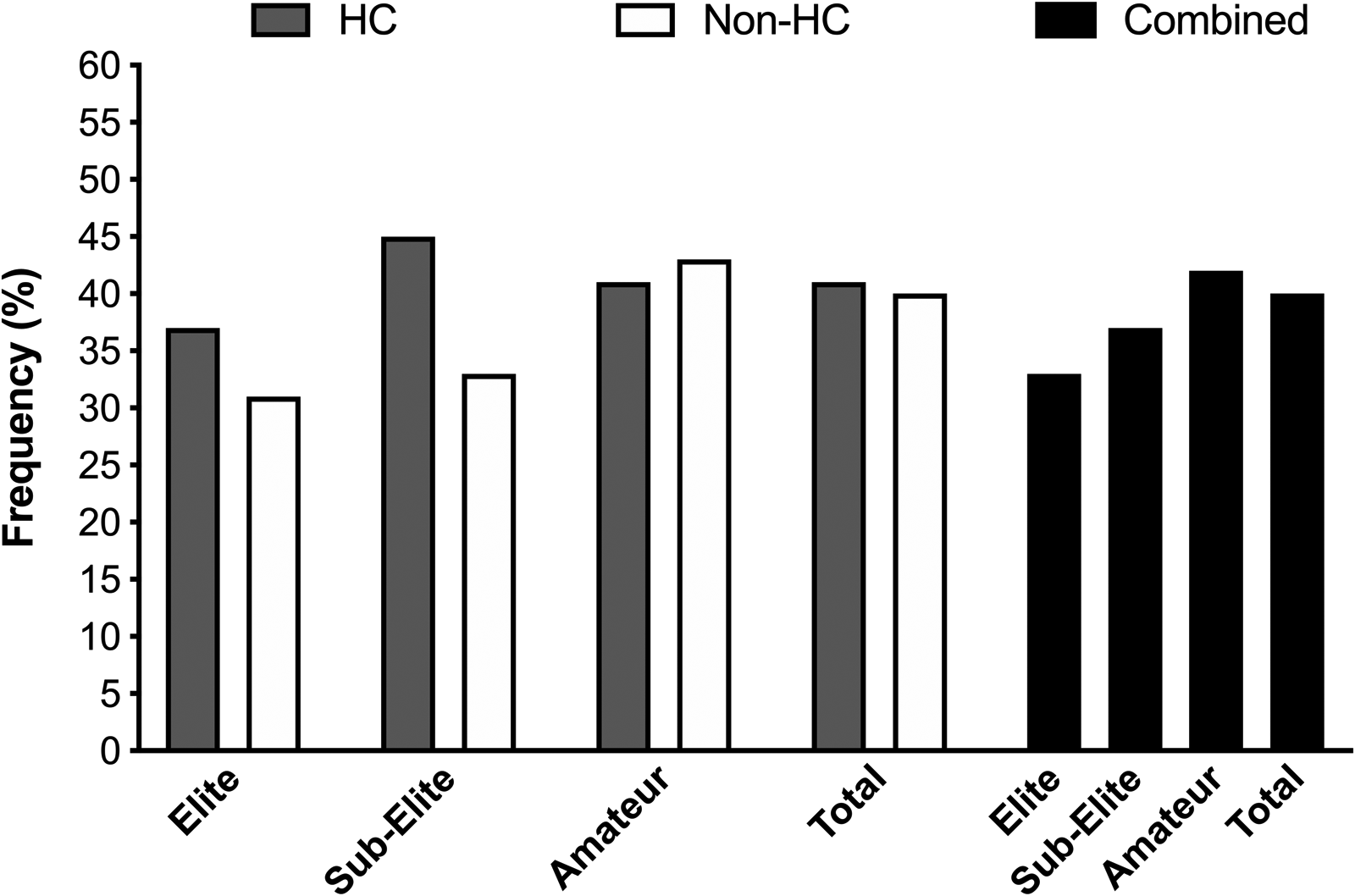
The frequency of respondents reporting skipping meals within the elite, sub-elite and amateur cohorts (HC: hormonal contraceptive users; non-HC users: non-hormonal contraceptive user; combined: collated HC and non-HC users).
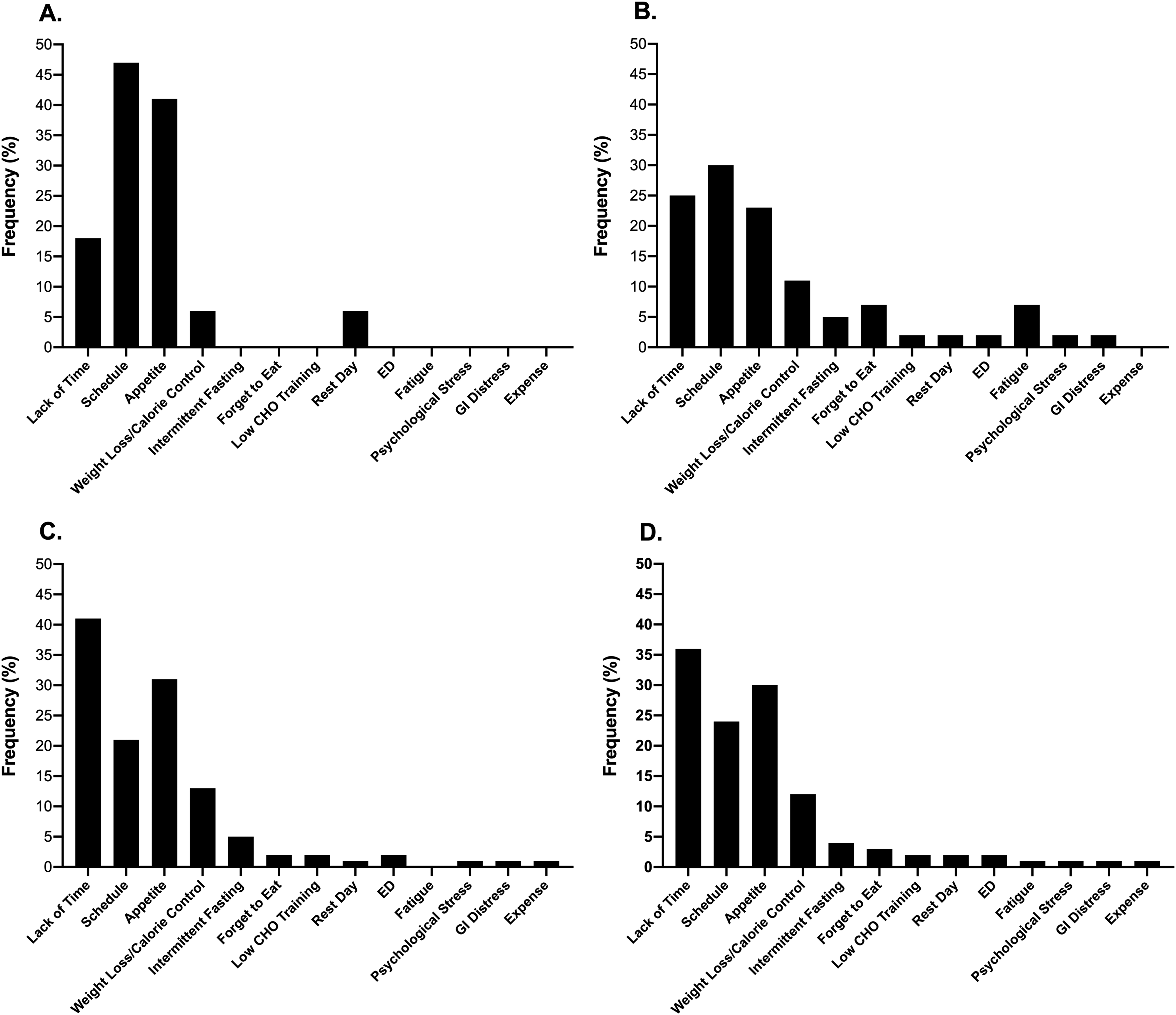
The frequency of reasons cited by respondents for skipping meals within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D).
Perceived changes in taste preferences throughout the menstrual and HC cycles
The frequency of participants who self-reported changes in taste preferences across the menstrual and HC cycles is presented in Figure 5. Neither contraceptive status (P = 0.17) nor competitive level (P = 0.48) affected the frequency of reporting for variations in taste preferences, respectively. The largest change in taste preference across both HC and non-HC users was towards sweet foods (as noted by ∼60% of respondents: see Figure 6), where the associated reasons were cited as changes occurring during pre-menses (14–35%) or menses (15–25%; see Figure 7).
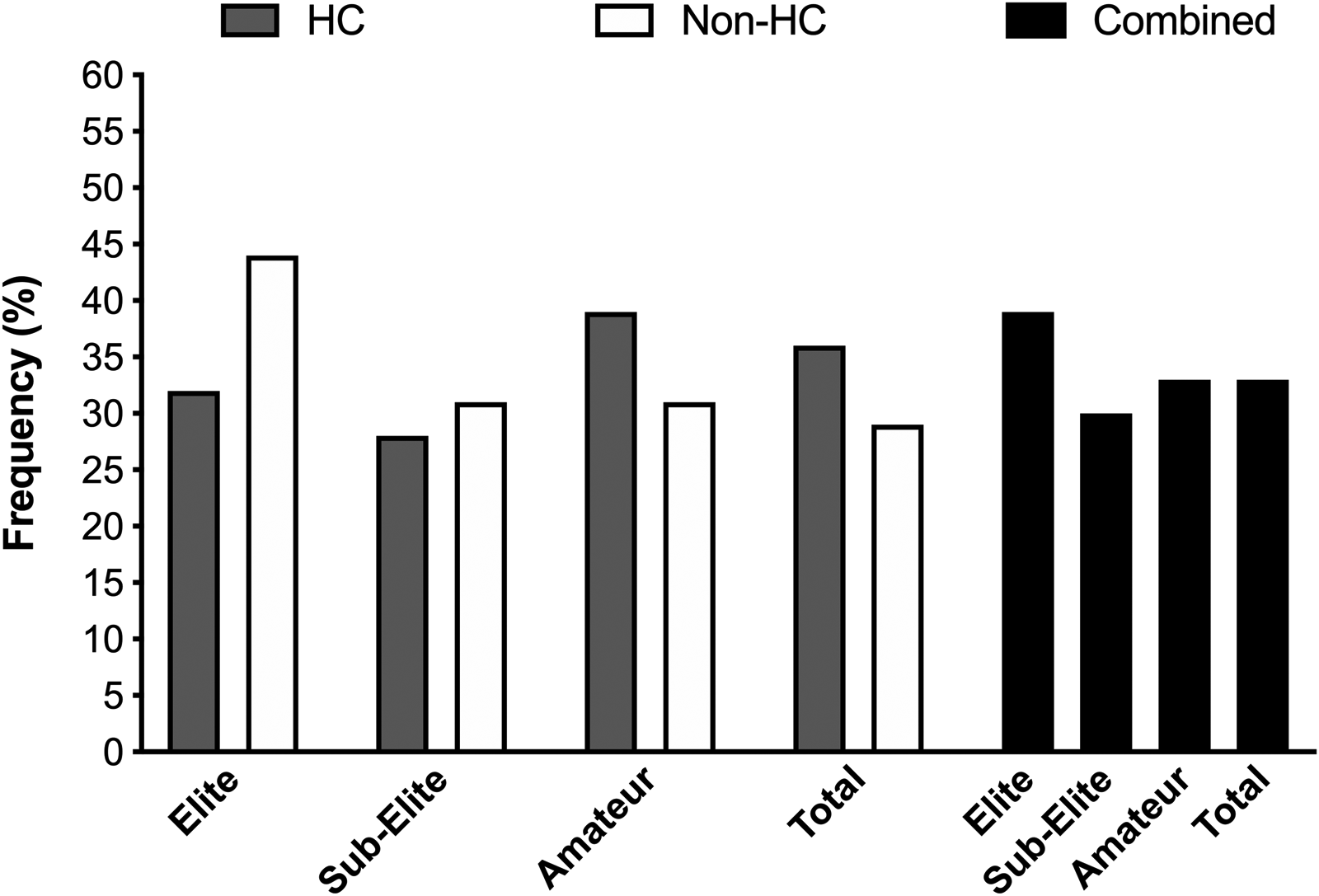
The frequency of respondents reporting changes in taste preferences across the menstrual and contraceptive cycles within the elite, sub-elite and amateur cohorts (HC: hormonal contraceptive users; non-HC users: non-hormonal contraceptive user; combined: collated HC and non-HC users).
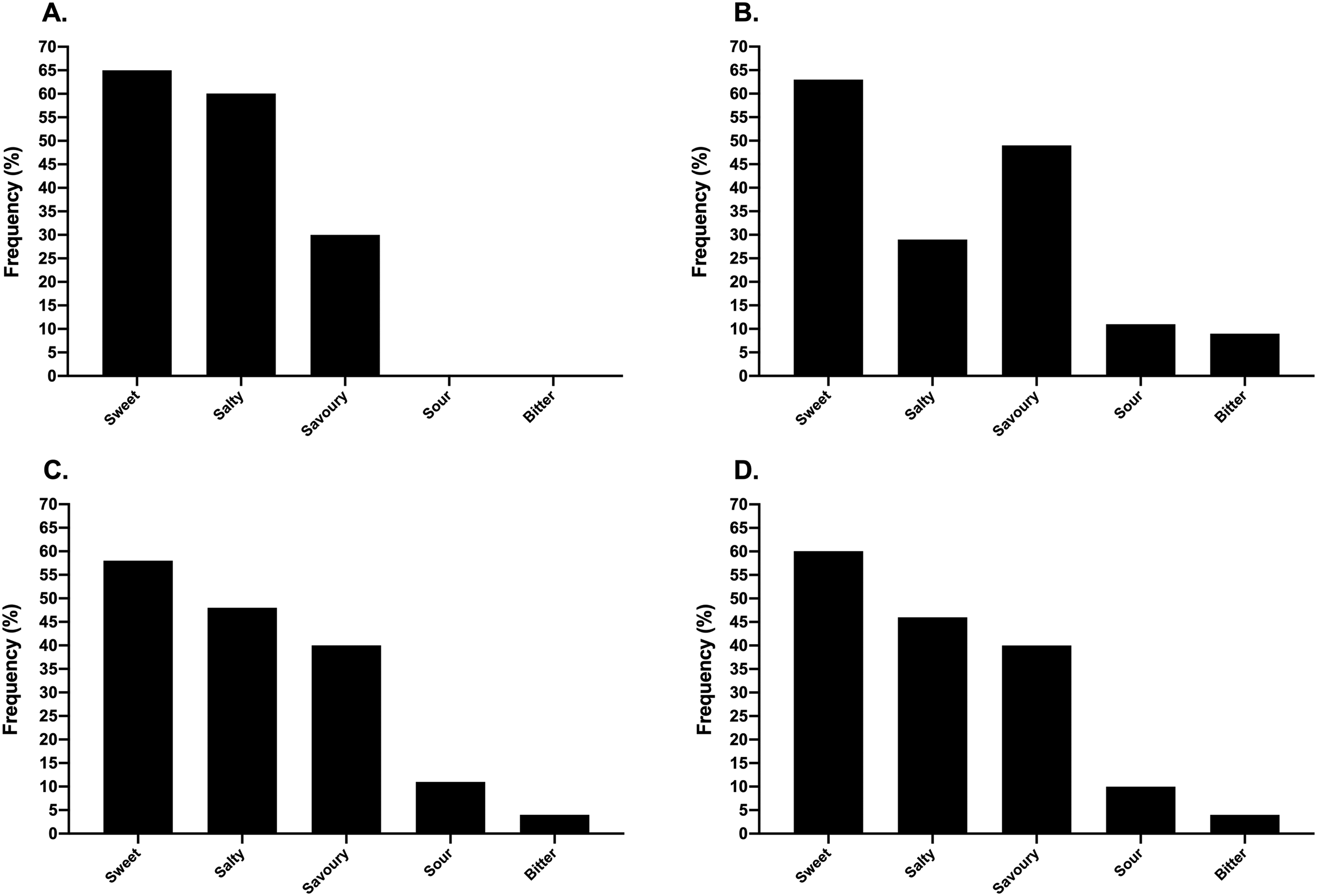
The frequency of respondents reporting types of taste preference across the menstrual and contraceptive cycles within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D).
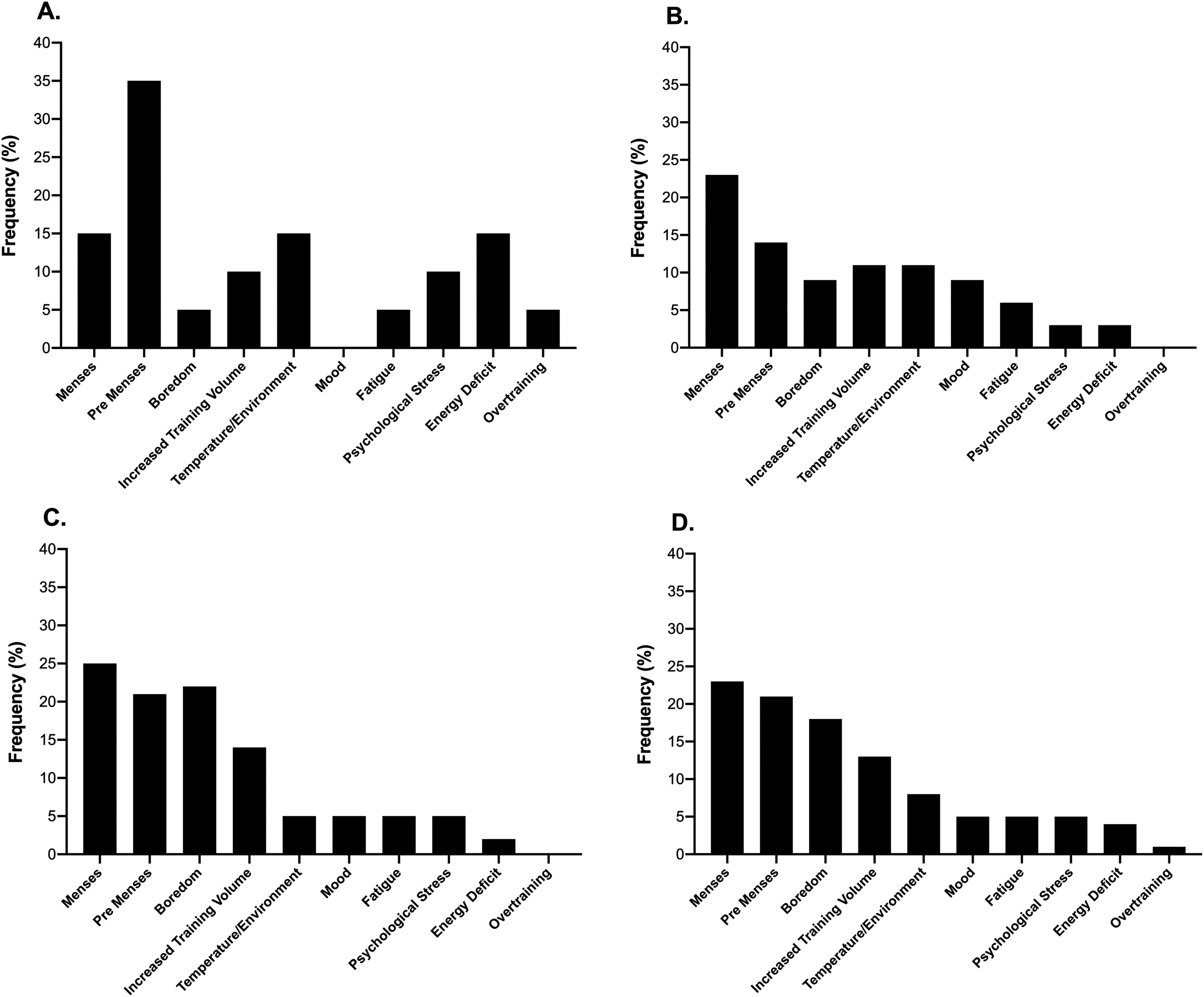
The frequency of reasons cited by respondents for perceived changes in taste preferences across the menstrual and contraceptive cycles within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D).
Perceived changes in food cravings throughout the menstrual and HC cycles
The frequency of those participants who self-reported changes in food cravings across the menstrual and HC cycles is presented in Figure 8. Neither contraceptive status (P = 0.20) nor competitive level (P = 0.52) affected the frequency of reporting for changes in food cravings. The most prevalent foods craved across both HC and non-HC users were chocolate and other confectionary (as noted by 25% of all respondents: see Figure 9), whereby the associated reasons were cited as changes occurring during pre-menses (21–30%) or menses (23–29%; see Figure 10).
The frequency of respondents reporting changes in food cravings across the menstrual and contraceptive cycles within the elite, sub-elite and amateur cohorts (HC: hormonal contraceptive users; non-HC user: non-hormonal contraceptive user; combined: collated HC and non-HC users).
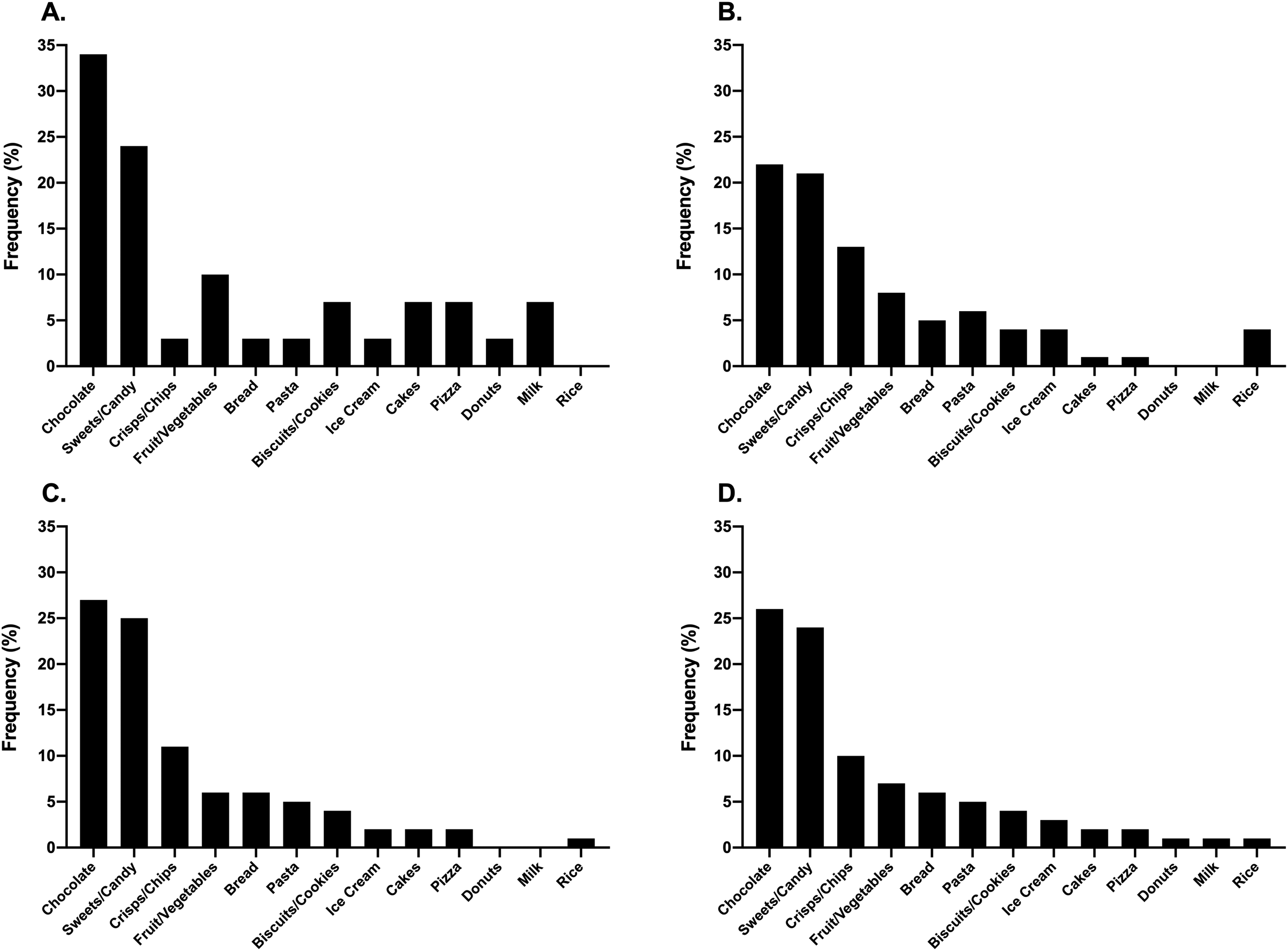
The frequency of respondents reporting changes in types of foods craved across the menstrual and contraceptive cycles within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D).
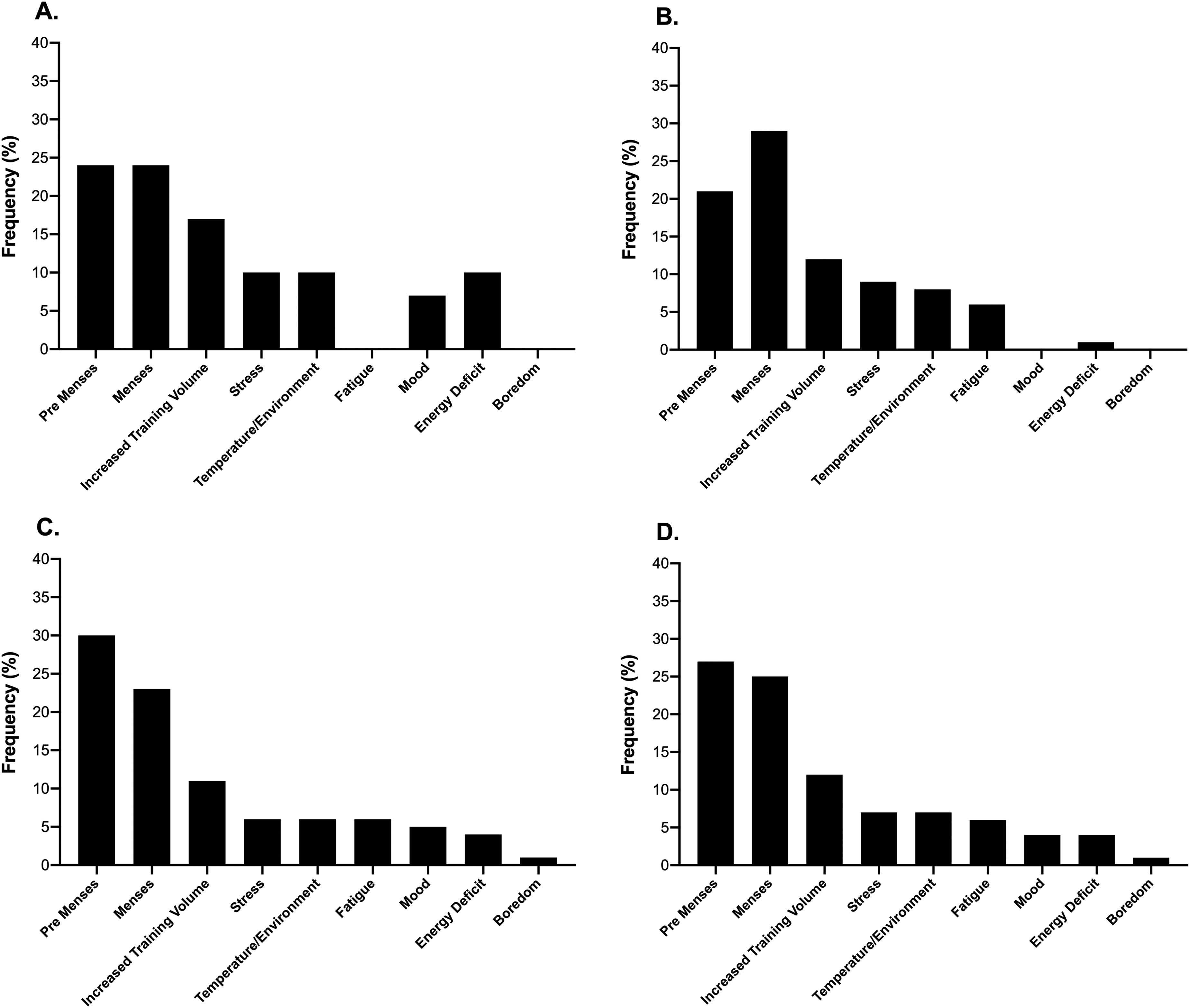
The frequency of reasons cited by respondents for changes in food cravings across the menstrual and contraceptive cycles within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D).
Frequency of dietary supplement utilisation and types consumed
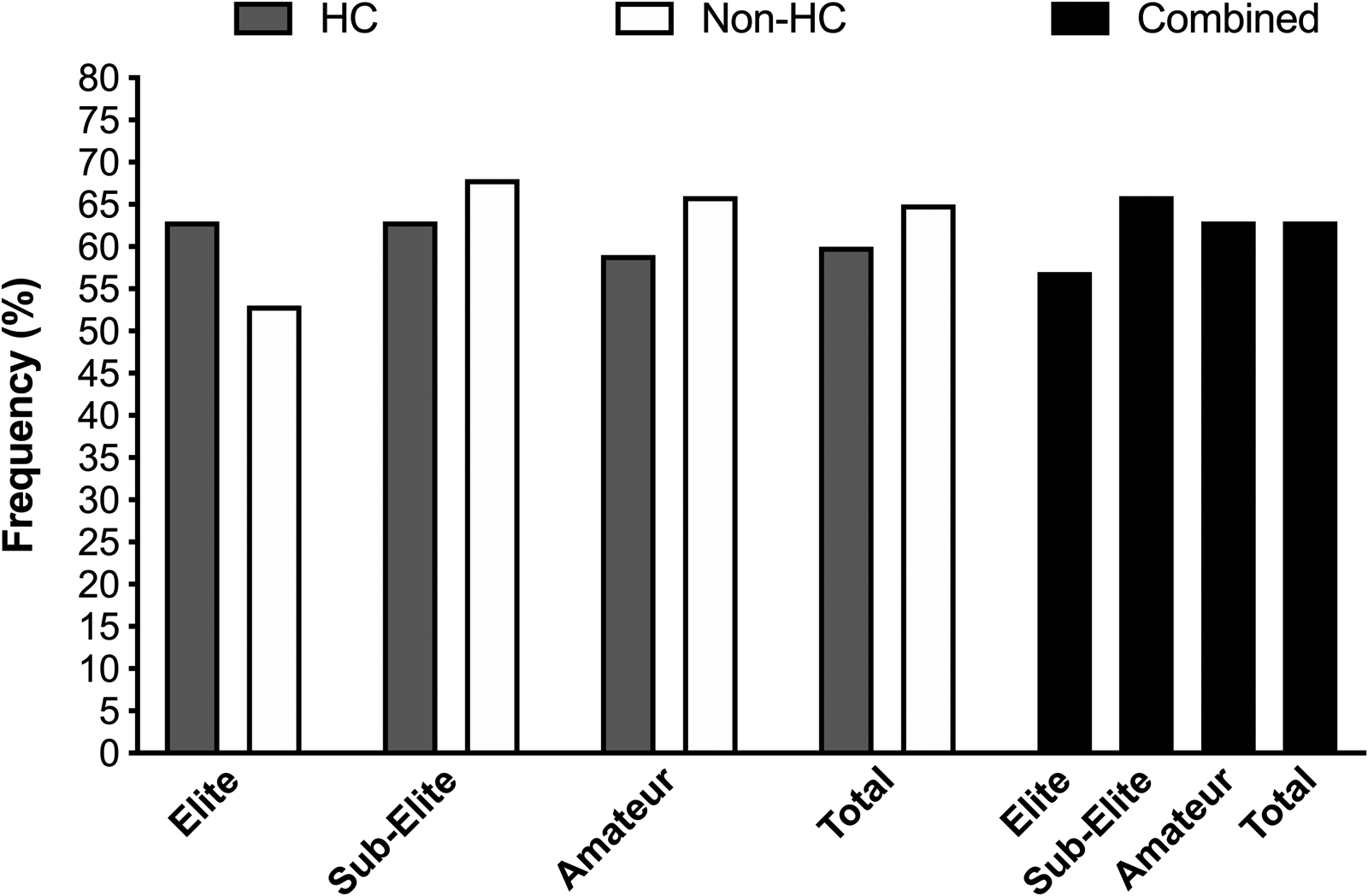
There was no effect (P = 0.31) of contraceptive status on prevalence of supplement use (see Figure 11) where 59 and 67% of HC and non-HC users reported overall use. In contrast, there was a higher prevalence of supplement use in the elite (76%) compared with the amateur (61%) groups (P = 0.04). The specific types of supplements consumed are also presented in Figure 12. The most popular nutritional supplement type across all competitive levels was whey protein (59–65%), which was then followed in prevalence by health-based supplements including vitamin D (18–33%), multivitamins (10–27%), omega 3 (11–23%) and iron (11–18%).
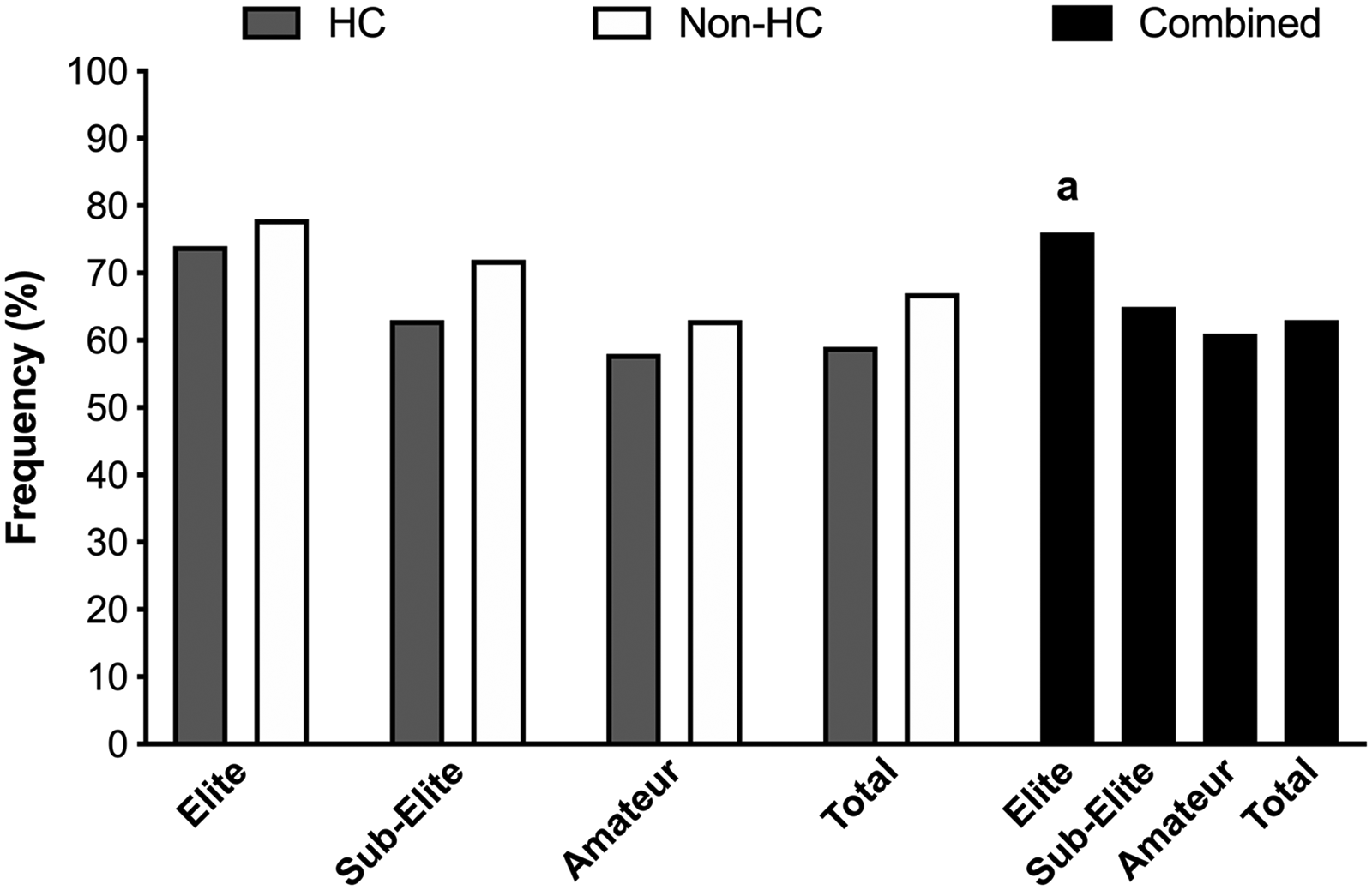
The frequency of respondents reporting supplement use within the elite, sub-elite and amateur cohorts. a denotes significant difference between elite and amateur cohorts (P < 0.05) (HC: hormonal contraceptive users; non-HC users: non-hormonal contraceptive users; Combined: collated HC and non-HC users).
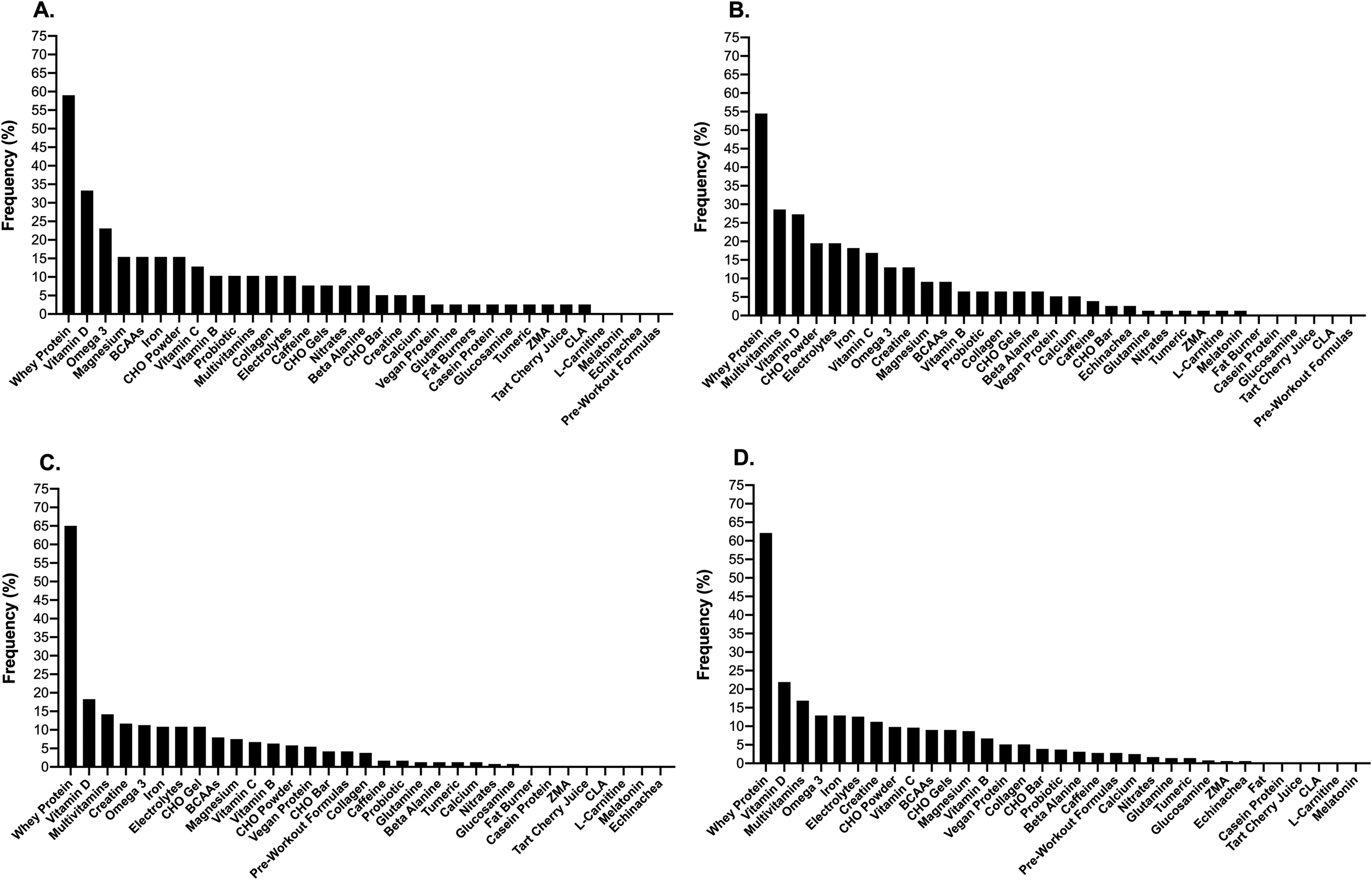
The frequency of respondents reporting specific types of supplement use within the elite (A), sub-elite (B) and amateur (C) cohorts as well as the combined total sample (D) (CHO: carbohydrate; BCAA: branch chain amino acids; ZMA: zinc magnesium aspartate; CLA: conjugated linoleic acid).
Discussion
The aim of the present study was to audit the HC use, menstrual cycle characteristics and training/nutrition related profiles of pre-menopausal female athletes and exercisers. Accordingly, we surveyed a cohort of elite participants, with a novel aspect of this study also including sub-elite and amateur participants for examinations between groups. To the authors’ knowledge, this represents the first time these individual contingents have been explored in tandem, with our data demonstrating that the prevalence and type of HC use was not different between these competitive levels. As expected, training profiles were different between competitive levels, though no differences were reported between HC and non-HC users. In non-HC users, we also observed that the frequency of respondents reporting menstrual cycle variability and irregularities was not influenced by competitive level. Although the elite and sub-elite groups tended to consume more meals and snacks per day than the amateur group, it is noteworthy that 40% of all respondents reported frequently skipping meals, predominately due to lack of time, schedule and appetite. Additionally, a high proportion of respondents reported alterations to taste preferences and food cravings during pre-menses and/or menses, particularly favouring sweet foods such as chocolate and other confectionary, the pattern of which was not affected by either contraceptive status or competitive level. When taken together, the present data highlights that competitive level does affect simple markers of training and nutrition profiles yet contraceptive status does not. However, given the high individual variability in HC type/use, menstrual cycle characteristics and associated symptomologies across the participant cohort, this provides further evidence that female athletes and exercisers cannot be treated with a one size fits all approach when attempting to formulate training and/or nutritional strategies.
The prevalence of HC use reported here (i.e. 41%) is largely similar to that highlighted from other recent studies on athletic populations (see Table 3). For example, Armour and colleagues 20 also observed HC use of 42% (n = 124) amongst a mixed sport cohort of Australian athletes, though it is noteworthy that the specific criteria qualifying athlete status in that study likely encompassed individuals who would fall under the sub-elite and amateur criteria specified here. 27 In evaluating HC use in populations that could be considered elite and sub-elite by definition, other researchers have observed higher prevalence of use in mixed sport athletes including 50% (n = 430, country of origin not specified), 57% (n = 186 Danish athletes) and 63% (n = 1086 Norwegian and Swedish athletes).8,15,19 Additionally, Parker and colleagues 29 recently reported a much lower HC prevalence of 20% amongst professional soccer players from the United Kingdom. Collectively, such data suggest that HC use in female athletes may reflect the culture, maturity and type of sport, intensity and frequency of competition, as well as nuances surrounding the criteria to define elite status. Interestingly, our data are also suggestive of a shift towards alternate contraceptive methods. For example, although OC was the most frequently reported form of contraception amongst our participants (63%) in parallel to the results of Ekenros et al., 8 both Martin et al. 15 and Oxfeldt et al. 19 reported a higher OC use of 70–80%. In relation to reporting of IUD/S, we observed a higher prevalence of use than that reported by Martin et al. (i.e. 21% versus 3%) but similar to that reported by Oxfeldt et al. and Ekenros et al. (i.e. 18 and 29%). In contrast, the frequency of implants reported here is comparable to Martin et al. and Ekenros et al. (i.e. 13 and 15%), but considerably higher than Oxfeldt et al. (i.e. 1%). Whilst it is tempting to speculate that subtle shifts in prevalence of HC use and type may be due to increased media driving the merits of a natural female ovarian cycle, or even geographical and/or cultural driven factors, future studies are required to evaluate the reasons underpinning the choices of specific contraceptive method particularly within differing representative samples. Finally, notwithstanding issues around privacy, future research should consider investigating the rationale for the use of HC, at least to ascertain whether it is being used as an improper countermeasure for secondary amenorrhea in athletes, which could also be masking menstrual cycle irregularities. Here, it is noted that HC use does not provide optimal hormonal replacement to manage bone health in amenorrhoeic athletes 30 and that breakthrough/withdrawal bleeding during the inactive phase of HC use may be mistaken as a sign of normal menstrual function. Further education around these points is needed, since studies have shown a limited level of knowledge surrounding the menstrual cycle and HC use amongst female athletes and support staff.7,31,32
In relation to non-HC users, a large proportion of each competitive level were tracking their menstrual cycle (83–91%), with the majority of the whole cohort utilising mobile phone apps. Whilst this study did not interrogate why these groups were gathering this information, future examinations should focus on (i) collecting these details and (ii) critically assessing the utility of current tracking processes (i.e. are current mobile phone apps fit for purpose) alongside the application of these data (i.e. how menstrual tracking data is used to modify training, performance, nutrition, etc.). Overall, 78, 65 and 64% of elite, sub-elite and amateur participants reported that their menstrual cycle varied in length (see Table 2). Although this prevalence is somewhat higher than the 51% previously highlighted in elite athletes by Martin et al., 15 the mean variability reported by the whole group cohort (i.e. 10 ± 11 days) is similar (i.e. 9 ± 9 days). Menstrual cycle length within and between individuals was highly variable (across the 2–72 day range: i.e. some cycle lengths varied by 2 days and are considered largely regular, whilst other cycle lengths varied by up to 72 days and are considered highly irregular) and in this regard, only 33% of the variability reported here was explained by self-reported instances of oligomenorrhoea, with a further 19% of all respondents self-reporting prevalence of secondary amenorrhea, in parity to the results of Ekenros et al.. 8 In a large-scale survey of 32,595 women evaluating 75,981 menstrual cycles, Soumpasis et al. 33 observed that only 13% of respondents reported a stringent 28-day cycle, with 87% displaying cycle variability within 5 days and 52% reporting variability of over 5 days. In considering such natural unpredictability of menstrual cycle length both within and between non-HC users, coupled with the aforementioned prevalence of HC use, there needs to be considerable focus by future researchers to scrutinise experimental design and address the complexities associated with female participation. 4 Furthermore, these factors also oppose the suggestion that generalised guidelines can be applied to female athletes and exercisers in relation to tailoring training and/or nutritional strategies based around phases of the menstrual or contraceptive cycles.12–14
In accordance with competitive status, the elite cohorts surveyed here reported higher training loads (e.g. longer training history, increased weekly training frequency, longer training duration, etc.) than the amateur athletes and exercisers. Although it has been demonstrated that an increased severity of menstrual symptoms (as experienced by non-HC users) is related to higher probability of missing training sessions,8,34 it is noteworthy that this does not always manifest as differences in training profiles between HC users and non-HC users. Indeed, previous cross-sectional comparisons of such groups, albeit within athletic populations, demonstrate no differences in weekly training frequency, average training session duration and total weekly training volume.8,15,19 In agreement, we also observed no differences in the training profiles reported by the HC users and non-HC users surveyed here (see Table 1), despite the increased prevalence of menstrual symptoms reported by non-HC users (see Figure 2). In this way, such data appear to agree with the recent reports that over 80% of athletes and coaches do not necessarily alter training schedules across stages of the menstrual cycle.8,20
The pattern of daily occasions of meal and snack intake reported here, is also consistent with that previously highlighted in female athletes.35–37 When evaluated via OR and frequency distribution, our data are suggestive of higher dietary intakes within the elite and sub-elite cohorts when compared with the amateur cohort, a feeding pattern that is perhaps expected given their higher training loads (see Table 1). Although a limitation of this study is that we did not objectively assess daily energy intake, a major nutritional goal for female athletes and exercisers is to simply ensure sufficient daily energy availability, an outcome of which is to reduce the risk of developing negative health and performance consequences arising from under fuelling.38,39 In this regard, it is noteworthy that between 33% and 42% of the elite, sub-elite and amateur participants surveyed here reported frequently skipping meals, with reasons largely centred around lack of time (40% of amateur respondents), schedule (45 and 30% of elite and sub-elite respondents) and low appetite (40% of elite respondents). Such findings suggest that factors associated with intense training schedules (i.e. elite cohorts) and busy lives (amateur cohort) may induce episodes of under fuelling. 40 Such insight may provide a starting point for which to formulate behaviour change strategies to improve the fuelling practices of female athletes and exercisers. Further studies are now required to explore this issue in greater detail and to subsequently develop, deliver and evaluate the efficacy of any associated behaviour change interventions.
On the basis that previous research has highlighted taste preferences and food cravings may change throughout the menstrual cycle,21–26 we also evaluated the prevalence of such issues in both HC and non-HC users. This was established on the premise that non-HC users would exhibit larger fluctuations in ovarian sex hormone profiles when compared with the more stable endocrine environment of HC users. Nonetheless, we observed no differences in perceived taste change preferences and food cravings between HC and non-HC users within the elite, sub-elite, and amateur cohorts. Such data agree with recent cross-sectional, laboratory and qualitative studies suggesting that food choices are not apparently affected by contraceptive use.7,41–45 In relation to specific taste profiles, we also observed that the present respondents reported changes in taste preferences during pre-menses (14–35%) or menses itself (15–25%), where the largest shift was in preference towards sweet foods (reported by 60% of all respondents) such as chocolate and other confectionary (reported by 25% of all respondents). Interestingly and despite consisting of a mixed contraceptive status cohort, HC participants accounted for 32% and 35% of responses for menses and 40% and 27% of responses for pre-menses across perceived taste change and food cravings, respectively, with only one respondent specifically referring to this as a withdrawal bleed.
In agreement with previous reports,46–48 we also observed that the elite cohort reported a higher prevalence of dietary supplement use compared with the amateur cohort (76 versus 63%), yet this was not different between HC users and non-HC users, suggesting again, that contraceptive status does not readily influence this dietary behaviour. 7 Whey protein was the most popular across all cohorts (55–65%), followed by micronutrients that are more commonly marketed towards general health such as vitamin D and multivitamins etc. Although we did not specifically evaluate the rationale for the use of dietary supplements, our data may suggest that the presented cohort of athletes and exercisers consider maintenance and/or growth of muscle mass alongside health-related goals as desired outcomes. Interestingly, despite the high prevalence of skipping meals, there was a low usage of dietary supplements to support daily energy and carbohydrate intake such as gels, powders or bars. Furthermore, there was also a low prevalence for use of evidence-based ergogenic aids such as creatine, caffeine, beta-alanine and nitrates. Such data demonstrate that female athletes and exercisers may benefit from targeted education regarding the use of specific dietary supplements to promote training adaptation, body composition and performance related goals.
Despite the presented novel findings, it should be noted that this study is not without limitations. Firstly, the data are based on self-reports which may contain participant biases/misunderstandings and given the survey tool items were generated from examinations of athlete participant groups, this may have limited the applicability to the amateur cohort within this study. Furthermore, the lack of information on nationality, race, gender identity or disability within the survey and potential specific questions that have been validated for generalised exercising population groups should be addressed in future investigations. We also acknowledge the limitation of not specifying the brand of contraception reported here, as well as types of OC (i.e. combined or singular, mono, bi, tri-phasic, etc.). Finally, these data were collected during the COVID-19 pandemic and may represent an altered lifestyle (e.g. modification of exercise and diet during lockdowns). Nevertheless, they may highlight the new normal and have ongoing relevance within present and future global circumstances.
In conclusion, we provide the first report to simultaneously survey HC use, menstrual cycle characteristics and training/nutritional related profiles in a cohort of females inclusive of elite, sub-elite and amateur athletes and exercisers. We observed that contraceptive status does not appear to readily affect training and nutritional profiles amongst all cohorts, the latter inclusive of changes in perceived taste change preferences, food cravings and dietary supplement use across the menstrual and contraceptive cycles. Nonetheless, the high prevalence of HC use coupled with the between and within participant menstrual cycle variability reported by non-HC users, challenge the suggestion of a generalised approach towards tailoring training and nutritional strategies to specific phases of the menstrual and HC cycles. Rather, the observation of a high frequency of skipping meals and low prevalence of use of evidence-based supplements and ergogenic aids, highlights that female athletes and exercisers would more likely benefit from targeted education and behaviour change strategies that address fundamental components of performance nutrition. To that end, the data from this study serves to highlight the unique specific contexts that HC and non-HC users may experience and the need for an individualised approach when considering modulation of training and nutritional practices in athletic and exercising female cohorts.7–9
Footnotes
Acknowledgements
The authors would like to thank Gina Kelly, Caraigh McGuiness and Poppy Hawe for their assistance during the data collection period.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: James P. Morton is a consultant for SiS plc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the Science in Sport plc.
