Abstract
Metal–organic frameworks are coordination polymers composed of inorganic nodes connected by organic linkers. The 2-carboxyethyl(phenyl)phosphinic acid (CEPPA) is a ligand capable of bonding to a wide variety of metal ions. This review presents data concerning CEPPA and its metal complexes, along with a few examples of applications of the acid in combination with other compounds. It also highlights the synthesis and characterization of CEPPA.
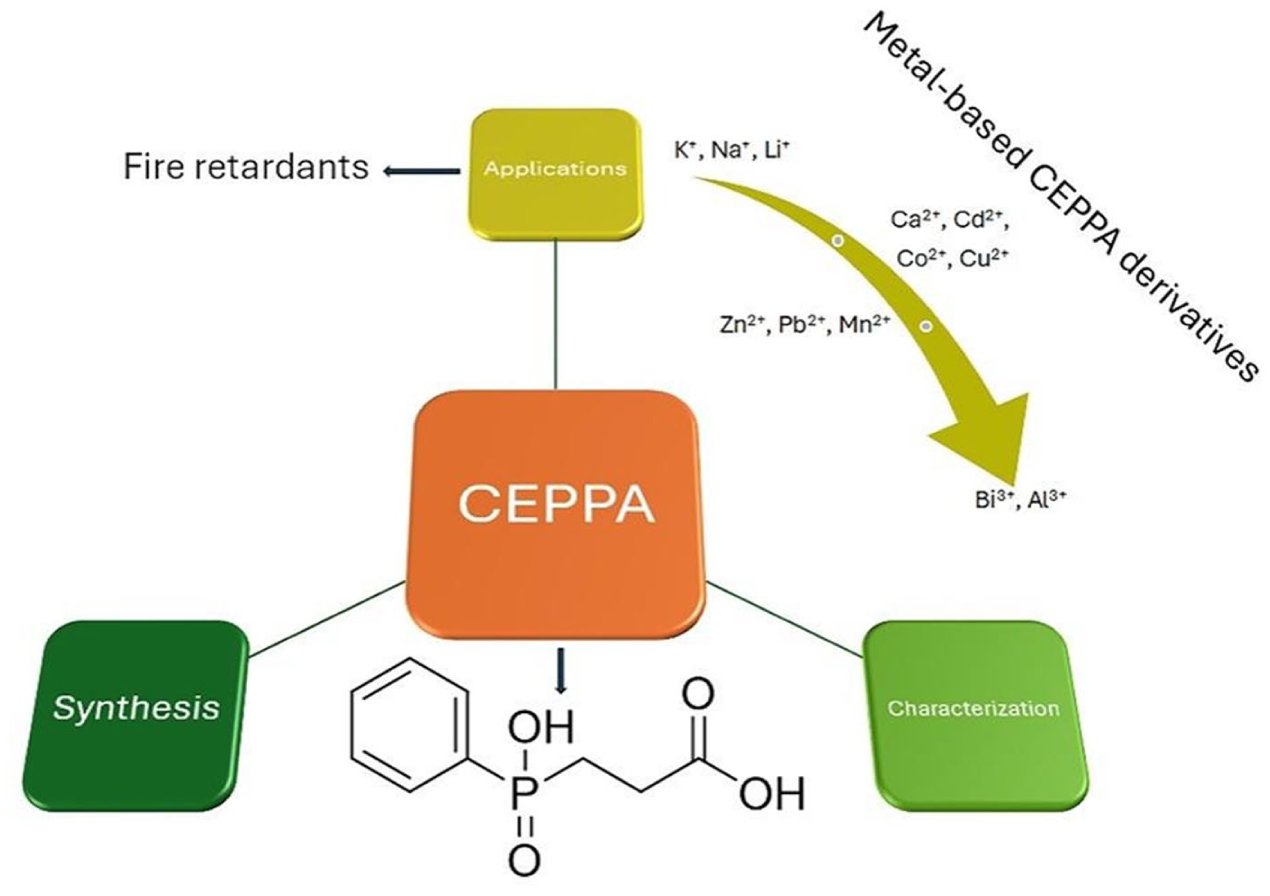
Introduction
The first natural phosphinic acid was isolated from Streptomyces viridochromogenes and Streptomyces hygroscopicus as a phosphinotricil-L-alanine-alanine tripeptide. 1 This tripeptide contains 2-amino-4-(methylphosphino)butanoic acid, which is very active against gram-positive and gram-negative bacteria. Another example of a natural phosphinic acid is phosphoenolpyruvate, which occurs from S. viridochromogenes. From 1818 to 1892, researcher A. W. Hofmann discovered many functional groups for organophosphorus chemistry, including phosphonic and phosphinic acids. 2
Phosphinic acids, or hypophosphorous acids (R1R2POOH) 3 molecules, have a tetrahedral geometry and represent a class of compounds with many different applications, such as floating agents,4,5 precursors for ionic liquids, 6 flame retardants, 7 coordination polymers, 8 separation processes, 9 metal extractions, 10 and peptide analogs. 11
Phosphinic acids differ from phosphonic acids in that the former present a secondary organic moiety that replaces a hydroxyl group (Figure 1). 12

Phosphinic and phosphonic acids’ molecular structures.
The electronic and electrostatic properties of the phosphinate ligand can be easily converted by changing the two organic moieties R 1 and R 2 attached to the phosphorus atom. 13
These compounds are guided by four fundamental characteristics: a tetrahedral transition state, the ability to form electrostatic interactions, hydrogen bonds, and the capacity to coordinate with metal ions. 14 Another important characteristic is their solubility in water because of the hydrogen bonds formed between the hydroxyl group from the acid and the hydroxyl group from the water. In addition, they are associated through hydrogen bonds both in solution and in the solid state. 15 Phosphinic acids are monoacids (pKa ∼ 1) that are weaker than their analog phosphonic acids (pKa ∼ 0.5–1.5), both of which are more acidic than carboxylic acids (pKa ∼ 2–3). 16
From a practical point of view, phosphorus-coordinated ligands offer a spectroscopic 13P NMR perspective that allows the characterization and identification of individual species even in complicated mixtures. 3 One of these applications is related to the identification of molecular intermediates during the synthesis of zinc oxide. 17 Nevertheless, NMR experiments in the solid state allow the differentiation and quantification of coordination models, such as acid coordinated by one oxygen atom or by both oxygen atoms. Another instrumental technique that can provide data about phosphorus bonds from these compounds is Fourier-transform infrared (FTIR) spectroscopy. This technique can be used to elucidate the orientation in which the molecules are attached.
In the last few years, 2-carboxyethyl(phenyl)phosphinic acid (CEPPA) gained interest in the research domain, with the accent on the obtaining of the new metal-based complexes and its use as reactive flame retardant.
Previous research has demonstrated that auxiliary ligands like 1,10-phenanthroline and transition metal ions like Mn²+, Cu²+, and Cd²+ can coordinate with (2-carboxyethyl)(phenyl)phosphinate to create octahedrally coordinated metal phosphinate complexes with discrete dimer or extended chain architectures. 18
Metal phosphinates occupy an intermediate coordination chemistry space between carboxylates and phosphonates, according to recent reviews on phosphinic acid derivatives. Because of their two oxygen donor atoms and organic substituents, which affect both steric and electronic coordination preferences, phosphinates, in contrast to phosphonates, usually coordinate as mono- or bidentate ligands but can nevertheless support prolonged frameworks. 14
Phosphinate coordination chemistry has uses in bioactivity and catalysis in addition to materials. Similar systems like copper carboxyphosphinates have demonstrated biomimetic enzyme-like activities and antibacterial properties, demonstrating the functional potential of carboxylate-phosphinate frameworks in catalysis and biomedical contexts, despite the fact that specific reports on CEPPA complexes are still rather rare. 19
This review presents data concerning CEPPA and its metal complexes, along with a few examples of applications of the acid in combination with other compounds. It also highlights the synthesis and characterization of CEPPA.
CEPPA: Synthesis, characterization, and applications
Phenylphosphinic acids are phosphorus derivative compounds that have many applications in different fields, such as flame retardants, 20 building blocks, 21 recognition of tyrosine phosphopeptides, 22 biological applications, 23 and corrosion. 24 CEPPA is a derivative of carboxylate-phosphinate compounds that present two carboxylate and phosphinate analogous fragments, both of which contain two potential oxygen donors. This asymmetric ligand can act as a bidentate, tridentate, tetradentate, or binding ligand to form multidimensional structures. The bond between the phosphinate ligand and the phenyl ring can be seen as a dipolar chromophore.
CEPPA synthesis
CEPPA (Figure 2) is a phosphorus copolymerizable, bifunctional monomer, 25 and its synthesis process involves two steps: the first is the reaction between dichlorophenylphosphine and acrylic acid, resulting in a three-intermediate mixture, and the second is the hydrolysis of the mixture to obtain the desired product. 26

CEPPA structure.
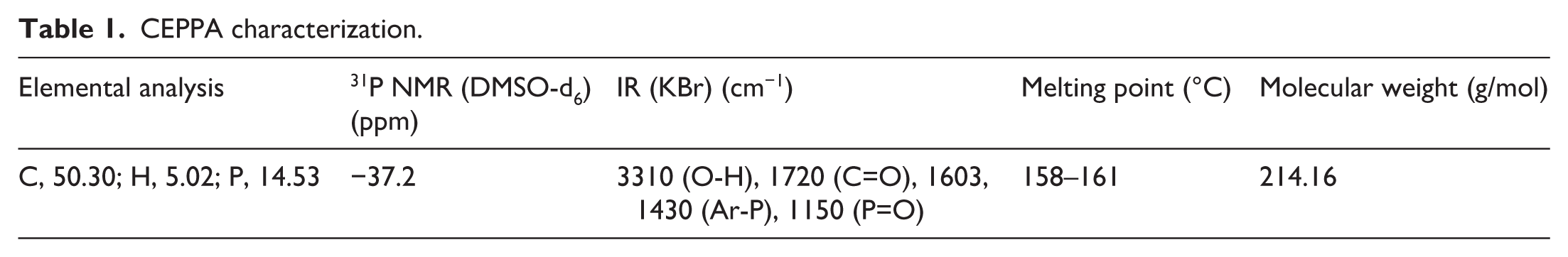
CEPPA characterization
Table 1 shows the characterization of the resulting compounds.
CEPPA characterization.
The solubility of this compound in water was tested at temperatures between 25 and 76 °C. The molar solubility of CEPPA in water increased from 1.782 × 10−3 M (25.1 °C) to 5.682 × 10−2 M (76.32 °C). 25
Hu et al. 27 studied the crystallization behavior of CEPPA and obtained two polymorphs with similar structures. Both polymorphs present similar hydrogen bonds (R43(20)), but the geometrical parameters are easily different, as shown in Table 2.
Hydrogen bond geometry.

The two polymorphs can be attributed to the different conformations of the molecules resulting from the rotation of the C–C bond from the two implicated groups. Flexible carboxyl groups and rigid phenylphosphinate groups can offer multidentate binding sites for metals but can also provide different coordination modes.
CEPPA applications
The synthesis of polymers with functional groups is one of the most significant realizations in polymer chemistry because those compounds are everywhere. 28 Polymer properties can be changed by choosing a wide range of functional monomers, depending on the desired applications. 29
Phosphorus flame retardants have shown good performance in many polymer matrices 30 because of their advantages, such as low smoke production, lack of toxicity, and low halogen content. Some reactive retardants have already been used to improve the flame retardancy of polymers. Among them, CEPPA was used as a reactive flame retardant for polyethylene terephthalate (PET), proving its efficiency,31,32 to its high thermal stability and antistatic properties 33 and its capacity to copolymerize with other compounds.34,35 Table 3 lists several examples of CEPPA derivatives used as flame retardants.
CEPPA derivatives used as flame retardants.

Beyond flame retardancy, CEPPA-like phosphinic acid derivatives are being investigated for coordination chemistry and adsorbent materials, where the building of heterogeneous networks for metal ion capture and selective binding is facilitated by polar ligands with numerous donor groups. 42
Khaled, Z. et al. studied the anti-neoplastic potential of CEPPA, along with the pharmacotoxicological profile of this compound, confirming its biological safety. 23 Another application is based on the covalent inhibition of bacterial urease, which is a type of antimicrobial therapy. 43
All these studies point developing role of CEPPA as a functional ligand for advanced hybrid materials as well as a flame-retardant additive. Research is still being done to maximize performance of CEPPA by using its multifunctional chemical structure and interaction with metal centers.
Metal-based CEPPA derivatives
Due to their high attraction, steric, and electronic properties—which may be readily altered by changing the groups linked to the phosphorus atom—P-donor ligands are essential in coordination, organometallic chemistry, and catalysis. 42
Diamagnetic metal complexes with organic ligands represent a unique category of molecules whose magnetic properties are used in materials science and biomedicine. The sign and effective mass of charge carriers in semiconductors can be determined by magnetically sensitive tests, and similar metal–ligand systems are frequently employed in diagnostic procedures to examine and evaluate pathological alterations in human tissue. 44
Metal–organic frameworks (MOFs) are porous coordination polymers consisting of inorganic nodes connected by organic linkers. 45 The use of phosphinate-based linkers in metal–organic synthesis represents a great advantage because of their ability to form strong bonds with trivalent metals, leading to very stable MOFs.46,47
Ligands with carboxylic groups offer a variety of coordination models that can interact with transition metals. Carboxylic groups may be seen as auxiliary groups for phosphinic acid to assemble new frameworks.
CEPPA with bivalents 48 and trivalent metals form MOFs with interconnected units, resulting in unique properties, such as high porosity, excellent adsorbent materials, high specific surface areas, thermal stability, and open channels, providing adjustable sizes and topologies.
According to the literature, the structure and properties involved in the formation of metal–organic chains depend on several factors, including the coordination geometry of the metal ions, the nature of the organic ligands, the counterions, and the reaction conditions. These are some of the most important aspects to consider when forming metal–organic frameworks. Moreover, metal ions that have unpaired electrons are involved in the synthesis of compounds with magnetic properties, whereas lanthanide ions with f-f transitions are involved in the preparation of compounds with luminescent properties. The magnetic properties of MOFs 49 vary based on different ligand conformations, functional group coordination patterns, metal ion coordination modes, and structural types. The influence of pH is another important factor in the synthesis of autoassembly processes. From an FTIR point of view, the bonding between the phosphinic acid and the metal ions or the surface is reflected in the changed vibration from O–H to O–P–O. In the case of phosphinates bearing hydrogen atoms as substituents, the P–H stretching mode occurs at approximately 2400 cm−1 and does not overlap with the vibrations of the common groups.
Cd2+ and Bi3+-based CEPPA
Dong et al. 50 studied the temperature-dependent crystal self-assembly, disassembly, and reassembly of three cadmium-based compounds: [Cd(L)(bipy)(H2O)2], [Cd(L)(bipy)] and [Cd(L)(bipy)0.5]·0.5H2O. In the [Cd(L)(bipy)] compound, the L2− ligand binds with four Cd2+ atoms through two phosphinate oxygen atoms and two carboxylate atoms, whereas the bipy ligand is monodentate. [Cd(L)(bipy)0.5]·0.5H2O contains two Cd2+ ion units, one L2− anion and a half 4,4’-bipyridine (bipy), as well as a half molecule of water. The L2− ligand binds two cadmium ions through two phosphinate oxygen atoms—one cadmium atom by one monodentate carboxylate oxygen and one cadmium atom by one bidentate carboxylate oxygen. In conclusion, the removal of two aqua ligands and the loss of one half of the bipy ligand from [Cd(L)(bipy)(H2O)2] lead to the collective reorientation of [Cd(L)] chains, as well as to the formation of two compact bilayers in the [Cd(L)(bipy)] and [Cd(L)-(bipy)0.5]·0.5H2O compounds.
All complexes exhibit fluorescence in the near-UV to visible region, with emission maxima at 429 nm [Cd(L)(bipy)(H2O)2], 369/388 nm [Cd(L)(bipy)], and 379 nm [Cd(L)(bipy)0.5]·0.5H2O. These emissions are attributed to intraligand (IL) and ligand-to-ligand charge transfer (LLCT) transitions.
TGA reveals three weight-loss steps for [Cd(L)(bipy)(H2O)2] and [Cd(L)(bipy)0.5]·0.5H2O and two for [Cd(L)(bipy)]. The initial losses of [Cd(L)(bipy)(H2O)2] and [Cd(L)(bipy)0.5]·0.5H2O correspond to lattice water removal, followed by ligand decomposition. Compound [Cd(L)(bipy)] is stable up to 167 °C, while [Cd(L)(bipy)0.5]·0.5H2O shows the highest thermal stability.
Zhang and coworkers obtained a complex formed between CEPPA and cadmium ions, which presents a two-dimensional (2D) layer structure containing two types of dimeric ring motifs. [Cd(HL)2] exhibits a 2D layered structure in which the Cd²+ center adopts a distorted octahedral geometry, coordinated by four phosphinate oxygen atoms and two carboxylate oxygen atoms from six HL⁻ ligands. The Cd–O bond lengths range from 2.170 to 2.388 Å, with near-linear axial O–Cd–O alignment. [Cd(L)(phen)(H2O)]2H2O features a Cd²+ center in a distorted octahedral geometry, coordinated by phosphinate and carboxylate oxygen atoms from deprotonated L²− ligands, two nitrogen atoms from phen, and one water molecule. The L²− ligands adopt a bidentate bridging mode, linking two Cd²+ ions to form discrete centrosymmetric dimers. These dimers assemble into one-dimensional (1D) chains via hydrogen bonding, which further stack through π–π interactions and C–H···π contacts to generate a three-dimensional (3D) supramolecular network with 1D channels containing lattice water molecules. The thermal stability of the compound increases at 290 °C. [Cd(HL)2] and [Cd(L)(phen)(H2O)]2H2O have a significant ultraviolet fluorescence emission, which makes them viable options for luminescent materials. 51
Xue, C.C. et al. studied the pH dependence of cadmium-based compounds. For synthesis performed under normal conditions, without changing the pH, the compound [Cd(HL)2]n was obtained, and when the pH was changed to 5 to increase the pH, a different compound was formed, namely, [Cd3(L)2(OH)2]n. 52 The influence of pH has an important role because, by increasing its value, the ligand used is completely deprotonated, allowing each µ3-OH− group to bind three cadmium ions, forming an inorganic chain that is strengthened by phosphine groups. Both compounds showed thermal stability up to 300 °C and complete decomposition at 533 °C. Table 4 shows the main characteristics of both complexes at different pH values.
Characteristics of the complexes formed at different pH values.

Liu and his team synthesized a new complex based on Bi3+, 3-amino-1H-1,2,4-triazole-5-carboxylic acid, and CEPPA. According to their research, the resulted complex, [Bi2(μ3-O)(CEPPA)2]n, is a 2D coordination polymer, which forms a Shubnikov tetragonal plane net double layer. The thermal stability of the compound is up to 300 °C, with two steps of weight losses: the first noticeable weight loss occurs at 373 °C, followed by the second one in the range of 417–597 °C. 53
Co2+-based CEPPA
The cobalt ions have six coordination sites, resulting in distorted octahedral geometries, whereas the incomplete coordination sites are occupied by oxygen atoms of the functional groups or water molecules. The stable O–P–C–C–O–Co and O–P–O–Co–O–Co rings are formed in an infinite chain with octahedral geometry.
The electrochemical properties of Co2[(O2P(CH2CH2COO)(C6H5)(H2O)]2·2H2O were evaluated by performing various heat treatments. Although hydrogen bonding interactions are formed with the involvement of water molecules and the hydrophilic groups of the ligand, the solid has low proton conductivity. This phenomenon is attributed to the absence of extensive hydrogen bonding networks caused by the presence of large, hydrophobic phenyl rings. 54
Li+- and Zn2+-based CEPPA
The reactions between Li(I) and Zn(II) with CEPPA took place according to the literature. 55 The Li-based compound presents a double-layered structure, with a Li–O distance of 1.907(4)–2.012(4) Ä. In the case of lithium, the ligand binds four Li+ ions through two phosphinate oxygen atoms and one deprotonated carboxyl oxygen atom. The two layers are assembled through π···π interactions between the phenyl groups of adjacent layers. The zinc ion is surrounded by two phosphinate oxygen atoms and two carboxyl oxygen atoms with a distance of 1.888(2)–1.959(2) Å, which is much smaller than that of Li-O. The zinc compound has greater thermal stability than the lithium compound.
Zhou and coworkers synthesized and characterized two Zn-based polymorphs using 1,4-bis(imidazol-1-yl)-butane as a coligand. Although there are similarities in terms of chemical composition, molecular connectivity between the metal and linker, and ligand conformation, the difference between the two polymorphs is based on distinct structural features due to different intramolecular propagations. 56
Another zinc-based compound was synthesized by Guan, namely, [Zn(CEPPA)(phen)·5H2O]n, which was used as the coligand 1,10-phenanthroline. In the absence of metal ions, CEPPA forms a wavy 2D supramolecular layer-like aggregate in its single-crystalline structure. For the formation of the ladder-like structure to take place, it is important to consider two essential factors: the coordination sites of the metal ion and the coordination pattern of the acid. In this case, the zinc ion, which adopts a five-coordinate geometry, and the two sites occupied by the two N atoms of 1,10-phenanthroline were considered. 57
A few years later, the same author obtained a new complex based on zinc and CEPPA, which used 4,4’-bipyridine as a coligand, and the pH value of the solution was 6. The thermal stability of the compound reaches 372 °C, and the decrease in mass is attributed to the decomposition of the ligand in the temperature range of 327–570 C. 58
Al3+-based CEPPA
Yao Z and coworkers synthesized and characterized a new complex based on Al(III) and CEPPA. The mechanism, surface characteristics, and flame-retardant qualities of the obtained compound on polyester fabric were examined. According to their studies, after a flame period of 0 s, self-extinguishment, a decreased burning distance, and a B1 rating in the vertical flame test, the polyester textile containing 14.7 weight percent 2-carboxyethyl-phenyl-phosphinate (CPA-Al) received a limited oxygen index (LOI) value of 24.5%.
The degradation temperature of the formed compound increases at 300°°C; at 600°°C, the compound decomposes completely. 59
Pb2+-based CEPPA
Sun, Y.-H. and coworkers synthesized a new compound based on the reaction between CEPPA and lead ions. 60 As already reported in the literature, the phosphinic acid group adopts η2μ2 coordination by binding two lead ions, whereas the carboxyl group is not coordinated. The interconnection of lead ions by ligands leads to a symmetric double-chiral chain.
Na+- and K+-CEPPA
The addition of a strong base, such as NaOH or KOH, to a solution of CEPPA leads to the formation of two new compounds based on monovalent metals. 61 These compounds are the first examples of phosphinates with symmetrical hydrogen bonds, O···H···O. The sodium-based compounds exhibit a layered architecture, whereas the potassium-based compounds present a chain-like matrix. These differences are attributed to the distinct modes of coordination of the phosphinate ligands.
Mn2+- and Cu2+-CEPPA
The L2− anion can provide different coordination modes through its four potential O-donor sites, even with the same number of nodes. The complexes [Mn(L)(phen)(H2O)]·2(H2O) and [Mn(L)(phen)(H2O)] are derived from different coordination modes of the tridentate L2− ligands, as shown in Figure 3.

Coordination modes of the tridentate L2− ligands.
[Mn(L)(phen)(H2O)]·2(H2O) has a dimeric structure, and [Mn(L)(phen)(H2O)] has a double-chain structure. Three of the six coordination sites are occupied by a phosphinate O atom and by a bidentate carboxylate group of the two L2− ligands, and the remaining sites are occupied by the bidentate phenyl chelate ligand and a terminal aqua ligand. 18 The [Mn(L)(phen)(H2O)] complex is bound by three L2− ligand manganese ions via a carboxylate group and two O phosphinate atoms.
Copper-based CEPPA was also synthesized and characterized by Xue et al. The magnetic properties of the resulting compound showed weak ferromagnetic interactions between the Cu(II) ions through O–P–O bridges. 62 The crystallographic data of the formed complexes are summarized in Table 5.
Selected crystallographic data for the formed complexes.

Conclusion
In summary, CEPPA is a versatile compound that can adopt different dentate (mono-, bi-, tri-, tetra-) coordination modes to generate new metal-based compounds. Depending on the desired compounds, this acid can be used alone or in combination with a coligand. This review aims to attract interest in organometallic chemistry to generate future applications with this type of new compounds.
Footnotes
Ethical considerations
Ethical approval is not applicable for the article.
Consent to participate
This paper has no human participants, and informed consent is not required.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the West University of Timisoara.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data discussed in this review are included in the published articles cited in the references.

