Abstract
The field of dental implants underwent a transformation with nanotechnology, which promises enhanced bone alignment and increased antibacterial properties. The systematic review evaluated the latest evidence about nanoceramic coatings in dental implants and their effects on clinical outcomes. The research team conducted a systematic literature search across multiple databases, including PubMed, ScienceDirect, Scopus, Google Scholar, and The Cochrane Library to locate studies published between January 2004 and June 2024 and performed this search following PRISMA guidelines. Selected studies evaluated dental implants enhanced with nanoceramic coatings or modifications concerning osseointegration rates, antimicrobial efficacy, biocompatibility, and implant stability. Two researchers individually evaluated data selection and quality using the QUIN quality assessment tool. This review included 10 studies, and different nanoceramic coating techniques, such as immersion coating, spin coating, and fabrication, were identified. The results showed that nanoceramic coating significantly improved osseointegration. Antimicrobial properties were remarkably enhanced, with nanoparticle-treated surfaces effectively inhibiting bacterial growth. Biocompatibility assessments demonstrated that nanoceramic-coated implants supported cell viability and proliferation, while stability tests indicated improved implant integration and mechanical strength. All studies were of medium methodological quality. Nanotechnology shows great promise for improving the function of dental implants. Advanced osseointegration, with high antibacterial properties and biocompatibility, suggests that nanoceramic-coating implants have better clinical outcomes. Future research should focus on long-term clinical trials to establish these findings and highlight and explore the potential of various nanoceramic materials in clinical settings.
Introduction
Nanotechnology has a significant impact on the field of dentistry, offering a variety of applications to improve the quality and efficiency of dental treatments, especially in the field of dental implants and can play various important roles, including providing antimicrobial and anti-inflammatory properties as well as promoting re-mineralization effects. 1 These developments have improved dental biomaterials through the addition of nanosized particles, which are helpful in improving implant surface properties and promoting better bone osseointegration. 2 The unique properties of nanoparticles, arising from their nanoscale size, position them as promising tools in dentistry for applications such as diagnosis, prevention, and treatment of oral diseases. 3 These developments are important because they can improve the durability of implant procedures and durations related to dental implants. 4 These material advancements contribute to the longevity and performance of dental restorations, ensuring better outcomes for patients undergoing restorative procedures. In addition, the production of nanocomposites and nanoceramics used for modifying dental implant surfaces has been facilitated by the use of nanotechnology and is helpful in enhancing their biocompatibility and performance. 1 These modifications are essential for improving the integration of implants with surrounding bone tissue, ultimately leading to better clinical outcomes for patients undergoing implant procedures.4,5 In addition, the utilization of smart nanoparticles in dental applications, such as nanoclays, nanofibers, and metallic nanoparticles, demonstrates the versatility and potential of nanotechnology in addressing various dental challenges.6,7 Overall, integrating nanoparticles into implant coatings, the goal is to offer osteoconductive and antimicrobial functionalities to prevent dental implant failure. 8
Researchers have explored the development of antimicrobial coatings using nanoarchitected materials to enhance the antibacterial properties of dental implant surfaces. By incorporating bioactive components and employing nano-coating techniques, efforts are being made to improve implant surface characteristics crucial for promoting osseointegration and preventing infections. 9 There are different types of nanoparticles, for instance, silver nanoparticles, known for their antimicrobial properties, have attracted attention for their potential in the prevention of infections associated with dental implants, 10 and these nanoparticles can influence cell behavior in culture mediums, indicating their potential in improving the performance of dental implants. 11 In addition, zirconium nanoparticles derived from plants and biomass have also shown promising outcomes in dental implant settings. 12 Meanwhile, the effect of titanium nanoparticles on dental implant surfaces has also been studied. Studies have shown that titanium nanoparticles at submicron-nanoscale sizes can affect cellular behavior, emphasizing the importance of understanding the cytotoxic effects of debris from tissue implants.11,13 In addition, antibacterial metal elements and metal oxide nanoparticles are used to modify dental implant surfaces, which has been proposed as a strategy to prevent bacterial infections post-implantation, highlighting the role of nanotechnology in improving implant success rates. 14 Moreover, iron oxide nanoparticles can also be helpful in the eradication of the infections and complications associated with implant procedures. 15 Furthermore, calcium phosphate nanoparticles utilized for coating implant surfaces present a novel approach for the enhancement of the dental implant osseointegration, further emphasizing the significance of nanotechnology in improving implant outcomes. 16 This antimicrobial effect is crucial for maintaining oral health and preventing peri-implant diseases, ultimately improving the longevity and success rates of dental implants.16,17 These diverse applications of nanoparticles highlight the versatility and potential of nanotechnology to enhance various aspects of dental implantology.
Overall, nanotechnology has brought a significant advancement to the field of dental implants by providing innovative solutions to enhance implant biocompatibility, prevent infections, and improve osseointegration. 18 The utilization of various nanoparticles, such as silver, titanium, copper, and zirconium nanoparticles, has shown promising results in enhancing the performance and durability of dental implants. Given the increasing application of nanotechnology in medical and dental fields, it is crucial to synthesize current evidence to understand the potential of nanoceramic materials in enhancing the integration of implants with bone. This study evaluated various outcomes related to implant stability, bone–implant contact, osseointegration, and safety/antimicrobial activity, providing a rigorous analysis of whether nanoceramics improve clinical results. By consolidating findings from different studies, this systematic review seeks to offer a clear, evidence-based perspective that could guide future research and clinical practice in dental implantology, potentially leading to improved patient outcomes and advancements in implant technology.
Methodology
This study was performed according to the Preferred Reporting for Systematic Reviews and Meta-analysis (PRISMA) protocols, which provide clear steps for the study selection process for better transparency and reproduction of the research articles. 19 The study was registered on the International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) under registration number 202540113.
Search strategy
Using the PIO framework as a foundation, the search approach was developed: Population (P): dental implants that are utilized in dentistry, Dental implants that have been treated with nanoparticles constitute the intervention (I). In terms of outcomes, osseointegration, stability, compatibility, bone–implant contact, safety, and antibacterial activity are all considered. For the purpose of conducting an advanced search, a number of databases, such as ScienceDirect, PubMed, Scopus, Google Scholar, and The Cochrane Library, were utilized. The search was restricted to publications that were published between January 2004 and June 2024. Different keywords such as “Nanotechnology” OR “Nanoceramics” OR “Nanotopography” OR “Titanium dioxide nanoparticles” OR “Nano hydroxyapatite” AND “Osseointegration” OR “Dental implant stability” OR “Implant stability” OR “Long term stability” OR “Long term implant stability” OR “Dental implantology” OR “Bone implant contact” and combination of these keywords were also utilized (see supplemental Table 1).
Eligibility criteria
Inclusion criteria
Specific criteria for inclusion of studies were used: Studies utilized nanoparticles for the treatment of dental implants. Studies provide information at least for osseointegration parameters, including bone–implant contact, implant stability, safety, and antimicrobial efficacy. In vitro, randomized controlled trials (RCTs), cohort studies, and case-control studies published in English.
Exclusion criteria
Similarly, specific criteria for the exclusion of studies were also used. Studies with incomplete information and required outcomes were performed on animals, and utilized non-nanoparticles or ceramics. Reviews, case reports, and animal studies published in a non-English language.
Study selection
The reviewers (R.I.H.B. and V.N.V.M.) screened research article titles and abstracts during the initial phase while they followed the study’s objectives to make selections. The selection process excluded research paper copies from further consideration by using the EndNote X9 referencing program after the initial screening. Following this, full-text screening was carried out. Researchers selected publications for the study using predetermined inclusion and exclusion criteria and included those works that met the inclusion criteria. The third reviewer (V.G.) mediated discussions between other reviewers when disagreements about research papers emerged.
Data extraction
Pre-defined data variables were extracted using a Microsoft Excel sheet (see supplemental Table 2). The included variables were study characteristics (study ID, country, study design, sample size, age, and gender), intervention characteristics (number of implants, implant type, implant size, type of nanoceramics coating, NP diameter, composition, surface modification technique, and duration for process), and outcomes (osseointegration, bone-to-implant contact, bone remodeling, safety/antimicrobial activity, biocompatibility, stability, key findings, and conclusion).
Quality assessment
For the methodological quality assessment, the QUIN assessment tool was utilized, for in vitro studies and the quality assessment was done according to the 12 items, and each study was rated as yes (1–2 points), no (0 points) or not applicable [20]. It was purpose-built through a Delphi panel process and based on 11 items, along with scoring and grading options. Later, each research article was graded according to the points response. High risk of bias (RoB) scores <50%, 50%–70% were the medium RoB, and >70% were the low RoB. 20
Statistical analysis
Qualitative and quantitative data were organized using the Microsoft Excel sheet for the construction of tables and graphs.
Results
Literature searched
A literature search performed through multiple electronic databases found and retrieved 1295 research papers by employing different keywords. The initial step in identification involved finding 90 duplicate research papers and removing them. During the second phase of the screening process, researchers performed additional screening and evaluation of 1205 research papers by examining their titles and abstracts. The research team excluded 1182 articles from their study because they did not meet the relevance criteria and consisted of review designs or were published in languages other than English. Researchers analyzed full-text versions of 23 publications throughout the eligibility phase. Figure 1 displays 13 research publications that were eliminated during this phase for multiple reasons. The final step of the inclusion procedure resulted in the selection of 10 research papers to be included in the systematic review. The lack of sufficient uniform and necessary data prevented any study from being selected for inclusion in the meta-analysis procedure.
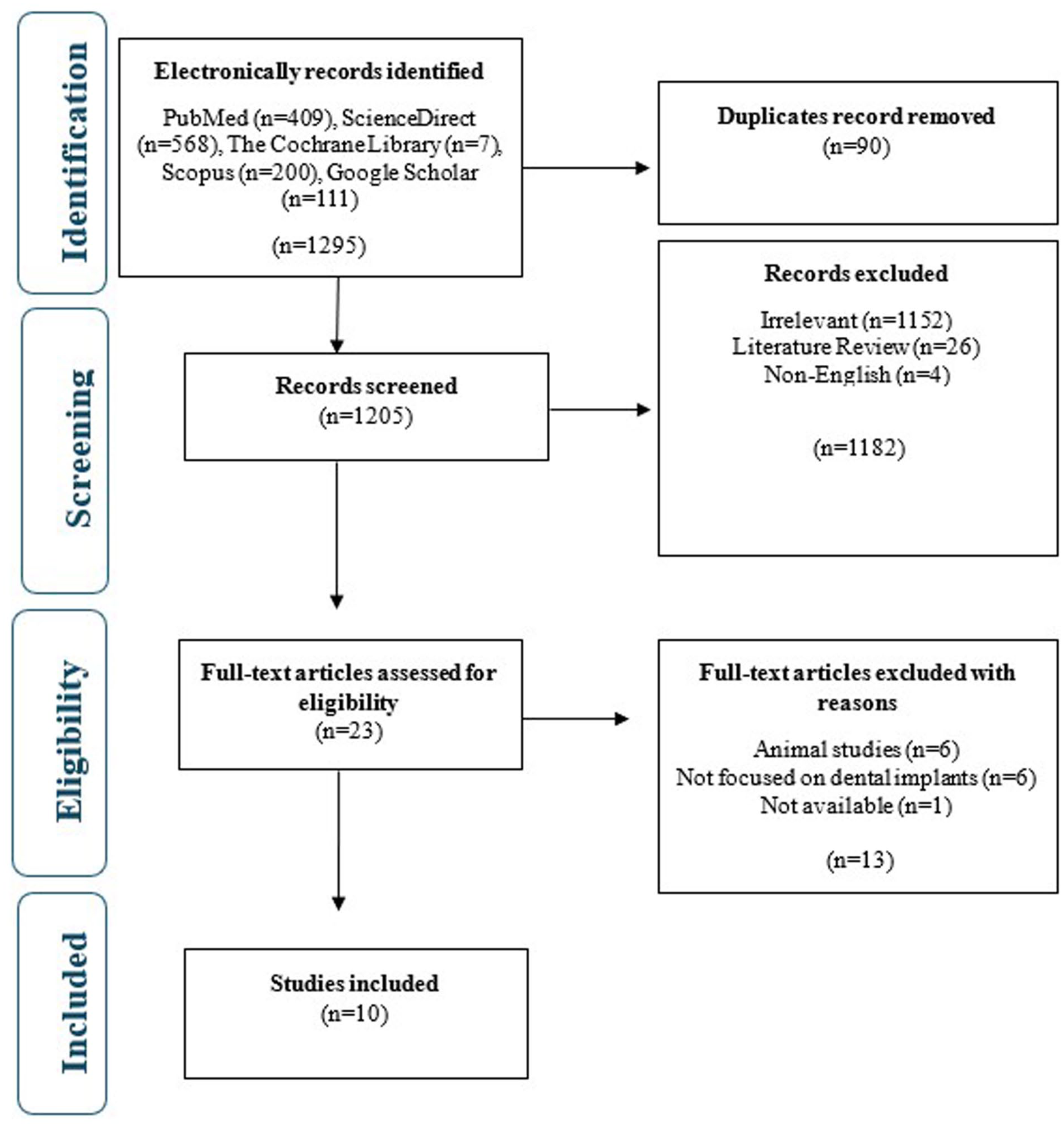
PRISMA flow chart for the selection of studies.
General characteristics
Most of the studies were reported from China,21,22 and each study from the United Kingdom, 23 Spain, 24 Malaysia, 25 Poland, 26 Greece, 27 Egypt, 28 South Korea, 29 and Romania. 30 All of the following in vitro study designs have a maximum of 40 dental implants 29 and a minimum of 6. 23 All of the studies utilized titanium-based dental implants, except one study, which utilized root-form implants. 28 In terms of implant size, 3.3 mm was the smallest dental implant 29 while 10 mm was the longest diameter. 26 Likewise, various types of nanoceramics/nanoparticles coatings were identified as dental implant surface modifiers; for instance, silver was utilized in most of the studies.22,26,27 Similarly, different surface modification techniques and their durations for enhancing dental implants were utilized in the included studies like immersion coating for 30 s 23 and 5 min, 27 coating method for 30 min,24,28 spin coating method for 3 s,21,30 fabrication for 120 min, 25 Tollens reaction immersion for durations of 30 s, 2, 5, and 10 min. 26 While Shin Bae 29 used a coated sandblasted, large-grit, and acid-etched (SLA) method (Table 1).
Summary of general characteristics of dental implants and nanoceramics.

CHX = chlorhexidine, HMP = hexametaphosphate, D = diameter, L = length, H = height, rGO = reduced graphene oxide, HA = hydroxyapatite.
Outcomes
Table 2 provides a summary of various studies examining the effects of different surface modifications on dental implants, focusing on factors such as osseointegration rate, bone-to-implant contact, safety/antimicrobial effect, biocompatibility, stability, and overall conclusions. Due to the variation in materials, methods, and outcome reporting, findings were summarized narratively by examining the direction and consistency of the reported effects rather than through numerical pooling or meta-analysis.
Summary of outcomes of dental implants treated with nanoparticles.

rGO = reduced graphene oxide, HA = hydroxyapatite, NA = not available.
Overall, dental implants coated through the utilization of nanoceramics demonstrated enhanced antimicrobial efficacy against different types of pathogens, including
Quality assessment
The risk of bias of selected studies is evaluated and mentioned in Table 3. All of the studies were found to have a medium risk of bias as their quality scores fall between 50% and 70% (Table 3). Two of the domains were not applicable to the study, that is, operator details and outcomes assessor’s details. However, all studies had zero scores in the domain of randomization and blinding.
Methodological quality assessment of included studies.

NA = not available, M = medium, Yes = 2, No = 0.
Discussion
The first promising avenue for dental implant coating is through surface engineering that would include nanoscale coatings for faster and better osseointegration, bone–implant contact, stability, compatibility, and safety/antimicrobial activity of implants designed to function for the lifetime of the individual. Overall, a positive impact of nanoceramics coating was highlighted in the present study. However, the included studies had a moderate risk of bias. This may be attributed to the predominance of in vitro studies, where traditional clinical studies’ methodological features are not always applicable, routinely implemented or documented. In many in vitro studies, samples are usually allocated based on the material preparation protocols rather than using a random approach. Likewise, blinding is considered unnecessary, as outcomes are measured using objective mechanical or microscopic techniques.
The overall methodological quality of included studies was rated as having a moderate risk of bias due to the majority of in vitro design studies. The QUIN domains of randomization, blinding, and sample size calculation were most commonly not reported or were scored low. In implant studies in vitro, allocation of specimens is frequently based on the order of material preparation rather than randomization, and objective mechanical, physicochemical, or microscopic testing of the response variable does not allow for blinding. Moreover, sample size calculations are not usually performed for in vitro implant studies, as an exploratory screening of materials is typically followed by confirmatory testing of a smaller number of selected materials. These methodological aspects do not negate the biological trends observed in these studies, but they do affect the overall level of certainty of the evidence and, therefore, the conclusions of this review. The current findings must be considered mechanistic and hypothesis-generating for future confirmatory in vivo and clinical studies using standardized protocols, appropriate controls, and better reporting that can provide insights into the translational impact of nanoceramic surface modifications.
Even though a single study reported the impact of nanoceramics coating on osseointegration and found positive with 100% improvement was observed because the coating increase implant surface area and enhance cell synthesis and proliferation, which is important for bone healing and integration can mimic the natural bone environment, and provide large positive surfaces for cells responsible for bone formation after specific nanoceramic coatings are known to be bioactive, and osteoconductive, which promotes direct contact with bone. 31 As it is well established that osseointegration requires 8–12 weeks on micro roughened titanium implants. 32 Therefore, the combination of mechanical and chemical interactions facilitated by the nanoscale topography of the bone may have resulted in stronger and faster osseointegration. Several histological studies provide evidence that intraosseous coatings provide more contact with bone than mechanical osseointegration of titanium.33,34 Moreover, with the application of nanoparticles, dental implant surface energy, roughness, composition, and topography can be modified, which is also helpful for better and faster osseointegration. 35 Likewise, another review also aligns with our findings that nanoceramics coating induces osseointegration, osteoconduction, osteoinduction, and osteogenesis. 36 Another study also highlights the same outcomes, which are that nanoparticles utilized for surface modification of dental implants can induce and improve osseointegration. 37
Similarly, nanoceramic coatings were also found to be very effective against pathogens and postoperative infections. This may be due to their unique biochemical properties and multiple mechanisms of action. They have a surface size-to-volume ratio due to their small size, which facilitates interaction with bacterial cell contact.
38
These nanoceramic coatings can damage bacterial cell membranes by direct physical interaction, causing cell damage.
39
In addition, they can generate reactive oxygen species (ROS) that cause oxidative stress and damage cellular components such as DNA, proteins, and lipids.
40
Many nanoparticles, such as silver, also release metal ions that interfere with important microbial enzymes and metabolic processes and inhibit growth and reproduction.
41
Furthermore, the ability to penetrate biofilms and prevent their formation makes nanoparticles especially effective at implant-related chronic infections, reducing the chances of progression and thus ensuring their continued efficacy in dental settings.
42
Cytotoxicity findings highlighting dose-size and surface-dependent effects of nanoceramic coatings, and also associated with the release of particles, where increased cell viability and proliferation were observed in the slow release of nanoparticles.
43
However, clinically, no study reported adverse effects of nanoparticles, like silver, which can cause aesthetic problems due to discoloration of the mucosa.
43
Therefore, the adverse effects are dependent on the physiochemical properties of the released nanoparticles.
44
Meanwhile, several studies are in agreement with our findings, for instance, a review concluded that dental implant surface modification utilizing nanoparticles enhances antimicrobial properties and can be helpful in decreasing infection rates and improving clinical outcomes.
37
Similarly, fully characterized silver chitosan nanoparticles proved to have an inhibitory impact on the growth and development of major dental pathogens, including

Nanoceramic surfaces enhance osseointegration.
A systematic review of nanotechnology in dental implants highlights several strengths, including the potential to extend the current research, providing a clear overview of nanoceramics efficacy and safety to drive implant performance. The study emphasizes improved osseointegration, antimicrobial properties, and biocompatibility of nanoceramic-coated implants, and infection rates appear low. However, limitations include possible variation in study methods, heterogeneity in nanoceramic coatings types and surface modification methods, and inconsistent reporting of clinical outcomes in studies failing to perform meta-analyses. Another limitation may be the inclusion of studies with a medium risk bias. Further high-quality studies are needed to confirm the theory and safety of the study, and challenges are also encountered in translating in vitro findings into human clinical practice, emphasizing the need for standardized protocols and detailed human studies.
Biosafety, cytotoxicity, and long-term biocompatibility considerations
The biosafety profile of nanoceramic surface modifications, including potential cytotoxicity and immune response modulation, remains an important consideration for clinical application. Cytotoxicity of nanoceramic coatings has been reported to be influenced by factors such as particle size, concentration, surface chemistry, and release kinetics. Several in vitro studies have shown that controlled or slow release of nanoparticles, particularly silver- and calcium-based ceramics, can support cell viability and osteoblastic proliferation, while excessive release of ions or particles may lead to oxidative stress and cytotoxicity. In addition to direct cytotoxic effects, nanoceramic coatings may impact local immune and inflammatory responses. 50 Surface-modified implants have been shown to modulate macrophage behavior and cytokine expression, which could promote a pro-healing local environment; however, dysregulated immune activation is a concern in the absence of long-term in vivo data. Furthermore, long-term stability of nanoceramic coatings under functional loading and oral environmental conditions is not well studied. Progressive detachment of nanoparticles or release of ions over time may raise concerns regarding chronic inflammation, altered peri-implant–tissue response, or systemic exposure. 51 Due to these considerations, current data should be considered preliminary with respect to biosafety. Standardized testing for toxicity, long-term in vivo studies, and clinically relevant exposure models are needed to determine safety thresholds and long-term biocompatibility of nanoceramic-coated dental implants before widespread clinical use. 52
The included studies were somewhat inconsistent with respect to reporting on physicochemical characterization of nanoceramic synthesis routes. Nevertheless, the information provided in the studies may be qualitatively summarized by combining the described biological effects with well-known tenets of biomaterials science. Indeed, the choice of coating and fabrication strategy determines properties such as surface topography and surface energy, as well as nanoarchitecture and ion release, which are important to the effects on osseointegration, antimicrobial properties and biocompatibility. Table 4 provides an overview of the typical nanoceramic preparation and coating methods alongside their expected biological effects on dental implants.
The link between methods of nanoceramic synthesis and coating techniques in dental implants and their observed biological performance.

This table provides a qualitative summary based on reported trends across included studies, as well as from established principles in biomaterials science.
In order to help to place the nanoceramic surface strategies into a larger context and interpret their scope, it is possible to classify the ceramic materials considered for dental implant surface engineering into the classes described in Table 5 according to their structural and functional features. While the following ceramic classes were not all represented by the studies included in the systematic review, the subsequent classification suggests new material classes of potential importance for future developments in implant surface design and biological performance.
This study categorizes ceramic materials used in dental implant surface engineering while outlining their potential functional roles.

This classification is for reference only and does not suggest that the ceramic classes listed have all demonstrated established clinical efficacy in dental implant applications.
Author perspective and identified knowledge gaps
The aim of the present systematic review was to evaluate the current evidence regarding whether there are consistent biological benefits of nanoceramic surface modifications for dental implants across multiple experimental models. This evaluation is not intended to prove clinical efficacy but rather to provide a basis for developing future research directions. In addition to providing a synthesis of current study-level evidence, we found that the existing body of research on this topic suggests a number of important considerations. In general, the high variability in nanoceramic composition and fabrication methods reported across studies precludes definitive conclusions regarding the optimal design parameters for this type of implant surface modification. However, across most studies, in vitro trends in osseointegration and antimicrobial efficacy have been favorable and consistent, suggesting that nanoscale surface engineering is a broadly valid design principle, rather than being specific to certain materials or fabrication methods. At the same time, several important knowledge gaps currently limit translation. These include the need for standardized physicochemical characterization of nanoceramic coatings, more quantitative and standardized reporting of biological outcomes, increased focus on immune and inflammatory outcomes, and the near absence of long-term in vivo and human clinical data. In addition, there is currently a notable lack of direct comparisons between different nanoceramic systems under standardized conditions, which limits the ability to determine which may be the most effective. Finally, there is a clear need for future research to transition from primarily exploratory in vitro screening experiments to hypothesis-driven, translational approaches with standardized outcome measures and more clinically relevant models. By highlighting these gaps, we hope the present review can help to guide future research efforts and facilitate the responsible translation of nanoceramic technologies into dental implant practice.
Current challenges and potential strategies for clinical translation
In spite of the encouraging in vitro results, some factors are still hindering the clinical translation of nanoceramic surface modifications. 53 The long-term mechanical and chemical durability of nanoceramic coatings under functional loading and the oral environment is one issue. Degradation, delamination, or uncontrolled release of nanoparticles and ions over time can lead to diminished implant performance and peri-implant tissue health. In addition, there are concerns about the biosafety and cytotoxicity of nanoparticles. Even though they have shown acceptable biocompatibility in short-term in vitro studies, long-term cellular effects, immune system changes, and systemic exposure are not well understood. Because of the variability in particle size, surface chemistry, and release kinetics, it is difficult to determine safe exposure thresholds. Developing better coating techniques alongside controlled-release systems for nanoparticles and standardizing physicochemical characterization protocols could provide answers to current challenges. Furthermore, the incorporation of advanced in vivo models, long-term follow-up studies, and standardized biological outcome measures can improve comparability across studies and increase translational relevance. Collaborations between materials scientists, microbiologists, and clinicians will be crucial in the development of nanoceramic implant designs that optimize biological performance while also ensuring long-term safety. 54
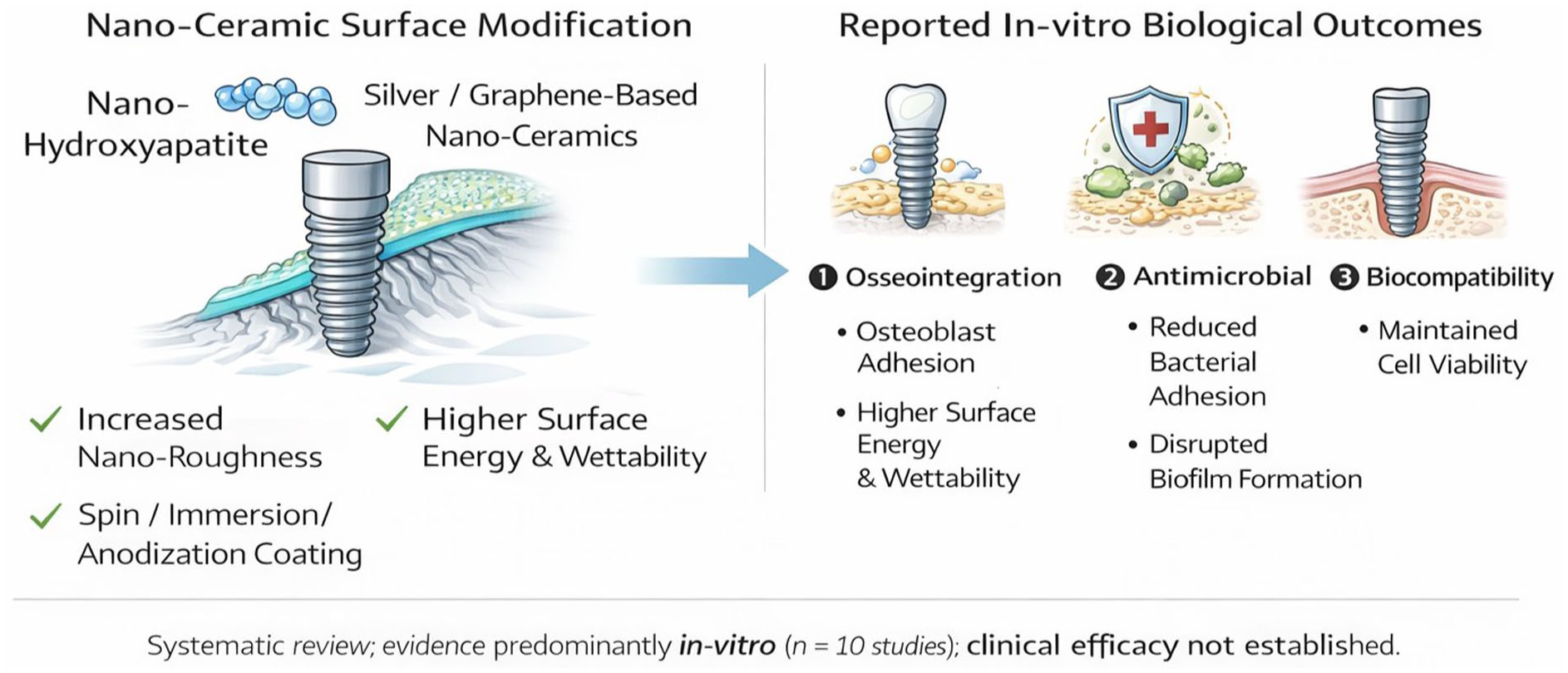
To increase clarity and aid in rapid visual interpretation, a conceptual schematic summarizing nanoceramic coating strategies and proposed biological interactions is provided (Figure 3). This figure incorporates data from the studies highlighted in this review and fundamental biomaterials concepts to summarize potential ways that nanoscale surface modifications could impact implant–tissue interactions; these are not meant to suggest direct clinical translation.

Various nanoceramic coating strategies used for dental implants with their intended impact on surface properties and biological response. The figure shows mechanistic interactions based on in vitro data and biomaterial concepts and should not be considered as evidence of clinical efficacy.
A limitation of this study is our restriction to English-language publications and the limited number of eligible animal or clinical studies which may introduce selection bias and may further limit the generalizability of our findings.
Future directions
Future research on nanotechnology in dental implants should focus on:
Long-term clinical trials to demonstrate the efficacy and safety of nanoparticle-based surface modifications in large patient populations.
Development of multifunctional nanoparticles combining antimicrobial and anti-inflammatory properties of the implant to enhance the success rate.
The development of biodegradable nanoparticles that also release the required bioactive compounds can lead to sustainable therapeutic effects.
Interdisciplinary collaboration integrating materials science, microbiology, and clinical dentistry will be critical to advances in this area.
Standardization of nanoparticle-based protocols and evaluation of cost-effectiveness will be important for greater clinical adoption and improved outcomes in patients.
Clinical implication
Results from this systematic review of the literature show that nanoceramic coating appears to in vitro consistently improve multiple important biological and physicochemical implant performance parameters, including ones directly involved in osseointegration, antimicrobial action, and biocompatibility. Nanotopographies are thought to potentially affect implant–tissue interactions due to their increased surface area, altered surface energy and wettability, and altered cell–material interactions. While it may seem that this is evidence of significant potential for nanoceramic coatings for dental implants, one must be careful in how the results of this systematic review are interpreted. The majority of the evidence supporting any of these conclusions comes from in vitro research of moderate quality with a lack of standardization in design, materials, and evaluated outcomes. For this reason, no conclusions can be made regarding clinical efficacy, long-term safety, or implant success at this time.
Conclusion
Results from this systematic review of the literature show that nanoceramic coating appears to in vitro consistently improve multiple important biological and physicochemical implant performance parameters, including ones directly involved in osseointegration, antimicrobial action, and biocompatibility. Nanotopographies are thought to potentially affect implant–tissue interactions due to their increased surface area, altered surface energy and wettability, and altered cell–material interactions. While it may seem that this is evidence of significant potential for nanoceramic coatings for dental implants, one must be careful in how the results of this systematic review are interpreted. The majority of the evidence supporting any of these conclusions comes from in vitro research of moderate quality with a lack of standardization in design, materials, and evaluated outcomes. For this reason, no conclusions can be made regarding clinical efficacy, long-term safety, or implant success at this time.
Supplemental Material
sj-docx-1-chl-10.1177_17475198261430494 – Supplemental material for Nanotechnology in dental implants: Impact of nanoceramics on osseointegration—A systematic review
Supplemental material, sj-docx-1-chl-10.1177_17475198261430494 for Nanotechnology in dental implants: Impact of nanoceramics on osseointegration—A systematic review by Ravinder S Saini, Rayan Ibrahim H Binduhayyim, VNV Madhav, Vishwanath Gurumurthy, Sarah Monserrat Lomelí-Martínez, Mario Alberto Alarcón-Sánchez, Javier Flores-Fraile and Artak Heboyan in Journal of Chemical Research
Supplemental Material
sj-xlsx-2-chl-10.1177_17475198261430494 – Supplemental material for Nanotechnology in dental implants: Impact of nanoceramics on osseointegration—A systematic review
Supplemental material, sj-xlsx-2-chl-10.1177_17475198261430494 for Nanotechnology in dental implants: Impact of nanoceramics on osseointegration—A systematic review by Ravinder S Saini, Rayan Ibrahim H Binduhayyim, VNV Madhav, Vishwanath Gurumurthy, Sarah Monserrat Lomelí-Martínez, Mario Alberto Alarcón-Sánchez, Javier Flores-Fraile and Artak Heboyan in Journal of Chemical Research
Footnotes
Acknowledgements
All the authors are thankful to the King Khalid University, Saudi Arabia, for the financial support.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through the Small Research Project under grant number RA/KKU/12/46.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
