Abstract
Straw biochar loaded with zero-valent iron, which could remove the tetracycline in the wastewater by liquid phase reduction and activated peroxydisulfate, was prepared at different pyrolysis temperatures (500 °C, 700 °C, and 900 °C). The zero-valent iron supported on modified activated carbon was characterized by scanning electron microscope, X-ray diffraction, and Brunauer−Emmett−Teller. Batch experiments were carried out to investigate the effect of different factors, such as the different peroxydisulfate concentrations, straw biochar loaded with zero-valent iron dosages, reaction temperature, initial pH, and various inorganic anions, on the degradation efficiency of straw biochar loaded with zero-valent iron. The straw biochar loaded with zero-valent iron prepared at a pyrolysis temperature of 900 °C/peroxydisulfate system exhibited excellent degradation effects on tetracycline, which was degraded and removed through the adsorption and sulfate radical oxidation of the novel catalyst. According to the experimental results, 0.05-g biochar-supported zero-valent iron prepared at a pyrolysis temperature of 900 °C catalyst could remove 93% of tetracycline within 30 min at a temperature of 323K and peroxydisulfate concentration of 0.5 g·L-1. And it was less affected by reaction temperature, initial pH, and various inorganic anions. In terms of stability and reusability, biochar-supported zero-valent iron prepared at a pyrolysis temperature of 900 °C remained stable and reusable after three adsorption-desorption cycles. Excellent catalytic activity and stability made it highly valuable for practical applications. This study provided a promising strategy for water treatment for antibiotic-containing wastewater.

Introduction
Global consumption of antibiotics has been steadily increasing due to their extensive use in treating diseases in humans and animals, promoting growth, and preventing infections. Nevertheless, antibiotics often fail to be fully metabolized by the body during use, thereby causing adverse environmental impacts on non-target species. 1 As a result, addressing ecological pollution caused by antibiotics has become a significant research priority.
In recent years, sulfate radical based advanced oxidation processes (SR-AOPs) have been proven to be effective in eliminating emerging organic pollutants such as antibiotics from water environments. 2 Persulfate containing peroxymonosulfate (PMS) and peroxydisulfate (PDS) possess advantages such as high solubility and stability in water, coupled with their extensive reactivity with environmental pollutants. 3 To achieve better activation and utilization, various efficiency-enhancing methods have been explored, such as combining metal-organic framework (MOF) materials, 4 transition metal oxide materials, 5 zero-valent metal materials, 6 or carbon materials, 7 with thermal, 8 photocatalytic, 9 and heterogeneous catalysts.
Compared with those energy-based activation methods, heterogeneous catalysts offer high economy and a simple reactor/system configuration in activating PDS. 10 Most transition metal ions can activate PDS to generate reactive oxygen species. Remarkably, the environmentally friendly, non-toxic, and cost-effective zero-valent iron (ZVI)-based catalysts are available. However, due to its high surface energy and magnetic interaction, ZVI is highly prone to agglomeration, thereby generating hydroxides and resulting in a decrease in activity. 11 To overcome this limitation, biochar produced under low-oxygen conditions has been recognized as an effective supporting material. Its porous structure and high specific surface area facilitate the dispersion and stabilization of nanoparticles. 12 Moreover, its high adsorption performance and abundant oxygen-containing functional groups promote mass transfer in catalytic reactions and accelerate chain reactions. 13 These characteristics make biochar a promising candidate for constructing composite catalysts. 14
Therefore, combining ZVI with biochar is expected to create a synergistic system for efficient pollutant degradation. Although the catalytic activity of Fe-doped carbon materials is well established, their practical application is often limited by complex synthesis routes, multiple procedural steps, and the requirement for additional reagents, which raise economic and environmental concerns. Thus, developing a simple and efficient method for synthesizing such composites remains a challenge.
In this study, we propose a straightforward strategy to fabricate a high-performance catalyst by supporting ZVI on waste straw-derived biochar. This approach aims to achieve highly efficient tetracycline degradation via activated PDS while simultaneously upcycling agricultural waste. This work not only offers a novel and practical technology for antibiotic removal but also offers important guidance for the development of sustainable environmental remediation materials.
Results and discussion
Characterizations of Fe@BC
The surface morphologies of the prepared composites were characterized by scanning electron microscopy (SEM) and are presented in Figures 1(a)–(f). As shown in the Figure 1(a), Biochar, after rapid pyrolysis at 900°C, is mainly in the shape of lumps with a smooth surface. As shown in the Figure 1(b), the ZVI particles are spherical particles with different sizes, but most of them form chain-like aggregates. After loading ZVI into biochar (Figure 1(d)–(f)), it can be observed that there are irregular spherical particles on its surface, which are iron oxides bound to the surface of the biochar, with a rougher surface and obvious pore structure. It could provide a large number of effective and active adsorption sites for adsorption, and help in the adsorption of tetracycline molecules at the monolayer level. Figure 1(c) shows the SEM images of the reacted Fe@BC900, and it can be seen that the surface of the reacted Fe@BC900 has become smooth with a lamellar structure, which indicates that corrosion occurred during its adsorption of activated PDS. The EDX element maps stated the distribution of C, O, and Fe on Fe@BC900, proving that each element is uniformly distributed on the surface of Fe@BC900 (Figure 1(g)–(i)). The primary elemental compositions of Fe@BC900 were C (35.3 wt%), O (29.8 wt%), and Fe (34.9 wt%).

SEM images of (a) BC900, (b) ZVI, (c) Fe@BC900 after the reaction, (d) Fe@BC500, (e) Fe@BC700, and (f) Fe@BC900. (g–i) Corresponding EDS elemental mapping images of Fe@BC900.
The XRD pattern of the Fe@BC in Figure 2 confirms a prominent characteristic peak at 44.7 corresponding to the (110) crystal plane of metallic Fe (Fe0) (JCPDS No. 87-0722). This revealed that Fe@BC was obtained by loading ZVI onto biochar, compared with the characteristic peaks of the composites corresponding to ZVI.

(a) XRD patterns of Fe@BC500, 700, and 900. (b) FTIR spectra of Fe@BC900.
To further investigate the structural characteristics of the Fe@BC catalysts, N₂ adsorption-desorption tests were conducted. The three samples have characteristic type IV N₂ adsorption-desorption isotherms at high pressure, together with unique hysteresis loops, as seen in Figure 3(a), suggesting that all catalysts have mesoporous characteristics. It can be seen that the specific surface areas and average pore sizes of Fe@BC500 (7.25 m2·g−1, 8.75 nm), Fe@BC700 (8.55 m2·g−1, 10.82 nm), Fe@BC900 (10.66 m2·g−1, 9.62 nm), and BC900 (61.34 m2·g−1, 2.91 nm) contain mesoporous structures. While the specific surface area of the biochar loaded with ZVI decreased compared to the biochar, this was due to the loaded ZVI particles occupying a certain number of adsorption sites of the biochar. FTIR spectra of Fe@BC900 are shown in Figure 2(b). The prominent peak observed at 3444 cm⁻¹ was primarily attributed to the stretching vibration of the O–H bond in the alcohol hydroxyl group. In addition, the C–O stretching vibration exhibited a characteristic peak at 1096 cm⁻¹, while the C=O stretching vibration corresponded to a peak at 1633 cm⁻¹. In addition to the absorption peaks mentioned above, the peak at approximately 618 cm-1 corresponds to the vibration of Fe-O, indicating that ZVI has been successfully loaded into the biochar, which aligns with the findings obtained from the XRD analysis in the previous section.

(a) Nitrogen adsorption/desorption curves. (b) BJH desorption dV/dD pore volume.
TC removal by Fe@BC
For a more detailed study, Fe@BC500, Fe@BC700, and Fe@BC900, which have single-crystal iron compounds, were selected as probe catalysts. As shown in Figure 4(a), the TC adsorption and removal curves demonstrate that Fe@BC900 exhibits the highest TC adsorption capacity, which can be attributed to its enhanced porosity and higher degree of graphitization, both of which result from the elevated pyrolysis temperature. Furthermore, the conversion of sp3-hybrid carbon brought about by the rise in pyrolysis temperature led to the degradation of the carbon framework and an increase in the defect structure of biochar, which raised the possibility of PDS activation. 15

(a) Adsorption efficiency and (b) degradation efficiency of TC by different materials.
A comparative study of the catalytic activation of PDS for TC degradation by the Fe@BC catalyst was carried out. As shown in the Figure 4b, a small amount of TC (4.43%) was removed when only PDS was added, indicating that PDS could hardly oxidize TC without catalyst activation. Meanwhile, the TC degradation efficiencies in the Fe@BC500/PDS, Fe@BC700/PDS, and Fe@BC900/PDS systems reached 46%, 64%, and 93%, respectively, which was attributed to the dual activation of PDS by ZVI and sp³-hybridized carbon. As ZVI was oxidized by persulfate, Fe²⁺ was steadily introduced into the system, initiating the conversion of persulfate into sulfate radicals. 16 As shown in Figure 4(b), Fe@BC500 and Fe@BC700 exhibited a reduced capacity to generate reactive species within the same reaction period compared to Fe@BC900, which resulted in their comparatively lower effectiveness in tetracycline (TC) degradation. The above results indicated that Fe@BC900 can promote the electron transfer process more effectively than raw ZVI.
Table 1 selects representative iron-based catalysts from the most recent literature and conducts a comparative analysis of their effects on the activation of persulfate for degrading organic pollutants, aiming to clearly demonstrate the performance differences and advantages of ZVI-loaded biochar in our research compared to other iron-based catalysts.
Comparison of Fe-biochar in removing organic pollutants by activating PS.
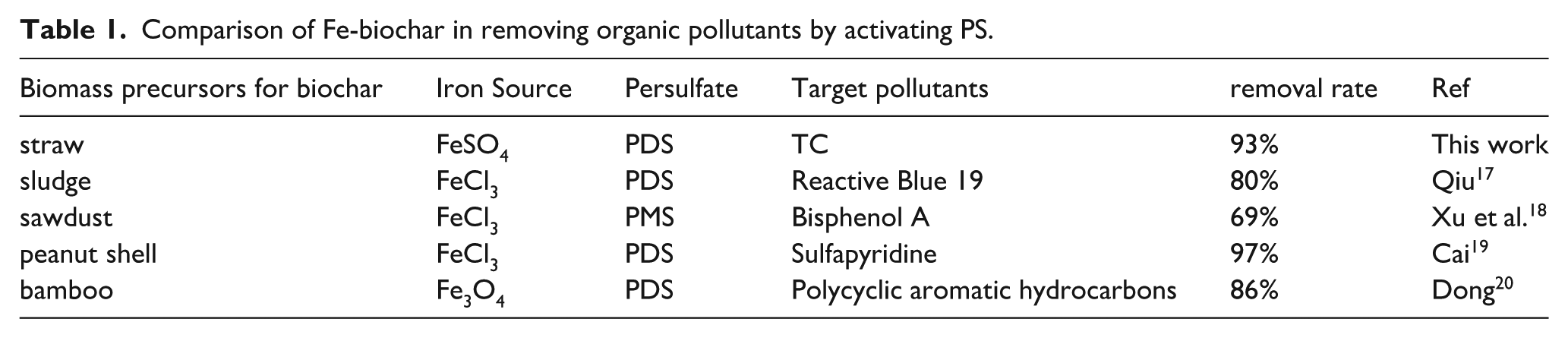
Therefore, optimized biochar (Fe@BC900) loaded with ZVI, abundant defects, large specific surface area, and porous structure was prepared in this paper and exhibited excellent catalytic activity for the oxidation of tetracycline by activated PDS.
Effect of reaction parameters on TC removal
To investigate the influence of the Fe@BC catalyst dosage on the degradation efficiency of TC, the concentrations of PDS and TC were maintained at 0.1 g·L⁻¹ and 20 mg·L⁻¹, respectively, under a reaction temperature of 25 °C. As shown in the Figure 5(a), the removal efficiency of TC improved with the increasing dosage of Fe@BC900/PDS. It might be that more catalysts could provide more active sites for adsorption and catalysis. 21 The degradation rates at 0.01g, 0.02 g, 0.04 g, and 0.05 g peaked at 68.90%, 65.28%, 89.09%, and 93.06%, respectively, at reaction times up to 30 min. As the reaction time increased to 60 min, there were more growth points. So the active site of the Fe@BC reaction with PDS almost reached saturation within 30 min.
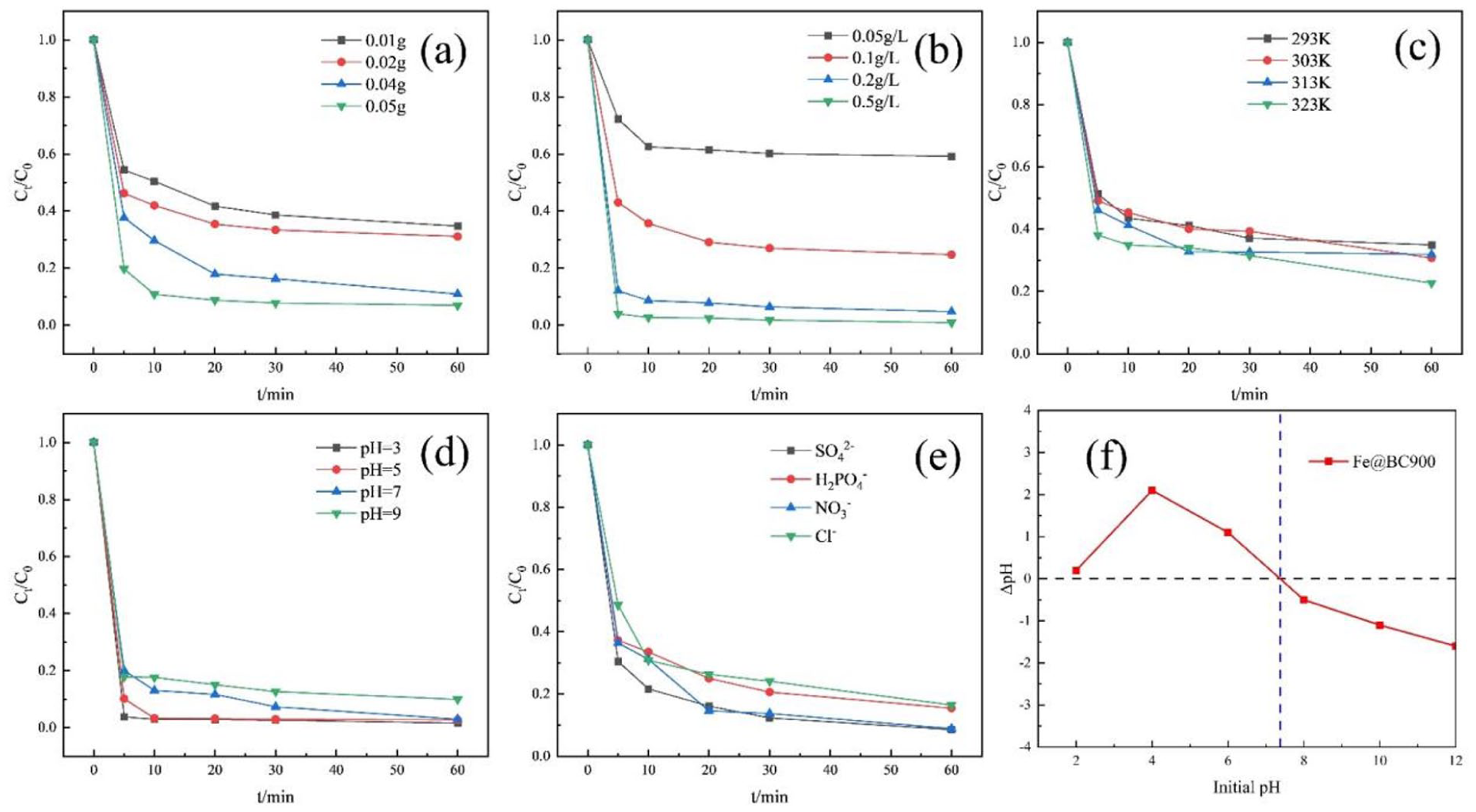
Effect of reaction parameters on TC removal: (a) carbon catalyst loading, (b)PDS concentrations, (c) temperature, (d) pH, and (e) anion and (f) pHPZC of FeBC900.
As shown in the Figure. Figure 5(b) also investigates the effect of PDS concentrations on the degradation efficiency of TC. As the PDS dosage increased from 0.05 g to 0.5 g·L⁻¹, the TC removal efficiency improved significantly, rising from 40.84% to 99.09%. It might be that a higher concentration of PDS produced more oxidative radicals, which accelerated the degradation of TC 2 .
One significant element influencing the reaction rate is temperature. High temperatures have been shown to increase molecular motion, 22 which could make it easier for oxidant and pollutant molecules to move to the surface of catalytic materials. However, the degradation efficiency was less affected by temperature, as depicted in the Figure 5(c). The removal of TC was 65.05%, 69.25%, 68.1%, and 77.41% at reaction temperatures of 293 K, 303 K, 313 K, and 323 K, respectively. It was discovered that the pH level of the solution significantly affects the catalyst’s surface characteristics and the production of radicals, which in turn affects the degree to which pollutants are removed. 23 The removal of TC was 98.41%, 97.28%, 97.02%, and 90.08% at pH 3, 5, 7, and 9, respectively, as shown in the Figure 5(d). Thanks to metal ions, which migrate more readily into the liquid phase under acidic conditions, this could activate the PDS to produce more reactive oxygen species, providing a more effective catalytic component 24 and ultimately leading to better degradation efficiency. When the initial pH value increased to 9, the degradation rate of TC decreased. This might be attributed to the fact that the pHzpc of Fe@BC900 was approximately 7.4 (Figure 5(f)). When the pH was greater than 7.5, the surface charge of Fe@BC900 was negative, which was unfavorable for the contact of PMS or PDS. At the same time, the electrostatic repulsion also led to a decrease in adsorption. 25 Recently, various inorganic anions (including SO42−, H2PO4−, Cl−, and NO3−), which significantly affected TC degradation, were frequently found in water. Therefore, the effects of SO42−, H2PO4−, Cl−, and NO3−(each at five mM) on TC degradation over the Fe@BC900/PDS system were further investigated (Figure 5(e)). In the Fe@BC900/PDS system, SO42−, H2PO4−, Cl−, and NO3−showed a slight influence role effect on TC degradation. The result indicated that the Fe@BC/ PDS system exhibited acceptable resistance to common inorganic anions during TC degradation.
Stability of Fe@BC900
In general, the stability of the heterogeneous catalyst plays a key role in TC degradation for further industrial applications. When TC degradation was over, the Fe@BC900 nanocatalyst was isolated and recycled by simple filtration. As shown in Figure 6, the catalyst exhibits good catalytic stability, maintaining a TC removal rate of 61.85% after three cycles of recycling. The decrease in catalytic performance could be due to the loss of ZVI from the magnetic biochar during the recycling process, along with surface oxidation of the catalyst and alterations in the biochar’s pore structure. These factors collectively diminish its reductive capacity, thereby impairing its ability to activate persulfate. In addition, the occupation of active sites on the surface by reactants or reaction by-products during the use of biochar might also lead to a reduction in the activation performance due to the covering of the surface. 26
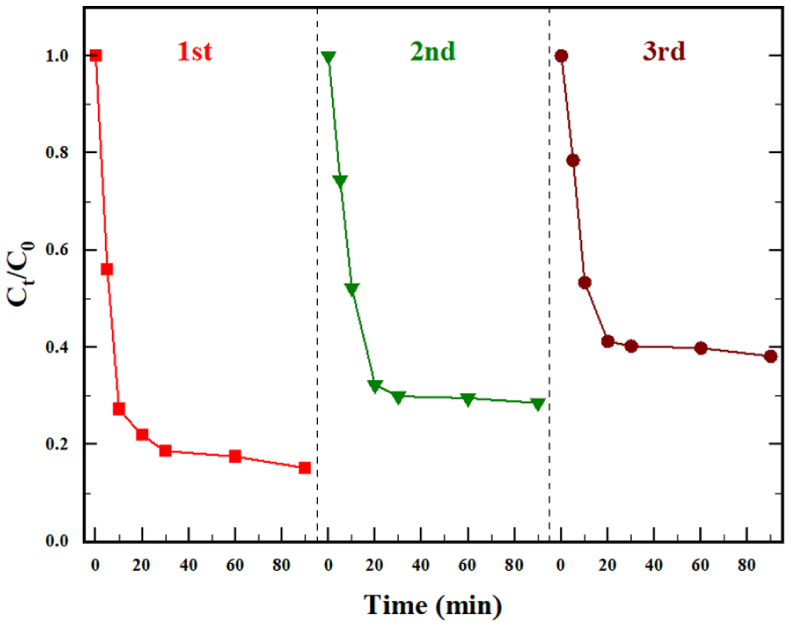
The reusability of Fe@BC900/PDS. Experimental conditions: [Fe@BC900] = 0.05 g, [PDS] = 0.5 g/L, and [TC] = 20 mg/L, T0 = 323K, no adjustment of pH.
Degradation pathway analysis
As shown in Figure 7, LC-MS was applied to identify the primary degradation intermediate products of TC to investigate further the degradation process of TC in the Fe@BC900/PDS system. According to relevant literature, electron-donating groups and ionizable functional groups (e.g. dimethylamine, amide, and double bonds) are prone to attack by reactive radicals, which facilitates the degradation of TC. 27 First, it is possible that after the removal of the amide group, the A ring of tetracycline becomes unstable, subsequently cracking and losing a small carbonyl fragment, accompanied by dehydration. Eventually, P1(m/z = 276) is formed. Then, the degradation has entered the deep oxidation stage, with the core skeleton destroyed, and fragments of P2 (m/z = 224) were obtained. The appearance of P3 (m/z = 151) indicates that the degradation is approaching the final stage of mineralization (formation of CO₂ and H₂O). 28 The second possible degradation pathway usually sees P4 (m/z = 224) corresponding to the early dealkylation and concomitant ring-opening products, which is a key indicator of the initial structural damage to tetracycline. P5 (m/z = 181) typically represents smaller molecular fragments resulting from further cleavage of the core skeleton, marking the progress toward complete mineralization (formation of CO₂ and H₂O).

The possible degradation pathways of TC in the Fe@BC900/PDS system.
Afterwards, these intermediates continue to undergo similar responses as described above, attacked by SO4•−, •OH, O2•−, 1O2, and oxidation due to loss of electrons, and through ring-opening and C-C bond breaking reactions, are broken down into smaller molecular structures. Eventually, as the catalytic process progresses, the aforementioned intermediates undergo further degradation into smaller molecular compounds, ultimately leading to their complete mineralization into CO₂ and H₂O.
Conclusion
After a series of experimental explorations, it was found that the novel catalyst Fe@BC was successfully prepared through one-pot synthesis with straw powder and FeSO4 as precursors. Novel material had excellent catalytic efficiency for activating PDS to degrade TC due to rich defects and synergistic effects resulting from Fe-doping. The optimum condition of this experiment was that 0.05 g Fe@BC900 catalyst could quickly remove 93% TC within 30 min at a temperature of 323K and PDS concentration of 0.5g·L-1. In terms of stability and reusability, Fe@BC900 remained stable and reusable after three adsorption-desorption cycles. So Fe@BC900 showed excellent degradation efficiency and stability, which was less affected by initial pH, reaction temperature, and various inorganic anions. The work established an efficient and low-cost Fe@BC900 catalyst for removing TC, which provided a significant reference for the degradation of organic pollutants in water.
Methods and materials
Materials and reagents
Tetracycline hydrochloride, ferrous sulfate heptahydrate, and ethanol were purchased from Shanghai Titan Technology. Sodium borohydride and polyethylene glycol-4000 (PEG4000) were purchased from Sinopharm Chemical Reagents Co., Ltd. The straw was taken from Lianyungang City, Jiangsu Province. Straw biochar was prepared in the laboratory. The solution’s pH was regulated through the addition of sodium hydroxide (NaOH) and hydrochloric acid (HCl). Ultrapure water was used in all experiments.
Preparation of Fe@BC sample
The straw was crushed to 200 mesh and then dried at 110 °C. To make sure there was no oxygen in the tube, the sample was then put in a tube furnace and purged with Ar for 10 min at room temperature. The target biochar (BC) was washed with deionized water and freeze-dried. After that, the straw powder was pyrolyzed in a tube furnace under oxygen-limited conditions at the set temperature (500 °C, 700 °C, and 900 °C). The Fe@BC was prepared by reduction of FeSO4·7H2O with NaBH4 as a reductant according to previously reported methods. 29 To prepare the Fe@BC under a nitrogen atmosphere, 4.0 g of biochar and 2.0 g of FeSO4·7H2O were dissolved in 100 mL of solution (30 mL ultrapure water and 70 mL absolute ethanol), and then 0.5 g of PEG4000 was introduced as a dispersing agent to minimize the agglomeration of ZVI particles in the ferrous sulfate solution. The mixture was subsequently subjected to mechanical agitation at 25°C for 1 h to obtain a homogeneously distributed solution. Next, 45 mL of 1 mol/L NaBH₄ was introduced into the mixture in a dropwise manner, followed by continuous stirring for 60 min. The solid residues were thoroughly washed with deionized water and ethanol, and subsequently subjected to freeze-drying. Finally, ZVI-loaded biochar (Fe@BC) was obtained and stored under dry conditions in brown airtight bottles.
Characterization
The surface morphology and structure of the catalysts were characterized using scanning electron microscopy (SEM). The catalyst’s crystal structure was characterized using X-ray diffraction (XRD) analysis. A specific surface area and porosity analyzer (BET) examined the biochar’s specific surface area, average pore diameter, and pore volume.
Experimental process
The static adsorption experiments were used to investigate and analyze the adsorption of different Fe@BC for tetracycline. 0.5 g · L-1 Fe@BC was dispersed in a 20 mg/L tetracycline solution in a beaker, placed in a thermostatic oscillator at 25 °C and 400 r/min to complete the adsorption experiment, and the resulting sample was analyzed at different time intervals by high-performance liquid chromatography (HPLC). The adsorption capacity q (mg/g) and removal efficiency R (%) were calculated according to equations (1) and (2), respectively.


Where C0 and Ct (mg/L) were the initial concentration of tetracycline and the residual concentration of tetracycline corresponding to the adsorption time t. m was the mass of adsorbent Fe@BC (g) and V was the volume of the solution (L).
Based on the tetracycline adsorption experiments to compare the adsorption effect, Fe@BC900, which has the optimal adsorption effect, was selected as the catalyst for the degradation experiments. The conical flask containing 50 ml of tetracycline (20 mg· L-1) was placed in a constant temperature air bath oscillator at a speed of 400 R/min speed. A certain amount of biochar and PDS was added to initiate the degradation reaction until the setting time, after which 0.50 ml of methanol was added to stop the reaction. Then the samples were filtered through a 0.22 μM filter membrane and analyzed by HPLC at a wavelength of 356 nm.
During the experiments, the factors for amount of biochar (0.01g, 0.02g, 0.04g, and 0.05g), concentration of PDS (0.05, 0.10, 0.20 and 0.50 g· L-1), reaction temperatures (293, 303, 313 and 323K), pH= (3, 5, 7 and 9) and solution anion (SO42-, H2PO4-, Cl-and NO3-) were explored to the influence for degradation experiment.
Supplemental Material
sj-docx-1-chl-10.1177_17475198251388223 – Supplemental material for Efficient activation of persulfate by biochar-supported zero-valent iron for enhanced tetracycline removal
Supplemental material, sj-docx-1-chl-10.1177_17475198251388223 for Efficient activation of persulfate by biochar-supported zero-valent iron for enhanced tetracycline removal by Peixuan Li, Haifeng Li, Yu Yao, Yunyi Tan, Yan Zhang and Jifen Wang in Journal of Chemical Research
Footnotes
Ethical considerations
This study does not involve human participants, and therefore, informed consent is not required.
Consent to participate
This paper has no human participants and informed consent is not required.
Consent for publication
Not applicable
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the Shanghai Sailing Program (grant nos 21YF1414400).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Data availability statement
Data will be made available upon reasonable request.
Supplemental material
Supplemental material for this paper is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
