Abstract
Inappropriate overactivation of the mineralocorticoid receptor by the steroid hormones aldosterone or cortisol contributes to the development of cardiovascular diseases. The new mineralocorticoid receptor antagonist can directly block mineralocorticoid receptor overactivation, inhibit inflammation and profibrotic gene expression, thereby reducing chronic kidney disease progression and cardiovascular risk, achieving dual benefits for both kidney and heart. Antergy of mineralocorticoid receptor is a proven therapeutic concept for the management of chronic kidney disease and type 2 diabetes associated diseases. Using finerenone (BAY 94-8862) as an inspiration, several novel naphthalidinamide analogues were designed, synthesized and their biological activities were determined as nonsteroidal mineralocorticoid receptor antagonists. In vitro results showed that
Keywords

Introduction
Cardiovascular diseases (CVDs) pose a significant threat to human health and life, ranking as one of the leading causes of mortality worldwide. 1 With the advancements in living standards, the issues of obesity and aging population are becoming increasingly serious, and the CVDs have become the main killer as well as tumor.2,3 The inappropriate overactivation of the mineralocorticoid receptor (MR) by steroid hormones such as aldosterone or cortisol is a critical contributor to the pathogenesis of CVDs. 4 Mineralocorticoid receptor antagonists (MRAs) have been shown to effectively reduce blood pressure in patients with primary aldosteronism and essential hypertension, particularly in individuals with resistant hypertension. 5 Moreover, combining MRAs with angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs) has demonstrated superior therapeutic benefits compared with conventional therapies alone, notably in reducing prote-inuria in patients with chronic kidney disease (CKD).6 –8 This combination has also been associated with reductions in markers of myocardial fibrosis. 9 Consequently, MR inhibition has emerged as a validated therapeutic strategy for managing CKD and diseases associated with type 2 diabetes (T2D).10,11
The first-generation MRA, spironolactone (IC₅₀ = 24 nM), and the second-generation MRA, eplerenone (IC₅₀ = 990 nM) 12 (Figure 1), have demonstrated significant efficacy in reducing morbidity and mortality in renal and cardiac diseases. Clinical studies further underscore the therapeutic potential of MRAs in treating CKD and diabetic nephropathy. However, the clinical utility of these steroidal spirolactones is limited by notable drawbacks. Spironolactone exhibits substantial off-target activity on androgen receptor (AR) and progesterone receptor (PR) due to its structural similarity to progesterone, resulting in poor selectivity and adverse effects such as gynecomastia, impotence, and menstrual irregularities. 13 Although eplerenone offers improved selectivity, it is less potent in vitro than spironolactone and has demonstrated lower efficacy in hypertensive clinical trials. 14

Chemical structures of the representative mineralocorticoid receptor antagonist.
To address the limitations of spironolactone and eplerenone, novel, potent, and selective MRAs have been developed.15 –17 Finerenone (BAY 94-8862), 18 a third-generation nonsteroidal MRA with high potency was identified through high-throughput screening of cyano-1,4-dihydropyridines. Finerenone has been shown to reduce the risk of sustained eGFR decline, end-stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with CKD associated with T2D.19 –22 In 2021, it received approval from the U.S. Food and Drug Administration, becoming the first nonsteroidal selective MRA to improve renal outcomes in adult patients with CKD and T2D. 23
Encouraged by the successful discovery of the marked finerenone, for development of new nonsteroidal MRAs that are more potent, selective, and orally available, reducing the risk of hyperkalemia, we disclose an extended SAR (structure-activity relationship) exploration for dihydronapthyridine derivatives by a pharmacophore fusion strategy of finerenone (
1,4-Dihydro-1,6-naphthyridine ring is a pharmacophore group that maintains MR inhibitory activity. Previous research has shown that the C3, C4, and C5 substituents on the naphthyridine have a strong impact on activity. 18 On the basis of retaining the pharmacophore of 1,4-dihydro-1,6-naphthoprim ring, C4 (A ring) was modified by introducing F and CF3, C5 (B ring) was modified by introducing cyclopropane (Figure 2). Therefore, in this paper, three new naphthalidinamide analogues were designed and synthesized as novel MRAs, and the antagonistic activity and selectivity were evaluated based on MR cell-based transactivation assay.
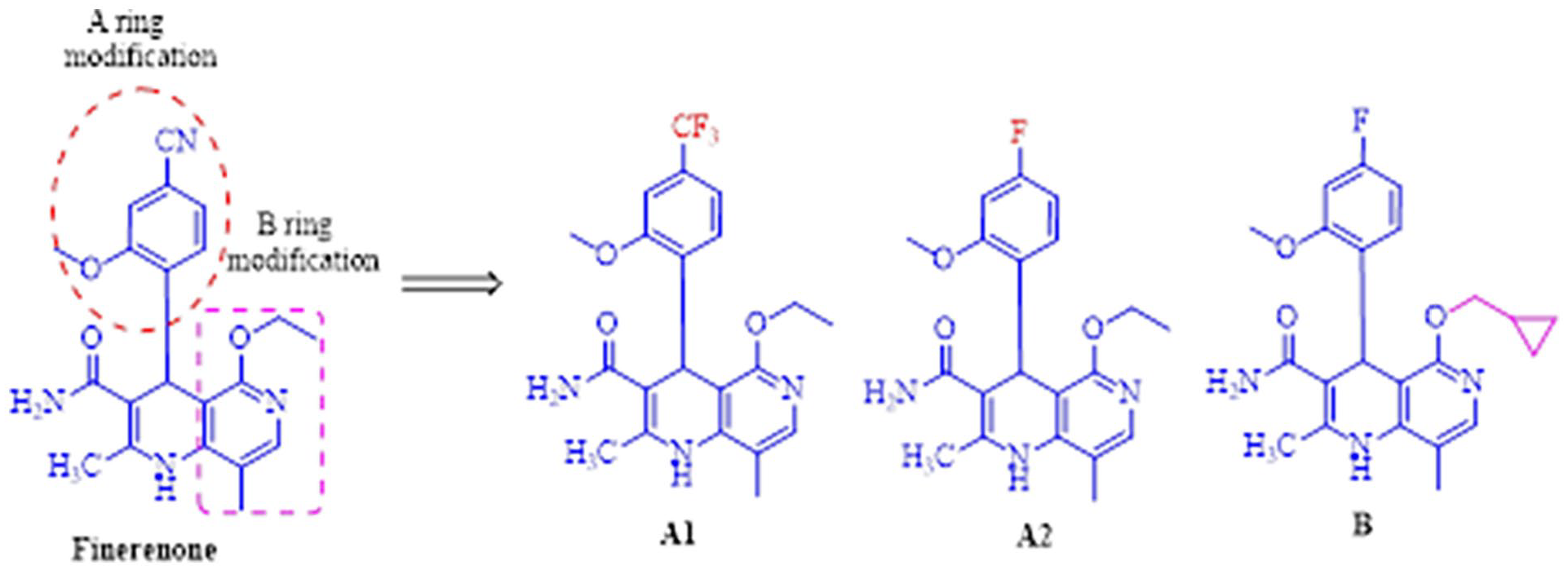
Design strategy of the novel naphthalidinamide analogues.
Results and discussion
Chemistry
The synthesis of targeted molecules was illustrated in Scheme 1. The protocols used for preparation of the intermediates and target compounds were described in detail in the “Experimental Section.” The classical Hantzsch reaction was successfully applied for the construction of dihydropyridines.24,25 Condensation of 2-methoxy-4-(trifluoromethyl) benzaldehyde (

Synthesis of naphthalidinamide analogues.
Haloalkane is frequently employed as an alkylating agent for preparation of alkoxy pyridine from pyridine.26
–28 Compound
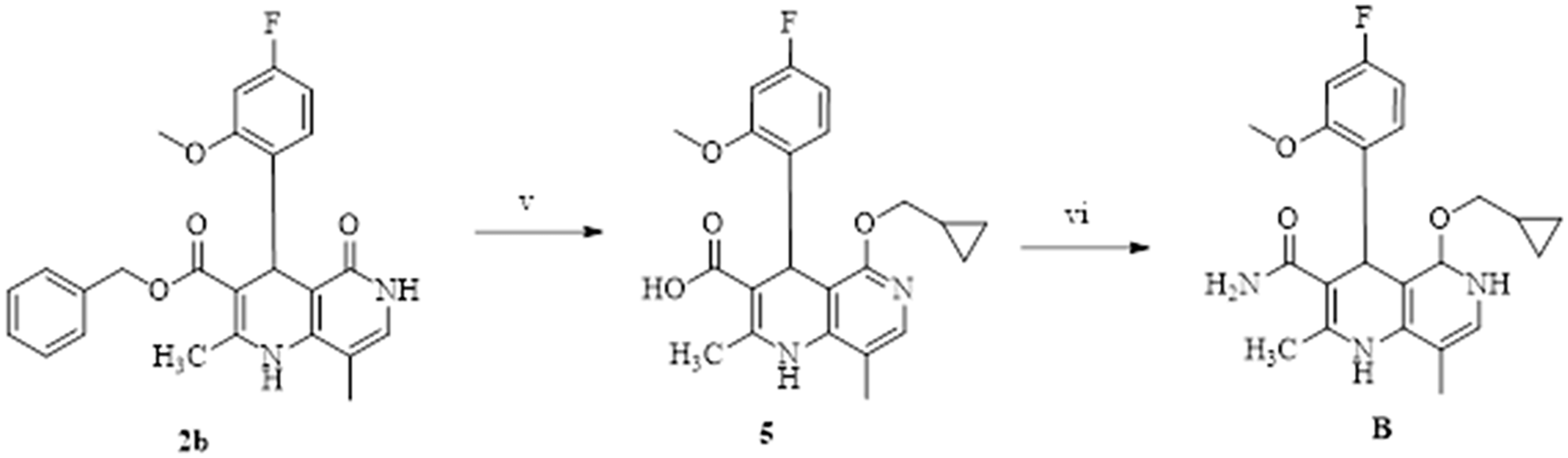
Synthesis of compound
Cell-based transactivation assay
The cell-based transactivation assay of the targeted compounds was evaluated and finerenone (
The inhibitory activities of target compounds against MR receptor. a
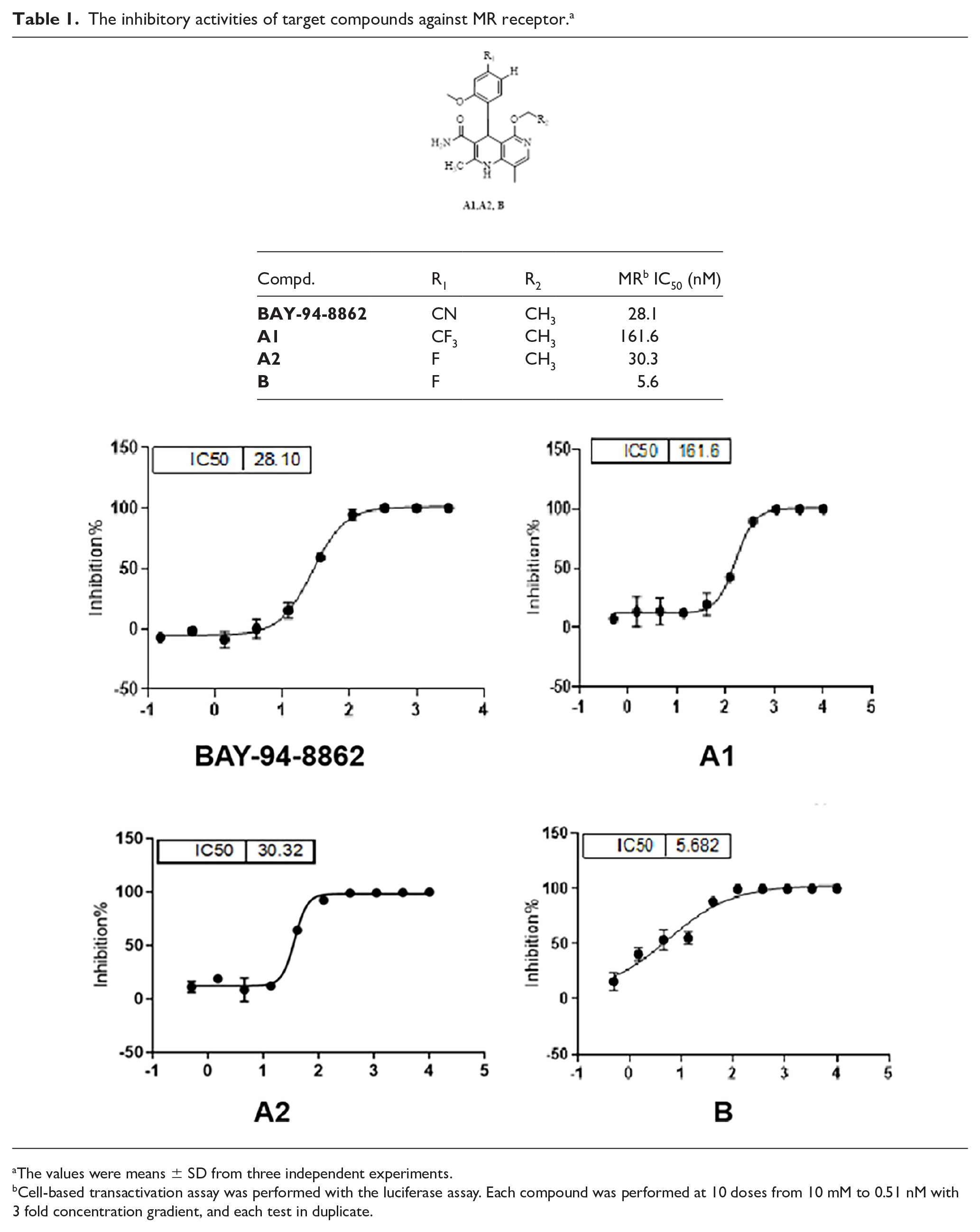
The values were means ± SD from three independent experiments.
Cell-based transactivation assay was performed with the luciferase assay. Each compound was performed at 10 doses from 10 mM to 0.51 nM with 3 fold concentration gradient, and each test in duplicate.
The development of novel nonsteroidal MRAs began with “A” ring modification using a pharmacophore fusion strategy of finerenone. Instead of CN in R1, CF3 and F as new hydrogen bond acceptor, the novel 4-aryl-1,4-dihydro-1,6-naphthalidinamide derivatives
CLogP represents the hydrophobicity of a compound, the larger CLogP means the greater hydrophobicity and the smaller hydrophilicity. TPSA refers to topological polar surface area, which is a physical parameter that describes the chemical properties of molecules. The higher tPSA value means the greater polarity and the higher solubility of the compound. The properties of the targeted compounds including molecular weight, CLogP and tPSA values is associated with activity and pharmacokinetic parameters.
29
Regarding to these synthesized naphthalidinamide derivatives, they were found to be potential in terms of potency, among them, the promising compound
Properties
a
of the target compounds and

CLogP as well as tPSA were predicted by ChemBioDraw 22. and the digitals were rounded to two digits.
The data indicated that the predicted CLogP values were less than 5.08 and the predicted tPSA values were less than 100.5 for all target compounds. According to the cell-based transactivation assay, compounds
Selectivity against other nuclear hormone receptors
In order to investigate the selectivity to other nuclear hormone receptors (NHRs), such as the glucocorticoid receptor (GR), AR, and PR, the promising compounds
Cell-based transactivation assay for MR, AR, PR, and GR. a

MR: mineralocorticoid receptor, GR: glucocorticoid receptor, AR: androgen receptor, PR: progesterone receptor.
All experiments were repeated at least three times.
The results in Table 3 indicated that compound
Generally, the potent compounds
Liver microsomes stability
The half-life time (t1/2) and intrinsic clearance (CLint) parameters were commonly used to evaluate their metabolic stabilities that could give good indications of the in vivo hepatic clearance.30,31
Stabilities of compounds

Table 4 revealed that compound
Theoretical docking analysis
To further explain the potent antagonistic activity against MR of compound

Interaction map of compound

The 2D diagrams of protein–ligand interactions between compound
Conclusions
In summary, based on the structure of the third-generation MRA, Finerenone (
Experimental Section
Chemistry
Instruments
Unless additional specification, all reactions were performed in air or moisture site. Reaction processes were monitored by thin-layer chromatography (TLC) with silica gel aluminum plates (60F-254) and spots were visualized with UV light at 254 nm or iodine. Flash column chromatography was performed using silica gel (300~400 mesh). 1 H NMR and 13C NMR spectra were recorded in CDCl3 or DMSO-d6 at room temperature on Bruker Avance spectrometers with Tetramethylsilane (TMS) as internal standard. Data of 1H NMR are reported as follows: chemical shift, multiplicity (s = singlet, br = broad, d = doublet, t = triplet, m = multiple), coupling constants and integration. High-resolution mass spectra were analyzed by Agilent Technologies 6540 UHD Accurate-Mass Q-TOF LC/MS.
Materials
Solvents for reaction, extraction and Flash column chromatography were reagent grade and used as received. Unless otherwise required, all the reagents were commercially obtained with analytical grade and used without further purification. Frequently dry solvents (CH2Cl2, Tetrahydrofuran (THF), DMF, etc.) were anhydrous packaged from suppliers with Sure/Seal bottles. Yields mean isolated yields unless otherwise stated.
Synthesis
General procedures for the synthesis of compounds
Benzyl 4-(2-methoxy-4-(trifluoromethyl)phenyl)-2,8-dimethyl-5-oxo-1,4,5,6-tetrahydro-1, 6-naphthyridine -3-carboxylate (
2-Methoxy-4-(trifluoromethyl) benzaldehyde
Benzyl 5-ethoxy-4-(2-methoxy-4-(trifluoromethyl)phenyl)-2,8-dimethyl-1,4-dihydro-1, 6-naphthyridine -3-carboxylate (
To a mixture of triethyl orthoformate (25.0 g, 168.7 mmol) and compound
5-Ethoxy-4-(2-methoxy-4-(trifluoromethyl)phenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxylic acid (
Intermediate
5-Ethoxy-4-(2-methoxy-4-(trifluoromethyl)phenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxamide (
Compound
Benzyl 4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-5-oxo-1,4,5,6-tetrahydro-1,6-naphthopyridine-3-carboxylate (
Compound
Benzyl 5-ethoxy-4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxylate (
Compound
5-Ethoxy-4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxylic acid (
Compound
5-Ethoxy-4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxamide (
Compound
5-(Cyclopropylmethoxy)-4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxylic acid (
To a suspension of 50 mL DMF,
5-(Cyclopropylmethoxy)-4-(4-fluoro-2-methoxyphenyl)-2,8-dimethyl-1,4-dihydro-1,6-naphthopyridine-3-carboxamide (
Compound
Biological assay
Determination of IC50 values in cell-based assays
IC50 determinations for humanized oxosteroid receptors (MR: mineralocorticoid receptor, GR: glucocorticoid receptor, AR: androgen receptor, PR: progesterone receptor) were performed with Bright-Lite Luciferase Assay from Vazyme according to the manufacturer’s instructions. Luc2P-GAL4-HEK293 (transient MR HEK293 cell, E5800) was purchased from ATCC and taken as an experimental cell line. Using JetPRIME transfection reagent, pBIND-MR as plasmids, all cells were cultured in DMEM medium (Corning), supplemented with 10% FBS (Invitrogen) and 200 μg/mL Hygromycin B (Solarbio), cells were connected to 96-well plates at a density of 3000 cells/well using complete medium. A gradient-diluted compound was added to the cells to dilute 9 concentrations from a final concentration of 3 μM, 3 replicates per concentration. Cells were maintained at 37 °C in a humidified atmosphere of 5% CO2, and exposed to compounds treatment for 24 h, luciferase activity was determined with a luminescence-detecting video camera system. GraphPad Prism Software (version 5.0, GraphPad Software Inc., San Diego, CA, USA) was used for curve fitting and calculation of IC50 values. The IC50 values were determined from at least three independent experiments performed in duplicate.
Compound stability test in liver microsome
Compounds were incubated with human (BIOIVT), monkey (RILD), dog (Corning), rat (Corning), and mouse (Corning) liver microsomes. The liver microsomes (20 mg protein/mL) was incubated in 37 ℃ water bath for 3 min, then, 25 μL β-NADPH solution (4 mM) was added to 75 μL incubation solution without β-NADPH, vortexed for 30 s with 100 μL total volume, and reactions were initiated in 0.05 M Phosphate buffer (pH = 7.4) at 37 °C for 0, 5, 15, 30, 60 min. The termination agent (including internal standard) was added, followed by vortex and centrifuge, the reaction was quenched, and the amount of the remaining compound was analyzed using LC-MS/MS (ESI+). LC-MS conditions are as follows: mobile phase A: acetonitrile, B: 0.2% acetic acid with water; gradient, time, and flow rate (A%): initial (0.4, 15); 1 (0.4, 95); 2 (0.4, 95); 3 (0.4, 15). Chromatographic column: Waters ACQUITY UPLC HSS T3 (1.8 μm, 2.1 × 50 mm), injection volume: 2 µL.
Protein modeling and ligand docking
The X-ray structure of the wild-type MR ligand binding domain complexed with deoxycorticosterone (PDB ID: 2ABI) was selected as a model for docking study. Autodock was used to determine the molecules rotatable bonds and torsion angles, then saved with pdb.qt format after charge treatment. Structure of the compound
Supplemental Material
sj-docx-1-chl-10.1177_17475198251351168 – Supplemental material for Design, synthesis, and evaluation of naphthalidinamide analogues as nonsteroidal mineralocorticoid receptor antagonists
Supplemental material, sj-docx-1-chl-10.1177_17475198251351168 for Design, synthesis, and evaluation of naphthalidinamide analogues as nonsteroidal mineralocorticoid receptor antagonists by Mingguang Zhang, Yan Pang, Yurui Zhao, Sibei Chen and Cheng Guo in Journal of Chemical Research
Footnotes
Acknowledgements
The authors gratefully acknowledge Jiangsu Medical College and Jiangsu Chia Tai Fenghai Pharmaceutical Co. Ltd. for their experimental support.
Ethical considerations
Ethical approval is not applicable for the article.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored by the Medical Research Project of Yancheng Health Commission (grant no. YK2024182).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data for this study are available from the corresponding author on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

