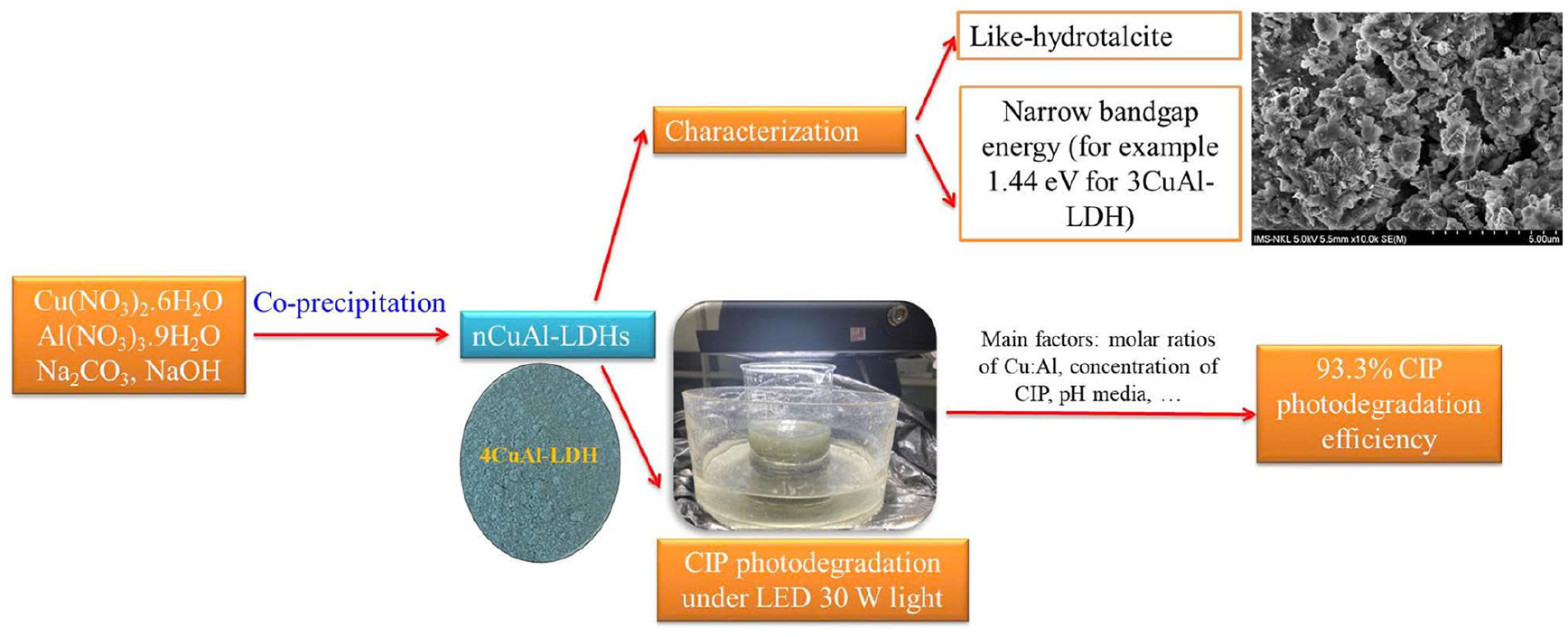
Abstract
Sets of nCuAl-LDH layered double hydroxide samples were successfully synthesized by the co-precipitation method, using precursors such as Al(NO3)3, Cu(NO3)2, and Na2CO3 salts to provide
Introduction
Antibiotics have been known to be one of the most successful drugs used to care health for humans. They have been also widely used in animal husbandry and agriculture. Ciprofloxacin is one of the effective antibiotics used widely. Ciprofloxacin belongs to the quinolone antibiotic group and is known to be an antibiotic used to treat severe infections. However, the residues of this antibiotic is reason of many particularly serious effects on human health and living organisms. It can cause drug resistance in humans. Ciprofloxacin can damage and affect the development of cartilage and joints in young animals, leading to the risk of harm to bone and joint development in fetuses and children. It can also cause muscle pain, neuritis, especially causing Achilles heel rupture, especially in the elderly with weak ligaments. It can affect on the nervous system. Less commonly, it can cause seizures, anxiety, depression, nightmares, or hallucinations, especially in people with a history of psychiatric illness. In addition, ciprofloxacin can also cause some serious hypersensitivity reactions such as facial swelling, laryngeal swelling, and life-threatening difficulty breathing, especially in people with allergies. Overuse of high doses of ciprofloxacin, like other broad-spectrum antibiotics, can upset the body’s bacterial ecosystem, causing intestinal dysbiosis and liver and kidney problems.1,2
In recent years, the layered double hydroxide materials modified have been widely studied as a new type of photocatalytic material.3 –8 Because these materials have many advantages: easy to fabricate, high catalytic activity, applicable in many fields (such as adsorption, catalysis, photocatalyst, pharmaceuticals, organic synthesis, antibacterial, and anticancer activity), low processing cost, especially high activity under visible light.9,10 Among the LDH materials, CuAl layered double hydroxide materials have been synthesized and applied as catalysts in a number of fields such as CO2 reduction to formic acid, 11 MO dye decomposition,12,13 phenol treatment, 3 adsorption of dye and red congo,14,15 CO oxidation, 16 organic synthesis.17,18
In the latest publications, the materials of CuAl-LDHs showed excellent photothermal antibacterial performance against gram-positive bacteria Staphylococcus aureus (S. aureus) and gram-negative bacteria Escherichia coli (E. coli) and Klebsiella pneumoniae (K. pneumoniae). 19 Especially in the field of antibiotic treatment, cellulose modified CuAl layered double hydroxide binary composites showed doxycycline adsorption efficiency of 40.36%, adsorption maximum capacity qmax of 150.16 mg/g, and photocatalytic degradation efficiency of 40 mg/L doxycycline reaching 96.2% after 120 min of illumination with 50 W LED lamp. 20 Thus, CuAl-LDHs materials have demonstrated the good abilities to adsorb organic substances (dyes, doxycycline) and they can photocatalytic degrade for doxycycline with high efficiency. However, in the field of treatment of emerging contaminants, especially antibiotics, there have not been many published studies on this series of materials. Therefore, preparing the CuAl-LDH materials at different molar ratios of Cu:Al and using them as photocatalysts for photocatalytic degradation of the antibiotic ciprofloxacin have not been mentioned up to now. In this report, we synthesized a series of CuAl-LDH materials according to the different molar ratios of Cu:Al and used the synthesized materials to study the ability to treat ciprofloxacin in aqueous environment under visible light. Through the obtained results, it was possible to select material samples with LDH-like structures, having the high photocatalytic activity in the photocatalytic degradation reaction of ciprofloxacin in water. In addition, main parameters affecting the photocatalytic activity of the synthesized materials such as molar ratio of Cu:Al in materials, Ciprofloxacin (CIP) concentrations, and environmental pH were investigated.
Materials and methods
Chemicals
The chemicals were used to synthesize the materials as Al(NO3)3.9H2O, Cu(NO3)2.3H2O, Mg(NO3)2.6H2O, Na2CO3, NaOH, HNO3, H2O2 produced by Sigma-Aldrich. All chemicals have analytical purity.
Ciprofloxacin is product of Shanghai Macklin Bio-chemical Co. (China). The chemical purity is 98%.
Synthesis of Cu-Al LDH materials
The synthesis processes of the materials were conducted following literature: 21 First, the nitrate salts of Al(NO3)3.9H2O, Cu(NO3)2.3H2O were simultaneously dissolved in 150 mL of double distilled water in a 500-mL glass beaker (the nitrate salts were determined according to the molar ratios in Table 1, the mass of Al(NO3)3.9H2O salt was fixed at 11.25 g, the mass of Cu(NO3)2.3H2O salt was varied from 4.84, 7.26, 9.68, 12.1, 14.52 g corresponding to the molar ratios of Cu:Al as 2.0:3.0; 3.0:3.0; 4.0:3.0; 5.0:3.0, and 6.0:3.0). Placing the beaker on a magnetic stirrer, mixture was stirred well to obtain solution A. Next step, 25 mL of Na2CO3 0.6M solution was slowly dropped into the reaction beaker and mixture was stirred for 60 min at room temperature.
Symbols and Molar Ratios of Elements in Synthetic Material Samples.

Then, the pH of the mixture was adjusted by NaOH 2M solution until the pH value as 9.5. These processes created a gel. The gel was then stirred on a magnetic stirrer for 60 min at room temperature, then aged in a 500-mL glass beaker at 80°C for 24 h.
After the gel had been aged, the product was filtered and washed with hot water (80°C) several times, then washed with distilled water until pH value decreasing to 7.0. The solids were dried at 80°C for 24 h to obtain the synthesized material samples. Finally, the samples were crushed with an agate mortar and pestle to obtain nCuAl-LDH material samples and were labeled according to Table 1.
MgAlH hydrotalcite material was also synthesized according to the above steps, from the nitrate salt precursors of Mg, Al (17.92; 11.25 g were the mass of Mg and Al nitrate salts used is 17.92; 11.25 g, respectively). The MgAlH hydrotalcite material sample was used to compare the structural characteristics as well as the photocatalytic activity with the nCuAl-LDH material samples.
Calibration curve to determine the concentration of ciprofloxacin in aqueous solution using UV-Vis absorption spectrum
0.1000 g of ciprofloxacin was diluted into a 1000-mL volumetric flask to obtain a stock solution containing the CIP concentration of 100 ppm. Diluting the stock solution into solutions with different concentrations of CIP (1.0; 2.0; 4.0; 6.0; 8.0; 10.0; 15.0; 20,0 ppm). The prepared CIP solutions were measured at a wavelength of 265 nm on the 1700 UV-Vis instrument. From the results obtained, the standard curve equation to determine the concentration of CIP in the solution was y = 0.069x − 0.0139, R² = 0.9998, LOD and LOQ of 0.47, 1.42 mg/L, respectively (Figure 1). Based on the standard curve equation, the CIP concentration was determined. The obtained results were used to evaluate the CIP adsorption and photocatalytic decomposition capacity in the aqueous environment of the synthesized material samples.

Standard curve for determining ciprofloxacin concentration in aqueous solution (a) and UV-Vis spectrum determining maximum absorption wavelength of CIP at concentrations as 8; 10; 15; 20 ppm (b).
Study on the structural characteristics and properties of the composite materials
The layered double structural characteristics of the materials were measured using a MiniFlex 600 machine from Rigaku—Japan. The morphology of the materials was determined by scanning electron microscopy (SEM) on a Hitachi S-4800 instrument. The chemical elemental composition was determined by energy dispersive X-ray spectroscopy (EDX) using a HORIBA instrument (MODEL 7593-H) (Kyoto, Japan). The UV-Vis DRS spectrum of the materials was determined using a U-4100 Spectrophotometer (Hitachi, Japan). The Apparent Zeta Potential of the material was measured on a Zetasizer Nano ZS90 (Malvern Panalytical Ltd).
Investigation of CIP adsorption and decomposition capacity of materials
Investigation of CIP adsorption capacity of materials in the dark
0.2 g of materials added to 250 mL of CIP solution at a concentration of 50 ppm placed in a 500-mL glass beaker and covered by a black plastic bag. The reaction mixtures were stirred on a magnetic stirrer at room temperature. Then, the volumes of 5–7 mL of the reaction mixtures were taken out after certain time intervals (30, 60, 90, 120, 150, 180 min). Next step, the CIP solution was centrifuged, dilluted within the concentration range of the standard curve (five times) and measured on a UV-Vis 1700 at a wavelength of 265 nm. The survey experiments were all repeated three times. The CIP adsorption efficiency of the material was calculated as follows

In which C0: Initial concentration of CIP; C: Concentration of CIP at the time of survey.
Surveying the adsorption capacity of the six synthesized materials above, the CIP adsorption capacity of the samples was evaluated. Furthermore, time to reach adsorption equilibrium, adsorption efficiency, and the influence of the Cu2+:Al3+ molar ratio in the material sample were also determined.
Investigation of CIP photocatalytic degradation ability of the material under visible light
Effect of illumination time and Cu:Al molar ratio on the catalytic activity of the materials
0.2 g of the material was added into a 500-mL beaker containing 250 mL of 50 ppm CIP solution. Covering beaker with a black bag and stirring mixture on a magnetic stirrer at room temperature for about 60 min to reach adsorption equilibrium. Then a sample was taken out to determine the adsorption efficiency.
1.2 mL of 30% H2O2 was added into the beaker. Then, the reaction mixture was illuminated by a 30 W LED lamp (6500K, manufactured by Philips, λmax = 464 nm), stirred with a magnetic stirrer at a speed of 500 rpm. Every 30 min of irradiation, the mixtures were taken out, centrifuged, diluted five times to determine the photocatalytic degradation efficiency of the material according to the formula

In which: C0: Equilibrium adsorption concentration of CIP; C: Concentration of CIP at the time of survey.
The CIP treatment efficiency of sample was caculated by formula (the treatment efficiency includes both adsorption and photocatalytic degradation)

In which C0: Initial concentration of CIP; C: Concentration of CIP at the time of survey.
The simultaneous surveys were conducted to evaluate the photocatalytic degradation ability of six synthesized material samples. Based on the results obtained, the influence of illumination time on the CIP treatment efficiency of the synthesized material samples had been evaluated.
Effect of CIP concentration
The material sample with good catalytic activity (3CuAl-LDH) was selected to investigate the effect of CIP concentration on CIP decomposition ability under LED light. 0.2 g of material were added to CIP solutions with different concentrations (10, 25, 40, 50, and 75 ppm). The reaction mixtures were conducted the decomposition processes under light and H2O2 conditions similar to section “Effect of illumination time and Cu:Al molar ratio on the catalytic activity of the materials.”
From the investigation results obtained, the effect of CIP concentration on the catalytic activity of the material was evaluated.
Effect of environmental pH
The environmental pH of CIP solution of 50 ppm concentration (5.8—initial pH value) was adjusted by using solutions of NaOH 0.1M and HCl 0.1M to achieve the required values of 3.0; 4.0; 8.0; 10.0; and 12.0. The investigation processes were conducted similar to the steps in section “Effect of illumination time and Cu:Al molar ratio on the catalytic activity of the materials.” Basing on survey results, the influence of environmental pH on CIP decomposition efficiency had been evaluated.
Investigate the role of catalyst material and H2O2 in the photocatalytic decomposition process of CIP
To evaluate the role of catalyst material and H2O2 oxidizing precursor for the advanced oxidation process of CIP in aqueous solution, we designed three experiments and conducted them simultaneously as follows: the first survey was conducted in the presence of both catalyst material and H2O2; the second survey was conducted in the presence of only catalyst material, without H2O2; the third survey was conducted in the presence of only H2O2, without catalyst material.
In all three cases, we investigated with 250 mL of 50 ppm CIP solution, 0.2 grams of 3CuAl-LDH material and 1.2 mL of 30% H2O2. The investigation steps were conducted similarly to section “Effect of illumination time and Cu:Al molar ratio on the catalytic activity of the materials.”
Results and discussion
Structural characteristics of materials
Results of X-ray diffraction analysis of material samples
The XRD patterns in Figure 2 showed that the three material samples MgAlH, 2CuAl-LDH, 3CuAl-LDH all had diffraction peaks at scales 2θ = 11.58o (003), 22.86o (006), 35.02o (012), 39.06o (015), 48.68o (018), 60o (110) which characterized for the hydrotalcite and the layered double hydroxide. These results were consistent with the authors’ publication.3,12,15,17,18 The peaks at 11.58o corresponded to carbonate (CO32-) intercalated in the LDH. 12 Thus, the three material samples above still retained the layered double structure characteristics similar to hydrotalcite materials. However, comparing the diffraction peaks characteristic of the layered double structure of the modified materials with the hydrotalcite sample (MgAlH), it can be seen that the 2CuAl-LDH sample had a much lower peak intensity at 2θ = 11.58o than the MgAlH sample. In the contrast, the 3CuAl-LDH sample had a higher intensity of the characteristic peak for the layered double structure at 2θ = 11.58o than the MgAlH sample. In particular, both 2CuAl-LDH and 3CuAl-LDH samples had peaks at 2θ = 35.5o and 38.8o characterized by the CuO phase.4,17 The three material samples 4CuAl-LDH, 5CuAl-LDH, 6CuAl-LDH had diffraction peaks at 2θ = 11.58o but the height and intensity of this diffraction peak strongly decreased. Therefore, the arrangement order as well as the layered double structure strongly reduced when compared with the three material samples above. In addition, the diffraction peaks characteristic of CuO phase did not appear on the three material samples 4CuAl-LDH, 5CuAl-LDH, 6CuAl-LDH. The results of XRD analysis of six synthetic material samples concluded that the two material samples 2CuAl-LDH and 3CuAl-LDH retained the basic characteristics of materials with hydrotalcite-like structure and the CuO phase appeared in these two material samples. The molar ratio Cu:Al equal 1:1 in the material sample 3CuAl-LDH ensuring the layered double structure characteristic like hydrotalcite.

XRD patterns of the synthesized samples.
Results of EDX spectrum analysis of material samples
The results of the atomic percentage composition of elements O, Mg, Al, Cu, in two material samples MgAlH, 3CuAl−LDH showed in Figure 3 and Table 2.
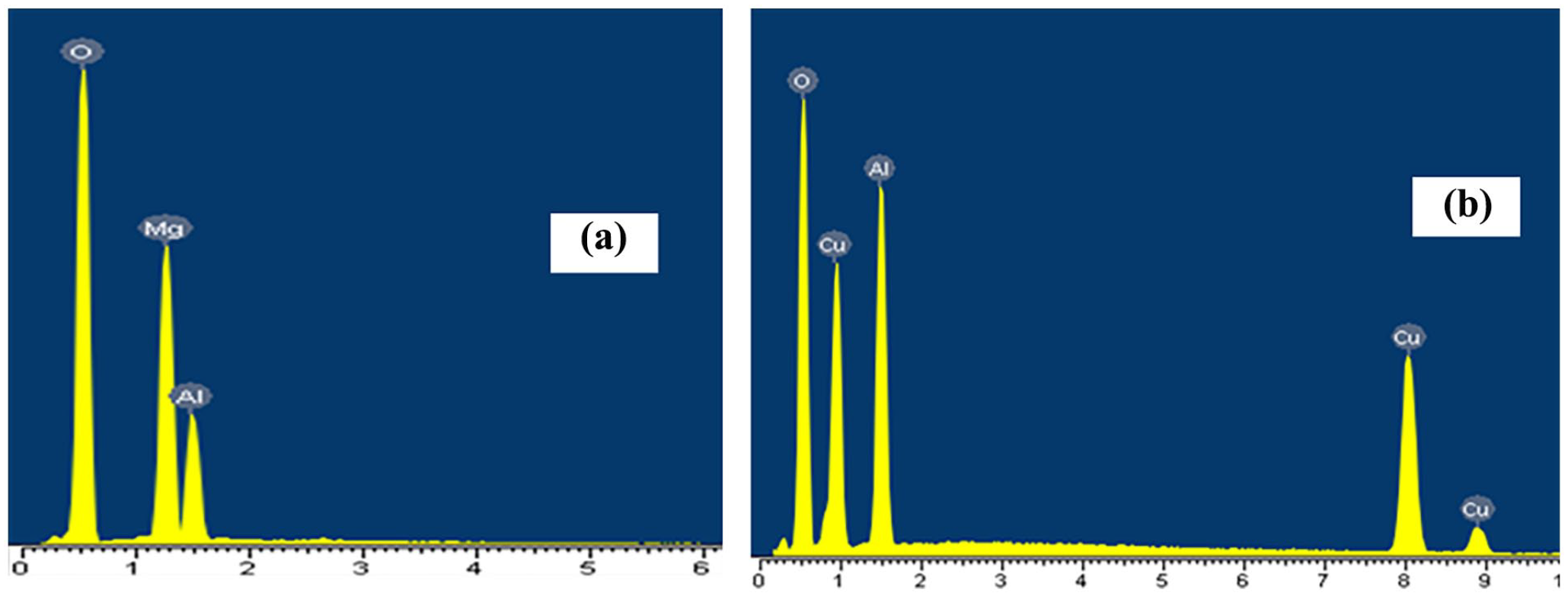
EDX spectra of two material samples MgAlH(a), 3CuAl−LDH (b).
% Composition of Some Elements in Synthetic Material Samples.
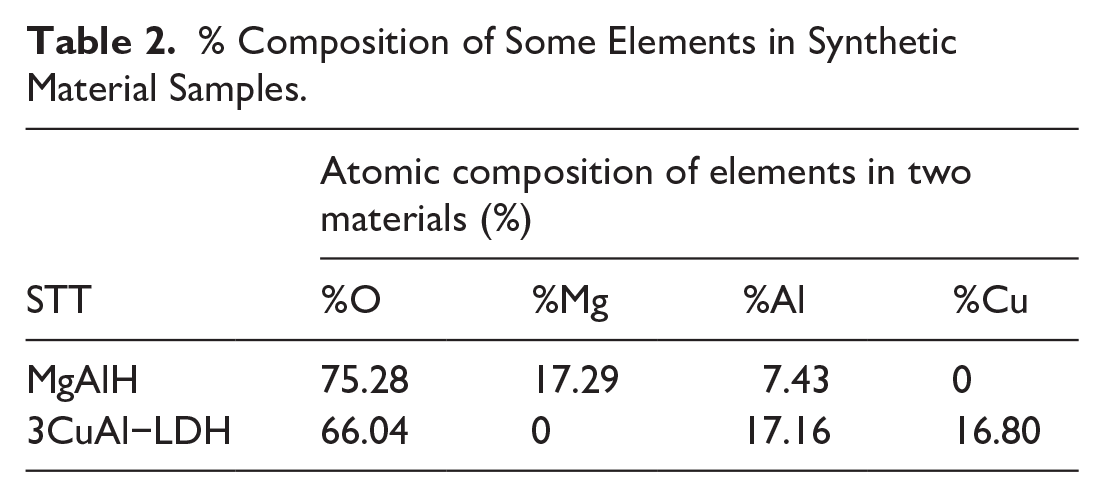
EDX spectrum showed the presence of three elements O, Mg, Al in MgAlH sample and three elements O, Al, Cu in 3CuAl-LDH sample with the atomic percentage composition of the elements listed in Table 2. The molar ratio of Cu:Al and Mg:Al in the two analyzed material samples was completely consistent with the theoretically calculated element ratios for synthesizing the material samples. Specifically, the Mg:Al ratio of MgAlH sample was 7:3 and the Cu:Al ratio of 3CuAl-LDH sample was 3:3.
SEM image of the synthesized materials
The SEM imaging of the two material samples MgAlH and 3CuAl-LDH showed the appearance of the characteristic lamellae of the hydrotalcite structure (Figure 4). It can be seen that the shape of the lamellae of the MgAlH material sample was more uniform and more ordered when compared with the lamellae of the 3CuAl-LDH material. These analysis results proved that when the Cu2+ ions replaced into the brucite network, the lamellae order of the layered double hydroxide materials had been reduced. However, the CuAl-LDH materials still maintained the layered double structure with a suitable Cu:Al molar ratio.

SEM images of two material samples MgAlH (a, b), 3CuAl-LDH (c, d).
UV-Vis diffuse reflectance spectrum (UV-Vis DRS) of the materials
The UV-Vis DRS spectrums of the synthesized material samples (Figure 5(a)) showed that the nCuAl-LDH modified material samples all had an absorption edge strongly shifted to the visible light region when compared with the MgAlH material sample. On the contrary, the MgAlH material sample had an absorption edge located in the UVA region. The material samples 4CuAl-LDH, 5CuAl-LDH, 6CuAl-LDH had absorption edges at wavelengths of 506.5; 488.8; 474.1 nm. The two material samples 2CuAl-LDH, 3CuAl-LDH had an extended absorption band in the range of 300–800 nm, due to the appearance of CuO phase according to the XRD analysis results. In addition, the comparison material sample MgAlH had an absorption edge at wavelength of 352.7 nm in the UVA region. As such, it can be concluded that the role of the Cu2+ ions in the layered double structure had strongly shifted the absorption edge of the nCuAl-LDH material samples. Therefore, these CuAl-LDH material samples were predicted that they had high photocatalytic activity in the visible light region.

UV-Vis DRS spectra of the synthesized samples (A) and Taucl plots to determine the band gap energy Eg of the samples (B).
To determine the band gap energy of the composite samples, the Kubelka–Munk equation and Taucl plots were used (Figure 5(b)) 8

In equation (4), α was the absorption coefficient, h was Planck’s constant and ν was frequency of vibration. Eg was the band gap energy and A was the proportional constant. The band gap energy Eg of the six synthesized samples was determined to be 3.52 eV (MgAlH), 2.65 eV (6CuAl−LDH), 2.54 eV (5CuAl−LDH), 2.40 eV (4CuAl−LDH), 1.44 eV (3CuAl−LDH), 0.95 eV (2CuAl−LDH). Thus, the two material samples 2CuAl–LDH, 3CuAl–LDH had the most similar structure to hydrotalcite, had the CuO phase, had small band gap energy, so they were predicted to have good photocatalytic activity under visible light.
Results of survey on CIP adsorption and decomposition capacity of material samples
Results of survey on CIP adsorption capacity in the dark of material samples
The results of the adsorption capacity survey of six synthesized samples showed that all synthetic material samples had the ability to adsorb the CIP 50 ppm in aqueous environment (Figure 6). The CIP 50 ppm adsorption efficiency on the synthesized materials decreased in the following order: MgAlH; 3CuAl−LDH; 2CuAl−LDH; 4CuAl−LDH; 5CuAl−LDH; 6CuAl−LDH. The maximum adsorption efficiency at 60 min of survey was about 23; 12; 16; 9; 8; 8% corresponding to the 6 material samples in the above order. In addition, 60 min of stirring in the dark can be selected as the adsorption equilibrium time of the synthesized material samples.

The CIP 50 ppm adsorption efficiency over time of the MgAlH, nCuAl−LDH samples.
Results of the investigation of the ciprofloxacin photodecomposition ability of the synthesized materials
Results of the investigation of the influence of illumination time and dosage of Cu2+ ion in the synthesized materials
After stirred in the dark for 60 min to reach adsorption equilibrium, 1.2 mL of solution H2O2 30% was added to the six reaction mixtures to survey the CIP 50 ppm degradation ability of these materials. Based on the results obtained in Figure 7, it can be seen that the CIP 50 ppm photocatalytic degradation efficiency of the five nCuAl-LDH materials all increased with the illumination time. The CIP treatment ability of the materials was arranged in decreasing order as follows: 3CuAl-LDH; 2CuAl-LDH; 4CuAl-LDH; 5CuAl-LDH; 6CuAl-LDH. The 3CuAl−LDH sample had the best CIP photocatalytic degradation ability, the photocatalytic degradation efficiency of 50 ppm CIP achieved about 76% after 240 min of illumination (the total CIP 50 ppm treatment efficiency achieved about 95% after 300 min of survey). When compared with the Cu-AlLDH.Bi2O3_CO32- (denoted as LDHB-C) and Cu-Al LDH.Bi2O3_SO42- (denoted as LDHB-S) material samples, the photocatalytic degradation efficiencies of the two material samples for the 50 ppm diclofenac sodium were 65% and 83%, respectively, the synthesized nCuAl-LDH material samples could also decompose CIP well at a concentration of 50 ppm. 22
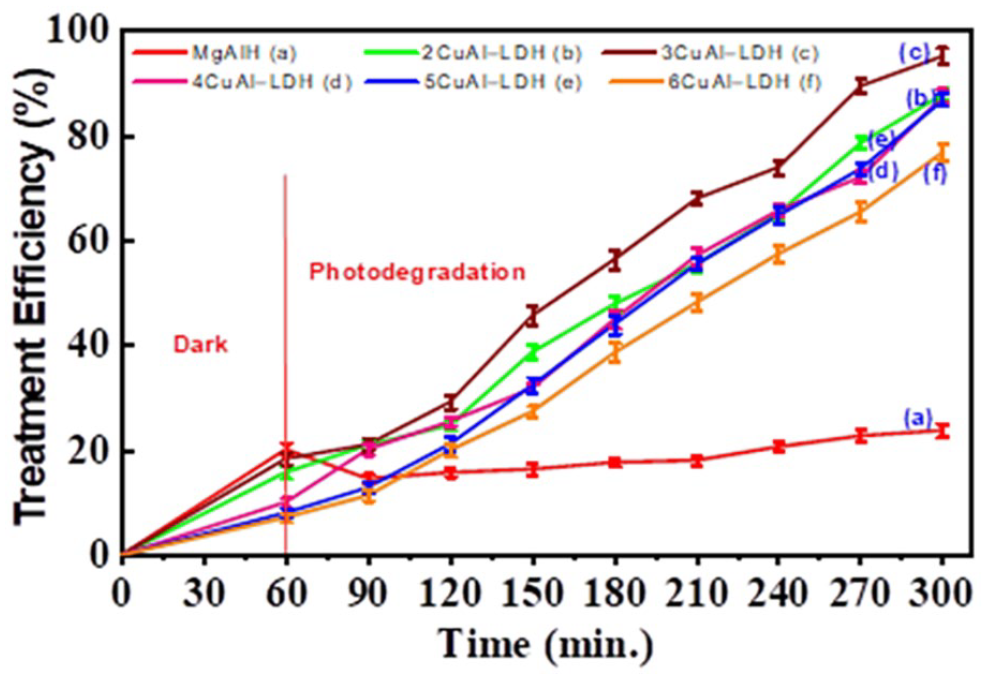
The CIP 50 ppm treatment efficiency over time on MgAlH, nCuAl-LDH material samples.
These survey results were consistent with the structural characteristics, light absorption edge shift and band gap energy of the composite samples. Based on the above survey results, the material sample 3CuAl−LDH was chosen to conduct a survey of the factors affecting the photocatalytic activity of the material (CIP concentration, environmental pH, etc.).
The CIP decomposition mechanism on nCuAl-LDH materials could been explained by the following equations 21 and Figure 8 23





CIP photodecomposition diagram of 3CuAl-LDH material sample. 23
Results of the investigation of the influence of ciprofloxacin concentration on the photocatalytic activity of the synthesized material samples
From the original concentration of CIP of 100 ppm, a series of solutions with CIP concentrations of 10, 25, 40, 50, and 75 ppm were prepared to investigate the influence of CIP concentration on the photocatalytic activity of the 3CuAl−LDH material sample. The obtained results in Figure 9 showed that the CIP concentration in the solution clearly affected the photocatalytic activity of the 3CuAl−LDH material sample. When increasing the CIP concentration from 10 to 75 ppm, the CIP treatment efficiency gradually decreased. After 300 min of investigation, CIP treatment efficiency decreased from 93.3–92.2–90.2–89.2–64.5% corresponding to concentrations of 10, 25, 40, 50, and 75 ppm.

The treatment efficiency at different concentrations of CIP on the 3CuAl-LDH sample.
The above results were explained as follows: at low concentrations, CIP molecules had the ability to disperse well on the surface of the material, and at the same time, the ability to receive energy to activate the material was higher due to less shielding by substances in the mixture, resulting in higher CIP decomposition efficiency. When increasing the CIP concentration in the solution, due to the shielding effect of CIP molecules on the material, the amount of HO• radicals decreased, reducing the photocatalytic decomposition efficiency of the material.
When comparing the photocatalytic activity with NiAlCe LDH/RGO, CoAl-LDH/BiOBr, NiAlFe LDH/RGO, Pt/LDH materials in the photocatalytic degradation reaction of CIP 10 ppm and ZnAl-LDH/g-C3N4 material decomposing CIP 20 ppm, the 3CuAl-LDH material sample also had very good CIP degradation ability, the treatment efficiency of CIP 10 ppm, 25 ppM achieved 93.3% and 92.2%, respectively.24 –28 However, the outstanding feature of the 3CuAl-LDH material sample was ability to decompose CIP at much higher concentrations than materials’ feature in documents above. The CIP treatment efficiency of 40 and 50 ppm had been reached over 90% after 240 min of illumination. In addition, the experiments were surveyed by our group, the light source of the LED 30 W lamp was used to irradiate, so it was more energy-saving, reducing the cost of electricity consumption.
Results of the investigation of the influence of environmental pH on the catalytic activity of the synthesized material samples
The media was one of the important parameters that mainly affected the adsorption and photocatalytic activity of materials. For ciprofloxacin, the environment determined the forms of CIP in solution because CIP had two values of pKa1 = 6.09 and pKa2 = 8.62. 29 In acidic medias, CIP existed in the form of a cation due to the cationization of the amine group (pH ⩽ pKa1). In basic environments, CIP was in the form of an anion as pH ⩾ pKa2. In addition, CIP was form of zwitterion ion in the pH range of the environment between the two pKa values above. Furthermore, the environment also affected the surface charge of the 3CuAl-LDH material sample. The results of the zeta potential analysis of the 3CuAl-LDH material sample was shown in Figure 10. In the range of four pH values used to determine the zeta potential of the 3CuAl-LDH material sample, the zeta potential measurement results were as follows: 42.8 mV at pH = 4.0, 41.8 mV at pH = 6.0, 30.2 mV at pH = 8.0, and 10.9 mV at pH = 10.0. Thus, it can be concluded that the zeta potential of the 3CuAl-LDH material sample was always positive in the range of pH ⩽ 10.0. The zeta potential tended to decrease when increasing the pH value of the environment from acidic to basic environment. Therefore, the positive charge density on the material surface decreased when the pH values increased. In addition, the zeta potential of the 3CuAl-LDH sample was fit with the zeta potential of the CuAl-LDH material sample (41.6 mV) in the literature. 19

Zeta potential of 3CuAl-LDH material sample at investigated pH values.
The 50 ppm CIP solutions were adjusted from 5.8 pH value to the pH values to be investigated by using NaOH and HCl solutions with concentrations of 0.1 and 0.5N. The medias of the 50 ppm CIP solutions were obtained having pH values as 3.0, 4.0, 8.0, 10.0, 12.0. The 3CuAl-LDH material sample was used to investigate the influence of environmental pH on the photocatalytic activity of the material. The results obtained in Figure 11 showed that the the 3CuAl-LDH sample had a high photocatalytic activity in a wide pH range from 4.0 to 8.0. The photocatalytic activity of the investigated material decreased when the environmental pH was in the strong acid region (pH = 3.0). In particular, the ability to decompose CIP 50 ppm decreased sharply in a strong base environment (pH = 12.0). At low pH value, the material was dissolved in the acid solution, forming Cu2+ ions, reducing the number of Cu2+ catalyst sites, thus reducing the photocatalytic activity. 30 In addition, both CIP and investigation material were positively charged in a strong acidic environment, so the ability to CIP adsorb and decompose over 3CuAl-LDH sample in this environment was not favorable. In a strong base environment (pH > 10), due to the increased viscosity of the solution, the ability of the material to receive light energy was reduced, leading to a decrease in photocatalytic activity. Therefore, the most suitable environment for CIP decomposition was in the pH range from 4.0 to 8.0. This survey result was in complete agreement with the zeta potential and pKa values of CIP as analyzed above.

The degradation efficiency of CIP at 50 ppm concentration over time on 3CuAl-LDH sample at different pHs.
Results of investigation on the role of catalyst and H2O2 in the AOPs
To evaluate the role of catalyst material, H2O2 and light source, the following three investigations were carried out: (1) The reaction mixture was added with 0.2 g of 3CuAl-LDH material and 1.2 mL of H2O2 30%; (2) The reaction mixture was added with only 0.2 g of 3CuAl-LDH material; (3) The reaction mixture was added with only 1.2 mL of H2O2 30%.
As comparing the CIP 50 ppm degradation capacity in the three cases above, the obtained results showed the role of the combination of the 3CuAl-LDH catalyst material sample and H2O2 in the advanced oxidation process of CIP degradation under visible light (Figure 12). This combination had created many hydroxyl radicals OH• that directly participate in the CIP degradation process. In the case of full presence of the material, and H2O2, the photodegradation efficiency of the material was very high, reaching over 72% after 240 min of illumination. In the case of no material, only presence of H2O2, the CIP 50 ppm degradation efficiency was negligible, only reaching 7.4%. This result was explained by reason that the energy of visible light was not enough to convert H2O2 into OH• radicals. In addition, if there was only catalyst material without H2O2, the adsorption process occured mainly, so the highest CIP treatment efficiency only reached about 32%. Under the effect of visible light, a small amount of O2 dissolved in the solution was able to be converted into radicals O2•-. These free radicals had less activity than OH• radicals, the CIP decomposition efficiency increased insignificantly after 240 minutes of illumination.

CIP degradation efficiency in three cases: the presence of catalyst material and H2O2, only presence material, only presence H2O2.
Table 3 was the comparison of the photocatalytic activity of the 3CuAl-LDH synthesized sample with previous research results.
Comparison of CIP Degradation Ability of Materials.

From the comparison results table above, it can be seen that the 3CuAl-LDH material sample demonstrated high photocatalytic activity under the investigated conditions. The highlight of this material sample was that it could decompose CIP at concentrations higher than 10 ppm with a decomposition efficiency of about 90%, and at the same time, using a 30 W LED light source. This light source saved on electricity costs and lighting equipment purchase costs when compared with high-power Xenon light sources in the literature,24,25,27,28,34 metal halide lamp, 33 and tungsten lamp. 37 In addition, compared with seven catalyst material samples in the literature, 35 and samples in documents,36,37 the 3CuAl-LDH material sample also showed much better CIP treatment ability. Therefore, it can be concluded that the 3CuAl-LDH material sample had the ability to handle CIP at high concentrations, which will help ciprofloxacin antibiotic production plants to effectively treat this antibiotic before discharging into the environment.
Conclusion
The six samples of MgAlH and nCuAl-LDH were successfully synthesized by the co-precipitation method. The results of the study on the structural characteristics, surface morphology and material properties showed that the three synthesized samples all had a lamellar structure similar to hydrotalcite (MgAlH, 2CuAl-LDH, 3-CuAl-LDH). The presence of Cu2+ ions in the Bruxit network degraded the lamellar structure of layered double hydroxides, especially the materials with high molar ratios of Cu:Al (Cu:Al = 4:3, 5:3, 6:3). However, the nCuAl-LDH samples had a strong shift of the absorption edge to the visible region, leading to a sharp decrease in the band gap energy (from 3.52 to 0.95 eV corresponding to the two samples MgAlH, 2CuAl-LDH). The 2CuAl-LDH and 3CuAl-LDH material samples had better photocatalytic activity than the remaining material samples. The 3CuAl-LDH sample showed the best activity. The CIP treatment efficiency at concentration of 50 ppm reached over 90% after 240 min of illumination in the presence of 1.2 mL of H2O2 30%. The optimal pH environment for CIP degradation was in the range of 4.0–8.0. The survey results obtained will promise the ability to treat antibiotics in the aquatic environment. In particular, the nCuAl-LDH material samples can decompose CIP at high concentrations when using a 30 W LED light source.
Supplemental Material
sj-docx-1-chl-10.1177_17475198251344773 – Supplemental material for Enhanced photocatalytic degradation efficiency of ciprofloxacin by CuAl layered double hydroxide materials
Supplemental material, sj-docx-1-chl-10.1177_17475198251344773 for Enhanced photocatalytic degradation efficiency of ciprofloxacin by CuAl layered double hydroxide materials by Van Nhuong Vu, Thi Thu Trang Hoang, Thi Ha Thanh Pham and Thi Kim Ngan Tran in Journal of Chemical Research
Footnotes
Authors’ Note
The authors declare that this manuscript has been read and approved by all authors, and that the content represents honest and original work.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author Contributions
Van Nhuong Vu: Writing—original draft. Van Nhuong Vu and Thi Kim Ngan Tran: Writing—review & editing. Thi Thu Trang Hoang: Data curation. Thi Ha Thanh Pham: Conceptualization.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Thai Nguyen University of Education under grant number DH2023−TN04−05.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding authors upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
