Abstract
From the measured density values of the mixtures between the tire-derived oil and toluene and xylene between 293.15and 323.25 K, the excess molar volumes were determined, and it was seen that the excess molar volumes calculated with the Redlich–Kister polynomial equation were in good agreement. It was determined that the excess molar volume showed a positive deviation at each temperature and that physical interactions between the components forming the mixture were dominant. It was seen that chemical forces were dominant due to intramolecular and intermolecular interactions such as charge-transfer complexes in the pyrolytic oil consisting of electron donor and electron acceptor components. It was observed that the physical and chemical forces between the components varied depending on the temperature, and therefore, the excess molar volume changes were not linear at 323.15 K.

Introduction
The increasing amount of used tires in the world poses great risks to the environment, and recently, the pyrolysis method has come to the fore in the fight against waste tires.1,2 Pyrolysis is a method in which hydrocarbons are thermally decomposed in an oxygen-free environment. High calorific value of tire-derived pyrolytic oil (TDPO) is obtained from the pyrolysis of scrap tires.1 –3
More than 60% of pyrolytic oil is composed of monoaromatic and polyaromatic molecules, with the remainder consisting of straight-chain aliphatic hydrocarbons and a very small portion of olefinic structures. 4 Aromatic components include benzene, toluene, xylene, limonene, ninhydrin, and their alkyl homologues. 4 Due to its high sulfur content (1%–2%) and acidity, pyrolytic oil is a thermally unstable fuel. With low-boiling components, pyrolytic oil has a flash point of <30 °C. 4 Due to physical properties like high density (>900 kg m−3), high sulfur content, and low flash point, pyrolytic oil cannot be used directly as fuel. 4 Common upgrading methods for pyrolytic oils include hydrotreating, 5 fractional distillation, 6 adsorption, and chemical refining. 7
Excess molar properties represent the difference between ideal and observed properties of mixtures. 8 Physical interactions such as van der Waals forces9,10 and dispersive forces, 11 chemical interactions like dipol–dipol interactions, 12 hydrogen bonding 13 and charge-transfer complexes, 14 and structural interactions related to the geometric structures of molecules occur among the mixture’s components. 15 Positive deviations in excess molar volume values arise from physical forces, while chemical interactions reduce these values. Structural interactions can cause both positive and negative deviations in excess molar volume. Physicochemical properties such as excess molar volume are crucial in various biochemical and industrial applications, such as storage, transportation, interaction with drugs, and packaging. 16
For instance, Pawar et al. 16 examined the excess molar volumes of ethanol–water binary mixtures, highlighting ethanol’s role as an extractive solvent in the chemical industry, as a carrier and additive in food and cosmetics, and as an antimicrobial agent. In water–alcohol mixtures, hydrogen bonding and the molecular complexes formed by the −OH group of alcohol as a π-electron donor and hydrogen as an s-electron acceptor play a significant role. Tegegn et al. 17 explained that understanding drug interactions with large biomolecules like sugars, proteins, and carbohydrates is challenging but can instead be explored through solvent–solute interactions in aqueous solutions using properties like volumetric and acoustic conductivity data. Similarly, Barabas 18 emphasized that knowing the density and volumetric properties of fuel mixtures is essential for adjusting optimal air–fuel ratios and fuel dosing. He investigated the excess molar volumes and densities of biodiesel and ethanol mixtures over the range of 273.15–333.15 K, attributing positive deviations in excess molar volume to physical interactions between biodiesel and ethanol. Vural et al. 19 studied the excess molar volume and thermodynamic properties of biodiesel with toluene and benzene at 298.15 and 303.15 K, observing that physical interactions dominate due to biodiesel’s long-chain structure interacting with the smaller benzene and toluene molecules.
Because of pyrolytic oil’s high density, blending it with density-reducing components like toluene and xylene could make it a more suitable fuel. Adding low-boiling aromatic components to the whole pyrolytic oil without separating it into fractions can increase its aromatic character and improve the dispersion of high molecular weight resinous phases within the oil. In addition, adsorption–precipitation techniques could enhance the efficiency of physicochemical refining for pyrolytic oil.
In this study, the excess molar volumes of pyrolytic oil with two aromatic components, toluene and xylene, were determined in the range of 293.15–323.15 K. This allowed the investigation of physical, chemical, and structural interactions between the pyrolytic oil and these aromatic components, as well as the effect of temperature on the mixture.
Experimental
Anhydrous toluene used in the study was 99.6% pure, isomeric xylene was 75% pure (25% ethyl benzene) and was supplied by Merck, and no further purification was done. Anhydrous toluene is referred to as toluene, and isomeric xylene is referred to as xylene. The physical properties of both components supplied by Merck and experimental data are given in Table 1. The densities were measured with an Anton Paar DMA 38 vibrating U-tube densimeter. The densimeter consists of a built-in thermostat controller capable of maintaining temperature precisely to ±0.01 K and measuring density to ±0.0001 g cm−3. Before each series of measurements, the apparatus was calibrated at atmospheric pressure using double-distilled and degassed water. In constant temperature experiments, Elektro-mag M 96 KP PID Microprocessor Controlled, 96 Tube, Digital Thermostat Bacteriological Water Bath was used. Experiments were carried out in a fume hood to avoid toxic effects during the study.
Densities of xylene, toluene, and TDPO at different temperatures.

The tire pyrolysis test was performed according to the method in the literature. 12 In total, 100 g of 1–3 mm tire crumbs obtained from recycling companies were weighed and placed in a round-bottomed flask. A distillation head with a thermometer and a water-cooled cooler were added to the flask. The pyrolysis process was completed at 450 °C in a mantle heater. Non-condensed gases were passed through two series-connected wash bottles containing NaOH solution and burned in a fume hood. The condensed pyrolytic oil (yield 45%) was transferred to another flask, and the fraction between 60 and 280 °C was collected in a single container. It was treated with 1% active clay and filtered to remove impurities and moisture.
Samples consisting of pyrolytic oil + solvent mixture were placed in 10 cm3 test tubes by increasing 0.1 mole fraction in the range of 0.0–1.0. Before density measurements, the mixtures in the test tubes were brought to thermal equilibrium for 1 h in a thermostatic water bath at temperatures of 293.15, 298.15, 303.15, and 308.15 K, respectively. The density of each mixture and the pure components were measured separately and are presented in Table 2. MATLAB R2022b software was used to calculate excess molar volume and related parameters.
Densities of mixtures of xTDPO + (1-x) C7H8 and xTDPO + (1-x) C8H10 at different temperatures.

Result and discussion
Excess molar volume is the difference between the ideal volume of a mixture and its observed volume. The excess molar volume is mathematically defined 19

where VE is the excess molar volume, V is the volume of the mixture containing TDPO and toluene or xylene, and xi and Vi are the mole fraction of each component and the molar volume of the pure components, respectively.
Equation (1) can be written as

where ρi and ρ are the densities of mixtures and pure components, respectively, and Mi is the molecular weight of pure components.
The excess molar volume calculated from the experimental data is in agreement with the Redlich–Kister equation, 19 which is one of the best estimation equations according to the experimental data, and is provided by the equation given in equation (3)

where VE is the excess molar volume, xi is the mole fraction, and i is the degree of the polynomial equation. Ai values were calculated by using the non-linear extrapolation method in MATLAB, and curve fitting VE values to mol fractions. The adjustment of the parameter of the Redlich–Kister equation, Ai, and the corresponding standard deviations, σ(VE), were calculated by using the equation 19

where adj is the adjusted data, and n and p are the numbers of experimental points and numbers of parameters, respectively.
The measured and calculated values of the densities, excess molar volumes, and the calculation result of Coefficient of Redlich–Kister equation of the binary mixtures of xTDPO + (1-x) toluene and xTDPO + (1-x) xylene were given in Tables 2–4, respectively.
Excess molar volume (VE) of mixtures xTDPO + (1-x)C7H8 and xTDPO + (1-x)C8H10 at different temperatures.

Coeffcients Redlich and Kister equation and standard deviation of binary mixtures.

As seen in Table 2, the density of both mixtures decreases as the temperature increases as toluene and xylene are added. It is expected that the density decreases due to volumetric expansion with increasing temperature. The excess molar volumes (VE) of binary mixtures of TDPO + toluene and TDPO + xylene were calculated from the mole fractions, molecular weights, and densities according to equation (2) and listed in Table 2 and Figure 1. As seen in Figure 1 and Table 3, the excess molar volumes of binary mixtures of TDPO with toluene and xylene showed positive deviations. It can be clearly stated that physical interactions such as dipole interactions, van der Waals forces, and dispersive forces are dominant in the mixture rather than chemical interactions (such as hydrogen bonds, charge-transfer complexes) between TDPO and toluene or xylene. In addition, the sudden increase in excess molar volume for toluene and the decrease for xylene at high temperatures indicate that intermolecular and intramolecular effects also occur. TDPO contains many BTX molecules as well as multi-ring components in its chemical structure. Molecules with high aromaticity in TDPO increase the electron density of TDPO. σ-bonds originating from aliphatic structures in TDPO act as electron acceptors and can form charge-transfer complexes with aromatic components. Intramolecular interactions occur due to electron donor and electron acceptor components in the chemical structure of TDPO.
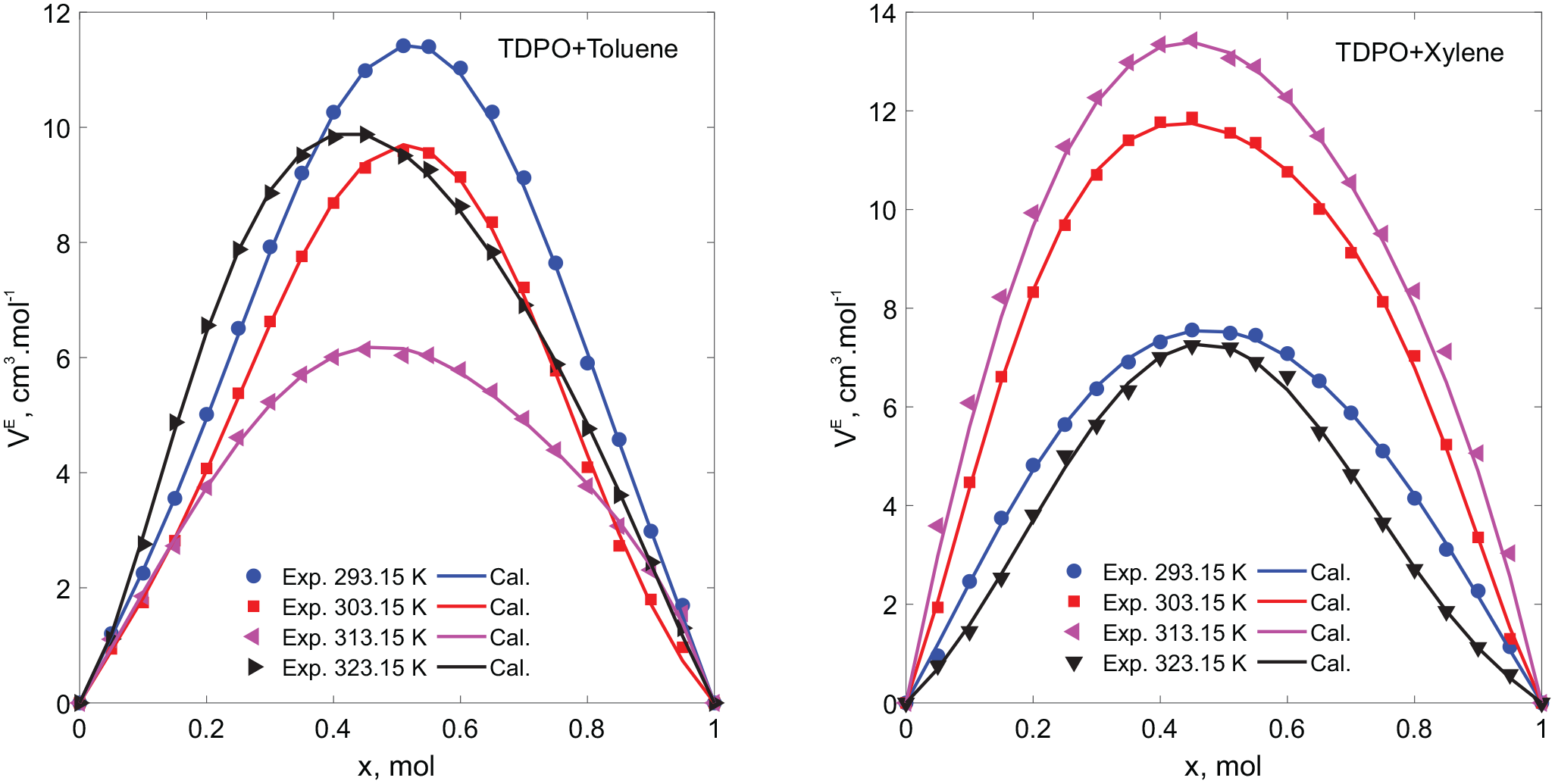
Excess molar volumes of TDPO + toluene and TDPO + xylene mixtures in the range of 293.15–323.15 K calculated from the Redlich–Kister equation and experimental data.
As seen in Figure 1 and Table 3, the excess molar volume values calculated from the density values of the TDPO + Toluene mixture decreased as the temperature increased. At 323.15 K, the excess molar volume values deviated from the linear decrease and showed a more positive deviation than at 303.15 K. This may be due to the fact that the dispersion of toluene in TDPO increases more with increasing temperature. It can be said that at high temperatures, the intramolecular or intermolecular interactions of TDPO weaken.
As seen in Figure 1 and Table 3, the excess molar volume values calculated from the density values of the TDPO + Xylene mixture were observed to increase as the temperature increased, contrary to what was observed in the TDPO + toluene mixture. The excess molar volume values at 323.15 K showed an unexpected decrease. The reason for the decrease in excess molar volume beyond 323.15 K means that chemical interactions such as charge-transfer bonds between xylene and TDPO molecules are strong. While physical interactions such as dispersion forces increase excess molar volume values, the reason for the decrease in excess molar volume values for xylene at high temperatures may be that chemical interactions are strengthened in addition to the strong physical interactions between molecules.
Vural et al. 19 investigated the excess molar properties of biodiesel with benzene and toluene, and the authors determined that due to the long-chain structure of biodiesel, the intermolecular interactions between benzene and toluene are weak and London dispersion forces are dominant. Similar to the studies of Vural et al., 19 it was observed that intramolecular interactions were strengthened at low temperatures and weakened at high temperatures in mixtures of toluene + pyrolytic oil. In mixtures of xylene and pyrolytic oil, on the contrary, intramolecular interactions were weakened at low temperatures. This may be due to the fact that xylene has more −CH3 groups and has a more positive structure than toluene.
The excess molar volume values calculated from the experimental data were determined by using the Redlich–Kister polynomial equation, the coefficients of the equation were determined by curve fitting with the least squares method and the standard deviation values were calculated, and the results obtained are given in Table 4. As can be seen from Table 4, the coefficients of the equation showed great agreement with the experimental data. As seen in Figure 1, the excess molar volume values obtained from experimental methods showed great agreement with the excess molar volumes calculated from the Redlich–Kister polynomial equation.
Conclusion
In this study, excess molar volumes of two-component mixtures between toluene and xylene with fraction <280 °C of pyrolytic oil obtained from tire pyrolysis were determined. It was observed that physical interactions such as dipole interactions, van der Waals forces, and dispersive interactions dominated the excess molar volume values of the mixtures. It was understood that charge-transfer interactions began to appear as the temperature of the mixture increased.
Toluene and TDPO mixtures showed good dispersion and volumetric expansion at low temperatures. At high temperatures such as 323.15 K, intermolecular and intramolecular interactions began to weaken, and volumetric expansion tended to increase. In xylene and TDPO mixtures, although the dispersion of xylene was weaker at low temperatures, chemical interactions began to strengthen above 323.15 K. In general, excess molar volumes of mixtures between toluene and xylene with TDPO showed positive deviations. Volumetric expansion should be taken into account in the transportation and storage of these mixtures. The positive deviation of the excess molar volume values provides preliminary insight into optimizing fuel–air ratios.
In this study, it is aimed to determine the effect of temperature on the volumetric behavior of toluene and xylene with pyrolytic oil. The study can be expanded with different fractions obtained by distillation of pyrolytic oil. In further studies, not only excess molar volumes but also thermal expansion coefficients, viscosities, and thermodynamic parameters in different fractions of pyrolytic oil will provide a better understanding of the mixtures. This study will shed light on the more detailed investigation of upgraded pyrolytic oil fractions.
Footnotes
Acknowledgements
The author thanks Süleyman Demirel University for their support in the study.
Ethical considerations
This paper does not contain any studies with human or animal participants.
Consent to participate
This paper has no human participants, and informed consent is not required.
Consent for publication
The authors declare that they have no known competing financial interests or personal relationships that may affect the work reported in this paper.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available on request from the corresponding author.
