Abstract
This work presents a comprehensive study on the effects of pyrolysis parameters (pyrolysis temperature, residence time, and heating rate) on the distribution of pyrolysis products of Miscanthus. Py-GC/MS (Pyrolysis-gas chromatography/mass) was conducted to identify building blocks of value-added chemical from Miscanthus. The results showed that the main pyrolysis products of Miscanthus were ketone, aldehyde, phenol, heterocycles, and aromatic compounds. The representative compounds of ketone and aldehyde compounds produced at different pyrolysis temperatures changed obviously, while the representative compounds of phenolic, heterocyclic, and aromatic compounds had no obvious change. Large-scale pyrolysis of Miscanthus had begun at 400°C, and the relative content of pyrolysis products from Miscanthus reached the maximum of 98.34% at 700°C. The relative peak area ratio of phenol and aromatic compounds reached the maximum and minimum at the residence time of 5 and 10 s, while the relative peak area ratio of ketone compounds showed the opposite trend. The relative peak area ratio of aldehyde compounds was higher under shorter or longer residence time. For heterocyclic compounds, the relative peak area ratio reached the maximum of 27.0% at residence time of 10 s. The faster or slower heating rate was beneficial to the production of aldehyde and phenol compounds. The relative peak area ratio of ketone compounds reached the maximum at 10,000°C/s, 70°C/s, and 10°C/s, and the relative peak area ratio tendency of heterocyclic compounds was similar to ketone. For aromatic compounds, the overall fluctuations were large, and the relative peak area ratio was the highest at the heating rate of 100°C/s.
Introduction
In the recent years, exhaustion of conventional resources and increasing greenhouse gases emission such as carbon dioxide, which have resulted in global climate change, push countries to find alternative solutions. Fawzy et al. 1 studied three main climate change mitigation approaches, including conventional mitigation technologies, negative emissions technologies, and radiative forcing geoengineering techniques. First, the conventional mitigation technologies employ decarbonization technologies and techniques that reduce CO2 emissions, such as renewable energy and fuel switching. Second, the negative emissions technologies are potentially used to capture and sequester CO2 from the atmosphere, including bioenergy carbon capture and storage, biochar, enhanced weathering, direct air carbon capture and storage, and so on. Sharma and Dhir 2 reviewed the CO2 adsorption on carbonaceous and non-carbonaceous solid adsorbents, because of their lower cost, higher energy efficiency, and easier versus solvent-based adsorbents. Carbonaceous adsorbents such as activated carbon and biochar have tunable surface properties, low moisture sensitivity, and high thermal stability. Finally, radiative forcing geoengineering techniques include stratospheric aerosol injection, marine sky brightening, cirrus cloud thinning, space-based mirrors, surface-based brightening, and various radiation management techniques. The principle of the techniques is to alter the earth’s radiation balance by managing solar and terrestrial radiation. The use of biomass energy is in the negative emission technologies, which can effectively alleviate climate change and energy pressure.
Miscanthus is regarded as the highly promising energy crop. Miscanthus is one of the energy plants with the highest dry matter output at present, which has strong resistance to barren and high net yield with an annual productivity of 10–40 t (dry matter) per hectare. 3 Miscanthus is originated from the tropical regions and subtropical of Asia and Africa, and China has extensive Miscanthus resource with a wide distribution through the entire climatic zone of the country.3,4 Same to other biomass resources, Miscanthus has low bulk energy density. It is therefore important to develop the efficient utilization of Miscanthus for optimizing its energy application.
Among various thermochemical processes, pyrolysis is considered as the most efficient process for recycling municipal solid waste due to its high fuel-to-feed ratio and environmental friendliness. 5 Pyrolysis can be described as the heating of biomass in the absence of oxygen in the range of 300°C–900°C, which results in the production of low-molecular-weight gases, biochar, and liquids (bio-oil).6,7 The production of bio-oil is usually accomplished through the fast pyrolysis. 8 Fast pyrolysis is characterized by very high heating rate, reaction temperature, and short residence time. The ratio of cellulose, hemicellulose, and lignin determines the degree of ease of pyrolysis. The contents of cellulose, hemicellulose, and lignin in Miscanthus are 43.1%–52.2%, 24.8%–34.0%, and 9.2%–12.3%, respectively. The content of lignin is lower than that of rice straw (12.43%), corn straw (15.38%), and wheat straw (15.0%).9–11 The cellulosic biomass is pyrolyzed easily if the content of lignin is lower. Compared with rice straw, corn straw, and wheat straw, Miscanthus is regarded as a desired bioenergy crop for production of biofuels, while there are challenges that come with Miscanthus thermal decomposition. Nitrous oxide such as N2O, NO, and NO2 are produced during the combustion of Miscanthus. Moreover, the mineral Si in Miscanthus can lead to the low temperature melting behavior of Miscanthus. 12 Yao et al. 13 studied the pyrolysis process of Miscanthus, corn stalk, rice husk, and pine by thermogravimetry. The results showed that corn stalk and Miscanthus were susceptible to pyrolysis because the activation energy of Miscanthus, corn stalk, rice husk, and pine were 46.7, 29.3, 54.3, and 58.1 kJ/mol, respectively. The ash content of Miscanthus was only 3.38% 14 (rice straw, corn straw, and wheat straw were 14.0%, 13.84%, and 6.77%, respectively), which could not only significantly improve combustion temperature and calorific value of biomass but also produced less dirt.15–17
Biomass pyrolysis is a very complex series of reaction, which depends on many different variables, such as pyrolysis temperature, heating rate, residence time, and feedstock. Many scholars have shown that the pyrolysis temperature is the most important variable in pyrolysis of biomass.
Zhang et al. 18 studied the pyrolysis characteristics of rubber seed oil at high temperatures in a pyrolysis system of fixed bed and the reduction products of Fe3O4 and carbon deposition were qualitatively analyzed. The result showed that Fe3O4 promoted the cracking of rubber seed oil and the reduction effect of Fe3O4 was improved with increasing temperature under injection conditions. The lowest conversion rate of carbon deposition at 1300°C was 39.99%. Liu et al. 19 performed detailed analysis on the solid products from fast pyrolysis of rice straw, bamboo culm, and reed straw under different pyrolysis temperatures in a fixed bed. It was found that the char yield of pyrolysis at high temperature (25.84%–28.84%) was lower than that of pyrolysis at low temperature (29.44%–34.4%). The PAHs content, H/C, and O/C of char decreased with the increase of pyrolysis temperature and the properties of the resultant biochar (p < 0.05) were significantly affected by the pyrolysis temperature. Pyrolysis of waste coffee grounds was carried out under temperature ranging from 400°C to 700°C at atmospheric pressure and inert atmosphere. The results showed that a maximum yield of bio-oil could be achieved at 540°C (36 wt%) where the gas reached a yield 9 wt% and the char reached the 29 wt%. At 700°C, the yield of gas, bio-oil, and char in oxidation reactions which mainly occurred against of cracking (gasification) reached 29%, 20%, and 26%, respectively. 20 Hidayat et al. 21 studied the influence of pyrolysis temperature (400°C, 500°C, and 600°C) on pyrolysis of Imperata Cylindrica in a tubular furnace. Py-GC/MS analysis revealed the presence of furfural, 2,3-dihydrobenzofuran, 4-vinylguaiacol, propenylguaiacol, guaiacol, and 4-ethylphenol. It was shown that the yield of bio-oil increased with the increase of temperature and it reached a peak of 37.16% at 500°C, and Imperata Cylindrica was suitable as feedstock to produce bio-oil via pyrolysis process.
Same to pyrolysis temperature, the pyrolysis characteristics of biomass is affected by the heating rate of pyrolysis. Chen et al. 22 investigated the effects of pyrolysis temperature and heating rate on pyrolysis of poplar wood, which showed that higher pyrolysis temperature and heating rate were beneficial to obtain higher mass and energy yield of bio-oil, while lower pyrolysis temperature and heating rate were favorable to obtain higher mass and energy yield of biochar. However, higher pyrolysis temperature and heating rate contributed to obtain both higher mass yield and energy yield of the non-condensable gas. In general, compared to the heating rate, the pyrolysis temperature had a significant effect on the product properties. The pyrolysis results of pinewood indicated that the output of charcoal decreased and the fast pyrolysis period of biomass moved to high temperature with the increase of heating rate from 0.11 to 10°C/min. 23 The lower heating rate was beneficial to the low temperature carbonization, and the initial charcoal acted as a catalyst for primary biomass decomposition. Lower heating rate was also associated with increased retention of pyrolysis vapor in the biomass which results in an increase in production of secondary charcoal and charcoal. Zhu et al. 24 studied the influence of different heating rates on the pyrolysis of dried sludge. The thermogravimetric analysis showed that the higher heating rate resulted in the increase of the maximum weight loss rate and the peak temperature. The activation energy of dried sludge under different heating rates (10, 20, and 30°C/min) were 70.0, 60.6, and 65.0 kJ/mol, respectively. Fourier transform infrared spectroscopy (FTIR) analysis of gas products conducted that the concentration of CO2 and CO increased gradually and the concentration of CH4 increased first and then decreased with the increase of heating rate. Moreover, the yield of gaseous products and bio-oil increased with the increase of the heating rate.
Some studies have shown that the change of pyrolysis residence time has a great influence on the pyrolysis reaction of biomass.25,26 Gautam and Chaurasia 25 investigated the effect of pyrolysis residence time on the production of bio-oil and individual gas components in the pyrolysis of rice husk, rice straw, bamboo, sugarcane bagasse, and neem bark using a fixed bed pyrolyzer. The results showed that the maximum bio-oil production of 46.9 wt% and the pyrolysis char of 26.2 wt% were obtained from bamboo at 450°C. The highest amount of clean syn-gas (carbon monoxide and hydrogen) was produced for neem bark (52.6 vol%). The optimal yield of bio-oil from rice husk was obtained at residence time of 33 s, while that of the best bio-oil yield from other biomass was 36 s. Kang et al. 26 studied the pyrolysis gas and soot formation characteristics of propane at different temperatures and residence times. It showed that propane began to pyrolyze around 970 K, and the main components of syn-gas were H2, CH4, C2H2, and C2H4. The soot formation rate increased obviously, the soot diameter increased with the increase in temperature when the temperature was higher than 1250 K, and the soot formation amount decreased with the decrease in residence time. Morgan et al. 27 reported the influence of residence time (1–10 s) and pyrolysis temperature (400°C–600°C) on the pyrolysis product distributions of bio-oil, char, and permanent gas produced by Banagrass pyrolysis. The study showed that the optimal bio-oil yield was recorded at 450°C with residence time of 1.4 s, which was about 37 wt% relative to the dry ash-free feedstock (excluding pyrolysis water). At higher temperature or longer residence time, less bio-oil and char were recovered and more gas and light volatiles were produced. Increasing the temperature had a more significant effect on product yields and composition than increasing the residence time of volatiles. The bio-oil yield from banagrass was significantly lower than that of woody biomass or grasses such as switchgrass or Miscanthus, but was similar to barley straw.
Considering all the previous works, the pyrolysis characteristics of rice husk, 28 microalgae, 29 straw, 30 and municipal solid waste 31 were studied, and there was limited research on the pyrolysis characteristics of Miscanthus, in particular comprehensively correlating the detailed pyrolysis mechanism and products. In order to study the reactivity and products of Miscanthus pyrolysis, it is necessary to clarify the factors that affect the pyrolysis process of Miscanthus first and analyze the pyrolysis reaction mechanism. At present, it is still unclear on how the pyrolysis conditions (pyrolysis temperature, residence time, and heating rate) affect the distribution of the pyrolysis products from Miscanthus, as well as the possibility for the directional control of the Miscanthus pyrolysis reactivity by adjusting the pyrolysis conditions. In-depth studies on these knowledge gaps are in need, in order to explore the fundamental reaction mechanism and improve the yield and utilization efficiency of the pyrolysis products.
In this work, Miscanthus is pyrolyzed to produce valuable products as a waste to resource option. Therefore, the effects of the pyrolysis temperature, residence time, and heating rate on the distribution of pyrolysis products of Miscanthus were studied by Py-GC/MS. A wide range of products can be produced under different conditions by pyrolyzing Miscanthus. This will encourage the usage of underutilized feedstock sources such as perennial grass for bioenergy utilization.
Experimental
Feedstock
Miscanthus sample was obtained from a local farm in Changsha City, Hunan Province, China. The sample was crushed and screened to a size of less than 74 μm and dried at 105°C for 6 h before use. The proximate analysis, ultimate analysis, and higher heating values (HHVs) measurement of the received sample was performed in accordance with Chinese standard methods GB/T 212-2008, GB/T 31391-2015, and GB/T 1574-2007, respectively. The results of proximate analysis and ultimate analysis are shown in Table 1.
Proximate and ultimate analysis results of Miscanthus.
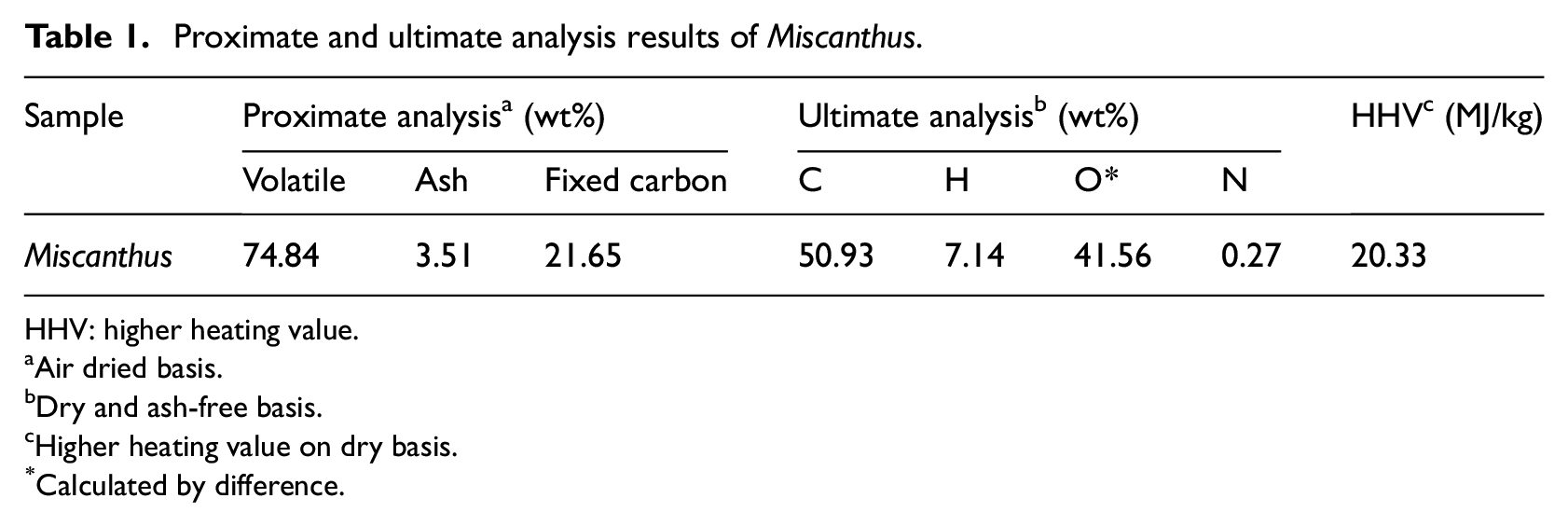
HHV: higher heating value.
Air dried basis.
Dry and ash-free basis.
Higher heating value on dry basis.
Calculated by difference.
Instruments and methods
Miscanthus samples (2 mg) were pyrolyzed using a CDS 5200 pyrolyzer close-coupled to a Shimadzu GC/MS-2010 Ultra gas chromatograph mass spectrometer (GC/MS), to a pyrolysis temperature of 600°C (held for 10.0 s at the pyrolysis temperature) at a heating rate of 10°C/min. The separation was carried out using a Rtx-5 polar column (30 m, 0.25 mm, 0.25
The effect of the pyrolysis temperature (200°C–900°C), heating rate (10,000°C/s, 1000°C/s, 500°C/s, 100°C/s, 70°C/s, 50°C/s, 30°C/s, 10°C/s, 1°C/s), and residence time(2, 5, 10, 15 s) on the distribution of pyrolysis products of Miscanthus was studied.
Results and discussion
Effect of temperature
The distribution of pyrolysis products of Miscanthus under different pyrolysis temperature are shown in Table 2. It could be seen that the relative yield of pyrolysis products of Miscanthus increased from 49.93% to 98.34% with the increase in pyrolysis temperature from 200°C to 900°C. Large-scale pyrolysis of Miscanthus had begun at 400°C, because the relative content of pyrolysis products from Miscanthus at 400°C reached 90%, which is consistent with the reported values of other researcher. 33 Table 2 shows that bio-oil products from the pyrolysis of Miscanthus were divided into seven categories. It can be found that the main products of Miscanthus pyrolysis were ketone, aldehyde, phenolic, heterocycles, and aromatic compounds, mainly due to their large relative peak area, and the relative content of aldehyde and heterocyclic compounds reached 15% and 30%, respectively. Therefore, studying the influence of pyrolysis temperature on relative peak area ratio of the above five compounds was valuable.
Distribution of pyrolysis products of Miscanthus at different pyrolysis temperature.
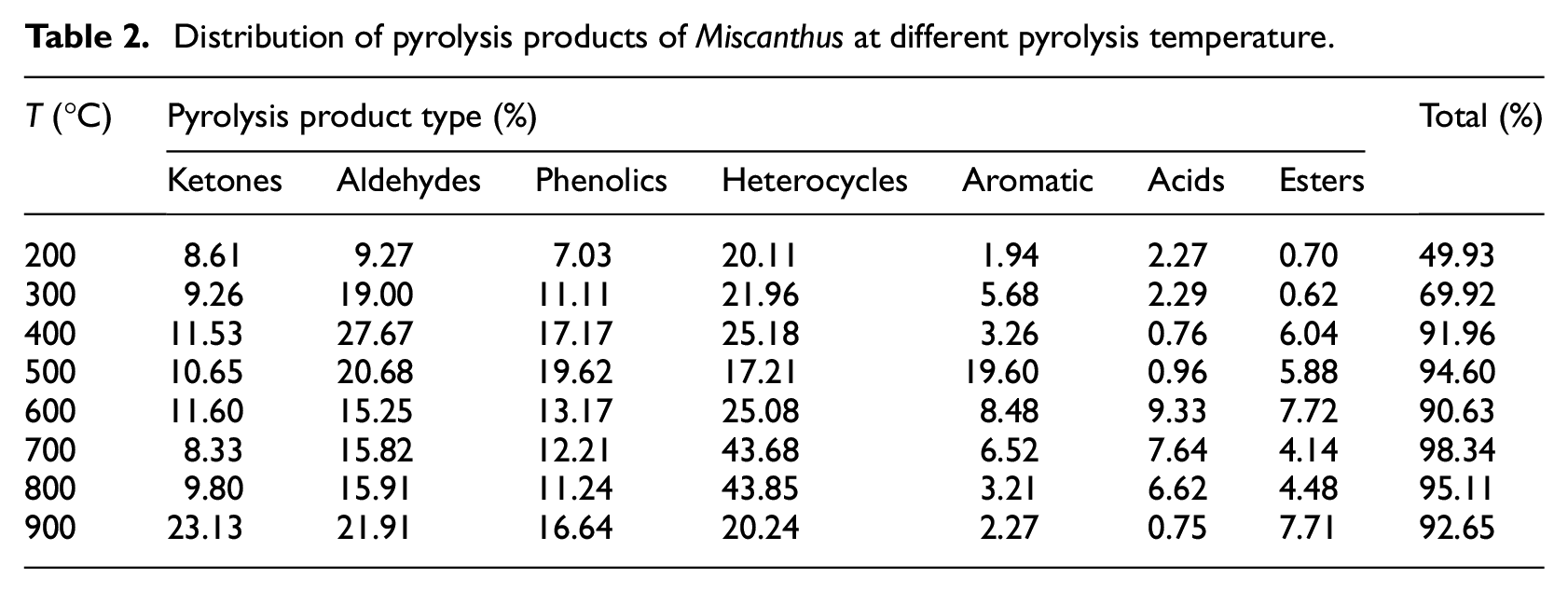
It could be seen that as the pyrolysis temperature increased, different species of products had different change laws. With the increase in pyrolysis temperature, the relative peak area ratio of heterocyclic compounds fluctuated dramatically and reached the peak in the temperature range of 700°C–800°C. The tendency of relative peak area ratio of ketone was that they increased from 8.61% to 11.6% during 200°C–600°C, then decreased from 11.6% to 8.33% in 600°C–700°C and increased greatly from 8.33%–23.13% in 700°C–900°C. The relative peak area ratio tendency of aldehyde was similar to ketone. The relative peak area ratio of aromatic compounds reached the maximum of 19.6% at 500°C. The reason is that lignin decomposes at 280°C–550°C and generates phenolic compounds via cleavage of ether and carbon–carbon linkages. 34 The progressive transformation from cellulose and lignin to a highly aromatic structure with the increase in temperature is shown in the literature. 35 The large number of small molecule compounds was produced by the pyrolysis of Miscanthus, resulting in the increase of ketone, aldehyde, phenol, heterocycles, and aromatic compounds. The relative content of heterocyclic compounds decreased because of the massive decomposition of heterocyclic compounds due to ring-opening reaction in the range of 400°C–600°C. In addition, it can be found that as the pyrolysis temperature continued to increase (500°C–900°C), the relative content of aromatic compounds decreased by eight times, which indicated that lignin occurred to significant pyrolysis with a plenty of aromatic and heterocyclic compounds produced by dehydration–condensation reaction. 33 Meanwhile, the side chain of cyclic ketone, aldehyde, and phenolic compounds broke with CO2 and other gases overflowed. When the pyrolysis temperature rose to 900°C, the relative content of small molecules (aldehydes and ketones) significantly increased. Therefore, it can be inferred that the transition from aromatic and heterocyclic compounds to small molecules is by dehydration–condensation reaction during the deep pyrolysis stage. Compared with other reaction conditions, different pyrolysis temperatures had a greater impact on the pyrolysis process of Miscanthus. 36
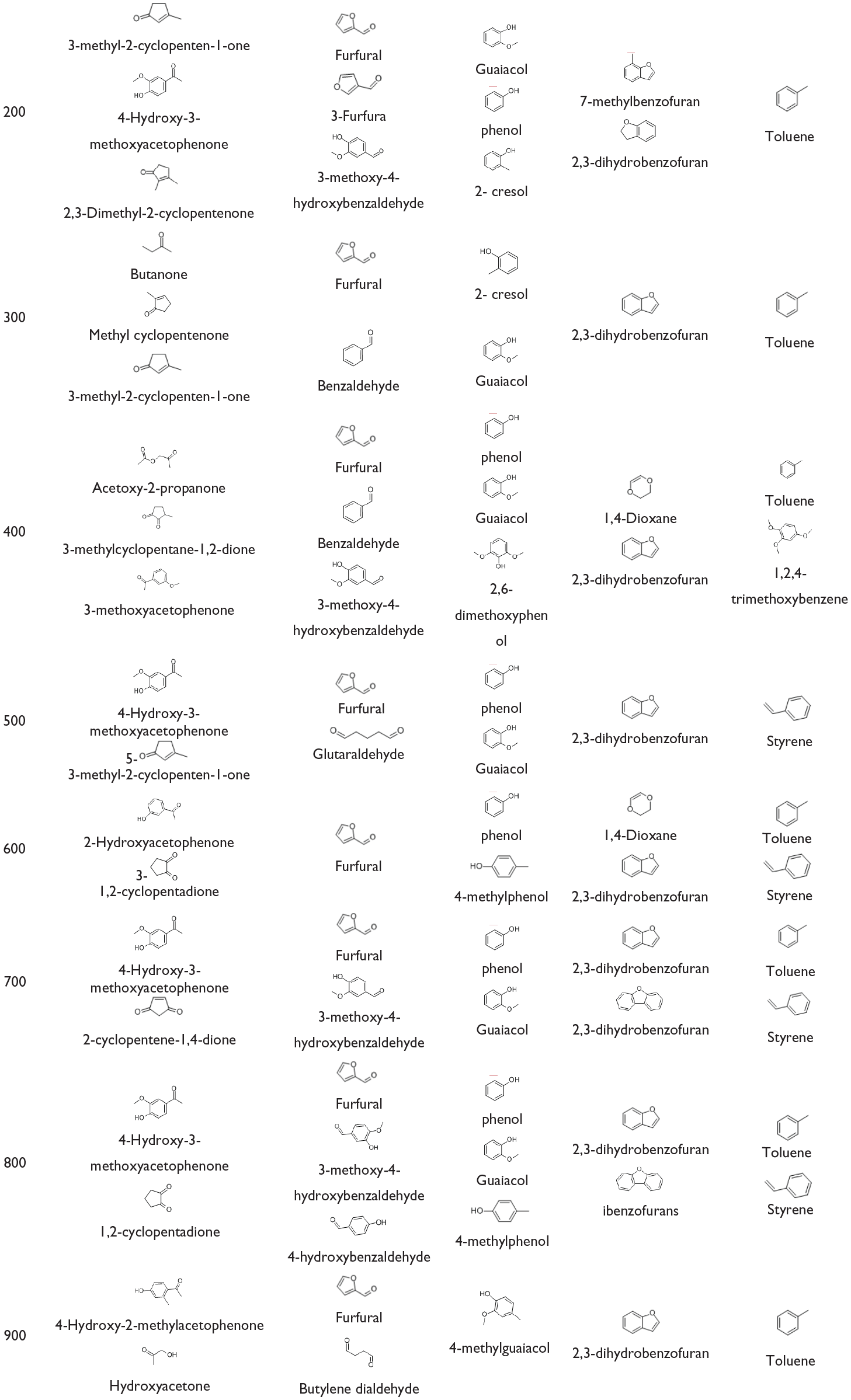
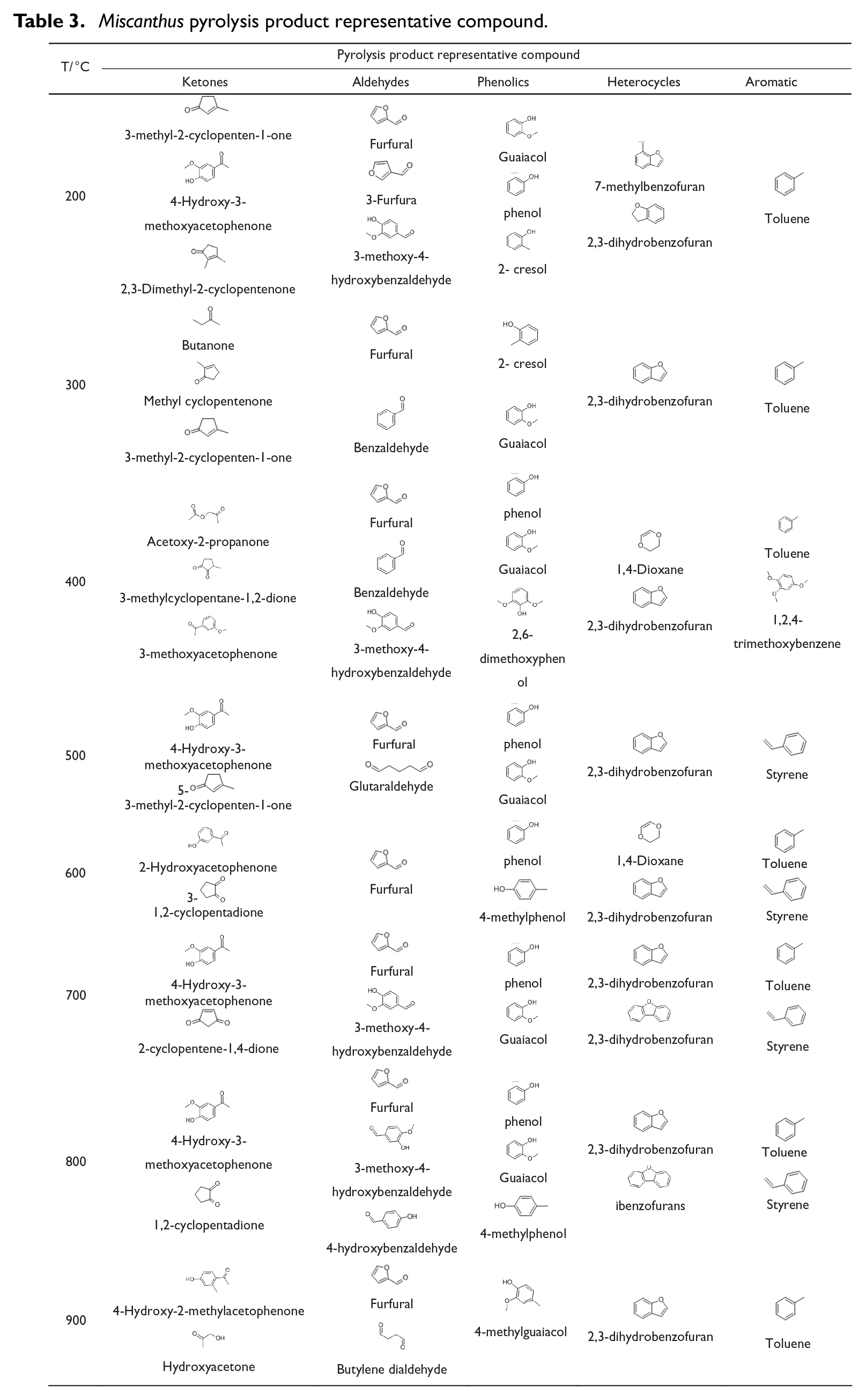
Table 3 shows representative compounds of pyrolysis products of Miscanthus at different pyrolysis temperatures (200°C–900°C). It could be seen that the representative compounds of ketone and aldehyde compounds produced at different pyrolysis temperatures changed obviously, while the representative compounds of phenolic, heterocyclic, and aromatic compounds had no obvious change, which indicated that the pyrolysis final temperature had little effect on these pyrolysis representative compounds. Phenols, aromatic compounds, and aldehydes with benzene rings were produced by pyrolysis of lignin among them, while heterocyclic compounds were produced by pyrolysis of cellulose and hemicellulose, such as aldehydes, ketones, and furans.
Miscanthus pyrolysis product representative compound.
Effect of heating rate
Table 4 shows the distribution of bio-oil products from the pyrolysis of Miscanthus at different heating rates. From the results of product distribution, it can be seen that the pyrolysis liquid products of Miscanthus can also be divided into seven categories: ketones, aldehydes, phenols, heterocyclics, aromatics, acids, and esters. As can be seen from Table 4, it was speculated that the main products of Miscanthus pyrolysis at different heating rates were ketone, aldehyde, phenolic, heterocyclic, and aromatic compounds because of their large relative peak areas, and the relative contents of ketone, aldehyde, and heterocyclic compounds were all reached about 20.0%. Therefore, the influence of different heating rates on the relative peak area ratio of the five compounds was studied. Figure 1 shows the variation curves of five types of pyrolysis products under different heating rates. The abscissas of 1–9 represented the heating rates of 10,000°C/s, 1000°C/s, 500°C/s, 100°C/s, 70°C/s, 50°C/s, 30°C/s, 10°C/s, and 1°C/s, respectively. In order to clearly present trend of five compounds, they were divided into two graphs by data analysis and processing.
Distribution of pyrolysis products of Miscanthus under different heating rates.

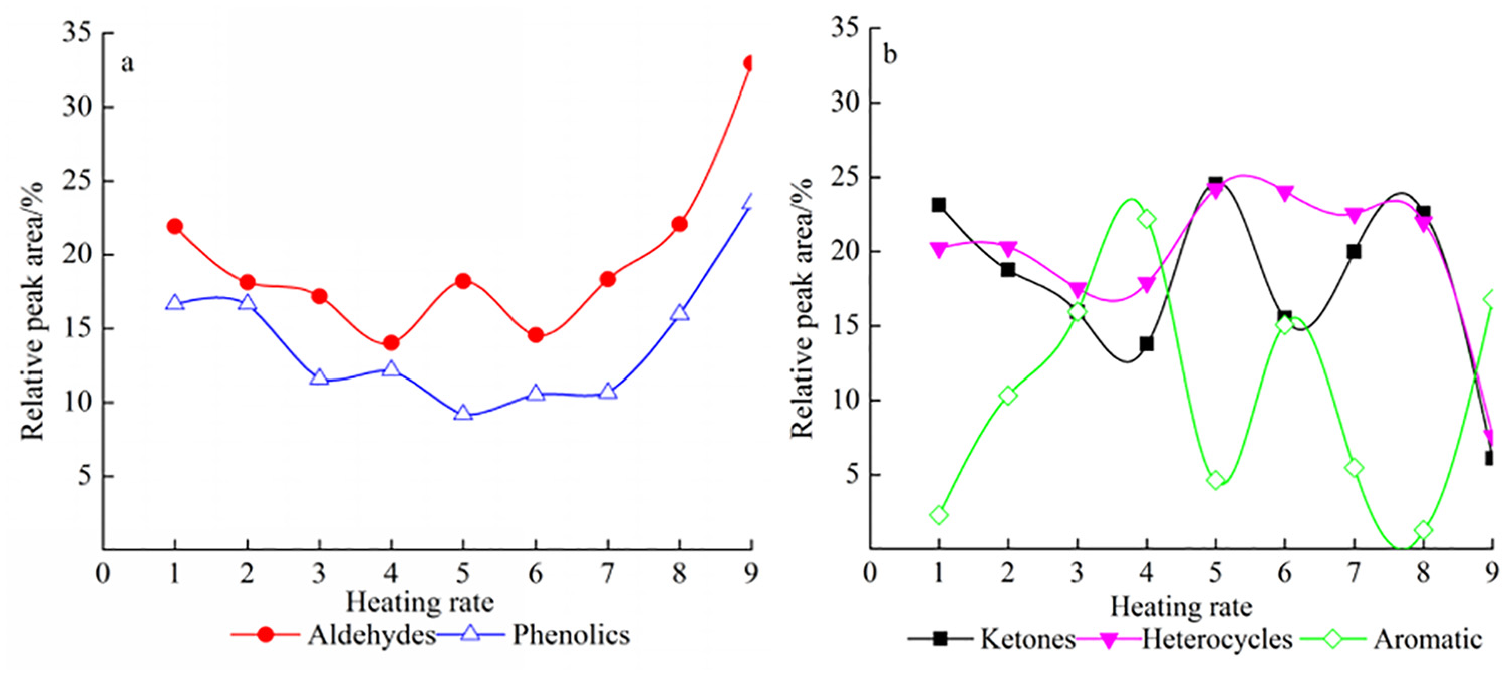
Changes of distribution of pyrolysis products at different heating rates: (a) changes of distribution of aldehydes and phenolics and (b) changes of distribution of ketones, heterocycles and aromatic. 1: 10,000°C/s; 2: 1000°C/s; 3: 500°C/s; 4: 100°C/s; 5: 70°C/s; 6: 50°C/s; 7: 30°C/s; 8: 10°C/s; 9: 1°C/s.
Figure 1(a) shows the tendency of relative peak area ratio of aldehyde and phenolic compounds under different heating rates. It was shown from Figure 1(a) that the overall peak area ratio of aldehyde and phenolic compounds showed a wave-like decrease and increase, respectively, as the heating rate decreased. The proportion of aldehyde compounds gradually decreased from 23.0% to 15.0% with the increase in heating rate from 10,000°C/s to 100°C/s. As the heating rate decreased from 100°C/s to 50°C/s, the proportion of aldehyde compounds increased from 14.5% to 18.0%. A small peak appeared when the heating rate was reduced to 70°C/s in aldehyde. The relative area ratio of the aldehyde compounds increased by two times as the heating rate decreased from 50°C/s to 1°C/s, which indicated that slow pyrolysis was favorable to the generation of aldehyde compounds. The variation of phenolic compounds was similar to that of aldehyde compounds. However, the proportion of relative peak area of phenolic compounds was the least at 70°C/s and then increased significantly from 9.5% to 22.0%, which also indicated that slow pyrolysis was beneficial to the formation of phenols. For phenolic compounds, the heating rate of 70°C/s was a turning point of the relative peak area ratio.
Figure 1(b) shows the tendency of total relative peak area of ketone, heterocyclic, and aromatic compounds at different heating rates. It was found from Figure 1(b) that the relative peak area ratios of ketone and heterocyclic compounds had a nearly simultaneous trend with the decrease in heating rate. The relative peak area ratio of ketone first decreased from 23.5% to 14.0%, then increased from 14.0% to 25.0%, and finally decreased from 25.0% to 16.0% with the change of heating rate in the range of 10,000°C/s–50°C/s. The relative peak area ratio tendency of heterocyclic compounds was similar to ketone. The relative peak area ratio of ketone compounds reached the maximum at 10,000°C/s, 70°C/s, and 10°C/s, and their peak area were close. However, heterocyclic compounds had a relatively gentle change in relative peak area ratio compared with ketone compounds and reached the maximum at 70°C/s. It was speculated that the relative peak area ratios of ketones and heterocyclic compounds occupied the maximum value at 70°C/s. The main reason is that a large number of aldehyde compounds converted to ketones and heterocyclic compounds at the heating rate, resulting in the decrease in relative peak area ratio of aldehyde compounds. It can be found from Figure 1(b) that the relative peak area ratio of aromatic compounds shifts considerably with the change in heating rate. Figure 1(b) shows two peaks at heating rates of 100°C/s and 50°C/s, which are 24.0% and 16.0%, respectively, and the relative peak area ratio reached the minimum during 10°C/s–30°C/s. It can be found that the tendency of peak area ratio of aromatic compounds is opposite to that of aldehyde compounds by observing the relative peak area ratio of aldehyde compounds, which indicated that the heating rate of increasing production of aldehyde compounds was not favorable to the production of aromatic compounds. The main reason is that the side chains of aromatic compounds turned into aldehyde compounds by dehydration condensation or aldehyde reaction at different heating rates, and the aldehyde with benzene ring turned into aromatic compound by addition or substitution reaction.
Effect of residence time
The fast pyrolysis experiments of Miscanthus with different residence times were carried out at a final temperature of 900°C and a heating rate of 10,000°C/s. The residence time was set to 2, 5, 10, and 15 s, respectively. Figure 2 shows the results of pyrolysis of Miscanthus under different residence times. It can still be seen that ketone, aldehyde, phenolic, and heterocyclic compounds were the main types of pyrolysis products at different residence times based on the previous paper on the influence of pyrolysis final temperature and heating rate on pyrolysis of Miscanthus. Therefore, the change laws of relative peak area of the five compounds under different residence times were also studied in this section.

Various types of Miscanthus pyrolysis products relative peak area ratio at different residence times.
Figure 3(a) shows the changes of relative peak area of phenol and aromatic compounds with the increase in residence time. It is found from Figure 3(a) that the relative peak area ratio of phenol and aromatic compounds had a completely consistent trend with the increase in the pyrolysis residence time. The relative peak area ratio of phenol and aromatic compounds reached the maximum of 25.0% and 9.0% at residence time of 5 s, which indicated that it was the most favorable to the production of phenolic and aromatic compounds. However, the relative peak area of phenol and aromatic compounds reached the minimum of 15.0% and 1.0% at 10 s. Figure 3(b) shows the changes of the relative peak area of ketone, aldehyde, and heterocyclic compounds with the increase in residence time. It was shown from Figure 3(b) that the tendency of relative peak area ratio of ketone compounds was the opposite to that of phenol and aromatic compounds. The relative peak area ratio of ketone compounds reached the minimum and maximum value, respectively, in 5 and 10 s. This indicated that the residence time of 5 s could promote the conversion of ketones to aldehydes in a certain extent. The oxidation of the carbon–oxygen double bond of ketone compounds resulted in the decrease in ketone content due to a long residence time at high temperature. Phenolic and aromatic compounds with benzene rings may convert to ketone compounds, leading to a gradual increase in the relative peak area ratio when the high-temperature residence time reached an extreme value. For aldehyde compounds, the ratio of relative peak area decreased from 18.0% to 13.5% and then increased from 13.5% to 21.4% with the increase in residence time, which indicates that the relative peak area ratio of aldehyde compounds was higher under shorter or longer residence time. For heterocyclic compounds, the relative peak area ratio reached the maximum of 27.0% at residence time of 10 s, which indicated that it took a long residence time at high temperatures for Miscanthus pyrolysis to transform phenol and aromatic compounds with benzene ring into heterocyclic compounds. However, the relative peak area proportion of heterocyclic compounds decreased when the residence time reached the limit value, which was mainly caused by the ring-opening reaction of the cyclic compounds during deep pyrolysis of Miscanthus.
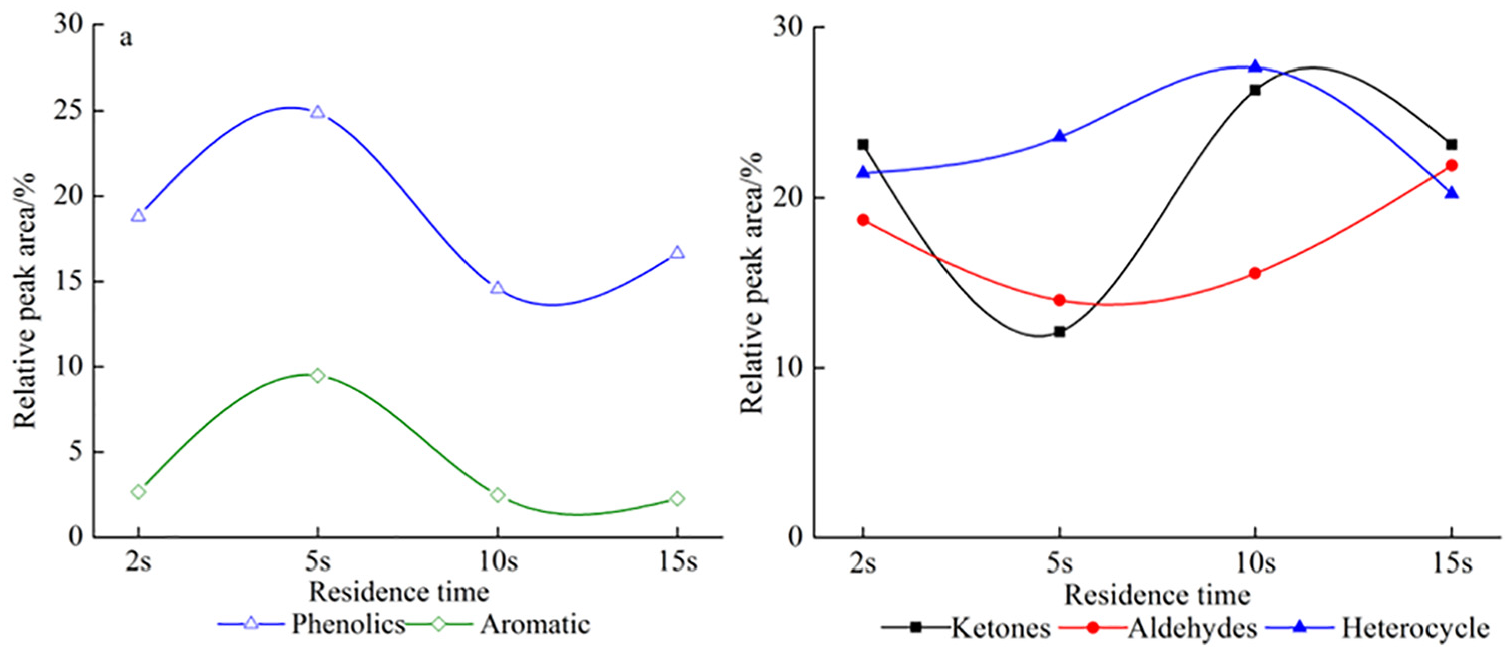
The total relative peak area of five main pyrolysis products of Miscanthus at different residence times: (a) the total relative peak area of phenolics and aromatic and (b) the total relative peak area of ketones, aldehydes, and heterocycle.
Conclusion
The fast pyrolysis of Miscanthus by pyrolysis gas chromatography was carried out to study the effect of different pyrolysis conditions (pyrolysis temperature, heating rate, and pyrolysis residence time) on the distribution of pyrolysis products of Miscanthus.
Regardless of the pyrolysis conditions, the pyrolysis products of Miscanthus were mainly ketone, aldehyde, phenol, heterocyclic, and aromatic compounds. The representative compounds of ketone and aldehyde compounds produced at different pyrolysis temperatures changed obviously, while the representative compounds of phenolic, heterocyclic, and aromatic compounds had no obvious change. Large-scale pyrolysis of Miscanthus had begun at 400°C, and the relative content of pyrolysis products from Miscanthus reached the maximum of 98.34% at 700°C.
The relative peak area ratio of phenol and aromatic compounds reached the maximum and minimum at the residence time of 5 and 10 s, while the relative peak area ratio of ketone compounds showed the opposite trend. The relative peak area ratio of aldehyde compounds was higher under shorter or longer residence time. For heterocyclic compounds, the relative peak area ratio reached the maximum of 27.0% at residence time of 10 s.
The faster or slower heating rate was beneficial to the production of aldehyde and phenol compounds. The relative peak area ratio of ketone compounds reached the maximum at 10,000°C/s, 70°C/s, and 10°C/s, and the relative peak area ratio tendency of heterocyclic compounds was similar to ketone. For aromatic compounds, the overall fluctuation was large, and the relative peak area ratio was the highest at the heating rate of 100°C/s.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the natural science foundation of China for young scholars (no.51706022), the Natural Science Foundation of Hunan Province of China for Young Scholars (Nos 2018JJ3545 and 2018JJ3552), Open Fund of Innovation Platform of Hunan Provincial Education Department of China (No. 17K002), Open Fund of Key Laboratory of Renewable Energy Electric-Technology of Hunan Province (No. 2017ZNDL007), and the Innovative Team of Key Technologies of Energy Conservation, Emission Reduction and Intelligent Control for Power-Generating Equipment and System at CSUST. We are grateful to Jingping Qin at Hunan Agricultural University, China, for the supply of samples.