Abstract
In this work, the inclusion complex of metoprolol with β-cyclodextrin was prepared and investigated using various spectroscopic techniques. The aqueous solution of the inclusion complex was examined using UV-visible, fluorescence spectroscopy, and 1HNMR, whereas the solid physical mixing was investigated by utilizing Fourier transform infrared. Confirmation of the existence of an inclusion complex is achieved through the observation of alterations in spectroscopic properties. Both 1:1 and 1:2 stoichiometry a host–guest interaction was formed between metoprolol and β-cyclodextrin. The constants that represent the inclusion the interaction was determined to be K1=9.09 × 10−4 μM−1 and K2=1.96 × 10−6 μM−1 by using typical double reciprocal graphs. The parameters affecting the inclusion complex formation were investigated and optimized. Based on remarkable increase on the fluorescence intensity, spectrofluorometric method for determination of metoprolol in pharmaceutical formulation was developed. The fluorescence intensity of metoprolol was measured at 297 nm, after excitation at 223. Under the optimum conditions, linear relationship in concentration range of 0.35–1.7 μM, with correlation coefficient of 0.9944, was obtained. The limit of detection has been shown as 0.11 μM, while the limit of quantification was shown as 0.34 μM. The proposed approach was successfully utilized to analyze the drug in its pharmaceutical formulation.

Introduction
Cyclodextrins (CDs) are cycle-like oligosaccharides created of glucose-based units. They possess the capacity to generate readily soluble in water complexes of inclusion of small molecules. They consist of glucopyranose units connected by

Chemical structure of β-CD.

Chemical structure of MET.
In this research work the inclusion complex of MET with β-CD was prepared in aqueous solution and solid was investigated fluorescence spectroscopy, UV–VIS spectroscopy, 1HNMR, and Fourier transform infrared spectroscopy (FTIR), respectively. To the best of our knowledge, spectrofluorometric methods based on inclusion complex formation of MET with β-CD have not been reported. Thus, due to enhancement of florescence intensity of MET spectrofluorometric method for determination of MET in pharmaceutical formulation based on inclusion complex was developed for the first time.
Experimental
Chemical and reagents
Dibasic sodium phosphate (99.0%), monobasic sodium phosphate (99.0%), MET standard (98.0%), HP-β-CD (97.0%), and β-CD (97.0%) were acquired from Sigma-Aldrich (St. Louis, MO, USA).
Instrumentations
The spectrophotometric investigations occurred using a Shimadzu UV-1650 pc double-beam ultraviolet-visible a spectrum analyzer (Kyoto, Japan), equipped with a 50 W halogen light and deuterium lamp. The fluorescence spectra and intensity analyses were conducted using the Shimadzu RF 6000 spectrofluorometer (Kyoto, Japan). This device is outfitted with a 150-W xenon lamp. Also, Bruker Ascend 400 spectrometer was utilized to obtain the 1H NMR spectra (Bruker, Billerica, MA, USA) with the Top Spin 3.5 software). Infrared (IR) measurements were performed using Cary 630 FTIR (Bruker, Billerica, MA, USA).
Preparation of standardized solutions of MET and β-CD
MET standard solution was prepared by dissolving 17 mg of MET in a phosphate buffer with a pH of 7.0. Consequently, a stock solution was formed with a concentration of 1000 μM. The solution was then transferred to a standard flask of 25 mL, then to accomplish dilution more phosphate buffer with an identical pH was inserted. In order to make a stock solution of β-CD with a concentration of 15,000 μM, 1.7 g of β-CD was dissolved in water that is deionized and transferred into a standard flask of 100 mL. Additional more water was added to obtain the desired dilution.
Buffer solution
The solution of buffer having a pH of 7.0 was generated by utilized both 0.1 M of Na2HPO4 and 0.1 M of NaH2PO4 solutions.
UV spectroscopy measurement
The MET sample of 0.1 mL was transferred into volumetric glass with a volume of 10 mL. Subsequently, at the same container 4 mL of β-CD solution was introduced. Afterward, the flask became filled with a buffered phosphate solution with a pH of 7.0. The resulting solution was subjected to vibration at ambient temperature by placing it in a sonicator at duration of 10 min. Afterward, the intensity of absorption was obtained at a precise wavelength of 223 nm.
Fluorescence measurement
In a volumetric container of 10, 0.1, and 4 mL of MET and β-CD were combined, respectively. For reaching the desired volume, the last combination was diluted by including a phosphate buffer solution with a pH of 7.0. Afterward, the flask ended up in the sonicator at room temperature over a period of 10 min. Finally, the MET-β-CD complex ’s fluorescence intensity was quantified at λex/λem 223/297 nm.
Determination of stochiometric ratio
According to the experimental parameters that had been previously computed, the stoichiometry of the inclusion complex was determined by analytical approach called Benesi–Hildebrand plots. The assumption was made that the complexes exhibited a 1:1 ratio. The equation expressed in the form: 1/F–F0 = 1/(F∞-F0) K[β-CD0] + 1/F∞-F0. In the above formula, F∞ denotes the fluorescence intensity when the majority of MET molecules remain locked in β-CD, F indicates the detected fluorescence intensity at each measured concentration of β-CD, and[β-CD0] reflects the initial concentration of β-CD.
Limit of detection and quantification
For calculating limit of detection (LOD) or limit of quantification (LOQ), the value 3.3 is used for LOD and 10 is used for LOQ. The formula is K. SDa/b, where K represents the respective value. The SDa symbol represents the standard deviation of the intercept, while the symbol b indicates the slope.
Results and discussion
Analysis of absorption spectra
The MET absorption spectra were measured in without and with the addition of 6 × 103 μM of β-CD which presented in Figure 3. The research results indicated that the wavelength at which MET exhibits which the peak absorption wavelength was 223 nm at pH 7.0. By introducing β-CD to the solutions, the wavelength at which the maximum absorbance occurs stays the same, but there is a minor rise in absorbency. This results in a rise in the molar absorptivity coefficients ε (L mol−1 cm−1) from 19,000 to 29,700.

Absorbance spectra for MET- β-CD, MET 10 μM, β-CD 6 × 103 μM at room temperature, time 10 min, pH 7.0.
Infrared spectroscopic analysis
Metoprolol’s infrared spectrum in Figure 4, the detected peaks are displayed as follows: 3500 cm−1 for OH stretching, 3032.6 cm−1 stretching band for = C-H, 2871.19 cm−1 for N-H stretching, 1577 cm−1 for C-O stretching, 1511.47 cm−1 for C=C stretching, 819 cm−1 for scissoring = C-H. These agree with the FT-IR spectrum corresponding to MET reported in the literature.30,31 Significant alterations in the FTIR spectra were seen following the establishment of the MET-β-CD inclusion complex. The OH stretching band at 3500 cm−1 exhibits significant broadening and a red shift. Also, the peak at 3032.6 cm−1 stretching band for =C-H has a red shift to 3200 cm−1. The band associated with the stretching of NH at 2871 cm−1 was moved to a higher wavenumber of 2873 cm−1, while the band associated with the stretching of C-O was both weaker and shifted to a higher wavenumber of 1583 cm−1. In addition, the band assigned to C=C stretching at 1511 cm−1 looked shorter with blue shift to 1512 cm−1. It is possible that the changes mentioned earlier are due to differences in the microenvironment. The interaction between β-CD and MET in the solid physical mixture had a considerable influence on both hydrogen bonding and van der Waals forces.

IR of β-CD, MET, and MET-β-CD.
Proton nuclear magnetic resonance spectroscopy ( 1 H NMR)
1 HNMR spectroscopy is a useful method for examining the complexation of CDs.32,33 The interactions between host and guest molecules are non-covalent bonds such as van der Waals forces, hydrophobic interactions, and hydrogen bonds through.34,35 If the guest molecule is incorporated into the b-CD cavity their chemical shifts will be changed. Non-covalent bonds, such as hydrophobic interactions, van der Waals forces, and hydrogen bonds, play a crucial role in connecting host and guest molecules. The 1H chemical shift values of MET before and after forming the inclusion complex are shown in Table 1. As can be seen from Table 1, the complexation caused several chemical shifts of MET protons. It confirmed that the complex was formed.
NMR of MET and the shift after complex with MET-β-CD.

Characterizing emission spectra
Fluorescence spectroscopy is an extremely effective technique for investigating host–guest molecular systems, offering sensitivity and selectivity. Fluorescence analysis offers notable benefits due to its heightened sensitivity and capacity for accurately measuring substantially smaller quantities compared with spectrophotometric analysis. At first, the spectral characteristics of MET was examined. According to the study results, the MET exhibited a maximum emission wavelength of 297 nm at pH 7.0. After the addition of β-CD to the drug flack, there was an observed rise in fluorescence intensity, although the emission’s maximum wavelength remained unchanged (Figure 5). This phenomenon may be attributed to the engagement by non-covalent interactions like hydrogen bonding and van der Waals forces between MET and of β-CD in the hydrophobic interior of β-CD’s core.

Emission spectra MET-β-CD, MET 10 μM, β-CD 6×103 μM, at room temperature, time 10 min, pH 7.0.
Optimization of variables affect inclusion complex formation
The study examined multiple variables that influence the creation of inclusion complexes, the kind of CD used (either native β-CD or derivative β-CD), including the pH of the buffer, the duration of complexation, and the concentration of β-CD.
Optimizing of CD type
The present study investigated the impact of CD type on absorbance and fluorescence intensity. Particularly, native β-CD and modified HP-β-CD, were used for this purpose. The outcomes obtained are displayed in (Figure 6 (a) and (b)). As can be observed, the absorbance and the fluorescence intensity were higher when the native β-CD was added to MET solution than HP-β-CD added to the same drug solution. Because of the steric hindrance provided by the HP-β-CD structure, which makes it less favorable for the inclusion complex, which make native β-CD is the optimum CD type for MET-β-CD inclusion complex.36,37

(a) Emission spectra of MET, MET-β-CD Inclusion complexes, concentration of MET 10 μM, β-CD 6 × 103 μM, in a 10 min, pH 7.0, at room temperature, and (b) MET-HP-β-CD Inclusion complexes, concentration of MET 10 μM, HP-β-CD 6 × 103 μM, in a 10 min, pH 7.0, at room temperature.
Optimizing the pH of the inclusion complex
The pH of the solution has a greatly influences on the intensity of fluorescence of the complex that builds up between MET and β-CD. The pH was checked throughout the spectrum of 2 to 9, and the outcomes can be seen in Figure 7. MET has a basic pKa value which is 9.6. 38 A rise in fluorescence intensity was observed when the pH values increased from 3.0 to 7.0, followed by a decrease in pH 8.0 and 9.0. According to this behavior, the optimum pH for the formation of the complex is the pH with the highest fluorescence intensity, which is pH 7.0. As the drug is a basic drug, any pH which is less than its pKa is considered as an acidic media. As a consequence of this, the inclusion complex of MET-β-CD was suitable in this media, which is where the protonated and ionized form of the drug is favorable for the inclusion complex.

Emission spectra of MET-β-CD in series of pH from pH 3 to pH 9, concentration of MET 10 µM, β-CD 200µM, at room temperature, time 10 min, the higher fluorescence intensity showed at pH 7.0.
Optimizing of the complexation time
The research examined the optimization of the time required of complexation and its impact on the fluorescence intensity of the MET-β-CD complex. The experiment was carried out at ambient temperature, with a duration ranging from 5 to 25 min (data not shown). The findings suggest that the highest fluorescence intensity value occurred at the 15 min. Consequently, the fluorescence intensity reduced, indicating that it was the optimal period for complexation.
Optimizing of β-CD concentration
In order to optimize the concentration of β-CD, the focus will be on examining the impact of β-CD concentrations on formation of MET complex. The drug concentration was maintained at a constant level of 10 μM, while the β-CD concentration was altered within the range of 50–400 μM (Figure 8). The fluorescence intensity exhibited a continuous and gradual rise until it achieved an unchanging complex at a concentration of 300 μM. Therefore, this specific point has been chosen as the most favorable concentration.

Emission spectra of MET 10 µM with different concentration of β-CD from 50 to 400 µM, at room temperature, time 10 min, pH 7.0.
Stoichiometry of inclusion complex
It was obtained by examining the previously estimated experimental parameters. The complex was presumed to have a 1:1 ratio. The research used the analytical approach known as Benesi–Hildebrand plots. The equation is expressed in the following manner: The equation may be written as 1/F–F0 = 1/(F∞-F0) K[β-CD0] + 1/ F∞-F0. The equation specifies F∞ as the fluorescence intensity when a large number of MET molecules are immobilized inside β-CD. The symbol [β-CD0] denotes the starting concentration of β-CD, whereas F indicates the fluorescence intensity seen at each detected concentration of β-CD. Figure 9(a) depicts a graph illustrating the relationship between 1/F–F0 and 1/[β-CD0]. The graph shows that there is a 1:1 ratio between MET-β-CD, with an inclusion constant of K1 = 9.09 × 10−4 µM−1. To precisely ascertain the stoichiometric ratio, the assumption of a 1:2 inclusion complex with β-CD. The assessment was conducted by graphing the reciprocal of the difference between F and F0 as a function of the reciprocal of the square of β-CD (Figure 9(b)). This resulted in a linear correlation, confirming the possibility of forming both 1:1 and 1:2 inclusion complexes of MET with β-CD simultaneously and K2 was found to be 1.96 × 10−6 µM−1. 23

(a) Plot of 1/(F–F0) vs 1/[ β-CD] of MET- β-CD complex; MET 10 µM, pH 7.0, and (b) plot of 1/(F–F0) versus 1/[β-CD] 2 of MET-β-CD complex; MET 10 µM, pH 7.0.
Validation of the procedure
The analytical procedures were validated accordance with International Conference for Harmonization (ICH) guidelines with respect to linearity, LOD, LOQ, accuracy, and robustness. 39
Linearity and LODs and LOQ
The suggested techniques showed linear relationships with strong correlation coefficients (n = 8) with varying concentrations from 0.35 to 1.7 μM. The calculated values and data from statistics display in Table 2. The equations used to compute the LOD and LOQ are as follows: LOD or LOQ is equal to K. In the context of calculating LOD, the symbol K is 3.3, but for calculating LOQ, it will be 10. The term “SDa” denotes the standard deviation of the intercept, whereas “b” indicates the slope. The LOD and LOQ were found to be 0.11 and 0.34 μM, respectively.
Parameters for MET-β-CD spectrofluorimetric method.

Accuracy of spectrofluorometric method
The evaluation was conducted by evaluating three sets of duplicate measurements at three different concentrations levels. Table 3 demonstrates a notable degree of consistency and precision in the acquired data, suggesting that the process is reliable. The exceptional level of accuracy was perfect for ensuring the quality analysis of MET in their pharmaceutical product formulations.
The accuracy and precision of MET-β-CD inclusion complex.
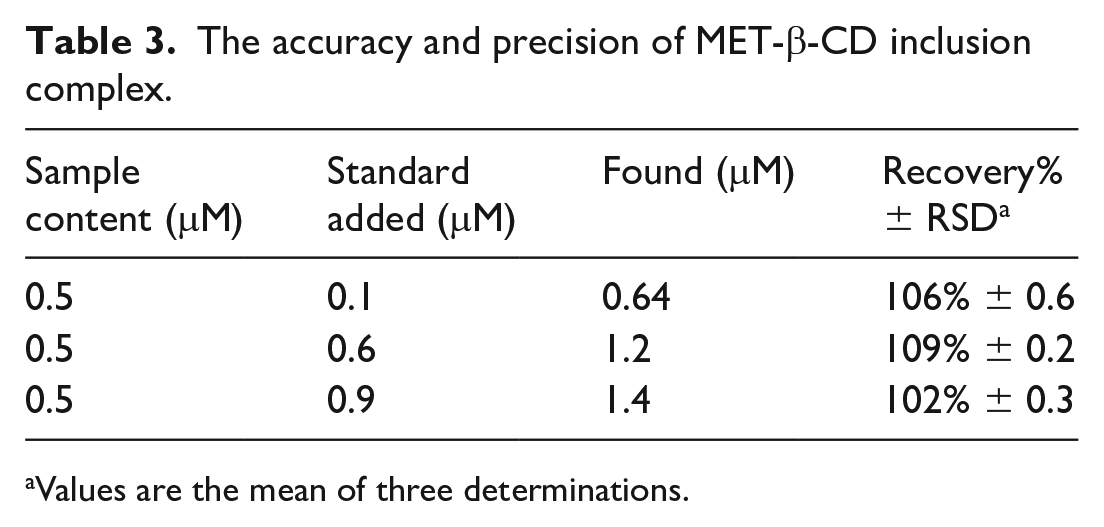
Values are the mean of three determinations.
Robustness of spectrofluorometric method
The robustness of the analytical results was evaluated by assessing the impact of small changes in the parameters. In these tests, the recovery was determined for each example by isolating and modifying a single parameter while leaving all other variables constant. Therefore, it was discovered that little modifications in the procedure parameters had negligible consequences for the operations. The values of recovery values can be seen in Table 4.
Robustness of the spectrofluorometric method for MET-β-CD.

Values are mean of three determinations.
Pharmaceutical formulation analysis
The suggested technique effectively examined MET pharmaceutical formulations with acceptable accuracy in drug detection. The percentage values were 95% calculated from five determinations, as presented in Table 5. Recoveries were accurately calculated as the amount found divided by the amount taken multiplied by 100 values are mean ± RSD.
The method application of MET-β-CD inclusion complex.

Value is the means of five determinations.
Conclusion
In the present study, we demonstrated that β-CDs can be used as guest complexing agent, which acted as substrate reservoir in a dosage-controlled manner. The results showed that the inclusion process was occurred. The formation of the inclusion complex MET-β-CD was confirmed by FTIR, 1H NMR, fluorescence, and UV/visible spectroscopy. Based on the enhancement of the fluorescence intensity of MET produced through complex formation a simple, sensitive, and accurate method for determination of MET in the presence of β-CD was developed. The proposed method is fully validated and successfully applied for the analysis MET pharmaceutical formulations.
Footnotes
Acknowledgements
The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this research work through the Project No. (Grant No. KFU241279). The authors thank Dr Abuzar Albadri, Qassim University for his help in the NMR analysis.
Author contribution
Conceptualization, A.A.E.; Methodology, H.A.; Validation, H.A.; Investigation, H.A.; Writing—original draft, H.A. and A.A.E.; Writing—review & editing, A.O.A.; Supervision, A.A.E. and A.O.A.; Funding acquisition, H.A. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Research Council (Grant No. KFU241279). The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia for funding this research work through the project number (Grant No. KFU241279).
Ethical consideration statement
This article does not contain any studies with human or animal participants.
Consent to participate
The manuscript has been read and approved by all the authors, and that each author believes that the manuscript represents honest work.
Consent for publication
Not applicable

