Abstract
Nowadays, cinnamon essential oil is one of the most popular spices for food flavoring and also for medicinal uses such as anti-inflammation agent, but its low solubility, irritations, and allergic reactions limit its applications. To overcome these limitations, cinnamon essential oil-loaded nanocontainers were prepared via inclusion complexation between the cyclodextrins and cinnamon essential oil under two different conditions, i.e., at room temperature or under sonic energy. The resulting supramolecular nanocontainers were characterized by XRD, SEM, DSC, and FTIR. Successful insertion of cinnamon essential oil in the cyclodextrins’ cavity was confirmed by the significant differences between the FTIR, DSC, and XRD spectra of initial mixtures and those of the complexes. XRD results indicated that crystalline complexes adopted a mixture of head to head channel-type and cage conformation for cinnamon essential oil/cyclodextrins at various conditions. Solubility, bioavailability, and in vitro dissolution of obtained cinnamon essential oil nanocapsules were investigated, and results show that by encapsulation of cinnamon essential oil, solubility improved and its release is controllable. The solubility of cinnamon essential oil increased linearly as the concentration of cyclodextrins was increased, confirming the 1:1 stoichiometry of the complex. Electro-spun nanofibers of poly-ɛ-caprolactone containing cinnamon essential oil/cyclodextrins inclusion complex at various conditions indicated that these nanofibers did not show beading defect with controlled release of cinnamon essential oil.
Introduction
Phytochemicals are a large group of chemicals that are naturally present in plants, fruits, vegetables, nuts, legumes, and grains which provide health benefits for humans; they are biologically active and can be responsible for much of the disease protection and medicinal uses such as asthma, arthritis, cancer, etc. [1,2]. Phytochemicals are being widely examined for their vital role in medicinal uses by people, and phytonutrients appear as potentially and generally effective products and may solve health problems and, as they are biologically active, and can be useful for many kinds of disease protection, such as from asthma, arthritis, cancer etc. and they also have medicinal uses without any side effects [1,2]. Various groups of phytochemicals can be used for the prevention of chronic degenerative diseases, which are used widely in food industry and dietary products and also led to healthy food with good taste such as phenolic compounds. Phenolic compounds can neutralize free radicals, block or suppres active carcinogens, prevent oxidation of lipids (lipoxidation), glucose (glycation), and proteins (carbonylation) which is important for diabetes mellitus II and inflammation, and correspond to important activators of inflammation in various tissues [2–4]. Cinnamon essential oil (CEO) is a phytochemical which is one of the most popular spices and food additives. CEO shows various applications such as an antiemetic, antidiarrheal, anti-flatulence, and general stimulant and can also kill bacteria, heal wounds, and relieve stomach flu caused by harmful bacteria like salmonella [2–4]. Bioavailability, solubility, and stability of phenolic compounds determine the absorption of these compounds in the body which CEO show very low water solubility. Because of low water solubility of cinnamon oil, its skin irritation, sensitivity to light and heat, poor biocompatibility, allergic effects, and its bitter taste, this compound must be encapsulated [4–6]. Encapsulation involves coating or entrapment of a pure material or a mixture into another material, which protects the active ingredient from its surrounding environment for a specific period of time or a triggering event including rupture, dissolution, and change of pH and temperature [4,5]. Bioactivity and antioxidant properties of polyphenols depend on the stability of polyphenols and chemical, physical, and biological conditions. Encapsulation technology on natural compounds has gained great interest and necessary to apply delivery or carrier systems in order to maximize the potential medical benefits of antioxidants. Encapsulation protects plant extracts from devastating environment effects such as undesirable effects of light, chemical reactions, moisture and oxygen, oxidation, and dehydration, which reduce the shelf life of compounds and improve the processing step, delivery of bioactive, and stability of phytochemicals [7–9].
Cyclodextrins (CDs) are cyclic water-soluble, non-reducing, non-toxic macrocycle oligosaccharides consisting of glucopyranose subunits bound through α-(1,4) links which result from starch degradation by bacterium Bacillus macerans. CDs have a hydrophobic inner cavity and hydrophilic outer surface, which enables them to form inclusion complexes with a wide range of organic and inorganic compounds [10–13]. The most important and amazing property of CDs is their ability to form inclusion complexes with several hydrophobic and hydrophilic compounds. CD’s special shape with hydrophobic cavity and hydrophilic surface led to the formation of an inclusion complex with various molecules with a wide range of chemical properties, which is different from non-complexed guest molecule [10–13]. Encapsulation of hydrophobic compounds in the CD’s cavity produced some new properties such as chemical reactions’ prevention, mask undesirable flavor, enhance stability of formulations, improve solubility, controlled release for drugs, vitamins, agrochemicals, live cells, active enzymes, flavors, pharmaceuticals, and inks [14–16]. Encapsulation immobilizes polyphenol molecules in a CD cavity and revealed to improve thermo-oxidative stability, chemical stability, bioavailability, photo-stability, and water-soluble compound. CDs have an empty cavity which can entrap guest molecules with appropriate geometry and polarity when encapsulation is done [14,15].
One of the important parameters in the formation of the inclusion complex is the lipophilic cavity of CD molecules which obtain a microenvironment and lead to the entry of appropriately sized non-polar moieties into a cavity. Formation of complex depends on the hydrogen bonding, and no covalent bonds are broken or formed during the formation of the inclusion complex, but various thermodynamic factors affect the formation of the inclusion complex. Removal of water molecule from a hydrophobic cavity and formation of Vander Waals bonding, hydrophobic, and hydrogen bond interactions are driving force for the formation of the inclusion complex. When the guest molecule is hydrophobic, water molecules can be displaced by guest molecules present in the solution, obtain an apolar–apolar association, decrease CD ring strain and cause more stable structure with lower energy state [3]. The binding of guest molecules within the CD as host is not fixed or permanent but is a dynamic equilibrium and inclusion strength depends on dimensional fit of guest molecule and CD cavity and on specific local interactions between surface atoms [15]. By formation of inclusion complex, reversible hydrogen bonding led to a controlled release of CEO from nanocontainers and continuing delivery of the CEO to target sites. Encapsulation of CEO also improves solubility, phase solubility, bioavailability and in vitro dissolution of obtained CEO nanocontainers. The encapsulation of cinnamon oil into CDs cavity is an effective delivery system of phenolic compounds for masking allergic effects, irritations, odor and slow release from the molecular cavity of CDs ensures the long-term delivery. Incorporation of CEO/β-CD in PVA electrospun nanofibrous film showed uniform nanofibers with excellent antimicrobial activity against both Gram-positive and Gram-negative bacteria, and also the thermal stability of the nanofilm improves significantly [16].
In the present work, CEO nanocontainers were synthesized via inclusion complex formation between CDs (α- or β-CD) at various conditions (at room temperature and under sonic energy). The obtained nanocontainers were characterized by FTIR, SEM, DSC and XRD techniques. After characterization of nanocontainers and confirmed inclusion complex between CDs and CEO, the obtained inclusion complexes were incorporated into poly(ɛ-caprolactone) (PCL) electrospun nanofibers for drug-delivery system and their structure was studied using SEM and EDX. The obtained nanofibers also showed controlled release of CEO with time. The slow release of CEO from the nanofibers ensures the long-term delivery of CEO to target site.
Experimental
Material
Poly (ɛ-caprolactone) (PCL) with a molecular weight of 60,000–80,000 was purchased from Sigma-Aldrich. Cinnamon essential oil (CEO, Aldrich, ≥98%), α-CD (α-CD, Aldrich, ≥98%), β-CD (β-CD, Aldrich, ≥98%), dimethyl sulfoxide (DMSO, Merck, ≥98%), ethanol (EtOH) (Merck, >98%) and dichloromethane (DCM, Merck, ≥98%) were used as received.
Inclusion complex formation of CDs/cinnamon oil
CEO/CDs inclusion complexes were prepared using CEO and CDs (α- and β-CD) at a molar ratio of 1:1. An aqueous solution of CDs was prepared by dissolving CDs (α- and β-CD) in water (5.0 mL of 5.0 wt %), mixed with vigorous agitation for 2 hours at room temperature. The inclusion complex between CDs and CEO was prepared by adding the CEO into the aqueous solution of CDs, mixed at various conditions (stirring at room temperature for seven days by magnetic stirrer or 30 min under sonic energy). The obtained solution was maintained at room temperature for 24 h; the solid part was collected and then dried at 50℃ in an oven for 48 h. The dried complex was characterized by XRD, SEM, FTIR, and DSC [15–18].
Characterization
X-ray diffraction (XRD) was used to study the diffraction patterns of CDs/CEO crystals at ambient conditions; a Xpert Philips diffractometer (USA) with nickel filtered the Cu Kβ radiation at a rate of 5°
Phase solubility analysis
The phase solubility study was carried out by the method reported by Higuchi and Connors [19]. For this purpose, different concentrations of aqueous CD solutions were prepared, filled in test bottles, and then the CEO solution was added to these solutions to bottles until attain saturation. Bottles were capped and placed in an ultrasound bath for 48 h at 27 ± 2℃, and then the obtained solutions were filtered using 0.45 µm nylon disk filter, diluted, and measured for the total dissolved CEO content by UV analysis at 360 nm. Each sample was determined in triplicate and the samples were protected from light. The phase solubility diagram was constructed by plotting concentrations of dissolved CEO against CDs concentration. The binding constant,
Dissolution profile of CEO and its inclusion complex at various conditions
The inclusion complexes containing 20 mg of CEO (calculated based on the content of CEO found in the inclusion complexes) were accurately weighed and placed in a vessel containing 250 mL of simulated dissolution medium and the temperature was controlled at 37 ± 1℃ and the paddle was set at 100 r/min throughout the study. At fixed time intervals (0, 5, 10, 15, 30, 45, and 60 min), 5 mL aliquots were withdrawn and equal volumes of fresh medium were replaced. The samples were filtered, suitably diluted, and assayed for CEO content by using UV-visible spectroscopy at 376 nm. The release profiles of CEO and the inclusion complexes at various conditions were also evaluated for comparison [20].
Electrospinning of PCL containing CEO/CDs nanocontainers
Electrospinning was carried out by preparing solutions of PCL and IC. For this reason, PCL solutions were prepared in DMSO/DCM (35/65 v/v%) mixture. After formation of a homogenous solution, 0.075 g CEO/CDs inclusion complex was dissolved in PCL solution. The solution was drawn into a 5 mL glass syringe with a 30 G needle and electrospinning was carried out at 15 kV and 2000 r/min on a cylindrical collector with fixed flow rate at 1 mL/h. The distance between the needle tip and the collector (aluminum foil covered collector) was fixed as 18 cm. Electrospinning experiments were carried out in an enclosed box at 23–26℃ and 18% relative humidity [21].
Results and discussion
Stoichiometry
The complex formation of CDs (α-CD and β-CD) with CEO was studied quantitatively. The amount of the complex formed increases with an increase in the amount of CEO added to the aqueous solution of CDs. The amount of the complex showed similar values even if excess amounts of CEO were used; this indicates the stoichiometric complexation. The continuous variation plot for the formation of the complex between CDs and CEO is at maximum level at 0.50 (monomer unit: CD = 1:1) stoichiometry (Figure 1). This result suggests that 1 units were bound in each CD cavity [10].
Continuous variation plot for complex formation between CDs and CEO (a) at room temperature for 7 days and (b) under sonic energy.
Phase solubility
The content of CEO was determined using UV-visible spectroscopy at a wavelength of 360 nm. The standard CEO solution was prepared in ethanol and diluted to the desired concentrations by 50% v/v ethanol. CDs did not show any absorption at the wavelength of 360 nm which is the λmax of CEO. The calibration curve of the method was linear. Increasing CD concentration showed a linear increase of the CEO concentration (Figure 2) with the slope between 0 and 1 which indicates an AL-type of phase solubility according to Higuchi and Connors concepts. Linear increasing indicates 1:1 CEO/CDs complex formation in all types of CDs studied. The complexation constant CEO/CDs was calculated from equation (1) [20].
Relationships between CEO and CD concentrations. Solubility of CEO and its complexes at various conditions.


FTIR results
Interaction between CD and the guest molecules in the solid state and formation of inclusion complex can be investigated by infra-red spectroscopy. Differences between CEO and, α-CD and β-CD FTIR with their inclusion complex and changes in the shape, position and intensity of the absorption bands indicates the formation of inclusion complex formation. FTIR of pure α-CD, β-CD, CEO, and their inclusion complexes with CDs at various conditions are shown in Figure 3. FTIR peaks of CDs observed at 3345, 2917, 1163, and 1045 cm−1 which corresponds to the symmetric and antisymmetric stretching of ν(OH), ν(CH2), ν(C–C), and bending vibration of ν(O–H), respectively. O-H stretching vibrations show a broad absorption band at about 3200–3600 cm−1. The spectrum for the inclusion complex looks almost similar to the pure CDs. The presence of a benzene ring in an aromatic compound often is determined by aromatic absorption for COH stretching near 3030 cm−1. Hydrogen bonding between the N-H of imides showed multiple absorption bands around 3000–3200 cm−1 [19–21].
FTIR of (a) α-CD, (b) β-CD, (c) CEO, (d) CEO/α-CD room temperature, (e) α CEO/α-CD sonic energy, (f) CEO/β-CD room temperature, and (g) CEO/β-CD sonic energy.
Characteristic peaks of CEO are seen at 3500 cm−1–3200 cm−1, 3000 cm−1–2850 cm−1 and 1666.90 cm−1 which is related to O–H stretch, C–H stretch for alkane and C=O bond respectively. Absorption band between 900 cm−1–675 cm−1 and 1680–1600 cm−1 indicated the presence of aromatic C=C and alkenes C=C stretch, respectively [19–21].
By formation of inclusion complex between CDs and CEO, the broad hydroxyl band of pure CDs at 3370.72 cm−1 was found to be narrowed and enolic COH bending in CD at 1362 cm−1 disappears which is a good indication of the formation of the inclusion complex and reported by various researchers in synthesizing the inclusion complex between CD (host) and a guest molecule and signifies involvement of enolic groups in some interaction between CUR and CDs [19–21,27].
X-ray diffraction
Figure 4 and Table 2 show the X-ray diffraction patterns of α-CD, β-CD, CEO and their inclusion complexes at various conditions. XRD is a powerful method to characterize the formation of the inclusion complex between CDs and guest molecules by disappearing of some peaks or formation of new peaks indicating the presence of new solid crystalline phases via this process [12–15]. CDs inclusion complexes with guest molecules are divided into channel-type, cage-type and layer type. Pure CDs are normally arranged in the cage type, where each cavity is blocked off on both sides by adjacent CDs. In channel-type conformation guest molecules included in CD cavities and stacked on top of each other. Cage structures inclusion complex shows characteristic peaks at 2 XRD of (a) α-CD, (b) β-CD, (c) CEO, (d) CEO/α-CD sonic energy, (e) CEO/α-CD room temperature, (f) CEO/β-CD sonic energy, and (g) CEO/β-CD room temperature. XRD peaks of α-CD, β-CD, CEO, and their inclusion complexes.

DSC study of complex
The DSC thermogram of pure α-CD, β-CD, CEO, and their inclusion complex at various conditions are shown in Figure 5. The thermogram of α-CD and β-CD showed a characteristic endothermic peak at 90 and 120℃, which is related to water loss from α-CD and β-CD, respectively. Second peak of CDs is seen at 310 and 320℃ for α-CD and β-CD, respectively which is due to thermal decomposition of CDs. CEO shows a characteristic endothermic peak at 178℃ corresponding to its hydrolysis or oxidation. By incorporation of guest molecule in CDs cavity and replacement of water molecules, active compounds were protected within the cavity of the CDs and peaks of inclusion complex of CEO were not detected in the thermogram. These results strongly suggested formation of strongly interaction between CEO and CDs and formation of stable inclusion complex in solid state [15–18].
DSC image of (a) α-CD, (b) β-CD, (c) CEO/α-CD room temperature, (d) CEO/α-CD sonic energy, (e) CEO/β-CD room temperature, (f) CEO/β-CD sonic energy, and (g) CEO.
By formation of inclusion complexes between CDs and CEO, new intermolecular or intramolecular bonds were formed between CDs and CEO, so the characteristic peak of CEO was shifted to lower values which indicated partially encapsulation of CEO in CDs cavity [15–18].
SEM images
Figures 6 and 7 illustrate the SEM of α-CD, β-CD, CEO, and their inclusion complexes at different conditions (at room temperature and under sonic energy). Pure CDs observed to be irregular in shape whereas CEO was irregular rather spherical. Formation of inclusion complex indicated by structurally and morphology changes of pure compounds and inclusion complexes. Original morphology of both the components had disappeared and irregularly crystals with a uniform particle size distribution and no aggregation was seen [14–18].
SEM image of (a) α-CD and (b) β-CD. SEM image of (a) CEO/α-CD room temperature and (b) CEO/α-CD sonic energy.

Discrete homogenous microcrystals are obtained under sonic energy which revealed the major effect of sonic energy on the formation supramolecular nanocontainers. At room temperature and without sonication, the inclusion compounds tend to be randomized without discrete homogenous crystals being created. Size of crystals strongly depended to ultra-sonication. Hence, the FTIR spectral study and SEM image analysis confirm the formation of an inclusion complex between CEO and CDs [14–18].
Dissolution study
The dissolution of CEO and its inclusion complex with CDs were investigated to evaluate the CEO release profiles (Figure 8). Dissolution medium was simulated for gastric (0.1 M HCl, pH 1.2) and enteric conditions (PBS, pH 7.4). In acidic pH = 1.2, hydroxyl groups of CDs are protonated, load a positive charge and more intense release as compared to the neutral conditions (pH = 7.4). Also in acidic pH, solubility of CEO is higher than neutral condition. The release of pure CEO and the physical mixture was very low (<2%) at every time point [20].
Release profiles of CEO and its inclusion complex with CDs at various conditions at pH = 7.4 and pH = 1.2.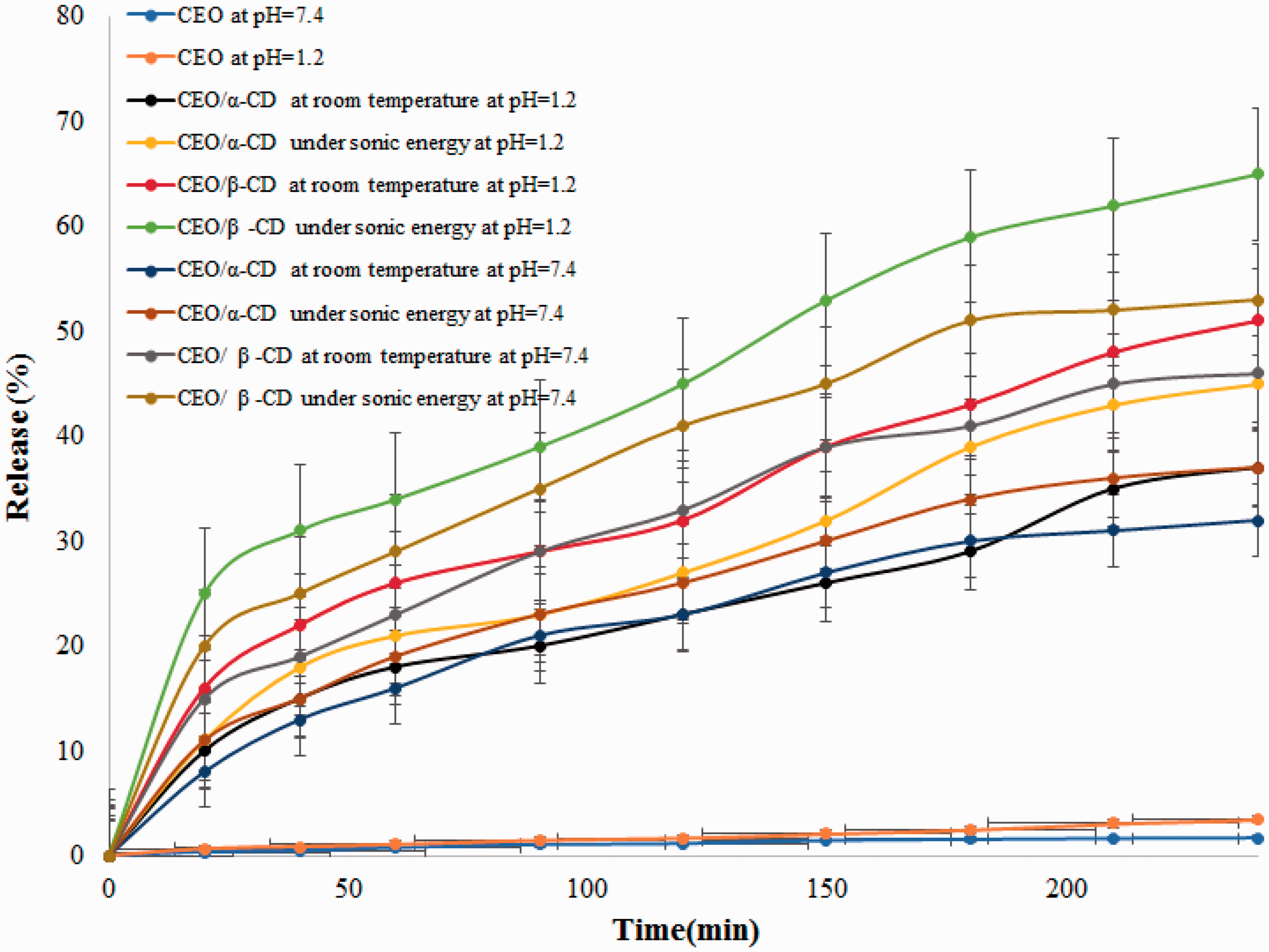
Morphology of PCL-based nanofibers containing inclusion complex of CDs/CEO
Morphology of obtained PCL-based electro-spun containing inclusion complex of CEO/CDs at various conditions were investigated by SEM (Figure 9). Bead-free and uniform nanofibers were resulted with average diameter around 300–600 nm for DCM/DMSO and a wide range of dispersion.
SEM of PCL-nanofibers and PCL-based nanofibers containing inclusion complex of CEO/CD at various conditions.
Under sonic energy, the obtained inclusion complexes of CEO/CDs are smaller than complexes at room temperature, so the diameter of the obtained nanofibers is smaller than nanofibers which are loaded with CEO/CD at room temperature. EDX analysis of PCL nanofibers loaded with CEO/CD inclusion complex under sonic energy confirmed the presence of the CEO/CD inclusion complexes trapped within the PCL and reached the collector along with the polymer solution during the electrospinning process. In neat PCL, distribution of carbon and oxygen elements is less than PCL nanofiber containing CEO/CD nanocontainers. Also, the EDX results showed that PCL nanofibers containing CEO/β-CD at room temperature and under sonic energy have a uniform distribution of carbon and oxygen elements than PCL nanofibers containing CEO/α-CD which is related to larger cavity size of β-CD than α-CD which can entrap higher CEO molecules in its cavity. With regard to all of CEO/CD inclusion complexes, no aggregation of CEO/CD was seen at the surface of nanofibers and nanofibers were more uniform and bead-free. A broad range of diameter distribution of fibers is because of incorporation of CEO/CD inclusion complex in nanofibers [20–22].
In vitro CEO release from the obtained nanofibers
The time-programmed release behavior of CEO from PCL-based nanofibers is shown in Figure 10. Under sonic energy, higher amount of CEO encapsulated in CDs cavity so higher amount of CEO released from PCL nanofibers loaded with CEO/CD inclusion complexes. On the other hand, PCL nanofibers which were loaded with CEO/CD at room temperature showed larger diameter than PCL nanofibers which were loaded with CEO/CD under sonic energy [20–22]. Incorporation of CDs inclusion complexes with various compounds into nanofibers matrix could control release of guest molecules which is attractive way for designing fibrous materials with advanced applications such as drug delivery and tissue engineering [18–20].
Release profiles of CEO from PCL-based nanofibers containing inclusion complex of CDs/CEO at various conditions at (a) pH = 7.4 and (b) pH = 1.2.
By incorporation of CEO/CD inclusion complex in PCL nanofibers, release of CEO/CD inclusion complex is slower than CEO/CD inclusion complex in solid phase. Various parameters controlled drug release from electrospun nanofibers such as drug diffusion rates, drug dissolution rates, drug physical desorption rates, diameters of fibers, and polymer degradation/erosion rates. By incorporation of CEO/CD inclusion complex in PCL nanofibers, several interactions are formed between PCL and CEO/CD. Release of CEO/CD from CDs cavity in the form of nanoparticles provided a burst release process followed by a drug diffusion manner, while during the release of CEO/CD from the nanofibers, the drug diffusion was the predominant factor in drug release [23]. PCL nanofibers showed slower release rate than nanoparticles which is indicative of the new route to further control of CEO/CD release for long time applications.
Conclusion
In the present work, inclusion complex formation between CEO with α-CD or β-CD was reported at two different conditions (under sonic energy and room temperature mixing) and formation of nanocontainers was investigated by SEM, XRD, DSC and FTIR which indicates the formation of inclusion complex between CDs and CEO. XRD results indicated that the obtained crystalline structures are a mixture of channel and cage type structures. By the disappearance of endothermic peaks of pure CDs and CEO, the DSC results proved the formation of inclusion complex. Pure CEO shows poor solubility and formation of inclusion complex significantly improved the solubility content as well as the dissolution rate. Inclusion complex of β-CD and CEO shows higher dissolution rate and control than α-CD and CEO. Controlled release of CEO from CDs-based nanocontainers is a result of reversible hydrogen bonds formation via inclusion complex formation of CD and CEO and can be simply repaired by bringing together the fractured ends for as little as a few minutes at ambient temperature. UV-spectra showed the controlled release of CEO from nanocontainers with time which is based on reversible hydrogen bonds formation in the inclusion complex of CDs and CEO. Acidic pH protonated the hydroxyl groups of CDs and loaded a positive charge to release CEO compared to neutral pH. The SEM and EDX images of PCL-based nanofibers containing CEO/CD inclusion complexes at various conditions indicated bead-free and uniform fibers with no aggregation of inclusion complexes at the fiber surface. Also, these nanofibers showed controlled release of CEO with time.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding is acknowledged from Iran Polymer and Petrochemical Inistitute. Award id (24793101).
