Abstract
A total synthesis of the natural polycyclic phenolic compound daldiquinone was reported derived from 1-naphthaldehyde. The synthesis utilizes regioselective peri-C-H oxidation as a key step, proceeding through a longest linear sequence of seven steps with an overall yield of 40%. The binaphthol intermediate is obtained via Baeyer-Villiger oxidative rearrangement in 81% yield, followed by IBX oxidation to produce naphthoquinone. The binaphthalene core is constructed using Suzuki coupling. This strategy, which relied on direct oxidation of the C-H bond of the naphthalene ring, significantly truncated the synthetic pathway and boosted efficiency. The regiospecific approach offers a promising pathway for obtaining a wide array of daldiquinone derivatives.

Introduction
Naphthalene-containing compounds represent a distinct class of molecules characterized by their unique structure and significant biological importance, found in numerous natural products and pharmaceuticals.1–5 The distinctive structure and properties of naphthalene provide broad opportunities for drug discovery.6–9 The aromatic polycyclic phenolic compounds with naphthalene skeletons, such as naphthols and naphthoquinones, have garnered attention for their wide-ranging pharmacological activities, including antioxidant,10,11 anti-inflammatory,12–16 antitumor,17–20 as well as their roles in regulating blood glucose levels21–24 and improving the intestinal microenvironment.25,26 Daldiquinone, a binaphthoquinone, was isolated by Koyama’s group in 2018 from Daldinia concentrica, and was discovered to possess potent anti-angiogenesis activity. 27 The first total synthesis was reported by Türkmen, 28 who employed 1,8-dihydroxynaphthalene as the starting material. However, the limited variety of 1,8-dinaphthol constrains the diversity of daldiquinone analogs and the synthesis of peri-naphthol is mostly relied on the substituted precursors of benzene containing C-O bonds, resulting in lengthy reaction sequences.
The development of C-H activation techniques29–31 has enabled efficient natural product synthesis through direct cleavage of inert C-H bonds and subsequent functionalization.32–34 It is recognized that natural polycyclic phenolic compounds would be obtained via direct C-H oxygenation of naphthalene material. Our previous studies demonstrated hydroxylation of the peri-position of naphthalene rings using aldehyde groups as directing groups, showcasing broad substrate applicability. 35 Herein, we presented the synthesis of daldiquinone via C-H oxidation as the central step, directly introducing the OH group at the peri-position of naphthalene. This approach facilitated the synthesis of 1,8-dinaphthol through Baeyer-Villiger oxidative rearrangement, enriching the diversity of dihydroxynaphthalene species and expanding the scope of daldiquinone derivatives (Figure 1).

(a) Natural products and drug molecules with a polyphenol structure. (b) Regioselective C-H oxidation of 1-naphtaladehyde with tunable TDGs. (c) This work is for synthesis of daldiquinone.
Results and discussion
Structural analysis of daldiquinone revealed that it was constructed by a Suzuki–Miyaura cross-coupling between naphthoquinone

Retrosynthetic analysis of daldiquinone.
The synthesis began with commercially available 4-bromo-1-naphthaldehyde. A hydroxyl group was smoothly installed at the peri-position of naphthaldehyde

The synthesis of daldiquinone.
Conclusion
We have devised an efficient synthetic route for the natural product daldiquinone, achieving an overall yield of 40% through a linear sequence of seven steps. This process begins with commercially available 4-bromo-1-naphthaldehyde and leverages Pd-catalyzed regioselective peri-C-H oxidation as a pivotal step. The application of regioselective C-H oxygenation proved critical for constructing the hydroxylated naphthalene core, facilitating access to active naphthols and their derivatives. This method establishes a versatile platform for the rapid synthesis of diverse natural polycyclic phenolic compounds. Ongoing research in our group focuses on expanding the synthesis of daldiquinone derivatives and exploring their biological activities.
Experiment
General information
1 H (500 MHz) and 13 C (125 MHz) NMR spectra were examined on Bruker AV-500 instruments at ambient temperature, using CDCl3 and DMSO-d6 as the solution with TMS as the interior label. Chemical shifts were reported as δ values and j (Hz). HRMS experiments were conducted in the positive mode using a Thermo Fisher Scientific Q Exactive Plus in ESI mode. Unless otherwise stated, all reagents were commercially sourced and utilized without further purification. The reaction progress was monitored by TLC from Jiangyou Chemical Co., Ltd. (Yantai, China), visualized using UV irradiation (254 nm). Silica gel (100–200 mesh) from Haiyang Chemical Co., Ltd (Qingdao, China) was used.
Procedure for synthesis of daldiquinone

4-Bromo-8-methoxy-1-naphthaldehyde (2)
4-bromo-1-naphthal-dehyde
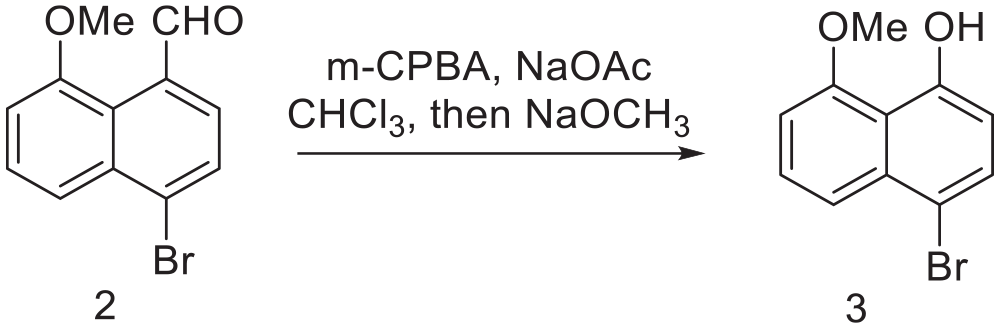
4-bromo-8-methoxynaphthalen-1-ol (3)
NaOAc (3.6 mmol) and 3-chloroperoxybenzoic acid (3.6 mmol, technical ~70%) were added to compound

4-Bromo-8-methoxynaphthalene-1,2-dione (4)
Compound

1-Bromo-4-(ethoxymethoxy)-5-methoxynaphthalene (5)
NaH (1.8 mmol) was added to compound 3 (1.5 mmol) in anhydrous DMF (3.0 mL) in an ice-water bath and maintained at 0°C with continuous stirring for 1 h. Chloromethoxyethane (2.25 mmol) was added at 0°C, then slowly warmed to ambient temperature and stirred continuously for another 2 h. The reaction was neutralized with H2O and extracted twice with EtOAc. The organic phases were combined, concentrated under vacuum, and subsequently purified to afford compound

2-(4-(Ethoxymethoxy)-5-methoxynaphthalen-1-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (6)

Under an inert N2 atmosphere, B2Pin2 (1.2 mmol), Pd(dppf)Cl2·CH2Cl2 (0.1 mmol), KOAc (4.0 mmol), and compound
Daldiquinone
Under an inert N2 atmosphere, compound
Supplemental Material
sj-docx-1-chl-10.1177_17475198251316139 – Supplemental material for Concise preparation of daldiquinone via palladium-catalyzed regioselective peri-C-H oxygenation of the naphthalene ring
Supplemental material, sj-docx-1-chl-10.1177_17475198251316139 for Concise preparation of daldiquinone via palladium-catalyzed regioselective peri-C-H oxygenation of the naphthalene ring by Jing Jiang, Yue Gao, Chenqi Wu, Suqin Zhang and Shuyang Hu in Journal of Chemical Research
Footnotes
Authors’ note
This article does not contain any studies with human or animal participants. Experimental procedures, characterization data for all products, and copies of the 1H NMR, 13C NMR are all in the Supplemental Material.
Author contributions
All authors contributed to the article.
Data availability
The data underlying the findings of this study are accessible in the Supplemental Materials accompanying this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Natural Science Research Foundation of Colleges and Universities of Jiangsu Province (no. 24KJD350001), PhD Faculty Special Program at Suzhou Polytechnic Institute of Agriculture (no. BS[2022]22), Special Funding for Suzhou Polytechnic Institute of Agriculture Innovative Research Team (CXTD202408) and Suzhou Industry-Education Integration Backbone Program.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
