Abstract
The development of novel antimicrobial agents is crucial in addressing antibiotic resistance. Combining ciprofloxacin with silver could potentially enhance antibacterial and antifungal properties, addressing current limitations in antimicrobial therapies. The study aimed to synthesize a ciprofloxacin–silver complex and subsequently evaluate its antibacterial and antifungal properties through in vitro assessments. The Schiff base of ciprofloxacin was synthesized by reacting methanolic ciprofloxacin (0.03 mol; 9.9 g) with o-toluidine (0.03 mol; 3.2 mL) in glacial acetic acid. The new compound was characterized using 1H NMR and IR spectroscopy and in vitro antibacterial and antifungal activity was studied. IR spectra of metal complexes revealed a significant shift in C=O absorption (1708–1692 cm−1), indicating complex molecular interactions. 1H NMR analysis of the Schiff base and its silver complex in DMSO-d6 showed the absence of the ligand’s –OH proton peak at 15.08 δ, suggesting oxygen atom involvement in complex formation. In vitro antimicrobial assessments demonstrated L1A’s enhanced antibacterial and antifungal efficacy compared to the ligand (L1) and ciprofloxacin, which may be attributed to synergistic metal complexation and increased lipophilicity. These findings suggest that the ciprofloxacin–silver complex represents a promising candidate for developing next-generation antimicrobial agents, potentially offering a new strategy to combat antimicrobial resistance.
Introduction
The human body is a complex ecosystem harboring diverse microorganisms in specialized niches such as the skin, mucosal linings, and gastrointestinal tract.1,2 Immunocompromised patients are particularly vulnerable to severe microbial infections, necessitating urgent broad-spectrum antibiotic interventions. This urgency reflects escalating challenges in modern healthcare, as morbidity and mortality rates continue to rise. 3
Antibiotics are essential tools in modern medicine, playing a crucial role in treating bacterial infections by either inhibiting bacterial growth or eliminating them entirely. 4 These vital drugs are categorized into various classes, including penicillins, cephalosporins, tetracyclines, fluoroquinolones, aminoglycosides, quinolones, streptogramins, and sulfonamides. Each category comprises specific derivatives designed to combat particular types of bacteria, reflecting the complexity of bacterial resistance mechanisms and adaptive strategies. 5
Within this landscape of antimicrobial agents, Schiff bases or imines have emerged as compounds of particular interest. Characterized by their azomethine group, Schiff bases exhibit robust chelating abilities, rendering them useful for various scientific applications. Their potential for complex formation, especially in conjunction with antibiotics, underscores the innovative pathways explored to enhance the effectiveness of existing antibiotics.6,7
Quinolones, a cornerstone of antibiotic therapy since the introduction of nalidixic acid in 1962, have demonstrated remarkable efficacy. Quinolones disrupt essential cellular processes by inhibiting bacterial DNA gyrase, a vital enzyme for DNA replication, thereby hindering bacterial proliferation.8 –10 Building on this foundation, researchers have explored metal complexes with quinolones and revealed promising avenues for enhancing their antibacterial potency. These investigations have opened new vistas, especially concerning synthetic fluoroquinolones such as ciprofloxacin, which are celebrated for their broad-spectrum activity. 11
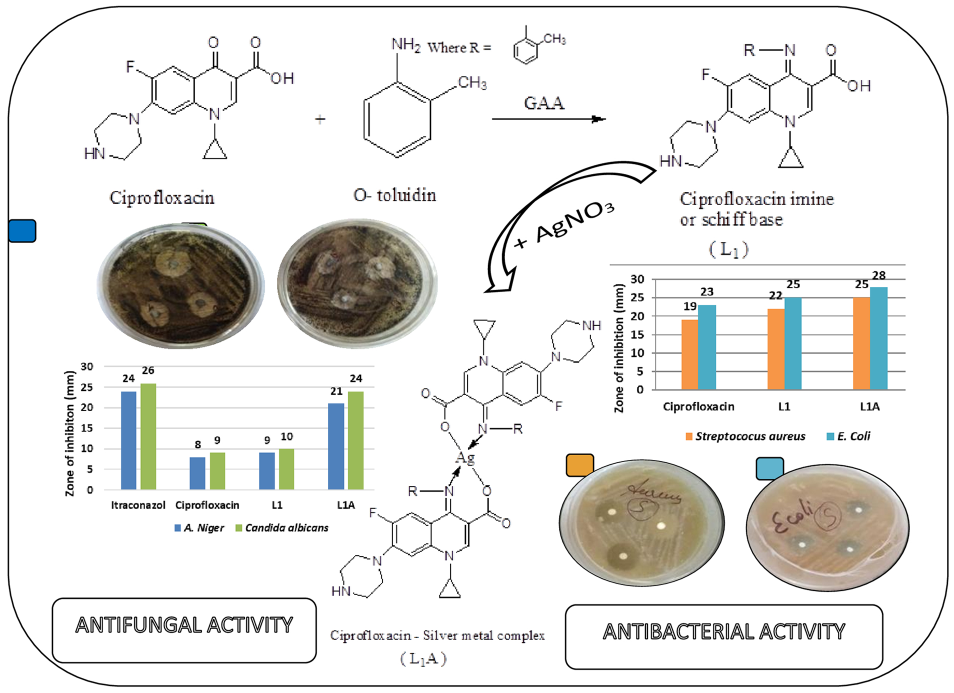
The present study aims to augment the stability and versatility of ciprofloxacin through intricate chemical transformations. Specifically, the keto and carboxylic acid (COOH) groups of ciprofloxacin were targeted to create a Schiff base. This novel compound, resulting from a combination of antibiotic- and metal-binding properties, has the potential for groundbreaking applications in antimicrobial therapy. A pivotal step in this exploration involves the complexation of this Schiff base with silver metal ions, a strategic approach aimed at amplifying its therapeutic efficacy and spectrum of action.
This study reports the synthesis and analysis of a new ciprofloxacin-based compound and its metal complex. Carefully designed experiments were conducted to assess their antibacterial and antifungal activities against a variety of microbial strains. These investigations aim to offer valuable insights into antimicrobial research, potentially advancing the development of more effective and versatile therapeutic agents.
Experimental
Materials and methods
Pure ciprofloxacin and itraconazole were obtained from Merck and were used without further purification. All the synthesis procedures employed analytical-grade laboratory reagents. The bacterial and fungal strains used in this study were provided by the DOLPHIN Institute of Dehradun. Infrared (IR) spectra were recorded by RC Saif at Punjab University, Chandigarh, using a Philips analytical PU 9800 FTIR spectrometer. UV-Vis spectra were measured with a spectrophotometer (ELICO LTD-200) and analyzed using the Spectra-Treats software at HIPR, Dehradun. Proton Nuclear Magnetic Resonance (1H NMR) spectra in DMSO were obtained using a Bruker Avance-II 400 MHz spectrometer at Punjab University, Chandigarh. The melting point of the synthesized compound was precisely determined using a digital Gallenkamp MP apparatus (Geminivi BV) at HIPR, Dehradun. The antibacterial and antifungal activities were evaluated using the disk diffusion method (Imran et al. 2007).
Synthesis of ciprofloxacin imine or Schiff base (L1)
The synthesis of the ciprofloxacin imine, also known as the Schiff base (L1), was achieved by carefully mixing ciprofloxacin (0.03 mol; 9.9 g) with o-toluidine (0.03 mol; 3.2 mL) in 25 mL of methanol. The mixture was then refluxed for 4 h with 1.8 mL of glacial acetic acid (0.03 mol). After the reaction, the solution was concentrated in a water bath and then cooled to 0 °C. The solid formed was filtered, washed with methanol and ethanol, and dried to obtain Schiff base (L1) (67% yield). The detailed synthesis procedure is shown in Figure 1. m.p.: 277–279 °C. Rf value: 0.62 (ammonia: methanol, 6:2:2). IR (v, cm−1): 1708 (C=O), 3377 (O-H), 1624 (C=N). 1H NMR (400 MHz, DMSO): δ 1.07 (s 1H, NH); 1.08–1.33 (m 4H, cyclopropyl); 1.9 (m 3H, CH3); 2.78 (d 2H, CH2); 3.46 (d 4H, CH2); 3.92 (s 1H, cyclopropyl); 7.5 and 8 (m 4H, benzylidenimine); 7.7 (d 2H, benzene); 8.8 (s 1H, ethylene); 15 (s 1H, OH).

Synthesis of ciprofloxacin imine or Schiff base (L1).
Synthesis of ciprofloxacin–silver complex (L1A)
The synthesis of the ciprofloxacin–silver complex (L1A) was initiated by carefully mixing the Schiff base ligand (0.015 mol; 6.30 g) with silver nitrate (0.0075 mol; 1.30 g) in 25 mL of methanol. The mixture was refluxed for 2.5 h, resulting in the formation of colored precipitates. The precipitates were then filtered, washed with methanol, and dried (62% yield). The complete synthetic process is illustrated in Figure 2. m.p.: 259–261 °C. Rf value: 0.57 (ammonia: methanol, 6:2:2). IR (v, cm−1):1692 (C=O), 1624 (C=N), 624 (O-Ag). 1H NMR (400 MHz, DMSO): δ 1.07 (s 1H, NH); 1.08–1.33 (m 4H, cyclopropyl); 2 (m 3H, CH3); 2.78 (d 2H, CH2); 3.46 (d 4H, CH2); 3.92 (s 1H, cyclopropyl); 7.5 and 8 (m 4H, benzylidenimine); 7.7 (d 2H, benzene); 8.8 (s 1H, ethylene).

Synthesis of ciprofloxacin–silver complex (L1A).
Molecular docking
Molecular docking studies were carried out to evaluate the binding energies of the synthesized metal complexes against microbial strains: Staphylococcus aureus, Aspergillus niger, and Escherichia coli. These analyses provided a detailed understanding of how the complexes interact with target protein binding sites. Using the Maestro 12.8 software package, the binding-free energies were estimated, offering essential insights into the binding affinities between the ligands and the model Target Preparation Protocol (TPP) in comparison to the standard drug, ciprofloxacin. 12
ADMET and drug-likeness predictions
The ADMET properties were evaluated using online Admetlab 2.0. The process began by drawing all structures using ChemDraw software to generate their InChI Key and isomeric SMILES strings. The analysis was consistently based on the SMILES strings. 13
Antibacterial studies (in vitro)
The in vitro antibacterial activities of the investigated compounds were tested against Streptococcus aureus and E. coli using the disk diffusion method with nutrient agar as the medium. 14 Nutrient agar was prepared by dissolving beef extract (1.5 g), peptone (2.5 g), agar (7.5 g), NaCl (2.5 g), and distilled water (500 mL) followed by sterilization in an autoclave. Sterilized medium was poured into Petri dishes and allowed to solidify. Sterile disks were arranged in Petri dishes containing an agar medium previously inoculated with microbial organisms. A mechanical pipette was used to dispense a fixed volume of ligand and silver complex 10 µL (10 µg/0.01 mL) onto each disk, ensuring that the pipette tip made light contact with the disk during the process. The Petri dishes were then incubated at 37 °C for 24 h. The resulting inhibition zones were carefully measured in millimeters (mm). Ciprofloxacin was used as a reference drug for comparative analysis.
Antifungal studies (in vitro)
The in vitro antifungal activity was assessed against Candida albicans and A. niger using the disk diffusion technique. The fungus was pre-cultured in a nutrient medium overnight and then incubated in a biochemical oxygen demand (BOD) incubator at 35 ± 2 °C, as this temperature is optimal for maximum organism growth. Sterile disks (Whatman No. 1 filter paper) were placed in Petri dishes containing Sabouraud dextrose agar (SDA) medium that had been pre-inoculated with fungal organisms. Using a mechanical pipette, 10 µL of the ligand and silver complex (10 µg/0.01 mL) were carefully applied to each disk, ensuring that the pipette tip gently touched the disk during the application. The Petri dishes were then incubated at 35 ± 2 °C for 24 h. The resulting inhibition zones were measured in millimeters (mm). Itraconazole was used as a reference drug for comparative analysis.
Results
A ligand (L1) and its corresponding metal complex (L1A) were synthesized, and their physical properties are listed in Table 1.
Physical properties of ligand and metal complex.

The purity and characterization of the compounds were confirmed through various physical properties, including MP, pH, and Rf values (solvent system: butanol: ammonia: methanol, 6:2:2). Elemental analysis, FTIR, and 1H NMR spectrometry were conducted for detailed characterization (see Table 2 and Figures 3 and 4, respectively).
FTIR data of L1 and L1A.


NMR interpretation for L1A.
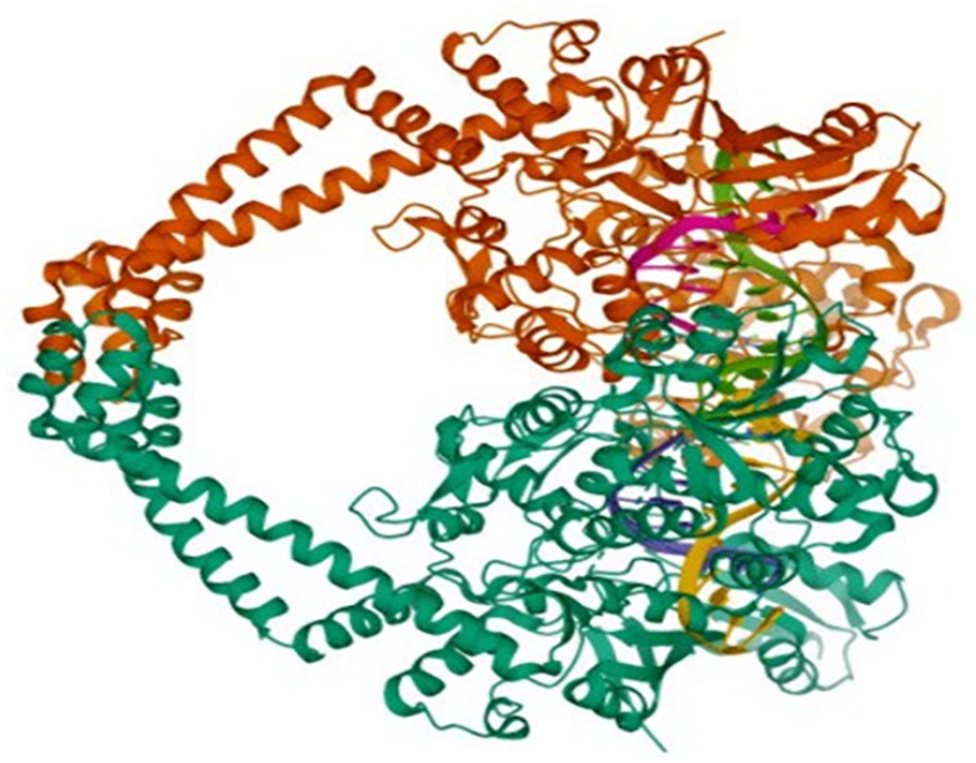
3D model of a protein (2XCT).
FTIR of ligand and ciprofloxacin–silver complex
The IR spectra of the unbound Schiff base ligand exhibited a prominent peak within the range of 1708 cm−1 (corresponding to a COOH functional group), 15 which shifted to 1692 cm−1 in the silver complex, suggesting that the carboxylic oxygen and silver were coordinated. The Schiff-based ligand also exhibited a prominent peak within the spectral range of 1624 cm−1, indicating the presence of an azomethine (C=N) functional group (Table 2). Previous studies have shown that including the N atom from the Schiff base ligand within the coordination sphere reduces bond order within the C=N group (Demehin et al., 2020). This reduction can be explained by solid electron density donation to the metal ion, which eventually decreases the stretching frequency of the C=N bond. Owing to the presence of ν (C=N), the IR spectra of the Ag complex exhibited a noticeable negative shift toward lower wavenumbers (1608 cm−1), indicating the coordination of azomethine N to Ag. A characteristic band in the complex at 1624 cm−1, absent in the Schiff base, suggested that both the carboxylic oxygen and nitrogen of azomethine were in coordination with the silver metal. 16 In addition, the metal complex exhibited a band at 624 cm−1, further confirming complex formation.17,18 The data are presented in Table 2.
1 H NMR of ligand and ciprofloxacin–silver complex
The proton peak observed at 15.08 δ in the downfield region of the spectra can be attributed to the presence of the –OH group in ligand.19,20 The chemical shifts of the uncomplexed ligands were compared to those of their Ag complexes, revealing that all signals fell within the anticipated range, except for the –OH group. The lack of this signal for the –OH group in silver complex indicated its deprotonation and the participation of its oxygen atom in complex formation (Figures 3 and 5).
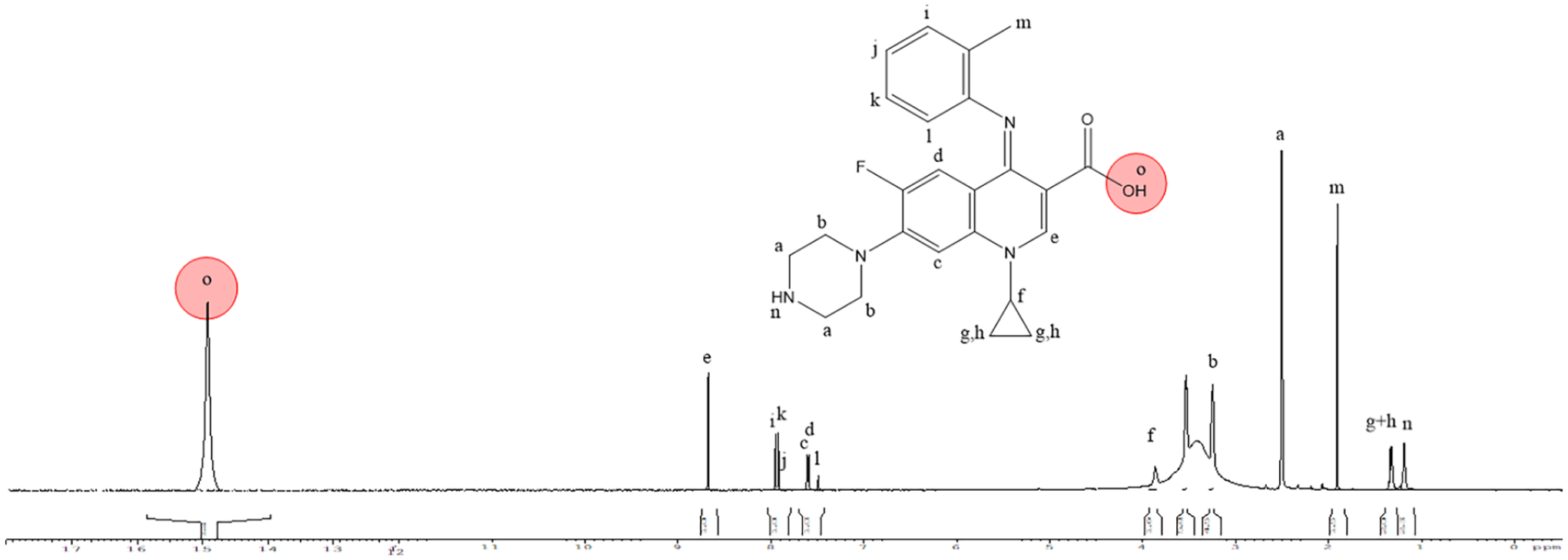
NMR interpretation for L1.
Molecular docking of the metal complexes
Ciprofloxacin metal ions, such as ciprofloxacin analog (L1) and ciprofloxacin–silver complex (L1A), showed the highest docking score and had significantly higher binding energies than the prescription drug ciprofloxacin, which had a docking score of (−7.8). Hence, ciprofloxacin metal ions as ligands exhibited superior docking and glide energies compared to those of the standard drugs. The ligands with the highest affinity for the TPP model are listed in Table 3. 21
Outcomes of molecular docking of different ligands.

Furthermore, a report revealed that the docking of calcineurin with inhibitors was examined using Maestro 12.8 and visualized by Discovery Studio, resulting in the prediction of binding affinity energies ranging from −9.6 to −7.8 kcal/mole. These predictions are consistent with the results of the present study. 22 The structure of the S. aureus gyrase complex with a ciprofloxacin analog (PDB ID-2XCT) exhibited a twinned arrangement (Figure 4). Other parameters such as drug efficacy, drug-likeness properties, pharmacokinetics, toxicity studies, and lipophilicity (in clogs) have been used for several years. For favorable drug-likeness parameters, a cLogP value ⩾5 within the acceptable range can be used. 23 Overall, the designed compounds were found to have clog values >4, implying their druggability for the oral drug administration route (see Table 4). This revealed that the metal complexes under investigation exhibited better antibacterial activity than uncomplexed ligands. The current study was conducted to identify a specific antibiotic rather than a broad-spectrum antibiotic.
Drug-likeness properties.

The paired 3.35A crystal structure of the S. aureus gyrase complexed with ciprofloxacin (depicted in Figures 6 and 7) and DNA. The classification of the subject under discussion encompasses isomerase, DNA, and antibiotics. The organisms used in this study were S. aureus subsp: aureus N315 and a synthetic construct. The expression system employed was E. coli BL21(DE3). The presence of mutation(s) is confirmed.

2D and 3D diagrams showing the docked conformation compound for ciprofloxacin.

Ciprofloxacin analog (L1) 2D and 3D diagrams of docked conformation compound.
Drug-likeness properties
ADMETLAB software (version 2.0) was used to study the drug-likeness of the complex and ligand. The results are presented in Table 4. Lipinski’s Rule of Five, Pfizer, GSK, and Golden Triangle rules were predicted for our compounds.13,24
Biological Activities
Antibacterial activity and antifungal activity
Table 4 presents the results of in vitro biological screening of the two bacterial strains. S. aureus and E. coli (shown in Figures 8 and 9) and the zone of inhibition values were measured by rounding of the well and using a scale to measure the diameters, as summarized in Table 5 and Figures 8 and 9.

Zone of inhibition for E. coli.

Zone of inhibition for S. aureus.
Zone of inhibition (mm) values of ligand and metal complex against for S. aureus and E. coli strains.

Evaluation of the ligand and its metal complex revealed better antibacterial activity than the uncomplexed ligand (Table 6 and Figure 10). The graph shows that the zone of inhibition for S. aureus were 19, 22, and 25 mm for ciprofloxacin, L1, and L1A, respectively. However, better results were observed for E. coli, where it was 23, 25, and 28 mm for ciprofloxacin, L1, and L1A, respectively. Our antibacterial results conformed to those of previous reported studies.25,26 The rationale behind the enhanced antibacterial study of the ciprofloxacin–silver complex is the concept of the overtone and chelation theory. According to the permeability of cell membrane overtones, the lipid bilayer that forms the cell membrane only enables lipid-soluble compounds to pass. Therefore, lipid solubility regulates bacterial activity. Chelation has been observed to substantially decrease the polarity of the metal ions. Consequently, the ligand orbital and partial positive charge of the metal ion interact, resulting in an overlap with the donor groups. Moreover, π-electron delocalization was observed to be distributed across the entirety of the chelate ring, resulting in an augmentation of the lipophilic nature of the complexes. 27 Based on these reports, increased lipophilicity of these complexes may facilitate their efficient traversal across lipid membranes, thereby enabling their binding to metal coordination sites within enzymes.
Zone of inhibition (mm) values of ligand and metal complex against for A. niger and C. albicans.


Comparative study of zone of inhibition for metal complex, ligand, and standard drug ciprofloxacin against S. aureus and E. coli.
Complexes such as these also interfere with the respiratory mechanism within the bacterial cell and suppress anabolic processes, such as protein synthesis, thereby stunting their growth and multiplication. The antifungal activity of ligands (L1) and their corresponding metal complexes (L1A) were assessed against strains of A. niger and C. albicans (shown in Figures 11 and 12).

Zone of inhibition for A. niger.
Zone of inhibition for C. albicans.
Our results revealed that the zone of inhibition for L1A (21 mm) was two times higher than that of ciprofloxacin (8 mm) and L1 (9 mm); however, it was not better than that of itraconazole (24 mm) against A. niger. Similarly, the measures of zone of inhibition revealed that only the L1A complex (24 mm) showed enhanced antifungal activity compared to ciprofloxacin (9 mm) and L1 (10 mm), and it was almost equivalent to that of itraconazole. Table 6 and Figure 13 present these findings.

Comparative study of the zone of inhibition for ligand and its metal complex and standard antifungal drug itraconazole against A. niger and C. albicans.
Discussion
Confirmation of ligand, and metal complex formation using FTIR and NMR spectroscopy
The synthesis and characterization of the ligand (L1) and its corresponding metal complex (L1A) were carried out, and its purity and stability were confirmed by evaluating the physical properties, including melting point, pH, and Rf value. The FTIR analysis revealed significant shifts in key functional groups, with the COOH group shifting from 1708 to 1692 cm−1, while the azomethine group shifted from 1624 to 1608 cm−1 upon metal complexation. These observations align with the previous studies by Konstantin et al. 28 and Al-Assafe et al., 29 which demonstrated that such shifts indicate the complexation of the carboxylic oxygen and azomethine nitrogen to the metal center, confirming complex formation. The appearance of a new band at 624 cm−1, which was absent in the Schiff base, further suggested that both the carboxylic oxygen and nitrogen of azomethine were in the complexation with silver.
1 H NMR spectroscopy analysis further corroborated the formation of this metal complex. The disappearance of the hydroxyl (–OH) signal at 15.08 δ in the complex spectrum indicates deprotonation and subsequent complexation of the oxygen atom to the metal center. Sadeek et al. 30 also reported a similar deprotonation in the complexation of metal ions. This structural evidence, supported by NMR and FTIR spectroscopy, collectively supports the conclusion that L1A has formed a stable metal complex, a crucial factor contributing to its biological activity.
Molecular docking and likeness analysis suggest improved efficacy of metal complex
Several studies have explored the role of molecular docking in elucidating the enhanced efficacy of metal–fluoroquinolone complexes. Abd El-Lateef et al. 31 demonstrated that metal-coordinated ciprofloxacin analogs exhibited superior binding affinities compared to the free drug. This study complements his findings, as L1A exhibits higher binding energies than ciprofloxacin, suggesting enhanced target interactions due to metal chelation. In addition, ADME was further evaluated using ADMETLAB software, which confirmed the drug-likeness properties of L1A. The cLogP values (>4) suggest that the designed compounds possess favorable oral bioavailability, adhering to Lipinski’s Rule of Five and other drug-likeness criteria. The improved binding affinity of L1A compared to ciprofloxacin could potentially lead to enhanced therapeutic efficacy and reduced dosage requirements.
L1A and antifungal properties
The pioneering work of Akinremi et al. 32 on metal–fluoroquinolone complexes established a fundamental framework for elucidating their broad-spectrum antimicrobial activity. The presented study extends this knowledge by specifically investigating the augmented antibacterial and antifungal efficacy of L1A. Mendonça et al. 33 previously reported the antifungal properties of metal–fluoroquinolone complexes. In addition, Măciucă et al. 34 suggested the synergistic potential of metal–fluoroquinolone complexes. Our findings not only confirm but also add further to this research by demonstrating L1A’s significant inhibitory activity against fungal strains like A. niger and C. albicans. The observed efficacy of L1A against these pathogens is particularly noteworthy, as they represent some of the most prevalent and problematic fungal species in clinical settings. This antifungal activity suggests a potential mechanism of action that may differ from or complement existing antifungal agents. This could be attributed to the unique structural and chemical properties of metal–fluoroquinolone complexes, which may enhance their ability to penetrate fungal cell walls or interfere with essential cellular processes.
L1A and antibacterial properties
The biological assays suggest improved antibacterial activity of L1A against S. aureus and E. coli compared to both the ligand (L1) and the standard drug ciprofloxacin. This improved activity may be attributed to the overtone and chelation theories, which posit that metal complexes exhibit increased lipophilicity, allowing them to penetrate cell membranes more efficiently.35,36 The chelation process in L1A reduces the polarity of metal ions, resulting in enhanced lipophilicity and more effective interaction with biological membranes. This observation aligns with the findings of Sabale et al, 37 who emphasized the importance of lipophilicity in the antimicrobial efficacy of metal complexes.
While previous studies, such as that by Efthimiadou et al, 38 primarily focused on the lipophilic nature of metal complexes and their impact on cell membrane permeability, our research extends this understanding by integrating molecular docking studies. This approach provides more comprehensive insight into the interactions between L1A and its target proteins, revealing the complex mechanisms underlying its antibacterial activity. This deeper exploration enriches the existing literature and offers new insights into the design of metal–fluoroquinolone complexes with enhanced therapeutic potential.
The implications of this research may extend beyond the immediate findings. L1A’s observed antimicrobial activity and the elucidation of its molecular interactions suggest potential for further investigation as a candidate for therapeutic development. Building on previous studies, this research contributes to the understanding of designing targeted antimicrobial agents. Further research is needed to fully explore these possibilities and validate the preliminary findings.
Future directions
Future research should focus on exploration of different metal ions and structural modifications of the ligand could optimize antimicrobial activity. Investigation of potential synergistic effects with existing antibiotics and evaluation against drug-resistant clinical isolates would provide valuable insights for potential clinical applications. In addition, expanding the investigation to in vivo toxicology studies, including cytotoxicity against mammalian cells and pharmacokinetic profiling, is essential for bridging the gap between laboratory findings and clinical applications. Comprehensive investigations into these aspects are essential for advancing L1A along the drug development pipeline, emphasizing the importance of a holistic approach to antimicrobial drug development. Moreover, exploring the synergistic effects of L1A in combination with conventional antibiotics could revolutionize combination therapies, providing potent solutions against resistant microbial strains.
Conclusion
This study demonstrates the successful synthesis and characterization of a silver–ciprofloxacin complex (L1A), providing insights into its structural and antimicrobial properties. Spectroscopic analyses confirm the formation of the metal complex, while molecular docking studies suggest enhanced binding interactions. The L1A exhibited improved antibacterial activity as compared to the parent compound, although its antifungal activity was moderate. While these findings contribute to our understanding of metal–antibiotic complexes, further investigations into the mechanism of action, toxicity profiles, and resistance patterns are necessary to fully evaluate its therapeutic potential. This work adds to the growing body of knowledge in metal-based antimicrobial development, offering a foundation for future research in this direction.
Footnotes
Acknowledgements
This work was supported by the Princess Nourah Bint Abdulrahman University Researchers’ Supporting Project number (PNURSP2024R89), Princess Nourah Bint Abdulrahman University, Riyadh, Saudi Arabia.
Consent to participate
All authors have agreed to participate.
Consent for publication
All authors have read the final manuscript and agreed to its publication.
Data availability
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP 2024R89), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia, for funding this work.
Ethical considerations
This article does not contain any studies with human or animal participants.
