Abstract
Due to the importance of heterocyclic rings in drug design, we synthesized a novel multicyclic heterocyclic compound, designated as (2,2′,2ʺ-(benzene-1,3,5-tricarbonyl) tris(6-bromo-4-chloroisobenzofuran-1,3-dione) (compound D), under mild conditions and within an acidic medium. The synthesis of this valuable heterocyclic compound commenced with the preparation of benzene-1,3,5-tricarbohydrazide from benzene-1,3,5-tricarboxylic acid and hydrazine hydrate. This transitional compound was subsequently interacted with 6-bromo-4-chloroisobenzofuran-1,3-dione to yield the target compound D. The synthesized compounds were purified, and their structures were elucidated through advanced spectroscopic techniques, including Fourier-transform infrared (FTIR) spectroscopy, nuclear magnetic resonance (NMR) spectroscopy (1H NMR, and 13C NMR). Gram-negative (Escherichia coli) and gram-positive (Staphylococcus aureus) bacteria were used to evaluate the antibacterial activity of the produced compound. The bioactivity of the new compound was compared to that of conventional antibiotics, such as ampicillin and streptomycin. Interestingly, the compound D exhibited antibacterial activity according to reference drugs. Furthermore, molecular docking studies have shown many connections with amino acid residues in the active sites, thus these results have the possibility to inspire further investigation in the field of heterocyclic compounds.

Introduction
Over the past decade, compounds containing heterocyclic atoms, such as phthalazine derivatives have played a crucial role in drug synthesis, biology, pesticides, agrochemicals, and pharmaceutical agents.1 –4 Furthermore, due to their versatile properties, phthalazine-based heterocyclic species exhibit a big attention from researchers in synthesis organic chemistry.5,6 Generally, these compounds are multi-ring components fused with a pyridazine-dione ring.7,8 It shows a number of biological activities, including anticancer, analgesic, anti-inflammatory, antibacterial, and antifungal properties. 9 According to the previous studies, these heterocyclic compounds prove a variety of biological and therapeutic activities, including anti-proliferative, antitumor, antiparasitic, antioxidant, and antiviral effects.10 –13
Targeting the synthesis of phthalazine derivatives could lead to develop a novel class of these compounds with substantial pharmaceutical and drug potential. 14 Nevertheless, due to limitations in traditional synthesis strategies, a wide range of synthesized heterocyclic derivatives remain inactive against some common diseases. It often involves long reaction times, high temperatures, low yields, and produce a complex crude reaction that affects the desired product and required excessive solvents for purification.15,16 Therefore, finding a new method is crucial to addressing the drawbacks of the traditional protocols. Recently, molecular docking studies have enriched drug discovery research by providing deeper insights into drug–receptor interactions.17,18 Here, we report an approach that predicts the possibility of binding modes and affinities of small drug molecules at their target sites. Accordingly, evaluating the biological activity of the target molecules is a crucial purpose for establishing this approach. Considering these factors, our research aims to synthesize tri-armed structure of 7-bromo-5-chloro- 2,3-dihydrophthalazine-1,4-dione based on benzene ring using economical and readily available starting materials, employing mild reaction conditions, and implementing an easy workup method to achieve high yields.
Notably, in this study, we aim to synthesize a library of polycyclic phthalazine derivatives, focusing on the ability to link at multiple positions on the enzymes to demonstrate their synthetic utility as inhibitory effects against fungi, bacteria, and other studied samples. Moreover, to evaluate their potential uses as antifungal, antibacterial, antioxidant, and anticancer, molecular docking studies were achieved using bioinformatics approaches. These studies were applied to the amino acid residues and their ability to bind with MET283 as a ligand. Furthermore, to ensure binding with ligands, Met283 was used, due to its ability to “affect the interaction between the ligand and the receptor.” Indeed, the substituted side chain of Met283 and its position are vital to provide clear information about the character and intensity of the ligand–receptor interaction. Therefore, the docking studies focusing on the relationship between Met283 and the ligand could improve our understanding of receptor–ligand interactions and thereby aid in drug synthesis development.19,20
Results and discussion
After optimizing the methods for synthesizing our target compounds, we turn our attention to confirming the structures and the properties of the compounds under study. The compounds under investigation were successfully synthesized, and their structures were characterized using Fourier-transform infrared (FTIR) spectroscopy and nuclear magnetic resonance (NMR). Reacting three equivalents of methanol with benzene-1,3,5-tricarboxylic acid led to the synthesis of compound B (trimethyl benzene-1,3,5-tricarboxylate) in mild conditions (see Scheme 1). Using thin-layer chromatography (TLC) with a convenient solvent system (3–7 mL (EtOAc) ethyl acetate-hexane) provides information about the progress of the reaction. After comparing the FTIR spectra of both the reactants and final product, we confirm the synthesis of compound D, which is crucial for biological study and the rest of characterization. From the comparison of the FTIR for compound B and A, a number of new stretching bands back to in compound B were detected, including the (C=O) and (O–H) bands of “benzene-1,3,5-tricarboxylic acid,” which were detected at 1677 cm–1 and 3050–2545 cm–1, respectively. In addition, carbonyl group (C=O) of ester shows a strong band at 1718.5 cm–1. Stretching bands for aliphatic (C–H), aromatic (C–H), and olefinic (C=C) functional groups were observed at 2933 and 2984 cm–1, 3033 cm–1, and 1610 cm–1, respectively. Therefore, the previous outcomes indicate the successful formation of compound B.

The general reaction for synthesis of compound B.
After the successful synthesis of compound B, the next step is to produce compound C (benzene-1,3,5-tricarbohydrazide), which can be achieved by mixing compound B with hydrazine hydrate in methanol at ambient temperature. The reaction’s progress was monitored using TLC in 3–7 mL EtOAc-hexane, as depicted in Scheme 2.
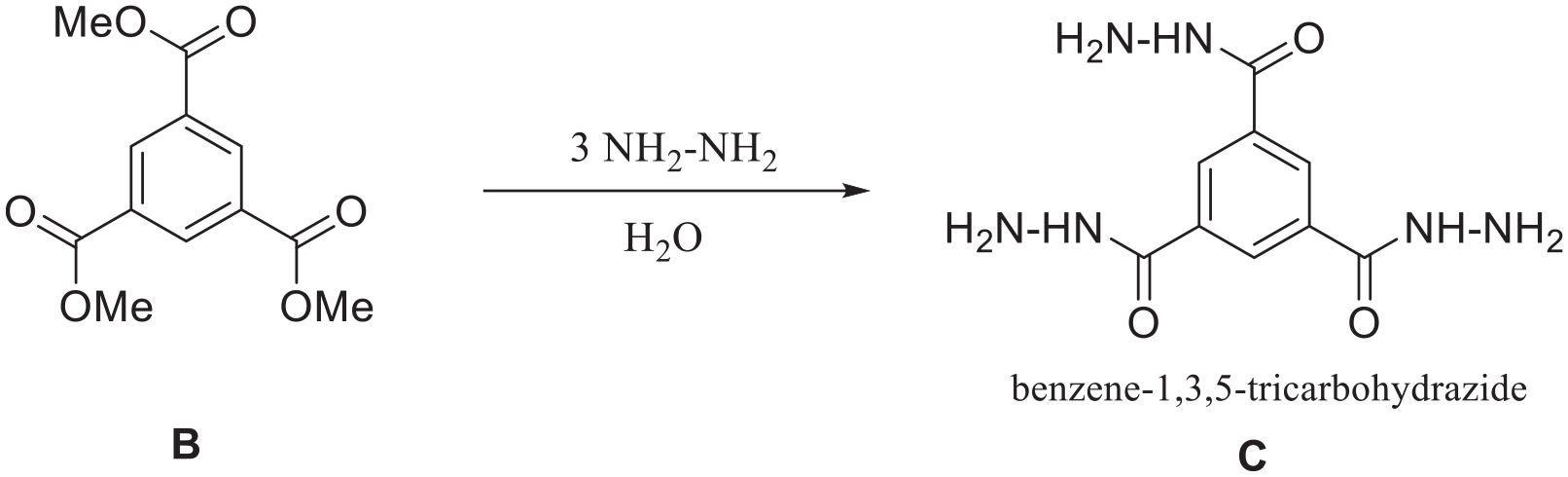
Synthesis of compound C.
The FTIR spectrum of compound C revealed an ester peak (C–O) at 1718.5 cm–1 and a carbonyl (C=O) band at 1654.8 cm–1. In addition, stretching bands corresponding to the carbonyl in the amide compound were observed at 1528 cm–1, while “the symmetric and asymmetric stretching bands” of the (NH, NH2) groups were seen at 3485 and 3248 cm–1, respectively. The FTIR spectra also exhibited aromatic (C–H) stretching bands at 3055 cm–1.
In contrast, compound D was synthesized by refluxing compound C with 6-bromo-4-chloroisobenzofuran-1,3-dione. The reaction progress was monitored using TLC solvent system ((3–7 mL) ethyl acetate-hexane) and after the complete consumption of compound C, the “crude reaction mixture” was processed to isolate the pure final product in high yield, as depicted in Scheme 3. The characterization data for compound D show clear evidence of stretching vibration bands for the C=C group at 1535 cm–1, the C=N group at 1658 cm–1, the aromatic (Ar-H) group at 3080 cm–1, and the NH group at 3274 cm–1. On the contrary, the structure of compound D was further characterized using 1H NMR and 13C NMR spectroscopy in CDCl3. The results revealed a signal at 6.72 p.p.m. corresponding to the (NH) functional group in the heterocyclic ring. The aromatic protons appeared as multiplet peaks between δ 7.35 and δ 8.35 p.p.m., while the aliphatic (CH) group was detected at δ 5.40 p.p.m. The 13C NMR spectrum of compound D showed peaks for the C=O of the diketone at δ 164.70 and δ 154.50 p.p.m., along with peaks at δ 135.66 p.p.m. and δ 80.67 p.p.m. related to the C–Cl and C–Br, respectively, in the chemical structure of compound D. In addition, the aromatic carbon atoms appeared as multiplet peaks between δ 122 and δ 140 p.p.m.

Synthesis of compound D.
After we produce our target compounds, we turn our attention to demonstrate mechanistic details of this reaction, based on our experience with the field of heterocyclic compounds.21,22 We suggest that using concentrated sulfuric acid as a catalyst is crucial for activating the carbonyl groups of the carboxylic acid in compound A. This activation leads to the conversion of all carboxyl groups to hydrazides through dehydration reactions. Subsequently, nucleophilic attack by methanol produces the corresponding ester, followed by nucleophilic attack by hydrazine to generate compound C. The carbonyl group of 6-bromo-4-chloroisobenzofuran-1,3-dione reacts with compound C to form an intermediate, which is considered the driving force in the synthesis of compound D (see Scheme 4).

Proposed mechanism for synthesis of D (2,2′,2ʺ-(bеnzеnе-1,3,5- triсаrbonyl)tris (6-bromo-4-chloroisobenzofuran-1,3-dione).
Experimental
Materials and methods
Without additional purification, the following solvents and compounds were bought from Merck: “2-aminophenol, isoniazid, cyanoguanidine, acetophenone, terephthalaldehyde, methanol, sodium hydroxide, tetrahydrofuran, and dioxane.” Silica gel was used for column chromatography, while Merck silica gel 60 (0.063–0.200 mm) was consumed for TLC on aluminum plates (20 × 20 cm2). The compounds were analyzed using 1H NMR (500 MHz) and 13C NMR (125 MHz) spectrometers (CDCl3, standard internal TMS), as well as FTIR (Shimadzu Prestige-21, KBr disks). Analysis of variance (ANOVA) was utilized to assess the results statistically, and Duncan’s multinomial test was used to compare the compounds’ arithmetic means at a significance threshold of p ⩽ 0.05.
Synthesis of compound B
A mixture of benzene-1,3,5-tricarboxylic acid (0.45 g, 0.002 mol) was combined with 2.5 mL of absolute methanol and 3 mL of concentrated sulfuric acid. The mixture was refluxed for 4 h. Subsequently, the solution was neutralized with 15% sodium bicarbonate. Afterward, the crude reaction mixture was washed three times with 10 mL each of ether and brine. The resulting ester layer was dried over MgSO4, filtered, and purified by column chromatography using a solvent mixture of n-hexane and EtOAc in a 40%/60% ratio. The ester exhibited a boiling point of 224°C –226°C (literature value: 225°C), appeared as a yellowish oil, and was obtained in 84.7% yield.
Synthesis of compound C
The ester compound B (3 mL, 0.012 mol) was mixed with hydrazine hydrate (1.2 mL, 0.035 mol) and stirred for 4 h. The crude mixture was then left to cool, and the resulting precipitate was recrystallized from ethanol. The melting point of the recrystallized product was found to be 174°C –175°C (literature value: 175°C), and the compound appeared as a colorless oil with a yield of 79.4%.
Synthesis of compound D
To a solution containing compound C (1.5 g, 0.006 mol) in 25 mL of acetic acid, phthalic anhydride (6-bromo-4-chloroisobenzofuran-1,3-dione; 4.63 g, 0.018 mol) was added. The reaction mixture was refluxed for 9 h, after consuming the starting material, the crude reaction mixture was cooled down using an ice bath. The resulting solid was then separated and recrystallized, yielding white crystals with a melting point of 118°C–120°C and 73.7% yield.
1 H NMR (400 MHz, CDCl3) δ 8.72 (s, 3H), 8.35–8.15 (m, 3H), 7.72–7.55 (m, 3H) p.p.m.; 13C NMR (101 MHz, CDCl3) δ 165.6, 160.7, 156.8, 153.6, 135.7, 132.4, 128.6, 122.5, 120, 117, 80.5, 40.5.
Biological activity
In plotting diagrams for dynamic systems, the “damping range” refers to the spectrum of damping ratios used to analyze and visualize how the system’s oscillations change over time. This range illustrates how varying levels of damping affect the system’s behavior. As an example, take a system with damping ratios less than unity. In this case, the system oscillates but its motion gradually decreases. One unity is the critical value of damping ratio, no oscillations are observed in such a case. On the contrary when a system is over damped greater than one, it comes back to equilibrium without oscillation but more slowly compared to what happens in case of critical damping. Such diagrams provide insight into how changes in damping affect stability and response characteristics for various values. 20
To evaluate the applicability of the target compound, we have studied biological activity of D compound using several bacteria types such as gram-negative Escherichia coli and gram-positive Staphylococcus aureus. 16 The study used Mueller–Hinton Agar (MHA) as a culture medium to determine how reactive samples were in terms of defining their minimum inhibitory concentrations (MICs). Dimethyl sulfoxide (DMSO) was primarily employed as a solvent for preparing sample solutions at 100 µg mL–1 concentration for the purpose of conducting biological experiments.
In distributing MHA media, the spotter prescribed a metric technique for dish wells, where 10 µL of each already prepared solution was slowly added.23,24 The samples were then placed at 37°C for 24 h and later examined. The inhibition zone of compound D around the wells was measured in comparison to ampicillin and streptomycin as well as DMSO. An increase in the diameter of the inhibition of compound D indicated higher biological activity of the tested sample as seen from Table 1. These results demonstrate that compound D exhibited high biological effect, with the most pronounced result observed at a concentration of 100 µg mL–1.
Antibacterial activity of compound D and the control antibiotics.

The biological activity of compound D may be attributed to its dihydrooxazepine structure, which inhibits with metabolic processes in the cytoplasm of bacterial cells. The most promising results from the biological studies indicated that compound D exhibited a larger inhibition zone against bacteria compared to the tested antibiotic references, suggesting its potential as an effective antibacterial agent (see Figure 1).
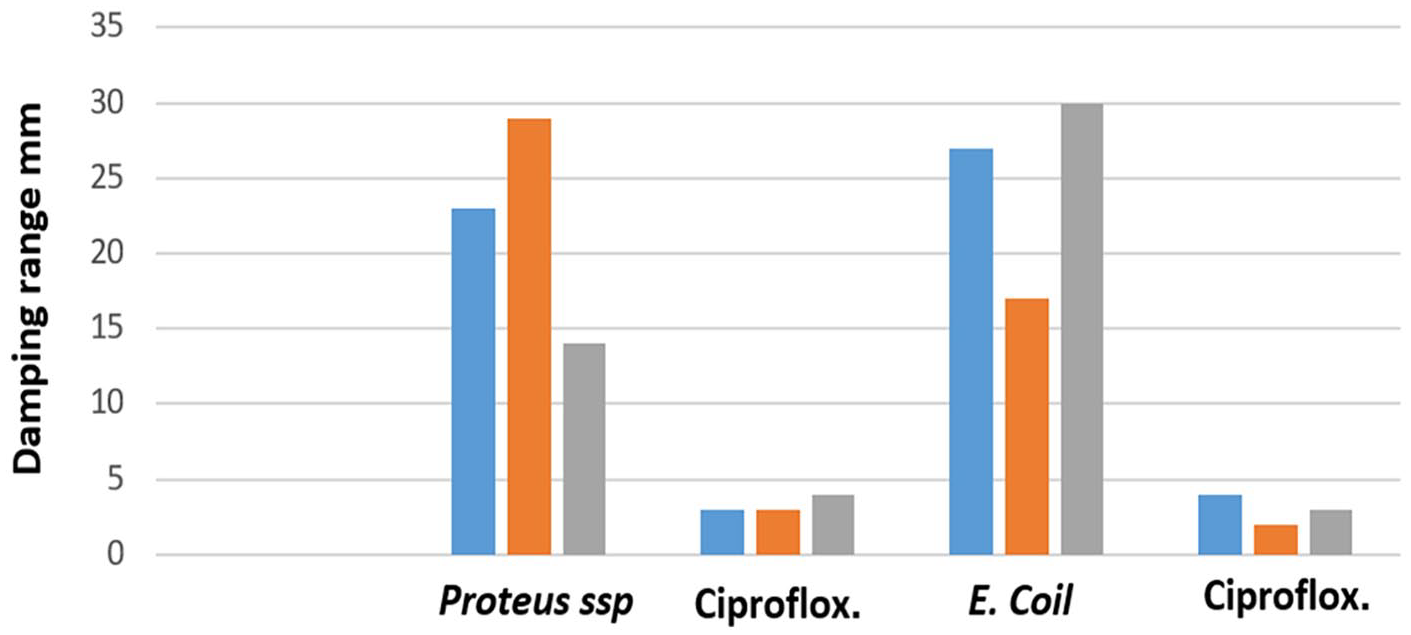
Evaluation of the inhibitory capability of compound D and the tested bacteria.
Molecular docking
To predict the target enzymes for the compounds under investigation and evaluate potential interactions, a molecular docking study was conducted for compound D. 25 Considering that compound D’s structure is chemically related to numerous drugs and pharmaceutical agents containing heterocyclic rings, it suggests potential similarities in their pharmacological properties or mechanisms of action. 26 In addition, amino acid residues that were discovered to be possibly important in ligand binding are the subject of docking research, such as MET283, which is an experimental compound with potential applications in medicinal chemistry. 27 Preliminary research indicates that MET283 may target specific biological pathways associated with various diseases. 28 Its mechanism of action involves modulating key enzymes or receptors, which makes it a promising candidate for further therapeutic research. These studies are focused on assessing its efficacy, safety profile, and potential side effects. 17 The π-sulfur bond of MET283 and the π-alkyl bond of LYS181, which connect the amino acid residues, are located in the active site where they interact with electronic pairs (see Figure 2).

Docked conformation of compound D with amino acids MET283 and LYS181.
The study proved that compound D cooperates with amino acid residues in the active site via establishing three bonds. Precisely, three hydrogen bonds were observed, linking the amino acid residues ARG98, LYS2, and GLN178 located in the active site, which bonded with the unshared pairs of electronics of the oxygen atoms in the carbonyl groups of compound D (see Table 2 and Figure 3).
Values of binding energies for the compound D.

RMSD: root mean square deviation.
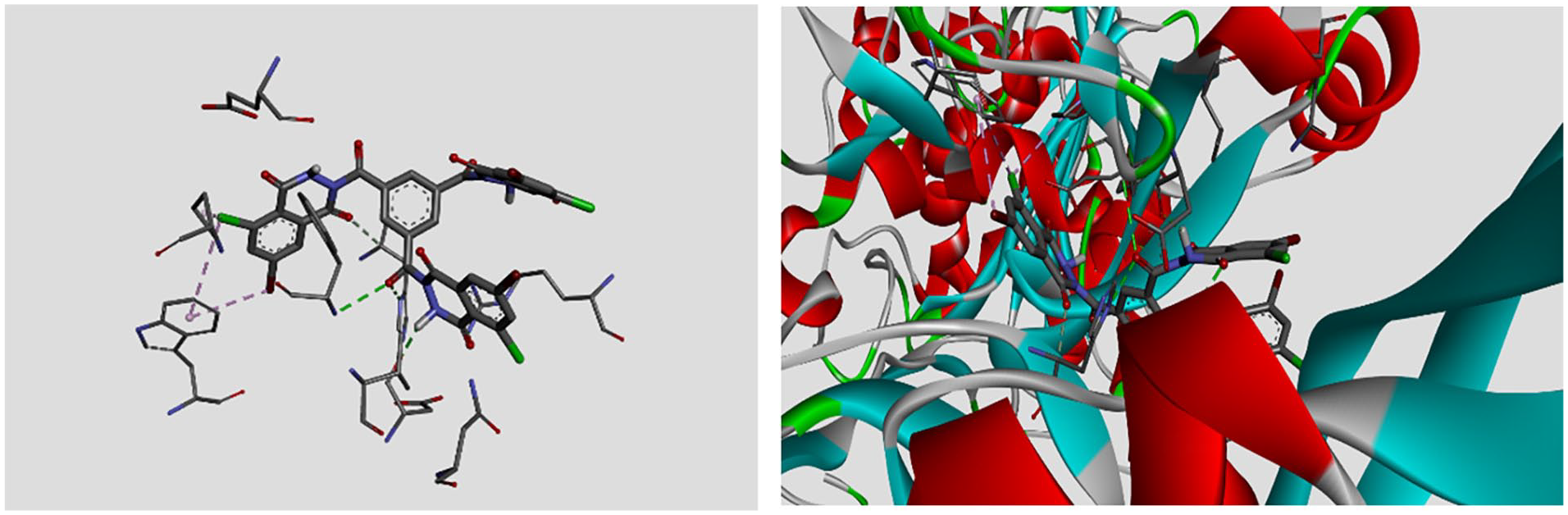
Docked conformation of compound D with amino acids residues HIS311, PHE211, and ARG110.
Notably, four hydrogen bonds were recognized: three linked the amino acid residues HIS311, PHE211, and ARG110, all located in the active site, with the electronic pairs of oxygen atoms from the carbonyl groups in compound D. In addition, the docking results indicated potential chemical interactions between the amino acid ASP209 and a hydrogen atom from the amine group in the hexagonal ring of compound D, as well as with the π-alkyl bond of TRP205, which is also present in the active site (see Figure 4 and Table 3).

The interaction between the compounds and the bacteria (a) Escherichia coli and (b) Staphylococcus aureus.
Values of binding energies for compound D.

RMSD: root mean square deviation.
Conclusion
A valuable heterocyclic compound was prepared successfully to form the target compound D. The compound was cleaned out, and their structures were determined using spectroscopic techniques such as FTIR and NMR. Their effects on two species of bacteria (gram-negative and gram-positive), Escherichia coli and Staphylococcus aureus, were investigated and described (see Figure 1). The bio-reactivity of the synthesized compounds was compared with common antibiotics such as ampicillin and streptomycin. Our new compound has shown clear inhibition against different types of bacteria, with some exhibiting higher biological activity than selected drugs. These promising results have prompted us to do more research on these substances. Interestingly, our molecular docking studies revealed a wide range of connections related to “amino acid residues in the active site.” As a result, these findings may have inspired researchers in this sector and prompted more research into a range of multi-heterocyclic compounds.
Supplemental Material
sj-docx-1-chl-10.1177_17475198241306505 – Supplemental material for Synthesis, characterization, and biological activities investigation of tri-armed structure of (7-bromo-5-chloro- 2,3-dihydrophthalazine-1,4-dione) based on benzene ring
Supplemental material, sj-docx-1-chl-10.1177_17475198241306505 for Synthesis, characterization, and biological activities investigation of tri-armed structure of (7-bromo-5-chloro- 2,3-dihydrophthalazine-1,4-dione) based on benzene ring by Marwan M Farhan, Mohanad A Hussein and Mohammed Hadi Ali Al-Jumaili in Journal of Chemical Research
Footnotes
Author contributions
M.M.F. and M.A.H. contributed to the conceptualization and writing of the original draft, while M.H.A.A.-J. reviewed, edited, and approved the final version. The authors have read and agreed to the published version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support this study are accompanying as supplementary material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
