Abstract
Water is directly or indirectly used in the industry for various processes. As a result, it is contaminated with different kinds of impurities. For the healthy life of all creatures on the earth, water should be free from all types of compounds. Various decontamination methods have so far been reported, but the adsorption process has emerged as the best method for the purification of water. An adsorbent named GG-g-P(AA)/grapheme oxide was prepared by free-radical initiator ammonium persulfate. The prepared adsorbent was utilized for the combined adsorption of metomorfin hydrochloride and sodium decofane drugs. Both drugs were efficiently removed from water by GG-g-P(AA)/GO.

Introduction
Water pollution has become a global problem today. Different kind of pollutants are found in water and water bodies. Water pollution is caused by industrialization, acid rain, discharging of industrial effluents and sewage without pretreatment, surface runoff from agricultural land (containing pesticides, fertilizers, and manures), global warming, deforestation, weathering of rocks, deposition by wind, and many others.1 –3 Water pollution leads to many life threatening diseases and environmental risk,4,5 hence nullifying harmful impurities from the water is so essential. Various water purification techniques, including conventional (boiling, distillation, and filtration) and advanced (chlorination, ultraviolet (UV) treatment, and reverse osmosis, etc.) were commenced for the removal of pollutants from water.6 –8 The adsorption process for water purification emerged to be a significant method due to its easy operation, high efficiency, and ability to remove a wide range of contaminants, but simultaneously high operational cost, regeneration, stability and degradability, and disposal of adsorbent are the main drawbacks of this method.9 –12 Hence finding a cheaper, efficient, and eco-friendly adsorbent is in continuous demand. Bio-based hydrogels as an adsorbent was reported for the adsorption of a wide range of pollutants owing to its high adsorption capacity.13,14 The high adsorption capacity of bio hydrogel is attributed to its three-dimensional structure which bears more water; various functionalities present in the bio hydrogel also contribute to interaction with various kinds of pollutants. Apart from this, biocompatibility, biodegradability, and easy availability of material are the main advantages of the bio-based adsorption method.15,16
Metomorfin hydrochloride (MF) and sodium decofane (DCF) are anti-diabetic and anti-inflammatory drugs that are detected in water and cause a negative effect on organisms and the environment.17 –21 Different kinds of adsorbents are used to remove MF and DCF individually,20 –24 but fewer works on the combined removal of MF and DCF 25 are reported. Hydrogel based on gum ghatti (GG) is used to remove a variety of pollutants.26 –31 In our work, GG-g-P(AA)/O-MWCNT, GG-g-P(NIPAM-co-AA)COFe2O4, and GG-g-P(NIPAM-co-AA)/GO for the removal of DCF and MF have been devised.25,32,33 In this context, GG-g-P(AA)/GO (GGAAGO-3) reported for the combined adsorption of MF and DCF.
Results and discussion
Fourier transform infrared spectroscopy analysis
The Fourier transform infrared spectroscopy (FTIR) spectrum of GG (Figure 1) indicates that the main functionality of GG such as -O-H stretching, -C-H stretching, asymmetrical carboxylate stretching, -C-O stretching(of alcoholic group), and pyranose ring (present in GG) corresponded at 3348.42 cm–1, 2926.01 cm–1, 1608.63 cm–1, 1016.48 cm–1, and 646.15 cm–1, respectively.29,34 –37 FTIR spectra of GO directed that a peak at 3220.20 cm–1 corresponded to -O-H stretching, asymmetric and symmetric -CH2 stretching observed at 2980.02 and 2850.50 cm–1, respectively, a peak at 1710.85 cm–1 and 1614.41cm–1 connected to -COOH and -C=C groups, and a peak appeared at 1381.03 cm–1 for the epoxide (-C-O-C).38 –40 The FTIR spectra of GG-g-P(AA) (GGAAGO-0) revealed the presence of -COOH (1699.28 cm–1), -O-H (out of bending-1016.48 cm–1), scissors and bending vibrations of -CH2 and -CHCO (1450.46 and 1400.32 cm–1).41–44 The band between 1600 and 1650 cm–1 has not been seen, which indicated polymerization of AA. Some peaks of GO found in the GG-g-P(AA)/GO (GGAAGO-3) supported the loading of GO on the hydrogel.

The FTIR spectra of GG, GO, GGAAGO-0, and GGAAGO-3.
X-ray diffraction analysis
Figure 2 presents the X-ray diffraction (XRD) spectra of GG, GG-g-P(AA) (GGAAGO-0), and GG-g-P(AA)/GO (GGAAGO-3). The characteristic peak of GG is observed at 18.84°; polymerization of AA caused to shift this characteristic peak from 18.84° to 19.48° and loading of GO also enhanced the characteristic peak of GG to 20.34°. It indicated about polymerization of AA and loading of GO to the GG.33,45 XRD analysis provide solid evidence of polymerization of AA and loading of GO on the GG rather than FTIR analysis.

XRD of GG, GGAAGO-0, and GGAAGO-3.
Adsorption activity
The adsorption activity of GG-g-P(AA)/GO (GGAAGO-3) is displayed in Figure 3 and Table S2–S6. GGAAGO-3 is preferred for the adsorption activity as it possesses high swelling capacity (Table S1). The effect of various amounts of GGAAGO-3 showed that as the amount of GGAAGO-3 increased from 10 to 50 mg, it led to enhance the removal efficiency of MF and DCF. The highest removal efficiency was found to be 64.94% and 75.92% for MF and DCF, respectively (Table S2). The removal efficiency of MF and DCF decreased as pH increased from 2 to 12 and the same trend happened in the case of the changing concentration of MF and DCF (Table S3 &S4). At pH 2, the highest removal of efficiency of MF and DCF was noted at 76.47% and 85.70%, respectively. At 25 mg/L (ppm), the highest removal of efficiency of MF and DCF was obtained at 61.50% and 90.02% for MF and DCF, respectively. The influence of time on removal efficiency showed that as time increased from 2 to 10 h, it led to an enhanced removal efficiency of MF and DCF, after which the removal efficiency became steady (Table S5). The removal efficiency of MF and DCF declined as temperature increased from 20°C to 60°C. The removal efficiency of DCF was found to be higher than MF. This is due to the difference in the structure of MF and DCF. MF drug is acidic in nature, while DCF drug is basic in nature. MF is less reactive than DCF with functionalities (hydroxyl, carboxylic acid, etc.) of GGAAGO-3 while DCF is more reactive than MF with various functionalities of GGAAGO-3. Under acidic pH, the surface of GGAAGO-3 could bear a more positive charge on its surface, which repealed MF drug and attracted DCF drug, while basic pH could generate a negative charge on GGAAGO-3 that can be repeal DCF and MF.

Adsorption activity of GGAAGO-3.
Adsorption isotherm and kinetic study
Adsorption isotherms (Figures 4 and 5) determined the nature of interaction of MF and DCF on the surface of GGAAGO-3. Langmuir, Freundlich, Temkin and Dubinin–Kaganer–Radushkevich (DKR) isotherm models were appraised for the examination of this purpose. Various parameters were evaluated by these four adsorption isotherm models, as given in Table 1. It showed that adsorption of MF and DCF drugs on the surface of GGAAGO-3 followed by Langmuir adsorption isotherm as the value of R2 is 0.99. Following the Langmuir adsorption isotherm revealed that adsorption of MF and DCF drugs on the surface of GGAAGO-3 is monolayer and homogeneous in nature. 46 The kinetic study deals with the rate of adsorption. For this, four kinetic models such as Pseudo-1st Order, Pseudo-2nd Order, Elovich, and intraparticle diffusion were examined (Figures 6 and 7). The various parameters from these four kinetic models were calculated and are depicted in Table 2. The adsorption of MF and DCF drugs on the surface of GGAAGO-3 lay with the Pseudo-2nd Order kinetic model. It indicated about chemisorption of MF and DCF drugs on the surface of GGAAGO-3 as it also supported by values of 1/n (relates to adsorption intensity or surface heterogeneity) calculated by the Langmuir adsorption isotherm.33,47,48 Thermodynamic parameters (Table 3) revealed that ΔG°, ΔH°, and ΔS° were calculated at temperatures of 20, 30, 40, 50, and 60°C, respectively. The increase in temperature led to an enhancement in the values of ΔG°, ΔH°, and ΔS°. This indicates that as the temperature increases, the adsorption process becomes unfavorable, non-spontaneous, and endothermic in nature. At room temperature (20°C), the adsorption process is favorable, spontaneous, and exothermic compared with higher temperatures (30, 40, 50, and 60°C).
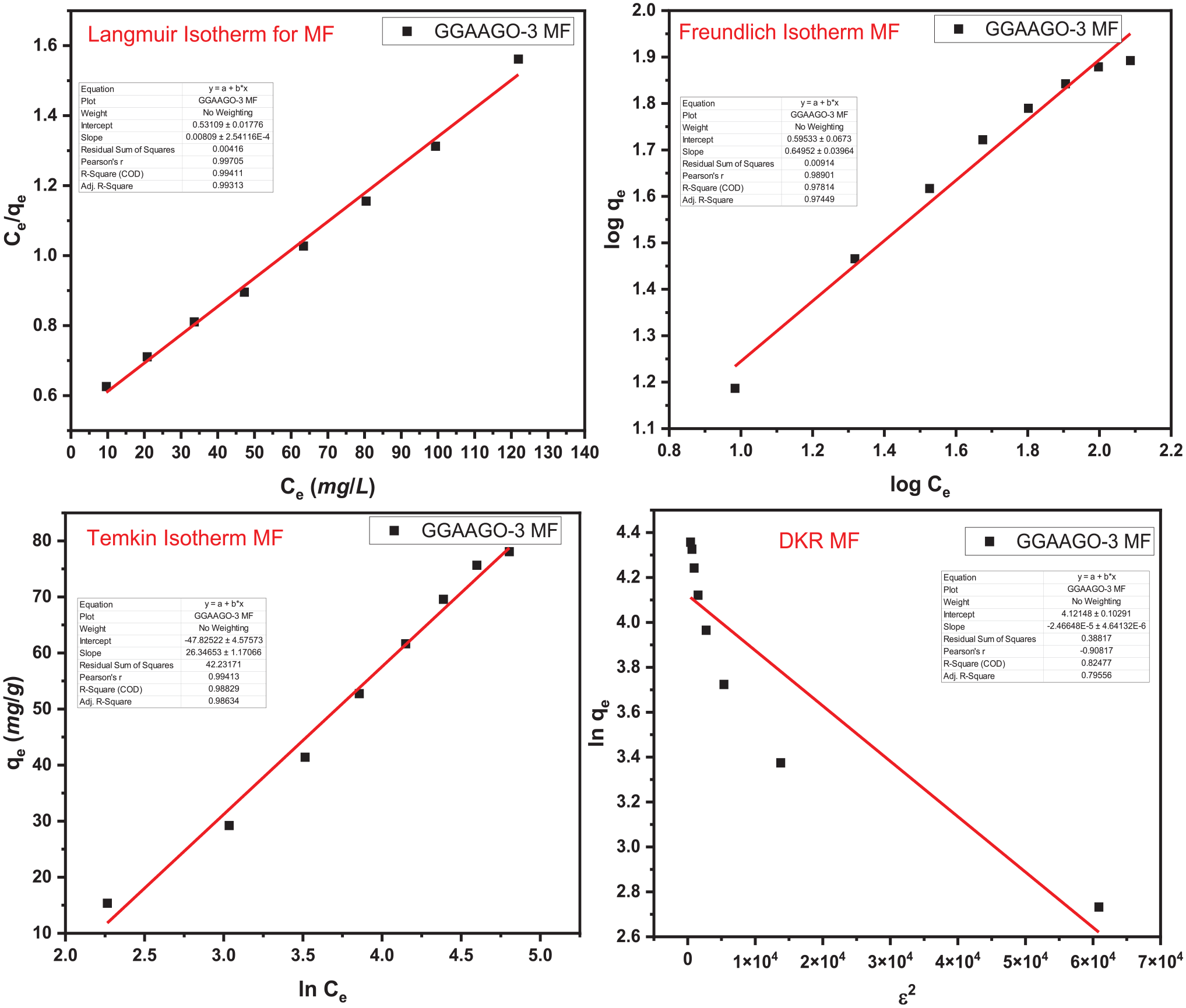
Adsorption isotherm of metomorfin hydrochloride (MF).
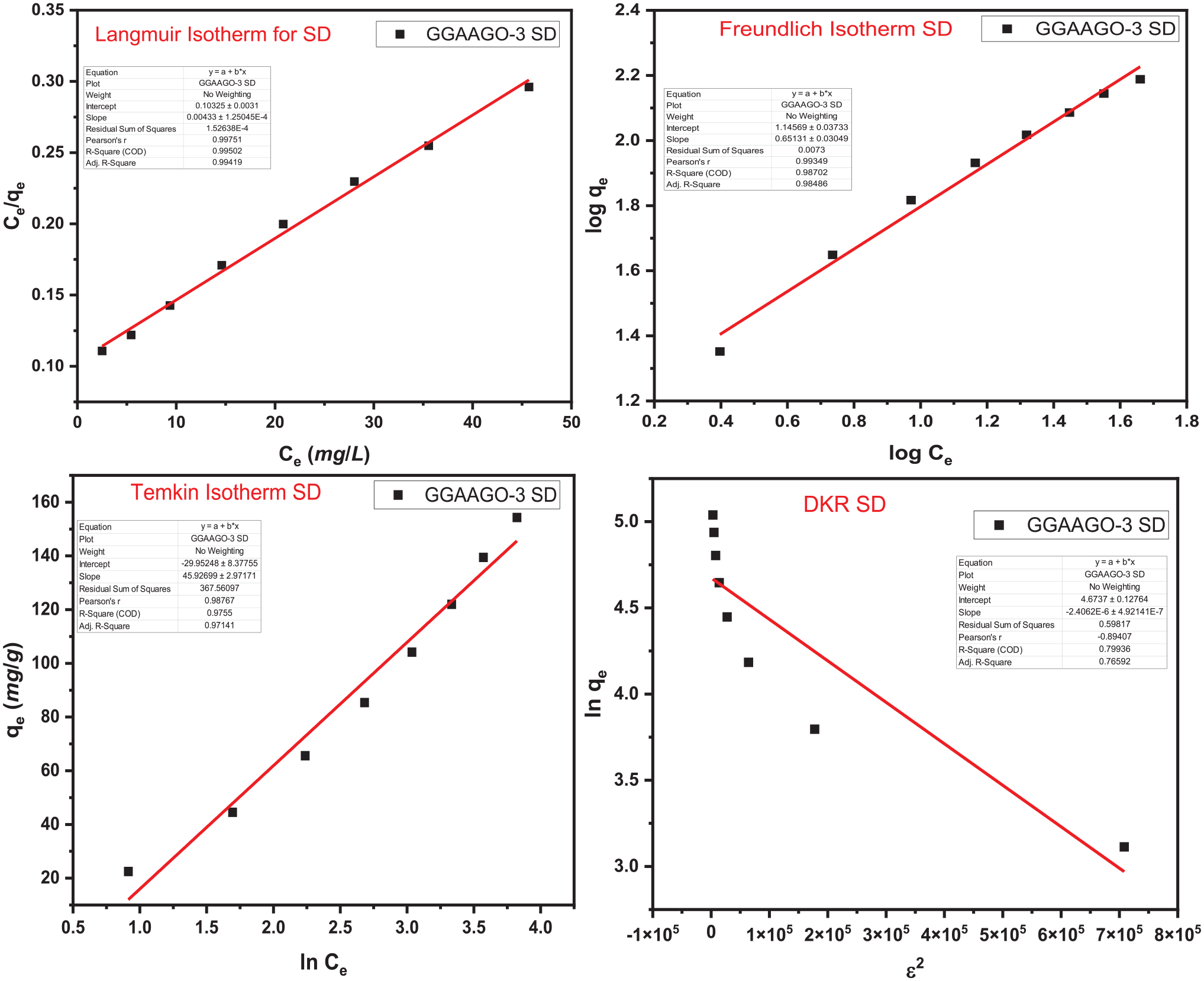
Adsorption isotherm of sodium decofane (DCF).
Equilibrium constants and parameters from the adsorption process of metomorfin hydrochloride (MF) and sodium decofane (DCF).


Kinetic models of metomorfin hydrochloride (MF).
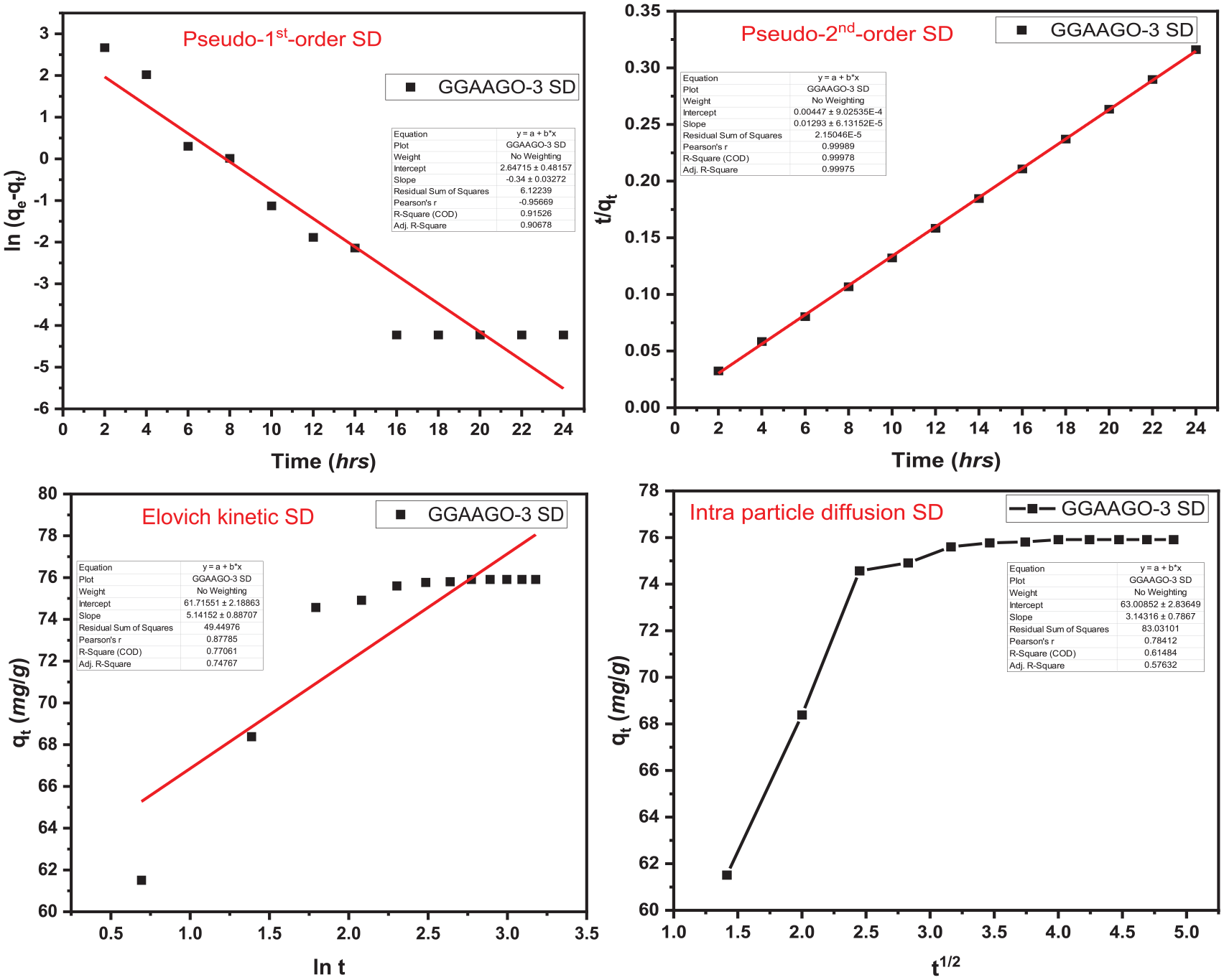
Kinetic models of sodium decofane (DCF).
Kinetic constants and parameters determined for the adsorption process of metomorfin hydrochloride (MF) and sodium decofane (DCF) drugs.

Thermodynamic constants and parameters for the adsorption process of metomorfin hydrochloride (MF) and sodium decofane (DCF) drug using GGAAGO-3 hydrogel.

Comparison of work
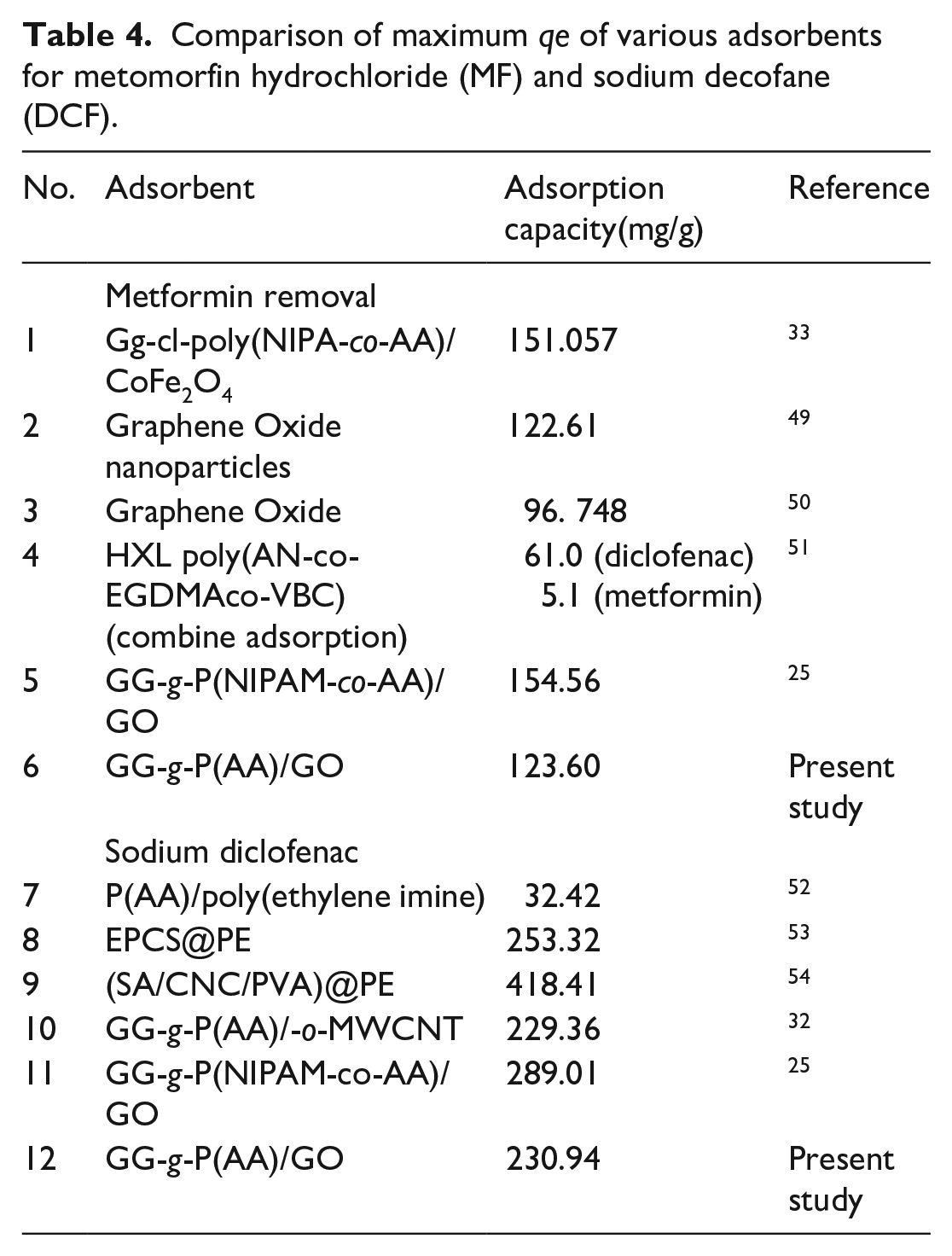
The comparison of maximum adsorption capacity (qe) of various adsorbents with GG-g-P(AA)/GO (GGAAGO-3) is presented in Table 4. It shows that undertaking adsorbent GGAAGO-3 exhibits the highest adsorption capacity (qe) for MF than GO, HXL poly(AN-co-EGDMAco-VBC), while it shows a lower adsorption capacity than Gg-cl-poly(NIPA-co-AA)/CoFe2O4 and GG-g-P(NIPAM-co-AA)/GO. In the case of DCF, with GG-g-P(AA)/GO (GGAAGO-3) performs a lower adsorption capacity than P(AA)/poly(ethylene imine) and GG-g-P(AA)/-o-MWCNT, while other adsorbents display a higher adsorption capacity than GG-g-P(AA)/GO.
Comparison of maximum qe of various adsorbents for metomorfin hydrochloride (MF) and sodium decofane (DCF).
Experimental
Materials
Required chemicals such as GG, acrylic acid (AA), ammonium persulfate (APS), tetramethyl ethylenediamine (TMED) and N, N′- methylenebisacrylamide (MBA) were sourced from Loba Chemie (Mumbai, India). GO was acquired from Sigma Aldrich (Munich, Germany). All the materials used in the present study were of reagent grade. MF was purchased from Himedia and DCF was supplied by Sigma Aldrich; triple distilled water was employed throughout the experiment.
Preparation of GG-g-P(AA)/GO
The preparation of GG-g-P(AA)/GO was done as per reported procedure (Figure 8).55 –57 The procedure followed the addition of GO (1–5 mg) into dissolved 0.5 g of GG in 10 mL deionized (DI) water in a 100 mL beaker. The combined solution was subjected to sonication at room temperature for 40 min for better dispersion of GO and agitated whole solution using a mechanical stirrer (250 rpm). Cross-linker 50 mg MBA was added to this solution followed by addition of 1 mL of AA and 10 mL of DI. The combined mixture was heated at 60°C for 20 min. Subsequently, 50 mg of APS (free radical initiator) and 0.05 mL of TEMED (accelerator) were supplied. After 25 min, gel formation was observed. The reaction was then continued for 3 h in a hot air oven for completion of the polymerization process. The non-reactive materials from the hydrogel were eliminated by repeated washing with DI water. The hydrogel was then allowed to dry at 50°C for 48 h in a hot oven. The preparation of material is presented in Figure 8 and composition of the material is depicted in Table 5.

Schematic presentation of material preparation.
Composition of materials.

Characterization
FTIR and XRD were used to analyze the materials. FTIR spectrum was recorded on PerkinElmer IR spectrometer in the range of 400–4000 cm–1 using KBR plates, while XRD of the materials was noted on the Rigaku MiniFlex 600 diffractometer (CuKa radiation (wavelength l1) λ = 0.15418 nm).
Stock solution preparation
1000 mg of DCF and MF were dissolved in 1000 mL triple distilled water for the preparation of 1000 ppm (mg/L) of MF and DCF solution. For the homogeneous dissolution of both drugs, ultrasonication and stirring on magnetic stirrer were done. The prepared solution was kept in a refrigerator 4°C and was used within 2 days. The stock solution was used to prepare the needed solution for adsorption.
Adsorption study
The combined adsorption of MF and DCF was conducted by batch method using GG-g-P(AA)/GO (GGAAGO-3).25,32 The adsorption study was carried out using 50 mL solution of both drugs in 250 mL standard sealed Erlenmeyer flasks. The maximum λmax of DCF and MF was noted at 276 and 232.5 nm, respectively, on a UV–Vis double beam spectrophotometer (TCC-240 A, Shimadzu). In the adsorption study, the effect of adsorbent (GGAAGO-3) amount (10-100 mg) was carried out. The adsorption experiment consisted of an addition of 10–100 mg of GGAAGO-3 into a 50 mL solution of both drugs at neutral pH, 100 ppm drug conc., vol 50 mL, temp. 20°C and at time 24 h. The reaction tube was put in the shaker and stirred at 250 rpm, room temp. (20°C) and contact time 24 h. The adsorption capacity of hydrogels was calculated by altering the concentration of both drugs before and after adsorption. The adsorption capacity and removal efficiency were calculated by equations (1) and (2), respectively


Here, C0 and Ce indicate the initial and equilibrium concentrations (mg/L) of DCF and MF, respectively; m shows the weight of the adsorbent and V is the volume of the solution (L). The concentration of DCF and MF was determined in mg/L. The effect of the other parameters of DCF and MF for such variations in pH (2–12) at dose 40 mg, 100 ppm drug conc., vol 50 mL, temperature 20°C(rt) and contact time 24 h, concentrations of 5–200 mg/L at neutral pH, dose 40 mg, vol 50 mL, temperature 20°C(rt) and contact time 24 h, times 2–24 h at neutral pH, dose 40 mg, 100 ppm drug conc., vol 50 mL and temperature 20°C(rt) and temperatures at 20–60°C at neutral pH, dose 40 mg, 100 ppm drug conc., vol 50 mL and contact time 24 h was done for the measurement of removal of combined capacity of both drugs. The pH of the solution was adjusted by adding 1 M HCl and NaOH solution.
Adsorption isotherm
For better understanding of the adsorption mechanism, studies of adsorption isotherm by various isotherm models are very essential. It provides intrinsic information about interaction of adsorbent on the surface of adsorbate. For this, four adsorption isotherm models such as Langmuir isotherm, Freundlich isotherm, Temkin isotherm, and DKR were evaluated.. The detailed equations of these four adsorption isotherms are shown below.
1. Langmuir Isotherm 58

Ce is the equilibrium concentration of DCF in the solution (mg/L), qe is the adsorption capacity at equilibrium (mg/g), qm is the maximum adsorption capacity (mg/g), and KL is the Langmuir constant (L/mg).
The value of qm and KL defined from the slope and intercept from the plot of Ce/qe against Ce, RL explains the favorability of the adsorption process and it is calculated from the following equation

2. Freundlich isotherm 58

Kf is the Freundlich constant (mg/g), and n is Freundlich exponent related to the adsorption intensity or surface heterogeneity (dimensionless).
The values of isotherm constants (Kf and n) are defined from the intercept and slope of the linear plot of log qe versus log Ce, respectively.
3. Temkin Isotherm 59

B is the heat of sorption constant, KT is the binding constant KT and B were determined from the slope and intercept of plot qe against lnCe.
4. DKR 58

β (mol 2 /kJ 2 ) is the constant related to the adsorption energy, qm (mg/g) is the theoretical saturation capacity, Ɛ (kJ/mol) is the Polanyi potential, qm and β were calculated from the intercept and slope of plot lnqe against Ɛ 2 , Ɛ and E (free energy of adsorption) calculated by the following equations


Kinetic study
Various kinetic models such as Pseudo-1st Order, Pseudo-2nd Order, Elovich and intraparticle diffusion were examined for the unveil rate of adsorption. The detailed equations of the four kinetic models are mentioned below.
1. Pseudo-1st Order and 2nd Order Models 58


qe (mg/g) is the adsorption capacity at equilibrium; qt (mg/g) is the adsorption capacity at time t; k1 (1/min) and k2 (g/(mg.min)) are the first-order and second-order rate constants, The rate constants (k1 and k2) were determined from the slope and intercept of the plots of log (qe − qt) against t and t/qt against t, respectively.
2. Elovich model 60

where qt represents the adsorption capacity at time t, and β and α are the Elovich constants equivalent to the extent of surface coverage and rate of adsorption at zero coverage, respectively.
3. Intraparticle diffusion model 58

where qt represents the adsorption capacity at time t, intercept CIP reflects the boundary layer thickness effect, and KIP indicates the rate constants of intraparticle diffusion.
Conclusion
Natural biopolymer known as gum ghatti, a dried exudate of Anogeissus latifolia was grafted by polyacrylic by using free-radical initiator APS and in situ supported by grapheme oxide (GO). The materials were characterized by XRD and FTIR. The material GG-g-P(AA)/GO (GGAAGO-3) was checked for the combined adsorption of MF and DCF drugs. The highest removal efficiency was obtained at 76.47% (adsorbent dose 40 mg, initial concentration 100 mg/L, contact time 24 h, 20°C, and pH 2) and 90.02% (adsorbent dose 40 mg, initial concentration 100 mg/L, contact time 24 h,20°C and neutral pH) for MF and DCF, respectively. The obtained results showed that DCF adsorbed more than MF by GGAAGO-3. The maximum adsorption capacity(qe) for MF and DCF was determined to be 123.60 and 230.94 mg/g, respectively.
Supplemental Material
sj-docx-1-chl-10.1177_17475198241301598 – Supplemental material for Adsorption of metomorfin hydrochloride and sodium decofane by grafting of polyacrylic acid/grapheme oxide on the gum ghatti
Supplemental material, sj-docx-1-chl-10.1177_17475198241301598 for Adsorption of metomorfin hydrochloride and sodium decofane by grafting of polyacrylic acid/grapheme oxide on the gum ghatti by Pragnesh N Dave, Lakha V Chopda and Bhagvan P Kamaliya in Journal of Chemical Research
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, Methodology, Formal analysis and investigation, data collection and analysis; Writing—original draft preparation was performed by S.B. The writing—reviewing and editing of the first draft; funding acquisition; resources and supervision of the manuscript were done by P.N.D. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The study data and materials are available with the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This research involved no human participants and/or animals.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
