Abstract
Preparing fluorocarbon surfactant for aqueous film-forming foam is a complex process. Overcoming steric hindrance when introducing imidazolium to the fluorocarbon chain is a key challenge. In this significant work, we proposed a simple one-step route to prepare imidazolium-contained fluorocarbon surfactant under mild conditions. The FT-IR, 1H NMR, 13C NMR, MS, and TG technologies characterized the imidazolium-contained fluorocarbon surfactant by analyzing functional groups, molecular weight, and thermal stability. An equimolar mixture of imidazolium-contained fluorocarbon surfactant and C6H13NaO3S demonstrated high surface activity, enabling the foam to spread effectively on commercial fuels. The spreading coefficient of foam on n-cyclohexane met the US Military Specification. Coordinatively, the foam drained in time to maintain the stability and completeness of the aqueous layer, with a moderate 25% drainage time of 317 s and a proper foam expansion ratio of 10.5. In extinguishing experiments, the imidazolium-contained fluorocarbon surfactant/C6H13NaO3 mixture extinguished test fires in 65 s, suggesting an essential potential in the aqueous film-forming foam formula.

Introduction
Based on extremely low surface tension, aqueous film-forming foam (AFFF) spreads and seals on the hydrocarbon fuels to suppress the fire.1,2 Developing surfactants with high surface activity is essential to improve the AFFF performance. 3 Fluorocarbon surfactants, known for their unique features due to the C-F bond, have garnered significant attention in AFFF formulations. 4 However, the long-chain fluorocarbon surfactants, perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) in particular, have been prohibited since their irreversible impacts on humans and animals.5,6 Weight loss, 7 gene induction, 8 hepatic damage, 9 and pulmonary lesions 10 are reported in cases using long-chain fluorocarbon surfactants. Shortening the hydrophobic chain is a convenient strategy for mitigating toxicity and durability.11,12 Some evidence proves that bioaccumulation and toxicity are significantly weakened when the fluorinated carbon atoms are no more than 6. When perfluorocarbon number is less than 4, bioaccumulation and short-term toxicity could be negligible.13,14 So, the design and development of short-chain fluorocarbon surfactants have become one of the research highlights.
Dobunt designed the Capstone series to replace long-chain fluorocarbon surfactants, and the classical Capstone 1157 with six fluorinated carbons shows impressive potential in AFFF. Yu et al. 15 composed an AFFF system upon Capstone 1157, which maintains a low surface tension of around 15.55 mN/m at CMC concentration. The hydrophilic head influences the surface activity of AFFF surfactants with the same tail structure. Zwitterionic Capstone 1157 has a lower surface tension than anionic DynaxTM DX1030, according to Hill et al. 16 He et al. 17 proposed that equimolar C4FI-C3F7COONa foam keeps considerable sealability, spreadability, and stability in extinguishing tests. Jia et al. 18 prepared a C4-based fluorocarbon surfactant (C4-Br) via two organic reactions, acylation and quaternization. The synergism effect of C4-Br/CH3(CH2)5NaO3S shows a surface tension of 16.58 mN/m to support smooth spreading to form the film-forming layer. Introducing ring structure provides a strategy to design branched short-chain fluorocarbon surfactants for AFFF, for instance, Yang et al. 19 designed three steps to prepare a surfactant with a carboxyl head and a C-4-based tail. Through ring-opening and sulfonation, Kang et al. 20 synthesized two novel fluorinated surfactants with acceptable surface tension ranging from 15.7 to 22.8 mN/m.
Particularly, an in-depth examination of the aforementioned surfactant structures reveals two key areas for improvement. First, all disclosed reactions are realized by at least two steps (Table 1), which involve complex conditions and unavoidable purifications. Developing a green one-pot process to prepare AFFF fluorocarbon surfactant is urgently needed. Second, the hydrophilic heads are common groups such as carboxyl, ammonium, and hydroxyl. Exploring a novel hydrophilic group of fluorocarbon surfactants to expand the AFFF family will be interesting. Hence, the imidazolium ring appears in our perspective for its attractive properties to provide a reasonable solution.
Structure properties of short-chain fluorocarbon surfactants for AFFF.

Identified as a crucial ionic liquid, the characteristic imidazolium ring contributes to many features: incombustible,21,22 nonvolatile, 23 biodegradable, 24 heat-resistant, 25 and excellently soluble. 26 Different reactions can be designed on imidazolium chemical sites, and even tetrasubstituted imidazolium compounds have been reported in recent research. 27 Numerous developed methods are discussed to prepare imidazolium hydrocarbon surfactants, demonstrating surface tensions above 25 mN/m.28–30 However, rare work has succeeded in synthesizing imidazolium fluorocarbon surfactant, 31 let alone a one-step method. It is indisputable that fluorocarbon surfactant containing imidazolium as a hydrophilic head will combine the merits of the C-F bond and stable imidazolium ring.
In this work, we first designed a one-step route to synthesize a novel imidazolium-head fluorocarbon surfactant (OS-TI) under mild conditions (Figure 1). FT-IR and NMR spectra identified OS-TI. MS spectrum measured the molecular mass, while TG analysis tested the thermal stability. Furthermore, we evaluated the surface activity, spreading ability, foam properties, and extinguishing performance of AFFF based on OS-TI.

Synthetic route of OS-TI.
Materials and methods
Reagents and materials
Synthesis of imidazolium-contained fluorocarbon surfactant (OS-TI)
In an 100-mL flask, 5.607 g (0.05 mol) of 1-(2-hydroxyethyl)imidazole was dissolved in 15 mL of
Structural characterization of OS-TI
We characterized the OS-TI structure by Fourier transform infrared spectroscopy (FT-IR) and nuclear magnetic resonance (NMR). A Nicolet 6700 spectrometer scanned and generated the FT-IR spectrum from 400 to 4000 cm.-1 A Bruker AVANCE III HD 400 NMR spectrometer characterized the H and F atoms, with tetramethylsilane (TMS) as the internal standard for obtaining 1H NMR and 13C NMR spectra. Molecular mass measurement was conducted on a Thermo Fisher UltiMate 3000 RS mass spectrometer. Finally, the thermal stability was tested by a Discovery 5500 thermal analyzer, and the thermogravimetry curve was described from 40 °C to 600 °C at a heating rate of 10 °C/min.
Performance tests of OS-TI for AFFF
The performance evaluation for OS-TI contained surface (interface) tension, spreading coefficient, foam performance, and extinguishing ability. Surface (interface) tension at 25 ± 0.5 °C was measured using the Lecomte du Noüy P method. 32 The average value for data discussion was obtained from three measurements. The spreading coefficient (S) could be calculated from the following equation 18 :

where γo represents the surface tension of the hydrocarbon fuel, γw represents the surface tension of the surfactant solution, and γ(o/w) represents the interface tension between hydrocarbon oil and surfactant solution.
We used a laboratory approach to simulate the foaming and drainage process. 33 10 mL of surfactant solution was sealed in a 100-mL volumetric cylinder. After shaking the cylinder for 1 min, record the expansion ratio through the ratio of initial foaming volume to prefoaming 10 mL solution. Correspondingly, the time from forming foam to drainage of 2.5 mL water was noted as 25% drainage time. The data for the graph were the average value from three measurements.
According to the requirements of the Chinese National Standard (GB 15308-2006), 34 the extinguishing time and the burnback time were determined. All the fire tests were executed inside a large burn room, utilizing a 0.25 m2 circular pan. Preburn of 9.0 ± 0.1 L of hydrocarbon oil in the pan lasted for 60 ± 5 s before extinguishing, and then the foam was sprayed at a rate of 750 ± 25 g/min for 180 ± 2 s. From the spraying, the extinguishing time was recorded when the fire was suppressed. After cooling the oil pan for 300 ± 10 s, a burning tank loading 1 L of fuel reignited the extinct oil pan. The time from the reignition to the complete reburning was recorded as burnback time.
Besides, we used the Ross-Miles method to estimate the foam stability. Fifty milliliters of surfactant solution was added and washed to measuring cylinder with water-bath circulation under a certain temperature for 5 min, and 200 mL of solution at the same temperature was flowed down from 900 mm height to collide with the bottom solution. The initial foam height and foam height at 5 min were recorded as h0 and h5, respectively. The ratio of h0 to h5 was noted as R5, meaning the foam stability.
Results and discussion
FT-IT analysis of OS-TI
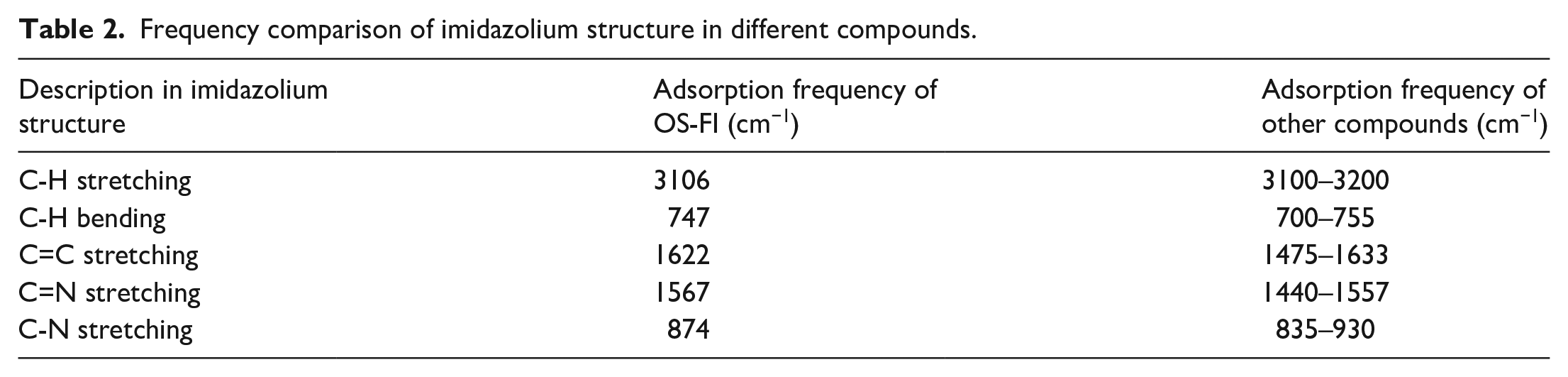
Figure 2 elucidates the hydrophilic head and hydrophobic tail of OS-TI. The characteristic C-F bond was identified at 1231 cm−1 (asymmetric stretching vibration) and 1168 cm−1 (stretching vibration). Various bond adsorptions confirmed the imidazolium ring. The C-H in imidazolium (N=C-H) was assigned to the peak at 3106 cm−1, while the C-H plane bending was detected at 747 cm−1. The stretching vibrations of C=C and C=N in imidazolium were observable at 1622 and 1567 cm−1, respectively. The band at 874 cm−1 was due to C-N stretching vibration. The various absorptions of imidazolium structure were widely discussed, and we also compared our data with other characterizations in Table 2.35,36 Expect for C=N stretching, most similar peaks indicated demonstrated hydrophilic imidazolium head in OS-TI. For the other groups, the terminal hydroxyl of OS-TI was found at 3392 (O-H stretching vibration), 1404 (O-H in-plane bending vibration), 665 (O-H out-of-plane bending vibration), and 1069 cm−1 (C-O stretching vibration). The C-H bond of methylene was also confirmed at 2950 and 2880 cm−1.

FT-IT spectrum of OS-TI.
Frequency comparison of imidazolium structure in different compounds.
NMR analysis of OS-TI
As shown in Figure 3, we used D2O (4.82 ppm) as a solvent to describe the 1H NMR spectrum of OS-TI. It was not hard to identify that the chemical shifts from 7.00 to 9.00 ppm were ascribed to H atoms in the imidazolium ring. Chen and Izgorodina 37 revealed that the substituent groups to N1 and N3 influenced the 1H NMR spectrum of H2, H4, and H5, which mostly oscillated between 6.00 and 10.00 ppm. The signals at 2.54 and 4.63 ppm corresponded to the methylene groups linking the imidazolium ring and perfluorinated carbons. The H atoms of methylene, between hydroxyl and imidazolium ring, were found at 3.72 and 4.28 ppm. The H atom of terminal hydroxyl was detected coincidentally around 4.64 ppm. Figure 4 (13C NMR) shows that the chemical shifts above 100 ppm originated from the imidazolium head and perfluorinated tail. The higher signals around 136.79, 123.38, and 122.85 ppm were due to three C atoms of imidazolium ring, and C atoms in the perfluorinated tail were distinguished at 121.71, 118.13, 116.35, and 110.14 ppm. The chemical shifts at 60.33 and 52.29 ppm were attributed to methylene C atoms between the hydroxyl and the imidazolium ring. Besides, the methylene C atoms that linked the imidazolium ring and perfluorinated tail were observable at 39.52 and 30.49 ppm.
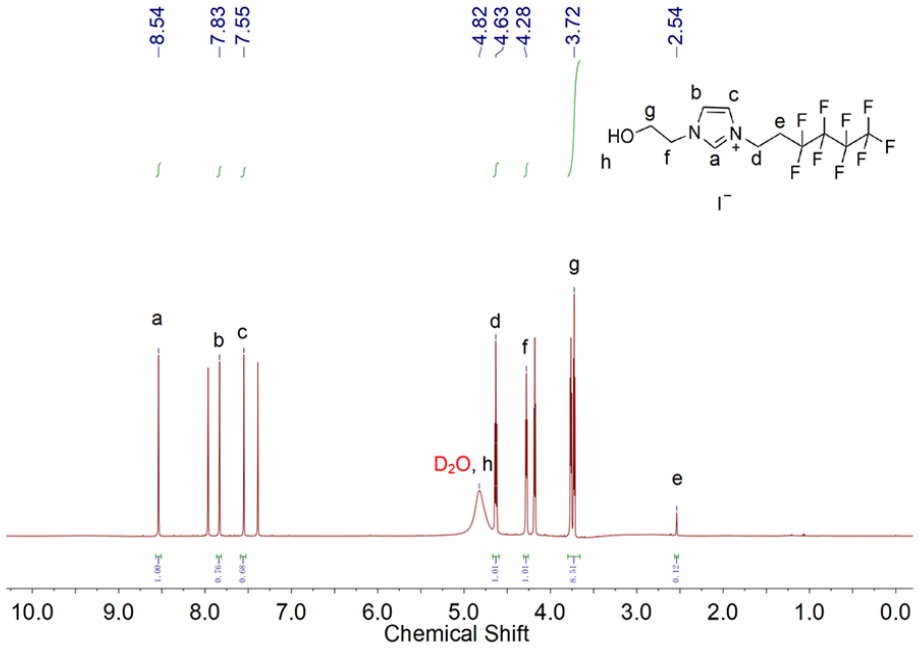
1H NMR spectrum of OS-TI.

13C NMR spectrum of OS-TI.
MS and TG analysis of OS-TI
As shown in Figure 5, two m/z signals appeared in the MS spectrum. The strongest signal at 359.08 was ascribed to the main cationic part of OS-TI, [C11H12F9N2O]+. We also found the structure fragment peak at 133.07, which implied an imidazolium [C5H8N2O]+ part. 1,1,1,2,2,3,3,4,4-Nonafluoro-6-iodohexane was not detected with considerable relative abundance, indicating the reactant was sufficiently consumed. The MS result ulteriorly completed the structural characterization based on FT-IR and NMR, suggesting that the synthetic scheme was realized as expected.
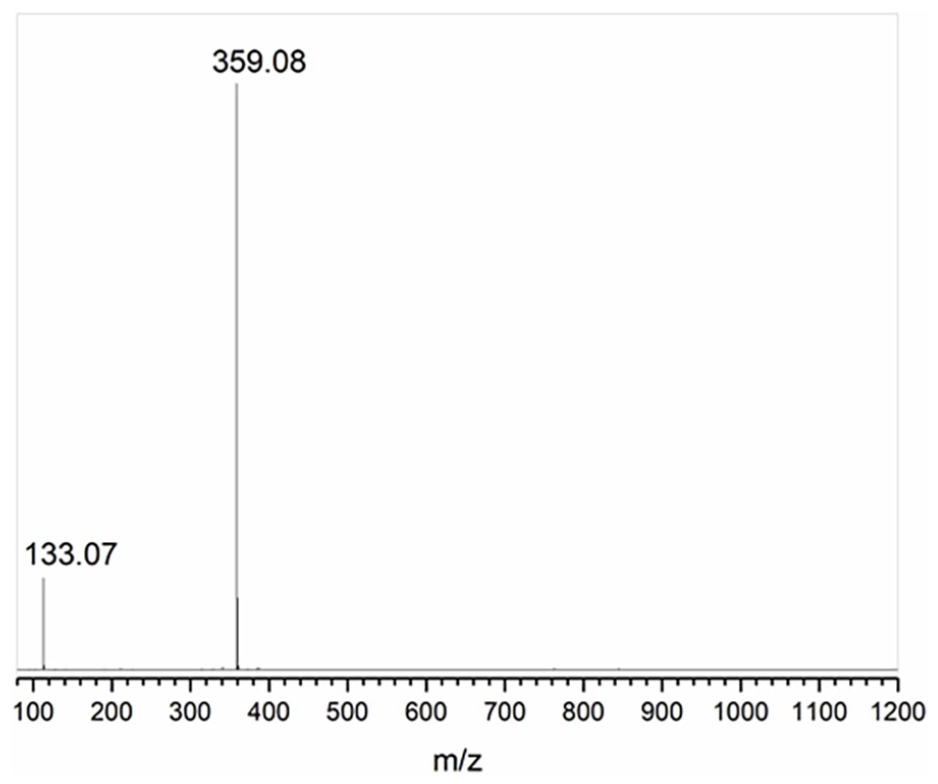
MS spectrum of OS-TI.
TG curve describes the heat resistance of OS-TI from 30 °C to 600 °C (Figure 6). Due to the excellent hygroscopicity of imidazolium ionic liquids, OS-TI showed the first weight loss in the stage before 130 °C.38,39 The second weight loss from 170 °C to 300 °C suggested a decomposition of connective methylene structure between the imidazolium and fluorocarbon chain. Most notably, OS-TI lost about 60% of its weight in a narrow interval (300–350 °C), demonstrating a signature thermal destruction of imidazolium. 40 Endothermic thermal decomposition happened by the anionic stable order PF6>Beti>Im≈BF4>Me≈AsF6>I-, Br-, Cl-. The heat resistance of OS-TI may be further improved by displacing the anion I-. The 350–550 °C weight loss stage hinted that OS-TI decomposed totally.

TGA curve of OS-TI.
Surface tension analysis
For fluorocarbon surfactant in the AFFF formula, the high surface activity is vital to spread on the hydrocarbon fuels to form the aqueous film. Given the statistical values for the surface tension of hydrocarbon fuels (20–25 mN/m) and interfacial tension between hydrocarbons and fluorocarbon surfactants (2–5 mN/m),41–43 it is necessary to design the surface tension below 20 mN/m at least for OS-TI to obtain the positive spreading coefficients. 2 Simultaneously, a low CMC is required to establish a green and economical AFFF formula. 44 Surfactants exert maximum advantage in decreasing surface tension at CMC concentration, which influences the AFFF cost in the application. CMC was confirmed by the intersection value of the two fitted straight lines before and after the CMC point.
Introducing an anionic hydrocarbon surfactant is a synergistic strategy to improve the efficiency of fluorocarbon surfactant. The strong attraction between positive and negative ions induces tighter micelle formation in a lower concentration, and the compactly arranged surfactant molecules contribute to lower surface energy. 45 In a nutshell, less fluorocarbon surfactant achieves the AFFF standard with a less environmental hazard. The mixture of hydrocarbon surfactant/OS-TI must balance the performance and solubility. The comparable length of two hydrophobic tails is preferred to obtain excellent surface activity, while asymmetric hydrophobic C atoms produce better solubility to form a homogeneous system. 33 In our work, we considered C6H13NaO3S to mix with OS-TI. Figure 7 shows the surface tension of C6H13NaO3S/OS-TI under different molar ratios. OS-TI alone decreased surface tension from 72.3 (pure water) to 27.4 mN/m (CMC concentration), with initial potential for AFFF application. C6H13NaO3S altered the CMC and surface tension, requiring further optimization. Most notably, the equimolar mixture acquired the lowest surface tension of 17.4 mN/m with the lowest CMC concentration (Table 3). These data were comparable to other published mixtures (i.e. CMC = 9.8 mmol/L, γCMC = 17.2 mN/m reported by He et al. 17 ; CMC = 7.72 mmol/L, γCMC = 16.58 mN/m reported by Jia et al. 18 ; CMC = 9.91 mmol/L, γCMC = 22.39 mN/m reported by Yang et al. 19 ; CMC = 0.31 mmol/L, γCMC = 5.6 mN/m reported by Kang et al.). 20 Although the electrical interaction between C6H13NaO3S and OS-TI remarkably improved the surface activity, it is interesting that the precipitation did not appear in the equimolar mixture even under three times of CMC. The precipitation phenomenon happened frequently due to the strong interaction under high concentrations of the equimolar mixture. 45 However, in our case, the flocculent complex only formed in the 1:4 mixture under a high concentration of over 30 mmol/L. This result could be explained by the large-sized cationic imidazolium ring, namely the higher steric hindrance, lowered the attraction between OS-TI and C6H13NaO3S. The excellent solubility remained as a consequence. A similar strategy to design the imidazolium-head fluorocarbon surfactant pairs with good solubility was proposed by Verdia et al., 31 and their products show potential in stabilizing the water/CO2 interface.

The surface tensions of mixture solutions under different OS-TI/C6H13NaO3S ratios.
The CMC and γCMC of mixture solutions under different OS-TI/C6H13NaO3S ratios.

Spreading performance analysis
The low surface tension of the C6H13NaO3S/OS-TI mixture satisfied the initial requirement of AFFF. Note that the spreading ability dominates whether the foam can spread smoothly on the fuels to form a stable aqueous film. 46 At the microscale, the fluorocarbon surfactant molecules prefer to adsorb on the air–water interface, and the hydrocarbon surfactant molecules tend to adsorb on the water–oil interface. 17 The comprehensive effect of hydrocarbon/fluorocarbon surfactants affects the interface tension γo/w, which determines the spreading coefficient (S). The positive S signifies the spreading phenomenon in many cases. 47 However, the spreading from spraying to the equilibrium state is a dynamic process that needs time, leading the dynamic surface tension to decrease from the pure solvent value (very high, around 70 mN/m) to approach the equilibrium value. 48 Apparently, the application of AFFF from a nozzle on fuel is a thermodynamically unstable nonequilibrium phenomenon, and the nonequilibrium phenomenon explains that AFFF applications with positive S, but no spreading phenomena are observable. 49 Hence, the simple observable method can verify whether ultimate spreading or not. 17
The spreading coefficients of the 1:1 C6H13NaO3S/OS-TI mixture on several standard fuels were calculated (Table 4). The positive S values demonstrated that the C6H13NaO3S/OS-TI mixture can form a sealed aqueous film. The S value on cyclohexane met the US Military Test Specification (>3 mN/m), 50 implying that the synergism between C6H13NaO3S and OS-TI decreased the foam solution–cyclohexane interface tension sharply. The S values on other hydrocarbon fuels were equivalent to some experimental foams.17–19 So, the C6H13NaO3S/OS-TI mixture has an opportunity to chase commercial products. Besides, it should be clarified that no spreading was observable on n-heptane with an S value of 0.6 mN/m. Increasing C6H13NaO3S/OS-TI density may solve this issue by shortening the equilibration time to achieve possible spreading, 51 but latent environmental hazards should be considered carefully.
Spreading coefficients of equimolar OS-TI/C6H13NaO3S foam on different fuels at CMC concentrations.

There is no absolute correlation between S value and spreading, not to mention under the high-temperature fire conditions. To modify Eq (1), some people used dynamic or quasi-equilibrium values to approach real spreading and sealing.52,53 In contrast, the cyclic “water drainage-cooling fuel-film creation” process facilitates the foam to cover the fuel rapidly. The insight discussion revealed that foam cooling on the oil/water interface is substantially effective. 54 Therefore, improving the calculation method to reflect the spreading is not indispensable.
Foaming performance
The foam above the aqueous film layer exerts multiple effects during extinguishing. Oxygen contact and fuel evaporation are blocked jointly by the foam and aqueous film. The aqueous film layer can absorb the thermal energy to cool fuel, and the steam dilutes the oxygen concentration. 50 In addition, the foam drains water to maintain the stability and integrity of the aqueous film layer. A too-short drainage process is relevant to the unstable foam structure, which is fragile to face high-temperature challenges in a fire scene. Conversely, the foam fail to supply water promptly to maintain the integral aqueous film layer during a too-long drainage process. 17 Hence, moderate foam performance is beneficial for exerting satisfactory extinguishing performance. With the increase in mixture concentration, sufficient surfactant molecules generated the compact foam structure, demonstrating the growing curves of foam expansion and 25% drainage time (Figure 8). Conversely, excessive OS-TI increased the foam viscosity so that the drainage process was delayed to support aqueous film completeness, further leading to extinguishing performance degradation. Subsequently, it is critical to determine the proper foam performance with reasonable parameters. According to the National Fire Protection Association, 55 the minimum standard indicates that AFFF should achieve an expansion ratio greater than 5 and a 25% drainage time over 2.25 min. Jia et al. 18 optimized a foam with an expansion ratio of 9.8 and a 25% drainage time of 313 s, and the mixture suppressed aviation kerosene fire in 41 s. After investigating many mixtures, Schefey concluded that the best foam should have an expansion ratio of 8 and a 25% drainage time of 6 min. 46 In the above comparable cases, researchers insisted on obtaining satisfactory foam performance to lay the foundation for extinguishing. To balance foam performance and cost, we finally decided on the 1:1 mixture of 16 mmol/L with an expansion ratio of 10.5 and a drainage time of 317 s.

The foam performance of equimolar OS-TI/C6H13NaO3S under different concentrations.
Extinguishing performance analysis
In an attempt to evaluate the application prospect, we conducted the extinguishing and reburn resistance tests of the 16 mmol/L equimolar mixture using cyclohexane, n-heptane, aviation kerosene, aviation gasoline, and gasoline #95 (Table 5). It was promising that the mixture suppressed fuel fires upon rapid spreading, effective cooling, and prompt drainage. All extinguishing happened in 65 s, and the extinguishing time against aviation kerosene fire (54 s) even reached the requirement of the International Civil Aviation Organization (60 s). 56 The data in Table 5 achieved the GB 15308-2006 34 standard (equivalent to EN 1568-3 2008) 57 , which stipulated the minimum values for extinguishing time (180 s) and burnback time (14 min). Though the optimal OS-TI/C6H13NaO3S foam seemed to be able to apply in the fire scene, the commercial products are still superior. For instance, classical DuPont FC1157 fluorine foam could suppress n-heptane fire in 40 s. 17 Our future work will focus on developing an integral AFFF formula based on OS-TI/C6H13NaO3S foam to improve its overall performance.
Extinguishing and burnback time of equimolar OS-TI/C6H13NaO3S (16 mmol/L).

Figure 9 shows the foam parameters (h0, h5, and R5) at different temperatures. Interestingly, the foam height at 40 °C is greater than that at 20 °C, suggesting that more surfactant molecules were activated to generate foam at a higher temperature before the thermal degradation. There was no obvious change of initial foam height before 70 °C, but the ratio R5 began to collapse in the range of 60–90 °C. Although the stable h0 signified the considerable thermal stability of the OS-TI structure, the foam structure was destroyed due to evaporation and drainage. 58 It could be expected that the fire scene was a more complicated environment, and high temperature would challenge the foam performance.

The foam stability of equimolar OS-TI/C6H13NaO3S (16 mmol/L) under different temperatures.
Conclusion
In this article, we designed a simple one-step reaction that overcame the steric hindrance between the fluorocarbon chain and imidazolium ring to prepare a novel surfactant (OS-TI). The FT-IR, 1H NMR, and 13C NMR spectra characterized the chemical structure of OS-TI. The MS technology qualified OS-TI with an accurate molecular mass of 359.08, while the characteristic decomposition signals in the TGA curve indicated OS-TI had thermal stability before 170 °C. We conducted a series of tests to assess the application potential of OS-TI in AFFF. The impressive surface activity of equimolar C6H13NaO3S/OS-TI suggested that the mixture can spread and foam on the hydrogen fuels. The spreading experiments revealed the successful spreading of the mixture foam on several hydrocarbon fuels, and the spreading coefficient on cyclohexane satisfied the US Military Test Specification. As foam features were investigated, the optimized foam contributed a comparable performance to the current cases. The synergism between OS-TI and C6H13NaO3S generated a dense foam with a prompt 25% drainage time. In the extinguishing experiment, the 16 mmol/L equimolar mixture extinguished several fuels in 65 s, and the burnback time on aviation kerosene fire met the International Civil Aviation Organization requirement.
Although the OS-TI/C6H13NaO3S showed promising potential in the AFFF application, there are some improvements we will make in the future. First, deepening the foam formula development is an essential strategy, tending to realize the field application by selecting the proper stabilizer, solubilizer, antifreeze, and preservative. Second, AFFF needs to mitigate its shock to the environment and health. Hence, our future study will focus on the toxicity investigation of OS-TI and on designing gemini surfactants to decrease the CMC concentration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by the Sichuan Natural Science Foundation Project of China (grant no: 2022NSFSC0302) and the General Program of Civil Aviation Flight University of China (grant no: J2021−096).
