Abstract
A novel route for C(sp3)-N bond formation via C(sp2)−H activation/reduction by metal-free which uses (E)-N′-benzylideneacetohydrazide and (vinylsulfonyl)benzene to synthesize derivatives of (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazide has been reported. This methodology features good functional group tolerance and convenient operation. In addition, it uses inexpensive and insensitive chemical reagents as raw materials and provides an atom- and efficient method for the preparation of (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazides.

A novel route for C(sp3)-N bond formation via C(sp2)−H activation/reduction by metal-free which uses (E)-N′-Benzylideneacetohydrazide and (vinylsulfonyl)benzene to synthesize derivatives of (E)-N′-Benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazide have been reported.
C−N bond formation is significant and plays an important role in synthetic chemistry because amines and their derivatives are privileged structures for alkaloids, pharmaceuticals, and organic materials.1–3 Many methods to construct C−N bonds have been reported, such as reductive amination, nucleophilic amination, electrophilic amination, hydroamination, and cross-coupling reactions such as Ullmann coupling, Chan−Lam coupling, and Buchwald–Hartwig coupling which catalyzed by transition metal. 4 More recently, many strategies for C−N bond formation through C–H bond activation have been gradually developed. For example, Kilian Muñiz’s group reported a method for an intermolecular C(sp3)H amidation via palladium-catalyzed. 5 And Alabugin developed a strategy that builds a C−N bond through C(sp3)−H aminations under metal-free conditions with unprotected anilines. 6 Also, Wang’s group has summarized several examples to construct C−N bonds by copper-catalyzed C−H functionalization. 7 Similarly, C−N bonds building between C(sp2)−H and N(sp2)−H bonds also have attracted the attention of synthetic chemists. For example, Cho’s group 8 has developed an efficient strategy to produce the corresponding nitrogen-containing scaffolds through the tandem reaction of 2-aminoazoles and 2-(2-bromoaryl)indoles promoted by CuI and base under microwave irradiation (Scheme 1a). Next, Wang and co-workers 9 delivered a method to synthesize isoquinolinones and pyridinones through palladium-catalyzed regioselective C−H functionalization/annulation reaction of amides and allylbenzenes (Scheme 1b). Besides, a strategy for photochemical C−N coupling was reported from 2-([1,1’-biphenyl]-2-yl)-1H-benzo[d]imidazoles 10 (Scheme 1c) or pyrazoles 11 (Scheme 1d) with arenes. Notably, an unprecedented approach of C(sp2)−N bond formation through deacetylation/intramolecular aryl migration under metal-free conditions was delivered by Han’s group 12 (Scheme 1e). In comparison with C(sp2)−N bond formation, metal-free C(sp3)−N bond building from C(sp2)−H and N(sp2)−H bonds is more attractive and challenging. Herein, we describe the strategy of C(sp3)−N bond formation via C(sp2)−H activation/reduction from (vinylsulfonyl)benzene with (E)-N′-benzylideneacetohydrazide under metal-free conditions. This methodology features good functional group tolerance and convenient operation (Scheme 1f).

Many C–N bond formation between C(sp2) and N(sp2)−H bonds.
First, various solvents were screened, and the best yield of 75% was obtained when using CH3CN as a solvent (Table 1, entries 1–6). A number of bases, such as Et3N, DBU, KOH, and C5H5N, were also tested, and all of the yields were poor (Table 1, entries 7–10). After this, the screening of different additives such as Et3N • 3HCl, C5H5N • HCl, and C5H5N • HF was performed at standard conditions, but the yields of
Reaction conditions:
Isolated yields.
Et3N • 3 HF (2.0 equiv.).
Et3N • 3 HF (4.0 equiv.).
8 h.
16 h.
O2.
N2.
The results of coupling/reduction reactions between various (E)-N′-benzylideneacetohydrazides
Reaction conditions:
Isolated yields.
Replaced the MeCN with DMSO.
To show the synthetic utility of this procedure, we next performed a gram-scale reaction with 10 mmol of
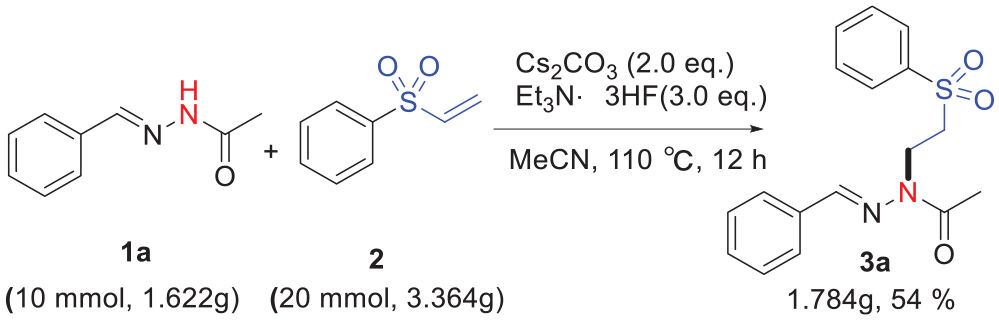
Synthetic applications.
Next, to learn about this reaction mechanism, a series of control experiments were conducted. First, it was known that no target product (
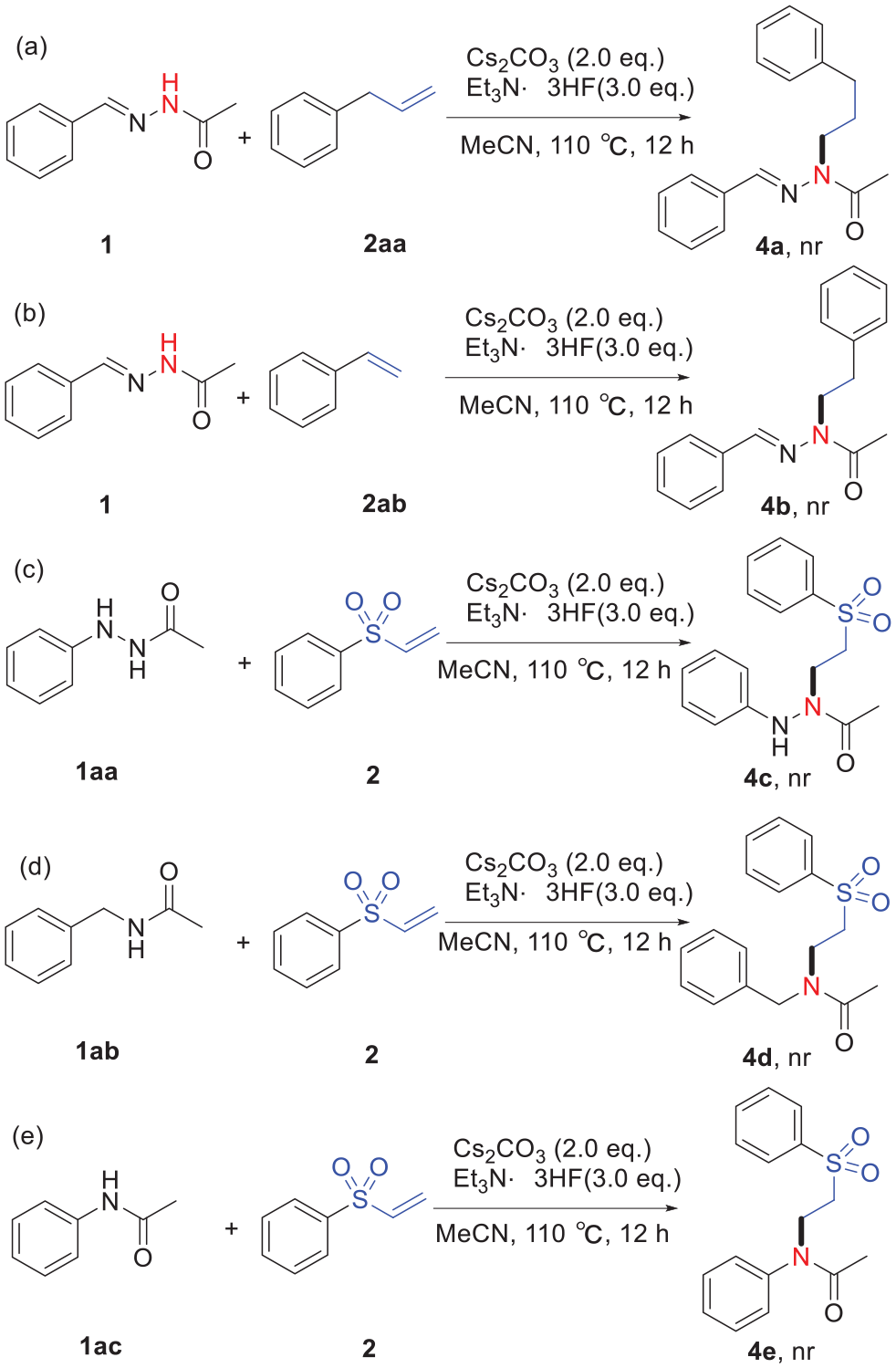
Control experiments.
According to the previous related studies13–17 and the above-mentioned results, a plausible mechanism of C(sp2)-H activation/reduction is depicted in Scheme 4. First, Et3N • 3 HF reacted with Cs2CO3 to produce H2CO3 which can release a proton, and then a proton reacted with

Postulated reaction mechanism.
In conclusion, we have developed a method of C(sp3)-N bond formation by metal-free via C(sp2)−H activation/reduction from (vinylsulfonyl)benzene and (E)-N′-Benzylideneacetohydrazide. This methodology not only provides valuable strategies to syntheses of various (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazides under mild reaction conditions, but also the materials are commercially available. Notably, the protocol provides progress in metal-free, atom- and step-economical nature which is considered as environmental friendliness and operational simplicity.
Experimental section
General information
All of the chemicals were obtained commercially and did not purify before use. The 1H NMR and 13C NMR spectra were recorded on a Bruker Avance II 400 spectrometer. All products were purified by silica gel column chromatography (200–300 mesh) and using ethyl acetate and petroleum ether (60–90 °C) as solvent.
General procedure to the synthesis of (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazides
A mixture of the (E)-N′-benzylideneacetohydrazide (
(E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3a)
White solid, 49.6 mg, 75% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-methylbenzylidene)-N-(2-(phenylsulfonyl)ethyl) acetohydrazide (3b)
White solid, 48.3 mg, 70% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-(tert-butyl)benzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3c)
White solid, 51.7 mg, 67% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-fluorobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3d)
White solid, 43.3 mg, 63% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-chlorobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3e)
White solid, 46.6 mg, 60% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-bromobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3f)
White solid, 47.3 mg, 58% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(4-cyanobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3g)
White solid, 33.4 mg, 47% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(3-methylbenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3i)
Light yellow solid, 46.9 mg, 68% yield, m.p. 151–153 °C. 1H NMR (400 MHz, DMSO-d6) δ 7.98 (d, J = 7.3 Hz, 2H), 7.80 (t, J = 7.4 Hz, 1H), 7.73–7.66 (m, 3H), 7.46 (d, J = 7.3 Hz, 2H), 7.33 (t, J = 7.7 Hz, 1H), 7.22 (d, J = 7.5 Hz, 1H), 4.24 (t, J = 7.2 Hz, 2H), 3.64 (t, J = 7.1 Hz, 2H), 2.35 (s, 3H), 2.22 (s, 3H). 13C NMR (101 MHz, DMSO-d6) δ 171.8, 140.3, 139.3, 138.3, 135.0, 134.5, 130.8, 129.9, 129.1, 128.2, 128.1, 124.6, 50.7, 33.7, 21.8, 21.4. HRMS(ESI+): Calculated for C18H21N2O3S, [M+H]+ 345.1267. Found 345.1276.
(E)-N′-(3-methoxybenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3j)
White solid, 47.6 mg, 66% yield, m.p. 170–172 °C. 1H NMR (400 MHz, DMSO-d6) δ 7.97 (d, J = 7.1 Hz, 2H), 7.80 (t, J = 7.3 Hz, 1H), 7.70 (d, J = 7.8 Hz, 3H), 7.36 (t, J = 7.9 Hz, 1H), 7.25 (d, J = 7.7 Hz, 1H), 7.21 (s, 1H), 6.99 (dd, J = 7.9, 2.5 Hz, 1H), 4.25 (t, J = 6.7 Hz, 2H), 3.80 (s, 3H), 3.65 (t, J = 7.0 Hz, 2H), 2.22 (s, 3H). 13C NMR (101 MHz, DMSO-d6) δ 171.9, 159.9, 140.0, 139.3, 136.5, 134.5, 130.3, 129.9, 128.2, 119.9, 115.9, 112.4, 55.6, 50.7, 33.8, 21.8. HRMS(ESI+): Calculated for C18H21N2O4S, [M+H]+ 361.1217. Found 361.1221.
(E)-N′-(3-fluorobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3k)
White solid, 39.7 mg, 57% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(3-bromobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide(3l)
White solid, 44.8 mg, 55% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(3-cyanobenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3m)
Colorless transparency liquid, 36.9 mg, 52% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(2-methylbenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3o)
White solid, 40.6 mg, 59% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(2-methoxybenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3p)
White solid, 48.2 mg, 56% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(2-ethoxybenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3q)
White solid, 40.4 mg, 54% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(2,4-dimethylbenzylidene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3w)
White solid, 43.6 mg, 61% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-(furan-2-ylmethylene)-N-(2-(phenylsulfonyl)ethyl)acetohydrazide (3x)
White solid, 33.9 mg, 53% yield (eluent: ethyl acetate/petroleum ether = 1:10);
(E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)benzohydrazide (3z)
Colorless transparency liquid, 50.2 mg, 64% yield (eluent: ethyl acetate/petroleum ether = 1:10);
Supplemental Material
sj-docx-1-chl-10.1177_17475198241275297 – Supplemental material for Access to (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazide via C(sp2)−H activation and reduction of (E)-N′-benzylideneacetohydrazide with (vinylsulfonyl)benzene
Supplemental material, sj-docx-1-chl-10.1177_17475198241275297 for Access to (E)-N′-benzylidene-N-(2-(phenylsulfonyl)ethyl)acetohydrazide via C(sp2)−H activation and reduction of (E)-N′-benzylideneacetohydrazide with (vinylsulfonyl)benzene by Haiyang Wang and Fusheng Liu in Journal of Chemical Research
Footnotes
Data availability statement
The data underlying this study are available in the published article and its Supporting Information.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully thank the Science and Technology Development Plan of Tan’an City (2019GX015) and Academic Promotion Program of Shandong First Medical University (2019LJ003) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



