Abstract
The potential risks associated with the high halogen content of typical meta-amide (m-diamide) insecticidals aroused concerns among researchers. Hoping to make discoveries by introducing sulfur elements with good environmental compatibility, a series of novel sulfur-containing m-amide derivatives
The novel sulfur-containing meta-amide (m-diamide) derivatives were synthesized through exploration, which had certain lethal rates against A. craccivora (38.89%–75.00%) and P. xylostella (93.33%−100.00%) at 500 mg.L−1. Notably, the sulfone-containing compound

Introduction
Broflanilide (BASF and Mitsui Chemicals, Inc.) and Cyproflanilide (Tahoe Group) (Figure 1) as the typical representatives of m-diamide insecticide that acts on γ-aminobutyric acid (GABA) receptors, which regulates the transmission of chloride ions into cells causing pests to vomit and excite until killing them.1 –3 The excellent insecticidal activities and unique meta-amide structure of these compounds have swiftly captured the attention of pesticide researchers.4,5 Structural analysis revealed that the halogen content in Broflanilide and Cycloflanilide accounts for more than 50% of the total molecular weight, primarily attributed to the presence of fluorine atom, bromine atom, trifluoromethyl, and heptafluoroisopropyl groups. The high halogen content in m-diamide insecticides raised concerns regarding their hydrophilicity and environmental compatibility. 6 In particular, the fluorinated groups were often introduced into drug molecules since their excellent biological activities. 7 However, the environmental impact of fluorinated organic compounds remains a significant concern. Consequently, the novel target compounds were designed to reduce fluorine content while preserving insecticidal efficacy, thereby enhancing environmental suitability.

The structures of two m-diamide and sulfur-containing drugs, and the designed target compounds in this paper.
The sulfur-containing structure has consistently been a focal point in scientific research due to its diverse biological activities and easily derivatized structural characteristics.8,9 Thioethers, sulfoxides, sulfones, and sulfoximine are common derivative structures of sulfur, which make the corresponding compounds show excellent biological activities in the fields of pesticides and pharmaceuticals. For example, Vaniliprole, 10 Fipronil, 11 Sulfoxaflor, 12 and Flubendiamide 13 (Figure 1) revealed effective insecticidal and acaricidal properties. Moreover, sudexanox with sulfoximide structure was utilized as an antibacterial agent. Recently, the Long et al. 14 group reported a series of compounds revealed good insecticidal activity against Nilaparvata lugens (98.92%, 100 mg.L−1) by substituting sulfur for the trifluoromethyl group in the leading compound—Cyproflanilide. Therefore, developing new m-diamide compounds containing sulfur derivatives as potential insecticidal could be a significant avenue in future pesticide research.
Based on the above reasons, this report detailed the introduction of sulfur derivative structures into the leading m-diamide insecticide—Broflanilide. The modification replaced the high-fluorine group—heptafluoroisopropyl with sulfur derivatives, aiming to reduce the halogen content. Exploring the synthesis route and insecticide activities of target sulfur-containing compounds (Figure 1), and summarizing the structure–activity relationship (SAR), which might provide valuable insights for the future research on sulfur-containing m-diamide compounds.
Results and discussion
Chemistry
In Scheme 1, the key intermediate thioether-containing m-amide compound (
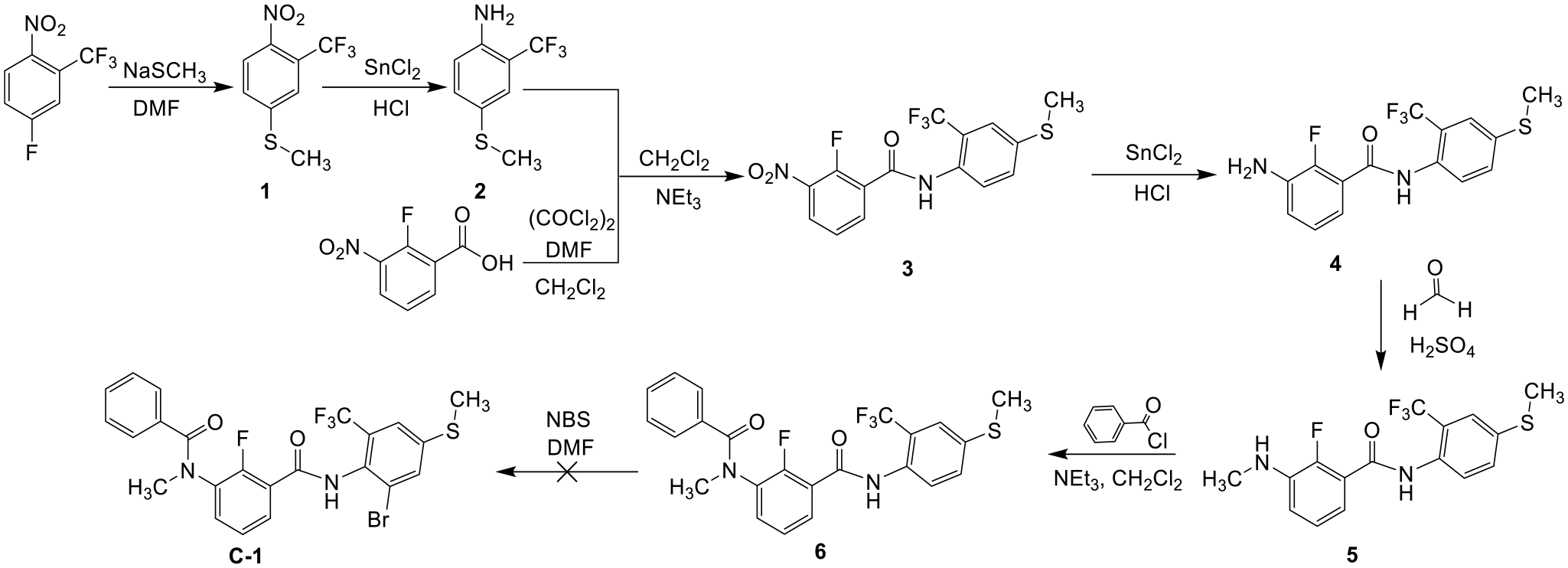
Linear synthesis route for a novel m-diamide compound containing thioether (
So, a bilinear synthesis route was proposed (Scheme 2) based on Aoki et al.
15
In the course of exploration (Table 1), the reaction cannot proceed as effectively as reported in the literature when NaBr was used as the initiator.
16
Therefore, a switch to KI was deemed necessary to facilitate the reaction,
14
considering the nucleophilic substitution mechanism. The choice of KI was influenced by the superior nucleophilic and leaving group properties of iodide ions. Ultimately, the target compound

Bilinear synthesis route for a novel m-diamide compound containing thioether (
Optimization of reaction conditions for the synthesis compound

Isolated yields.
The intermediate compounds
The sulfoxide-containing compound
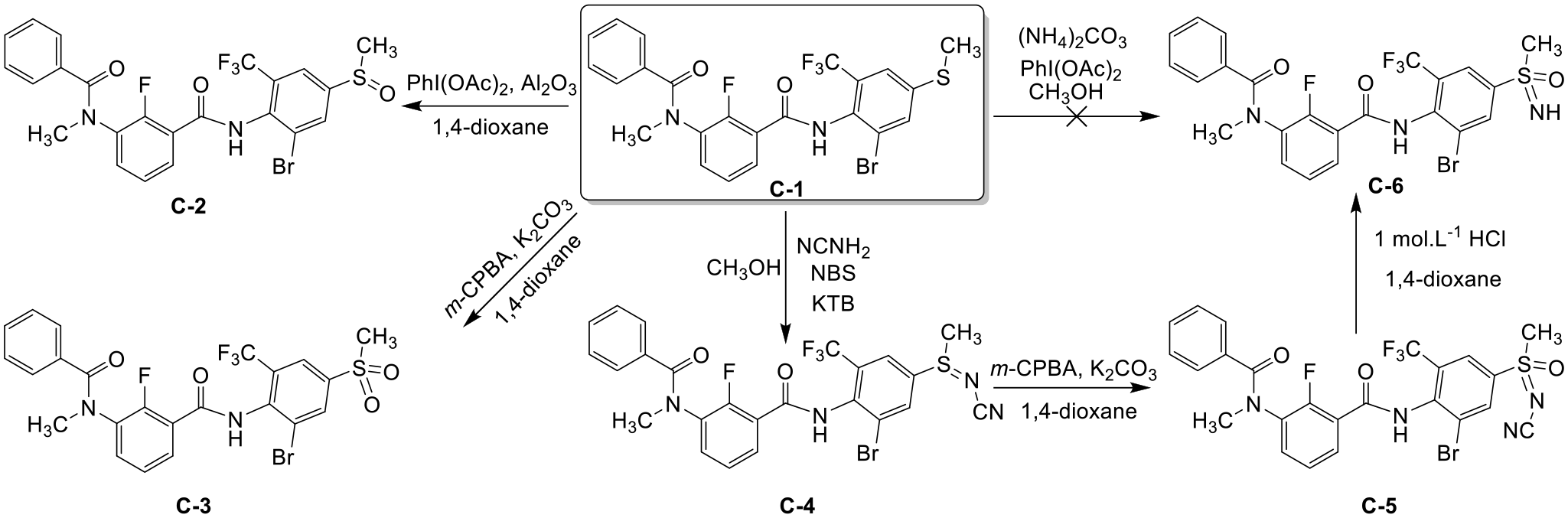
Synthesis route for novel m-diamide compounds containing sulfoxide (
Optimization of reaction conditions for the synthesis compound

Isolated yields.
All of the target m-diamide compounds (
Biology
The insecticidal activities data of target compounds
Insecticidal activity of target compounds against P. xylostella, N. lugens, and Aphis craccivora Koch.

-.: not test.
As shown in Table 3, the SARs can be deduced: For P. xylostella, the larvicidal activity sequence of the (X)n = S-CH3 was identified as sulfone (
Conclusion
Novel sulfur-containing m-amide target compounds
Experimental
Chemistry
Reagents and solvents were purchased from Titan Corporation and used without further purification. Melting points were measured by the SGWX-4B melting point analyzer and uncorrected. NMR spectra were recorded on a Bruker Avance NEO (400, 101, 376 MHz) spectrometer, using DMSO-d6 (TMS as the 0 point internal standard) as the solvent. EI-MS data were analyzed by LCMS-8040 with ESI ionization. HRMS data were obtained on Thermo Q Exactive Focus with ESI ionization.
Synthesis
Synthesis of compound C-1 ; general procedure
Method 1
The thioether-containing target compound
Method 2
The synthesis of the target compound
Synthesis of compound C-2 ; general procedure
Thioether-containing compound (
Synthesis of compound C-3 ; general procedure
Thioether-containing compound (
Synthesis of compound C-4 ; general procedure
In the presence of t-BuOK as a base, thioether-containing m-amide compound (
Synthesis of compound C-5 ; general procedure
N-cyanosulfilimine
Synthesis of compound C-6 ; general procedure
NH-sulfoximine (
Insecticidal activity assay25–27
Feeding conditions
P. xylostella: Raised indoors with radish seedlings at a temperature of 22 ± 2 oC and a light intensity of 12L:12D.25 –27
N. lugens: Raised indoors with water rice seedlings at a temperature of 26 ± 2 oC and a light intensity of 12L:12D.
A. craccivora: Reared indoors with silkworm bean seedlings at a temperature of 22 ± 2 oC and a light intensity of 12L:12D.
Drug preparation
Dissolve the raw materials in DMF to prepare a 1% mother liquor, and dilute with distilled water containing 0.1% Tween 80 to achieve the desired concentration.
Insecticidal activity methods
The lethal rates of the target compounds against P. xylostella, N. lugens, and A. craccivora were investigated in a greenhouse, compared to Broflanilide and a blank control group without medication.
Determination of the activity of P. xylostella 26 : Using the leaf soaking method, gently immerse radish leaves in the prepared test solution for 30 s. Then, transfer the leaves onto plastic culture dishes lined with filter paper and allow them to air-dry in the shade. Each dish was infested with 8 s-instar diamondback moths and placed in an observation room at a temperature of 25 ℃. Test results were observed after 48 h. Insects were considered deceased if they exhibited no response or were unable to crawl normally when lightly touched with a brush. Repeat three times for each sample.
Determination of the activity of N. lugens 27 : Using the spray method, select a rice seedling with two leaves and one core and place it in a 6-cm Petri dish. Then, quartz sand was spread on the Petri dish. Each dish was infested with 20 third-instar early brown planthopper nymphs and treated with 2.5-mL spray with Potter spray tower. The dishes were placed in an observation room at a temperature of 25 °C. Test results were observed after 48 h. Insect was deemed deceased if it exhibited no response or was unable to crawl normally when lightly touched with a brush. Repeat three times for each sample.
Determination of the activity of A. craccivora 27 : Using the spray method. Infested with 30 alfalfa aphid nymphs and treated with 2.5 mL spray with Potter spray tower, and then placed the treated aphids in a 25 °C observation room for cultivation. After 48 h of investigation, the insect bodies were gently touched with tweezers, and the absence of a response was considered an indicator of mortality. Repeat three times for each sample.
Data statistics and analysis
The number of deaths of each processed target was counted and the lethal rate was calculated as follows:

Supplemental Material
sj-pdf-1-chl-10.1177_17475198241234629 – Supplemental material for Design, synthesis, and insecticidal activities of the novel sulfur-containing meta-amide compounds as potential pesticides
Supplemental material, sj-pdf-1-chl-10.1177_17475198241234629 for Design, synthesis, and insecticidal activities of the novel sulfur-containing meta-amide compounds as potential pesticides by Yan Zhang, Jingwen Wu, Shuaihui Dang, Sha Zhou, Jifen Wang and Ruixue Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Here, we thank teacher Yaqin Jiang of the Biological Assay Center, Zhejiang A&F University, for kind bioassay assistance of compounds.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Sailing Program (grant no 21YF1414400).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
