Abstract
CuCo2O4@MoS2 microflowers have been synthesized on Ni foam using a hydrothermal method, and their electrochemical performances as supercapacitor electrodes are investigated. The influence of MoS2 loading on the specific capacity and cycle stability of the nanocomposites is systematically analyzed. It is found that the addition of MoS2 significantly enhances the specific capacity of the CuCo2O4 microflowers, while the cycle stability varies with MoS2 content. Competitive mechanistic insights into the interplay between MoS2 content, electrochemical behavior, and structural stability during cycling are provided. Notably, the CuCo2O4@MoS2-60 sample exhibits an impressive specific capacity of 11.1 F cm−2 at a discharge current density of 0.003 A cm−2, with a remarkable capacity retention rate of 94.6% after 1800 charge–discharge cycles. Moreover, the practical application prospects are demonstrated through the fabrication of symmetric supercapacitors. This study offers valuable guidance for the rational design and optimization of CuCo2O4@MoS2 nanocomposites for high-performance supercapacitor applications.
Keywords

Introduction
Supercapacitors have emerged as a promising energy storage technology due to their high power density, rapid charge–discharge rates, and long cycle life,1–5 making them ideal for a wide range of applications, including portable electronics, 6 electric vehicles, 7 and renewable energy systems. 8 The performance of supercapacitors is largely determined by the electrode materials, which directly impact the specific capacity, rate capability, and cycling stability. 9 Transition metal oxides have garnered significant attention as electrode materials for supercapacitors due to their high specific capacitance and abundant redox-active sites.10,11 Among these materials, bimetallic oxides have shown great potential for supercapacitor applications owing to their unique advantages. Bimetallic oxides offer the ability to combine the properties of multiple transition metals, allowing for synergistic effects that can enhance the electrochemical performance of the resulting materials.12–15
CuCo2O4, a spinel-type copper cobalt oxide, has demonstrated high theoretical specific capacity and excellent electrochemical stability, making it an attractive candidate for supercapacitor applications.16–20 However, the practical utilization of CuCo2O4 is hindered by its low electrical conductivity and poor rate capability. To address these limitations, the integration of CuCo2O4 with highly conductive and electrochemically active materials has been proposed to improve the performance of the electrode material.21–24 For example, Wang et al. 21 have synthesized urchin-like core–shell CuCo2O4@CuCo2S4 heterostructure on carbon cloth substrate through a hydrothermal-assisted electrodeposition method, which not only spatially separates the redox reactions but also enhances the electrochemical stability. Liu et al. 22 prepared porous CuCo2O4/CuO microspheres and nanosheets, demonstrating excellent electrochemical performance and significant electrochemical energy storage potential. Xiao et al. 23 coated CoSe shell on an urchin-like CuCo2O4 electrode for high-performance supercapacitors and demonstrated that the core–shell structure can greatly enhance the stability and electrochemical performance. By combining CuCo2O4 with other materials, the resulting composites can leverage the unique properties of both components, leading to enhanced supercapacitor performance. One such material, molybdenum disulfide (MoS2), has garnered attention for its high theoretical specific capacitance, excellent electrochemical activity, and good mechanical flexibility.25–27 MoS2 has been widely investigated for applications in energy storage and conversion devices, including lithium-ion batteries,28–30 hydrogen evolution reactions,31–33 and supercapacitors.34–36 For instance, Wang et al. 37 synthesized nanoflower-like MoS2@CuCo2O4 heterostructures and exhibited a high sensitivity for real-time glucose detection. Koohdareh et al. 38 investigated the synthesis of MoS2 nanoflowers decorated with CuCo2O4 and their catalytic activities toward the electrochemical hydrogen evolution reaction.
While the integration of MoS2 with different electrode materials has shown promising results, the potential of CuCo2O4–MoS2 nanostructures for supercapacitor applications remains relatively unexplored. This study aims to address this gap by investigating the synthesis and electrochemical performance of CuCo2O4–MoS2 composite materials for supercapacitor electrodes. The synergistic effects arising from the combination of CuCo2O4 and MoS2 are expected to provide increased active sites for charge storage, improve electrical conductivity, and facilitate charge transfer kinetics, leading to enhanced supercapacitor performance. The findings of this research could provide valuable insights into the development of advanced electrode materials for high-performance supercapacitors, contributing to the advancement of energy storage technologies.
Results and discussion
The X-ray diffractometer (XRD) patterns of CuCo2O4 and CuCo2O4/MoS2-60 are shown in Figure 1(a). CuCo2O4 sample exhibits characteristic diffraction peaks at 2θ values of 18.8°, 31.2°, 36.7°, 44.6°, 55.4°, 59.2°, and 65.0° corresponding to (111), (220), (311), (400), (422), (511), and (440) crystallographic planes in the standard spectrum of spinel CuCo2O4 (JCPDS card no. 37-0878), respectively. For CuCo2O4/MoS2-60 sample, in addition to the characteristic peaks of CuCo2O4, new diffraction peaks are observed at 2θ values of 32.8°, 33.7°, and 50.5°, which are indexed to the (100), (101), and (105) crystallographic planes of MoS2 (JCPDS card no. 87-2416), confirming the successful incorporation of MoS2 into the CuCo2O4 nanostructures without vulcanization.

(a) XRD pattern of CuCo2O4 and CuCo2O4/MoS2-60, (b) SEM image of CuCo2O4 on Ni form, (c) magnified SEM image of CuCo2O4 cluster, (d) SEM image of CuCo2O4/MoS2-60 microflower, (e) TEM image of CuCo2O4/MoS2-60 microflower, and (f) high-resolution TEM image.
Figure 1(b) is the scanning electron microscopy (SEM) image of CuCo2O4, revealing that the cluster-like CuCo2O4 structures are well formed on the Ni framework and uniform in size with a hierarchical morphology. Figure 1(c) is a magnified SEM image of a CuCo2O4 cluster, displaying the 2-µm diameter cluster consists of interconnected ultrathin nanosheets. The surface of CuCo2O4 cluster exhibits a porous and rough texture, providing a high surface area for electrochemical reactions and charge storage. The SEM image of the CuCo2O4/MoS2-60 composite shows the uniform distribution of MoS2 nanoflakes on the surface of the CuCo2O4 nanostructures with the diameter expanding from 2 to 10 µm (Figure 1(d)). The MoS2 nanoflakes appear to coat the CuCo2O4 clusters, forming a conformal and continuous layer with microflower morphology. Figure 1(e) is a transmission electron microscopy (TEM) image of a CuCo2O4/MoS2-60 microflower, showing that CuCo2O4 cluster is totally covered with MoS2 nanoflakes. Distinct lattice fringes can be observed in the high-resolution TEM image (Figure 1(f)). The irregular and lattice fringes with distances of 0.25 and 0.62 nm are in good agreement with the (311) plane of CuCo2O4 and the (002) plane of MoS2, respectively.
Figure 2(a) is the energy-dispersive X-ray spectrometer (EDS) spectra of CuCo2O4 and CuCo2O4/MoS2-60. Co, Cu, and O are originated from CuCo2O4, while Ni comes from the Ni foam substrate. In CuCo2O4/MoS2-60 spectrum, the peak belonging to the Mo and S can be observed, implying the formation of MoS2. The elemental mapping profiles of CuCo2O4@MoS2-60 microflower are shown in Figure 2(b), exhibiting a distinct spatial arrangement. Cu and Co are primarily concentrated in the core region, further indicating the dominant presence of CuCo2O4. Mo and S are found in the outer layer, indicating the existence of MoS2. This elemental distribution suggests the successful formation of a well-defined core–shell structure, where CuCo2O4 acts as the core material and MoS2 forms the shell.

(a) EDS spectra of CuCo2O4 and CuCo2O4/MoS2-60 and (b) elemental mapping profiles of CuCo2O4@MoS2-60.
X-ray photoelectron spectroscopy (XPS) is conducted to further elucidate the chemical bonding and oxidation states of the elements presented in CuCo2O4/MoS2-60 sample, as shown in Figure 3. The survey spectra of CuCo2O4 reveal the presence of Cu, Co, and O elements, consistent with the expected composition of the spinel-structured CuCo2O4 (Figure 3(a)). For CuCo2O4/MoS2, new peaks corresponding to Mo and S are observed, indicating the successful incorporation of MoS2 into the composites. The high-resolution XPS spectra of CuCo2O4/MoS2 are shown in Figure 3(b)–(f). In Cu 2p spectrum (Figure 3(b)), the peaks at 962.4, 943.8, and 942.1 eV correspond to the shake-up satellite peaks of Cu2+, while 932.6 and 952.7 eV correspond to Cu 2p3/2 and Cu 2p1/2, respectively. Six peaks appear in the spectrum of Co 2p, as shown in Figure 3(c). Generally, the peaks located at 793–803 and 780–785 eV belong to the Co 2p1/2 and Co 2p3/2, respectively. In this case, the two main pairs of peaks belong to Co3+ and Co2+, while the peaks at 786.6 and 803.1 eV are raised from satellite peaks, proving the hybrid chemical states and spinel structure of CuCo2O4. 39 Figure 3(d) shows the Mo 3d spectrum, showing that Mo 3d5/2 and Mo 3d3/2 are located at approximately 228.2 and 231.4 eV, respectively. In S 2p spectrum, the peak can be well bimodal fitted with the peaks at 163.3 and 161.9 eV (Figure 3(e)). 40 The fine spectrum of O 1s in Figure 3(f) can be devolved into three peaks, which are classified as metal–oxygen–metal bond (530.6 eV), metal–oxygen–hydrogen bond (531.4 eV), and oxygen–hydrogen bond (532.5 eV) in surface-adsorbed water, respectively. 41 These analyses provide comprehensive insights into the elemental composition, crystal structure, and morphology of CuCo2O4 and the CuCo2O4/MoS2 microflowers, highlighting the potential of the CuCo2O4/MoS2 composite for advanced energy storage applications.
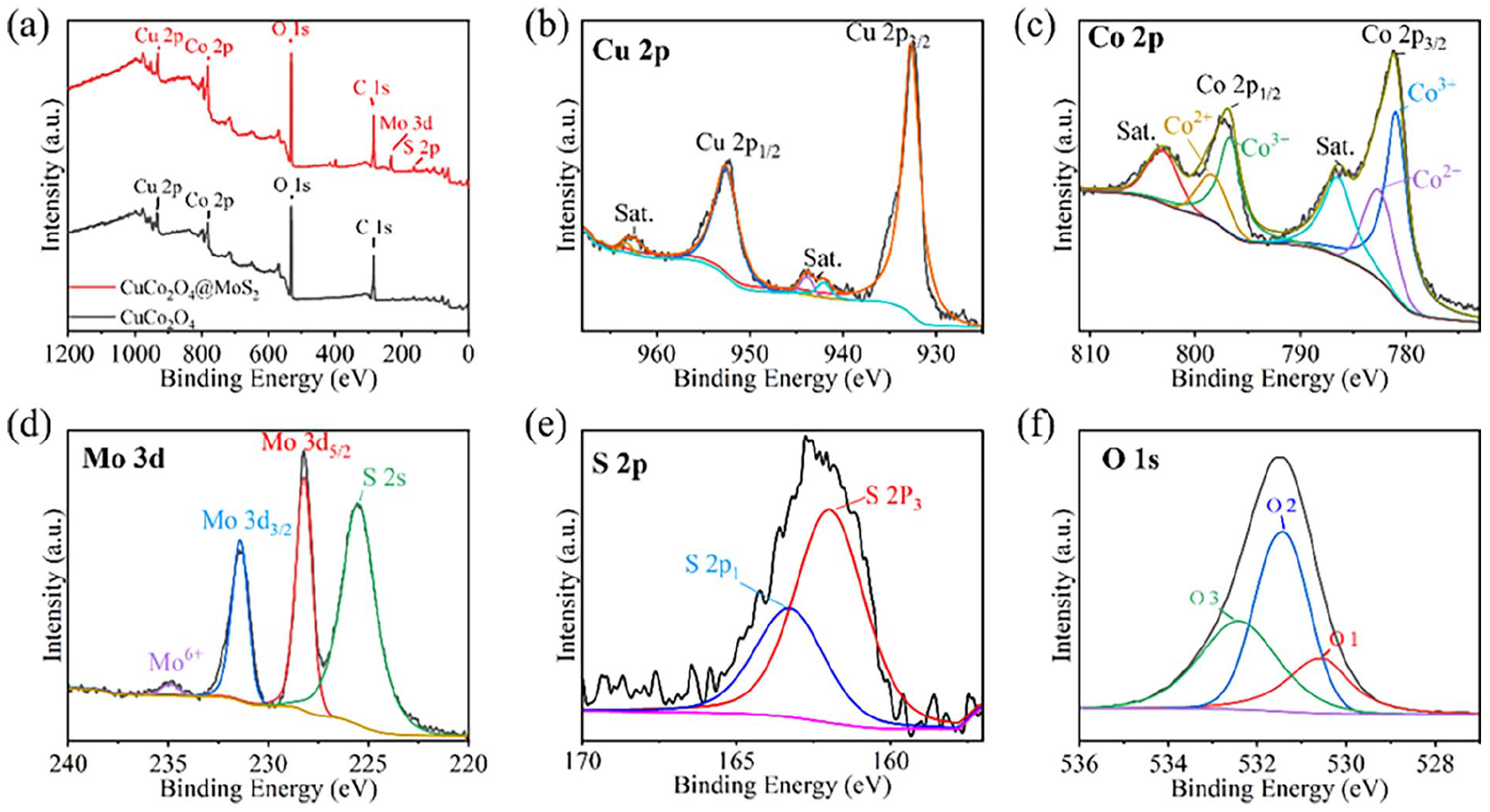
XPS spectra of CuCo2O4/MoS2: (a) survey, (b) Cu 2p, (c) Co 2p, (d) Mo 3d, (e) S 2p, and (f) O 1s.
The cyclic voltammetry (CV) curves of the CuCo2O4 and CuCo2O4/MoS2 samples are shown in Figure 4. Figure 4(a) exhibits one pair of reduction/oxidation peaks in a CV cycle of CuCo2O4, which remains almost unchanged and exhibits good symmetry, revealing the pseudo-capacitance nature of CuCo2O4 with good reversibility at different scan rates. Under the scan rate of 0.005 C s−1, the reduction/oxidation potential of CuCo2O4 is 0.19/0.40 V. With increasing scan rate, the peak current increases gradually. The anode peak shifts positively, while the cathode peak shifts negatively, which is caused by the polarization of the electrode and the uncompensated resistance. As shown in Figure 4(b)–(d), the CV curves of CuCo2O4/MoS2 remain one pair of reduction/oxidation peaks but the peaks shift. The oxide peaks of CuCo2O4/MoS2-40 are less positive than those of CuCo2O4 under the same scan rate, suggesting the coating of MoS2 can induce fast ion–electron transfer. Once CuCo2O4 is coated with MoS2, the closed area significantly increases and varies with MoS2 content, suggesting the improved capacity of CuCo2O4/MoS2.
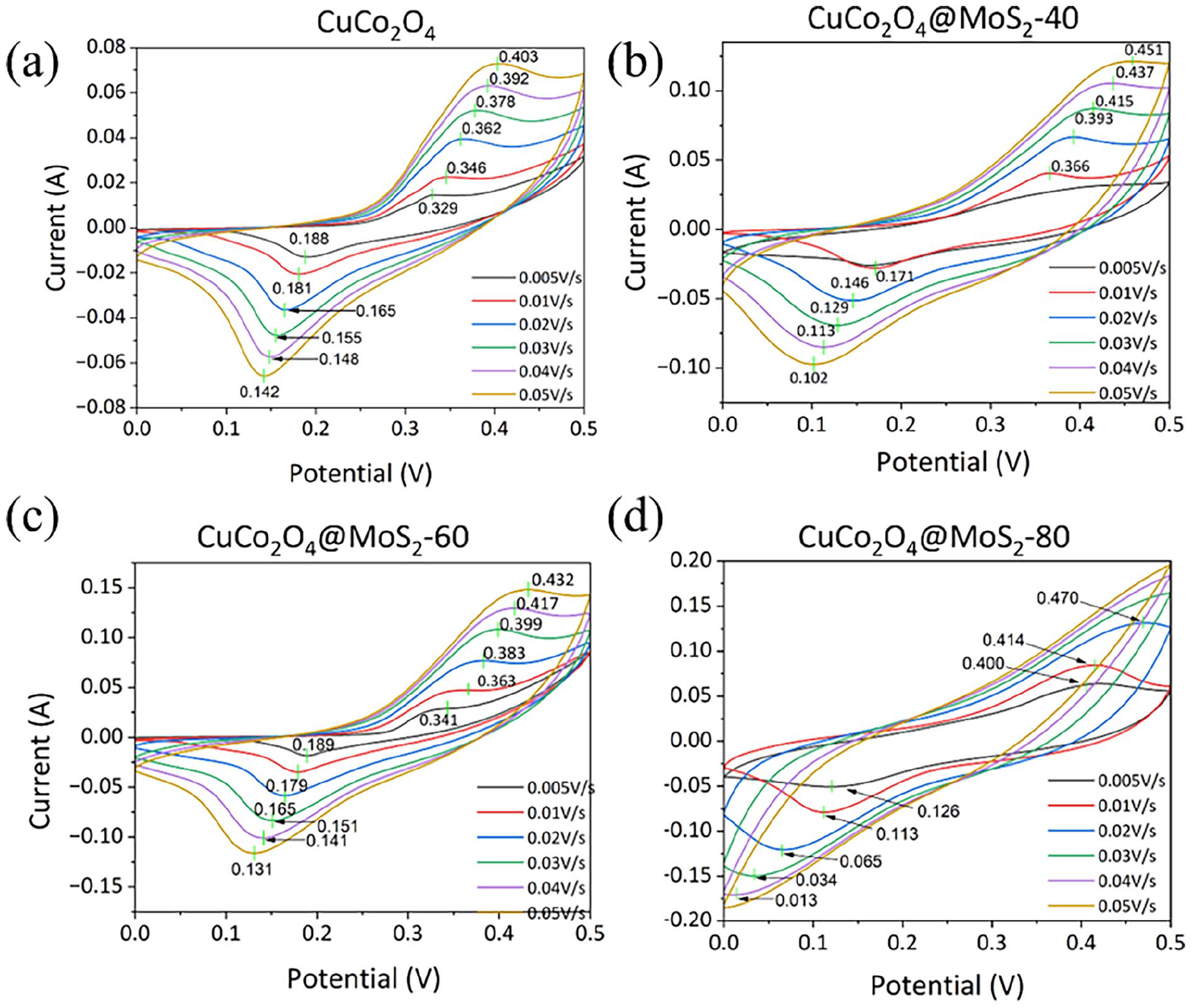
CV curves of (a) CuCo2O4, (b) CuCo2O4/MoS2-40, (c) CuCo2O4/MoS2-60, and (d) CuCo2O4/MoS2-80.
To determine the specific capacity of the samples, galvanostatic charge–discharge (GCD) curves are obtained by applying a constant current density during the charge and discharge processes, as shown in Figure 5. Each curve exhibits typical plateaus, which is the characteristic of the redox reactions of transition metal oxide materials. At current density of 0.003 A cm−2, CuCo2O4 exhibits a specific capacitance of 4.0 F cm−2 (Figure 5(a)), while CuCo2O4@MoS2-40, CuCo2O4@MoS2-60, and CuCo2O4@MoS2-80 demonstrate excellent specific capacity of 7.7, 11.1, and 21.1 F cm−2, respectively (Figure 5(b)–(d)). The high capacitance of CuCo2O4@MoS2-80 is mainly attributed to its outstanding morphological structure and good conductivity. 42 The rate capabilities are shown in Figure 6(a). As the current density increases to 0.01 A cm−2, the specific capacities of the four samples drop to 12.4, 6.3, 3.9, and 2.4 F cm−2, respectively. The specific capacity of the electrodes rapidly decreases with the increase in the current density, which may be attributed to concentration polarization and electrochemical polarization at high current density. Electrochemical impedance spectroscopy (EIS) analysis is conducted at the open-circuit potential and an amplitude of 5 mV with frequency ranging from 0.01 Hz to 100 kHz, and the obtained Nyquist plots are depicted in Figure 6(b). All the Nyquist plots consist of a semicircle in the high-frequency region and a straight line in the low-frequency region, representing the charge transfer resistance and diffusion resistance of the electrode, respectively. It can be observed that both the charge transfer resistance and diffusion resistance of CuCo2O4 are the highest. However, as the MoS2 content increases, both resistances decrease simultaneously. This suggests that MoS2 effectively enhances the conductivity of the electrode, leading to improved electrochemical performance and facilitating Faradaic redox reactions.

CV curves of (a) CuCo2O4, (b) CuCo2O4/MoS2-40, (c) CuCo2O4/MoS2-60, and (d) CuCo2O4/MoS2-80.
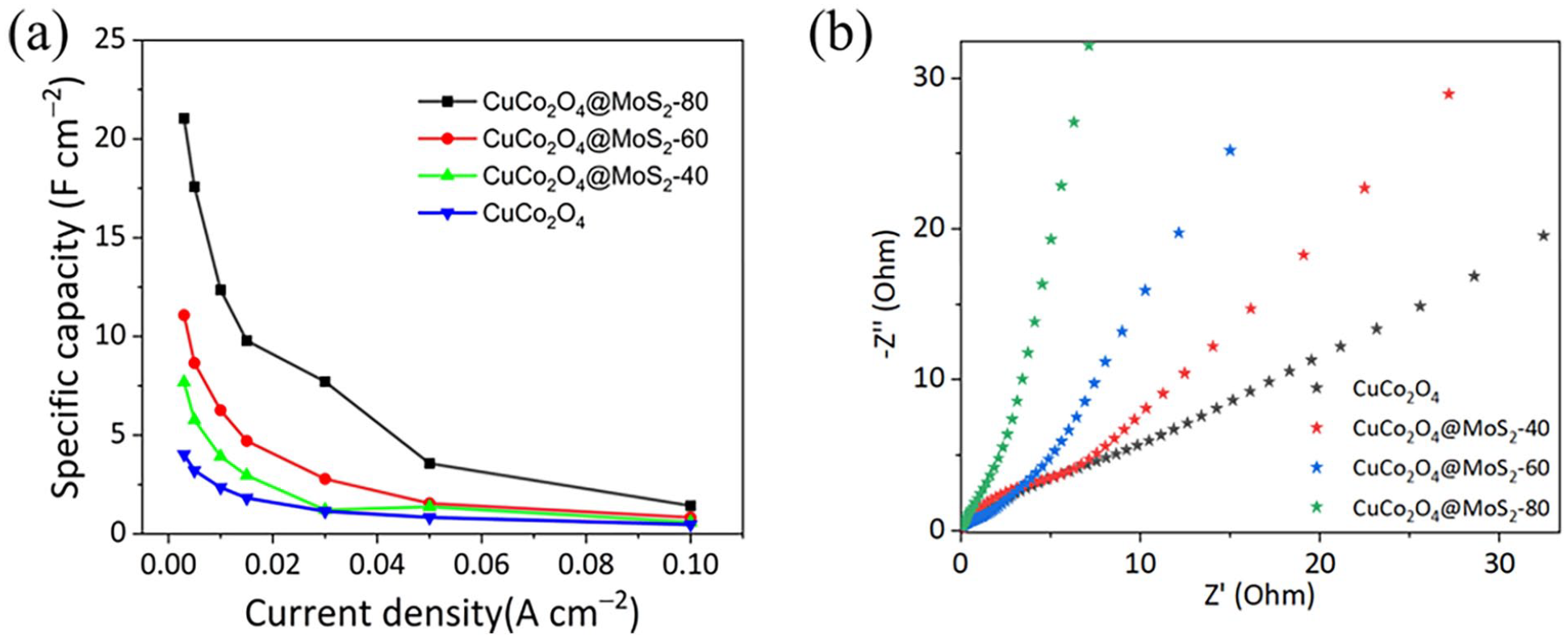
(a) Rate capabilities and (b) EIS curves.
To further study the sulfuration reaction, repeated charge–discharge experiments are conducted for 1800 cycles at a current density of 300 mA cm−2 to evaluate the cycle stability performance under rapid charge–discharge condition, as shown in Figure 7. With the increasing cycle number, the specific capacity of CuCo2O4 increases to a final retention of 142.5% (Figure 7(a)), indicating the slow self-activation process of pure CuCo2O4 clusters. The Coulombic efficiency remains ~100%, implying no irreversible chemical reaction occurs in charge–discharge cycle. For CuCo2O4@MoS2-40, CuCo2O4@MoS2-60, and CuCo2O4@MoS2-80, the specific capacities rise and fall alternately several times, but the whole shows downward trend in varying degrees, resulting in capacity retentions of 74.3%, 94.6%, and 70.8%, respectively (Figure 7(b)–(d)). It is also shown that the capacity retention is affected by the MoS2 content. Figure 8 shows the SEM images of the CuCo2O4@MoS2-80 sample before and after the 1800 charge–discharge cycles. It is clearly observed that plenty of microflowers detach from the Ni foam framework after the recycle test, leading to the rapid decline of specific capacity. It is possible that the excessive MoS2 shell layer leads to a decrease in the binding force of electrode materials, making them more prone to detachment from the Ni foam. In the future works, we should pay more attention to optimizing the synthesis and processing conditions to enhance the mechanical adhesion between microflower and framework, and balance the competition of capacity and stability.

Specific capacities and Coulombic efficiencies of (a) CuCo2O4, (b) CuCo2O4/MoS2-40, (c) CuCo2O4/MoS2-60, and (d) CuCo2O4/MoS2-80 in 1800 cycles.

(a) SEM image of CuCo2O4@MoS2-80 before cycling test and (b) SEM image of CuCo2O4@MoS2-80 after cycling test.
MoS2 is a two-dimensional material with a layered structure, allowing for high surface area and abundant active sites for charge storage. To quantitatively measure the active surface area, Brunauer–Emmett–Teller (BET) and electrochemical surface area (ECSA) tests are conducted, as shown in Figure 9. Figure 9(a) shows that the CuCo2O4@MoS2-80 exhibits the highest surface area of 93.65 m2 g−1 compared to the surface area of 60.64 for CuCo2O4 and 84.71 m2 g−1 for CuCo2O4@MoS2-60, indicating that the MoS2 coating contributes to the increment in surface area. Similar results are also observed from the ECSA test. The calculated double-layer capacitance (CDL) values of CuCo2O4, CuCo2O4@MoS2-40, CuCo2O4@MoS2-60, and CuCo2O4@MoS2-80 are 29.8, 51.7, 54.5, and 57.9 mF cm−2, respectively, as shown in Figure 9(b). The higher CDL values of CuCo2O4@MoS2 samples provide abundant active surface area for charge storage. The incorporation of MoS2 nanosheets into the CuCo2O4 enhances the charge storage capability of the composite material. As the MoS2 content increases, more active sites are available for charge storage, leading to an increase in the specific capacity of the composite. The efficient charge transfer enabled by MoS2 leads to a higher utilization of the active material, allowing for more charge to be stored and delivered during the charge–discharge cycles. In addition, the high conductivity of MoS2 helps to reduce the resistance at the electrode–electrolyte interface, ensuring better electrochemical kinetics.

(a) BET-specific surface areas and (b) CDL-related ECSA.
To verify the feasibility of CuCo2O4@MoS2 material as a supercapacitor electrode, the two-electrode CV and GCD performances are conducted using two CuCo2O4@MoS2-60 samples as the cathode and anode, respectively. In Figure 10(a), the CV curve shows an extended voltage window up to 1 V, maintaining excellent reversibility. In Figure 10(b), the GCD curves at different currents indicate a significant decrease in specific capacitance compared to the three-electrode method, but still exhibit a relatively high capacity. At a current of 0.003 A, the capacitance can reach 1.21 F cm−2. These results demonstrate the feasibility of CuCo2O4@MoS2 material as a supercapacitor electrode.

(a) CV curves of two CuCo2O4@MoS2-60 electrodes and (b) GCD curves of two CuCo2O4@MoS2-60 electrodes.
Conclusion
In conclusion, CuCo2O4@MoS2 microflowers are successfully synthesized on Ni foam using a hydrothermal method and their electrochemical performances as supercapacitor electrodes are investigated. The incorporation of MoS2 has been found to significantly enhance the specific capacity of the CuCo2O4 microflowers. In addition, the cycle stability of the nanocomposites is influenced by the content of MoS2, where the specific capacity and stability are competitive. Through competitive mechanistic insights, we have gained a deeper understanding of the interplay between MoS2 content, electrochemical behavior, and structural stability during cycling. Particularly, the CuCo2O4@MoS2-60 sample has exhibited impressive specific capacity and remarkable capacity retention rate after 1800 charge–discharge cycles. Furthermore, the practical application prospects of the CuCo2O4@MoS2 nanocomposites are demonstrated through the fabrication of symmetric supercapacitors. This study provides valuable guidance for the rational design and optimization of CuCo2O4@MoS2 nanocomposites for high-performance supercapacitor applications.
Experimental section
Synthesis of CuCo2O4 microflowers on Ni foam
The 2 × 4 cm2 Ni foam was cleaned by ultrasonicating in 3M HCl, deionized water, and ethanol for 30 min to remove the oxide layer on the surface, and then placed in dilute HCl for later use. CuCo2O4 microflowers were synthesized via a simple hydrothermal method. In a typical synthesis process, 4 mmol Co(CH3COO)2·4H2O and 2 mmol Cu(CH3COO)2·H2O were dissolved in 60 mL of deionized water to form a homogeneous solution. Subsequently, 18 mmol CH4N2O were added under continuous stirring for 30 min. The resulting mixture and as-cleaned Ni foam were transferred to a 100-mL Teflon-lined stainless steel autoclave and maintained at 120°C for 6 h. After the hydrothermal reaction, the Ni foam was thoroughly rinsed with deionized water and ethanol, followed by drying at 60°C in a vacuum oven.
Synthesis of CuCo2O4/MoS2 microflowers on Ni foam
A certain mass of Na2MoO4·2H2O and CH4N2S with mass ratio of 1:1.5 was dissolved in 30 mL of deionized water with continuous stirring to form a clear solution. Subsequently, the mixture and as-grown Ni foam were transferred to a 50-mL Teflon-lined stainless steel autoclave and heated at 200°C for 24 h. After the hydrothermal reaction, the Ni foams were rinsed with alcohol and deionized water, and dried in a vacuum oven at 60°C. The obtained composite materials with the Na2MoO4·2H2O usage of 40, 60, and 80 mg were named CuCo2O4@MoS2-40, CuCo2O4@MoS2-60, and CuCo2O4@MoS2-80, respectively.
Characterization
XRD (Bruker D8 Advance) was used to analyze the crystal structure and phase composition at a scanning rate of 0.02° s−1. TEM (JEM 2100F, JEOL) and SEM (Regulus 8100, Hitachi) were conducted to characterize the crystal structure and morphology. EDS (7021-H, Horiba) and XPS (K-Alpha, Thermo) were employed to examine the surface chemical composition and oxidation state. The specific surface area was measured by N2 adsorption–desorption analysis with a Micromeritics ASAP 2000 apparatus and calculated using the BET method.
Electrochemical measurements
The electrochemical performances of the CuCo2O4/MoS2 microspheres were evaluated in a three-electrode electrochemical workstation (CHI660E, CH Instruments) using GCD and CV methods. Ag/AgCl electrode and Pt foil were used as the reference and counter electrode, respectively. The Ni foam was cut into 1 × 3 cm2 in area used as working electrode with 1 cm2 immersed in electrolyte (3M KOH). CV measurements were cycled within a potential window ranging from 0 to 0.5 V at different scan rates of 0.005, 0.01, 0.02, 0.03, 0.04, and 0.05 V s−1. GCD was carried out with a working voltage between 0 and 0.42 V during the charge and discharge processes by applying a constant current density (0.003, 0.005, 0.01, 0.015, 0.03, 0.05, and 0.1 A cm−2). The area-specific capacity (C, F cm−2) was calculated following equation (1)
where
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Major Science and Technology Projects of Shanxi Province, China (grant no. MC2016-02); the National Natural Science Foundation of China (grant no. 11947133); and Fund for Shanxi Key Subjects Construction, “1131” Project Key Subjects.

