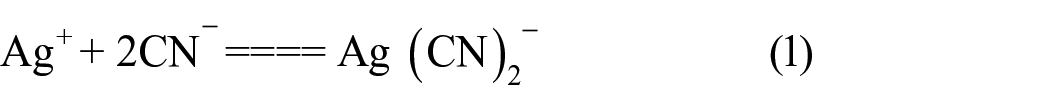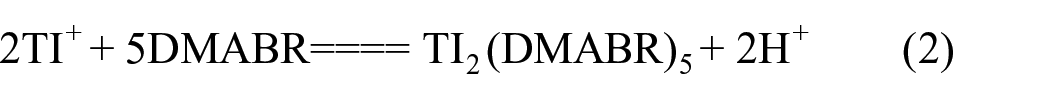Abstract
The novelty of immobilized p-dimethylaminobenzylidenerhodanine is successfully prepared by using TiO2-based nanoparticles as the carrier. It is applied to preconcentrate and separate trace thallium and detect by flow injection combined with graphite furnace atomic absorption spectrometer. The results show that immobilized p-dimethylaminobenzylidenerhodanine can preconcentrate and separate trace thallium and eliminate the interference of many other ions. The optimal adsorption occurs at pH = 3.5, the retention time is 2.0 min, and the flow velocity is 2.5mL/min. The optimal desorption takes place at a flow velocity of 1.0 mL/min, 1 mL mixed solution (thiourea and HCl were both 0.1 mol/L). The recovery rate of thallium is over 95%; the amount of immobilized p-dimethylaminobenzylidenerhodanine is 22.08 mg/g; the adsorption capacity of thallium for immobilized p-dimethylaminobenzylidenerhodanine is 7.2 mg/g; the molar ratio of thallium ions and p-dimethylaminobenzylidenerhodanine is 5:2. The immobilized p-dimethylaminobenzylidenerhodanine can be recycled and reused. For the developed method, the limit of detection is 8 ng/L, the relative standard deviation is 3.7%, and the recovery is 89%–107%. The method is successfully applied to actual samples with satisfactory results.

Introduction
Being highly neurologically toxic, hazardous, harmful, and an environmental pollutant, thallium has very strict limits. 1 Thallium in surface environments mainly originates from mining activities and iron-smelting, and is typically very low. It is currently challenging to pretreat and detect thallium in environmental samples. Therefore, the developments of new methods for the pretreatment and detection of trace thallium samples represent an important challenge.
Present methods for detecting thallium include square-wave anodic stripping voltammetry, 2 liquid–liquid extraction,3,4 solid-phase extraction, 5 flame atomic absorption spectrometry after preconcentration, 6 ICP-sector field mass spectrometry (ICP-SMS), 7 flotation spectrophotometric methods using iodide and rhodamine B, 8 further, hyphenated techniques in microfluidic to investigate ions, 9 micro-extraction by packed sorbent, 10 and so on.
p-dimethylaminobenzylidenerhodanine (DMABR) can make complex metal ions in acidic solution, and thus is widely used to determine the presence of platinum, 11 palladium, 12 silver, gold, 13 and other metal ions. The surface atoms of the nanomaterials can bind with other atoms easily because of the nanoparticles, which provides high chemical activity. 14 TiO2-based nanoparticles have a large surface area and are stable materials for the preconcentration and separation of metal elements. 15
In this study, DMABR was immobilized on the surface of the TiO2-based nanoparticles by using a simple procedure. Trace thallium was detected by furnace atomic absorption spectrometer (FI-GFAAS). The results show that immobilized DMABR exhibits good selective adsorption and separation ability for trace thallium. Under optimized conditions, our method can be applied to detecting trace thallium in water and urine samples with satisfactory results.
Results and discussion
Characterization of immobilized DMABR
The Fourier transform infrared (FTIR) spectra (Figure 1) show that the stretching absorption peak of S=C is shifted from 1153.223 cm−1 to 1091.512 cm−1. This indicates an interaction between immobilized DMABR and TiO2-based nanoparticles, but not chemical reactions. The scanning electron microscope (SEM; Figure 2) shows that immobilized DMABR exists as nanoparticles with a large surface area. It indicated that immobilized DMABR was successfully prepared.

The FTIR spectra of immobilized DMABR, DMABR, and TiO2-based nanoparticles.

Scanning electron microscope (SEM) image of immobilized DMABR.
The amount of immobilized DMABR
The amount of DMABR loaded on the surface of TiO2-based nanoparticles was calculated from equation (1) as
where
The immobilized DMABR was dissolved in hot ethanol, filtered, and the DMABR in the solution was determined by UV-9200 UV-Vis spectrophotometer at 458 nm with a solution of ethanol. The results showed that the amount of DMABR was 22.08 mg/g.
Effect of pH
The pH values of 1.0 μg/L thallium solutions were adjusted to a range of 2.0–7.0 with 0.1 mol/L CH3COOH/CH3COONa buffer solution to evaluate the effect of pH. We can observe from Figure 3 that the pH value plays an important role, with well-defined peaks located at pH = 3.5. Perhaps the thallium ions and immobilized DMABR form stable complexes at pH 3.5, which is favorable for adsorption by immobilized DMABR. Therefore, pH = 3.5 was chosen as the optimum value for this study.

Effect of pH on the absorption ratio.
Effect of the flow rate of the sample solution
The speed of the solution flow can affect the retention time of thallium and the analytical accuracy. As shown in Figure 4, we found that under optimum conditions (pH, eluent, etc.), when keeping the speed of the flow in the range of 0.5–2.5 mL/min, the recovery rate of thallium was virtually changed. The recovery rate of thallium dropped significantly when the speed of the flow was 2.5–4.0 mL/min. This result could be due to the adsorption kinetics at a higher flow rate. Based on the above results, the speed of the flow in this study was set at 2.5 mL/min.

Effect of flow rate on the recovery of thallium.
Effect of the retention time
By allowing different volumes of 1.0 μg/L thallium solutions to flow through the microcolumn at a speed of 2.5 mL/min, the effect of the retention time was investigated. The results (Figure 5) show that in the experimental range, the absorption signal increases with retention time by up to 2 min. The absorption signal starts to decrease when the retention time is over 2 min. Therefore, a retention time of 2.0 min was selected as the optimum.

Effect of the retention time on the adsorption of thallium.
Elution of the adsorbed thallium
To find the best effective eluent, different types of solutions including HCl, HNO3, HClO4, thiourea in HCl, thiourea in CH3COOH, NaClO3, and diphenylthiourea were investigated. We found that the above-mentioned solutions did not completely elute thallium ions from the microcolumn at a flow rate of 1.0 mL/min, except for the solution of thiourea in HCl.
Different concentrations of solutions of thiourea in HCl were investigated. We found that a mixed solution (thiourea and HCl were both 0.1 mol/L) was the best. The effect of the eluent volume from 0.5 to 2.5 mL was also investigated. The results show that the recovery rate of thallium was over 95% when the volume of the mixed solution (thiourea and HCl were both 0.1 mol/L) was over 1.0 mL. Therefore, 1.0 mL of a mixed solution (thiourea and HCl were both 0.1 mol/L) was selected.
Adsorption capacity
The adsorption capacity was investigated by Maquieira et al. 16 Five concentrations of thallium were prepared in pH = 3.5 buffer solution: 0.1, 0.5, 1, 5, and 20 μg/L, then, 50 mL thallium solution of each concentration by a 100 mL flask. The proposed separation and preconcentration procedure described above was applied. The adsorption capacity can be calculated from equation (2) as
where
The ratio of the thallium DMABR complex can be calculated from equation (3) as
where QDMABR (mg/g) is the amount of DMABR immobilized on a unit of the TiO2-based nanoparticle surface; MDMABR is the molar mass of DMABR; QTI (mg/g) is the adsorption capacity of the unit immobilized DMABR; and MTI is the molar mass of thallium.
The results showed that the adsorption capacity of immobilized DMABR for thallium was 7.2 mg/g, nDMABR:nTI = 5:2. This indicates that five DMABR molecules probably act as a bidentate coordinator for two thallium ions. The FTIR spectra shown in Figure 6 indicate that the characteristic peak for N-H is shifted from 1673.909 to 1732.551 cm−1. They also show that complexes of immobilized DMABR with thallium ion are formed. All the results showed that the structure of DMABR had a minor change. 17

The FTIR spectra of immobilized DMABR and immobilized DMABR with thallium.
Microcolumn reuse
The stability and potential regeneration of immobilized DMABR was next investigated. The microcolumn was rinsed with 10 mL of 1.0 mol/L HCl and 20 mL of distilled water and then dried, before being reused. The results showed that the adsorption capacity of immobilized DMABR was not obviously decreased after 10 cycles. These recycling studies show that immobilized DMABR can be efficiently recycled and reused.
Effects of coexisting ions
The effects of coexisting ions on the adsorption were investigated. In these experiments, the interfering ions were added to 1.0 µg/L thallium. The tolerance limits of the coexisting ions were defined as being the largest amount making the recovery of the investigated element less than 95%. The results show that a 10,000-fold excess of Na+, K+, Ca2+, Mg2+, NO3−, or Cl−; a 500-fold excess of Al3+ or Ni2+, a 200-fold excess of Zn2+, Cd2+, or Pb2+; a 100-fold excess of Cu2+ or Fe3+, a 50-fold excess of Pt4+, In3+ or Au3+ do not interfere. However, serious interference occurred in the presence of Ag+, which can be overcome by adding 0.001 mol/L of CN−. This indicates that immobilized DMABR shows better selectivity than DMABR, which can be attributed to the structure of DMABR having undergone minor change on immobilization. Meanwhile, the serious interference of Ag+ is attributed mainly to Ag+ and DMABR to form a red gelatinous precipitate, and to be overcome by CN− owes much to Ag+ which can react with CN− and form a stable complex (1). When the interference of Ag+ is overcome the complex reaction of DMABR and thallium ions was carried out (2)
Evaluation of the analytical method
Under optimized conditions, the thallium samples (0.02−40 µg/L) passed through the microcolumn at a rate of 2.5 mL/min and were detected by GFAAS. The linear regression equation was у = 0.5082χ − 0.0015, R2 = 0.9995. The linear calibration showed good linearity in the range of 0.02–40 µg/L. The triple background signal of the instrument (3σ)(LOD) was 8 ng/L. A prepared sample (1.0 µg/L) spiked with a standard solution at three different concentration levels was analyzed using three replicate measurements, and the recoveries were 89%−107%. The accuracy of our methodology was controlled by three repeating analyses of solutions containing 50 and 200 pg of 1.0 μg/L thallium solutions. The results were 3.2% and 3.8% (n = 3), respectively.
Sample analysis
The developed method was used to detect trace thallium in human urine and water samples; 24-h urine samples were collected from voluntary healthy laboratory personnel in sterile disposable poly (propylene) containers to which 1 mL of 0.1% v/v HNO3 had previously been added. Sub-samples of 10 mL were pipetted from these samples into 10-mL capped high-pressure polyethylene test tubes and stored at 4°C until the samples were investigated (usually within a few hours). The sample pH value was adjusted to 3.5 with 0.1 mol/L CH3COOH/CH3COONa buffer solution before being investigated. The river water and sewage samples were filtered through a 0.45-μm membrane filter and analyzed as soon as possible. The pH value of the sample was adjusted to 3.5 with 0.1 mol/L CH3COOH/CH3COONa buffer solution before being investigated.
The sample solutions were concentrated to a suitable volume by slowly heating and evaporation before comparison by analysis with ICP-MS. The results shown in Table 1 indicate that the difference in the detection of thallium using the developed method and ICP-MS were not very significant. Thus, the results obtained utilizing the developed method are satisfactory.
Determination of the quantity of thallium in different samples (n = 5).

Conclusion
In this study, recyclable immobilized DMABR was successfully prepared from DMABR and TiO2-based nanoparticles and was used to preconcentrate and separate trace thallium. Compared with other methodologies,18–20 immobilized DMABR was effective in selectively separating and preconcentrating trace thallium, and can eliminate the interference by numerous other ions. This new method is simple, accurate, and rapid for detecting and measuring trace thallium, which provides new ideas for determination of metal ions by complexes and a complex carrier.
Experimental and material
Reagents and apparatus
Thallium was detected by GFAAS (SOLLAR M6, ThermoElemental Instrument Corp, American); a GF95 furnace system and an FS95 furnace autosampler were employed; the characterization of immobilized DMABR was investigated by FTIR (IRXROSS, Shimadzu, Japan) and SEM(H-800, Hitachi, Japan); an LZ-2000 flow injection processor (ZhaoFa institute for laboratory automation, Shenyang, China) and a self-made PTFE microcolumn (45 mm×3.5 mm i.d.) packed with immobilized DMABR were used in FI manifold for thallium preconcentration and separation. The amount of DMABR loaded on the surface of TiO2-based nanoparticles was investigated by UV-9200 ultraviolet visible spectrophotometer (Beijing Rayleigh Analytical Instrument Corp, Beijing, China).
A solution (1000 µg/mL) of thallium was prepared by dissolving 0.1 g of thallium metal (99.999%) in a mixture of 40 mL of hydrochloric and nitric acid (1:3 (v/v)), and diluted to 100 mL with 0.1 mol/L HCl. DMABR (Beijing Chemical Plant, Beijing, China) and titanium tetrabutoxide (Ti(OC4H9)4) (Shanghai Yuanhang chemical plant, Shanghai, China) were used; all other reagents were of analytical reagent (AR).
Preparation of immobilized DMABR and microcolumn
Solution A: 10 mL of Ti(OC4H9)4 was added to 12.5 mL of ethanol. Solution B: a mixture of double quartz distilled water (0.5 mL), ethanol (12.5 mL), and HCl (0.25 mL). Solution B was slowly added dropwise to solution A while stirring at room temperature, for 2 h. The mixture was cooled to form a dry sol–gel at room temperature. The dry sol–gel was dried and calcined at 500°C for 2 h to give TiO2-based nanoparticles.
DMABR (0.05 g) was dissolved in 50 mL of ethanol with magnetic stirring, and 2.0 g of TiO2-based nanoparticles were added to the solution. The mixture was stirred for 1 h at room temperature, allowed to stand for 2 h, and filtered. The residue was washed at least three times with distilled water and it was kept at 80°C in a vacuum desiccation for 12 h to give the immobilized DMABR.
PTFE microcolumn (45 mm × 3.5 mm i.d.) with 0.1 g of immobilized DMABR was filled, and plugged with a small portion of glass wool at both ends. The microcolumn was rinsed with distilled water, and the acidity of the microcolumn was regulated by using 0.1 mol/L of CH3COOH/CH3COONa buffer solution.
Preparation and determination of samples
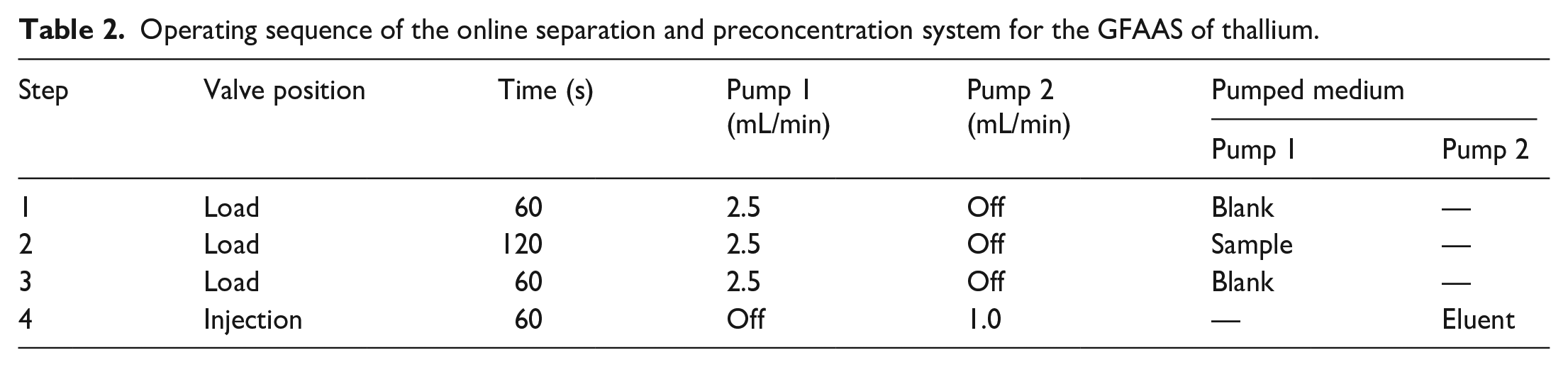
Thallium samples were prepared and adjusted pH to 3.5 with 0.1 mol/L CH3COOH/CH3COONa buffer solution. If necessary, an appropriate amount of HNO3 or NaOH solution was used to adjust the pH, then buffer solution was added. Details of the operation process are given in Table 2. According to Figure 7, in Step 1, the injection valve was in the “load position” and pump P 1 was activated. The blank solution (0.1 mol/L HCl) was pumped through the microcolumn for 1.0 min with a flow rate of 2.5 mL/min to eliminate the eluent remaining in the microcolumn in the previous elution step. Meanwhile, distilled water was pumped into the GFAAS autosampler to confirm the baseline. In Step 2, the sample solution was loaded into the microcolumn, while the other ions were pumped into the waste. In Step 3, the blank solution was pumped through the microcolumn again to remove the sample matrix. In Step 4, pump P 2 was activated, while pump P 1 was stopped and the injection valve turned to the “injection position” to introduce 0.1 mol/L of thiourea with 0.1 mol/L of HCl for eluting the analyte retained on the microcolumn. The flow rate of 1.0 mL/min was maintained for 1.0 min and the eluent was introduced into the GFAAS autosampler directly, with detection at 276.8 nm. If the thallium level was too low in the sample, then the time of Step 2 can be extended.
Operating sequence of the online separation and preconcentration system for the GFAAS of thallium.
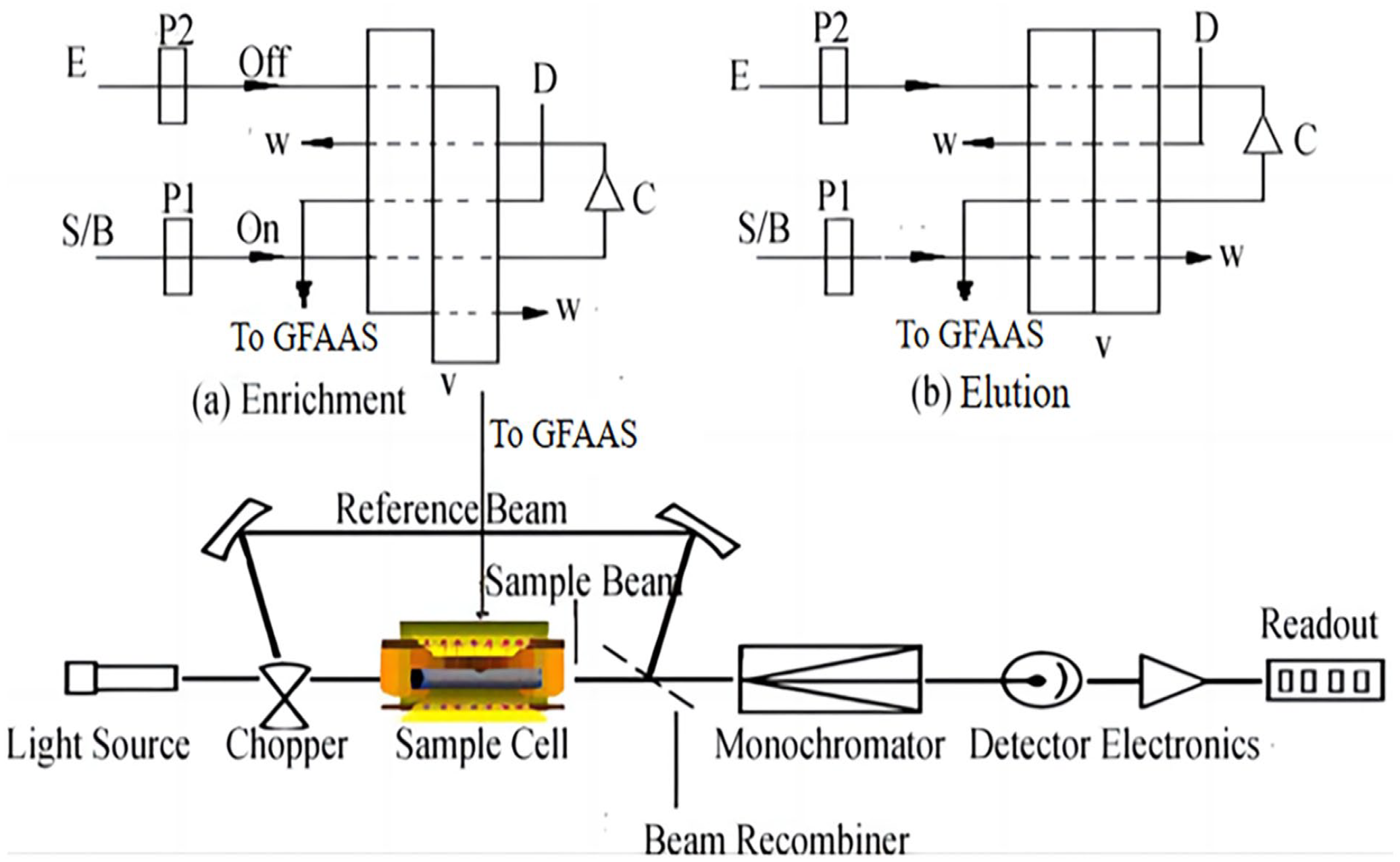
The schematic of the flow injection system combined with GFAAS: (a) enrichment; (b) elution.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hunan Provincial National Health Commission of China (No. 202212065516).