Abstract
2-[1-Oxo-1-(piperidin-1-yl)propan-2-ylidene]-N-(prop-2-en-1-yl)hydrazinecarbothioamide (

Introduction
Thiosemicarbazones are the subject of constant and multidisciplinary interest, not only because of their structural features and extremely rich coordination chemistry, but also due to their versatile pharmacological properties such as antimicrobial,1,2 antiviral,3,4 anticonvulsant,5,6 and carcinostatic activity. 7 Particular attention has been paid to thiosemicarbazones derived from N-heterocycles with α-carbonyl groups. This class of thiosemicarbazones and the corresponding metal complexes exhibit pronounced biological activity. Metals bonded to atoms such as N, O, and S can form a chelate ring, similar to the interactions between metal ions and many important biological molecules such as proteins, enzymes, and DNA.
In this context, pyruvic acid is one of the α-keto acids of the Krebs cycle and plays a critical role in important biochemical processes such as the metabolism of proteins, fats, carbohydrates, and amino acids in humans and animals.8,9 Pyruvic acid is a natural product found in Populus tremula, Macrobrachium nipponense, and other organisms. Pyruvic acid thiosemicarbazone and its complexes exhibit various types of biological activity: antimicrobial,10,11 antifungal,12,13 cytotoxic, 14 and antitumor. 15 The complexes were more active than the uncoordinated thiosemicarbazone and even exceeded the activity of medicinal compounds used as standards. Also, it was found that this thiosemicarbazone inhibits soluble ornithine carbamoyltransferase in vitro and the enzyme in a mitochondria-rich cell fraction. 16
The α-ketoamide moiety is a key structural unit present in many natural and non-natural products with a wide range of interesting biological activities. For example, they are effective as a reversible covalent inhibitors against cysteine proteases. Pyruvamides are representative of this class of substances. They also manifest different types of biological activity, which have been studied in vitro and in vivo. For example, N,N-diethyl-2-oxopropanamide has anti-inflammatory and antiexcitotoxic effects. It effectively blocks neutrophils that accumulate around damaged brain tissue and exacerbate tissue damage during a stroke. 17 N,N-Diisopropyl-2-oxopropanamide manifests anti-inflammatory and neuroprotective activity and is a potential therapeutic agent for improving ischemic brain injury and other pathologies associated with inflammation. 18
Thus, it is of interest to study thiosemicarbazones of pyruvamides and their coordination compounds, which are limited to only a few reports.19–21
Recent developments in the medicinal field report a number of diseases associated with free radicals. 22 Such free radicals are normally neutralized by efficient systems in the body that include antioxidant enzymes and nutrient-derived antioxidant small molecules. In healthy individuals, a delicate balance exists between free radicals and antioxidants. In some pathologic conditions, oxidative stress causes the levels of antioxidants to fall below normal. Antioxidant supplements for such conditions are expected to be of benefit. 23 Thiosemicarbazones and their coordination compounds manifest antioxidant activity, often exhibiting values that exceed the activity of medicines used as antioxidants.24–27 Studies of the antioxidant activity of pyruvic acid have shown that it has potential for controlling inflammation and preventing diseases caused by oxidative stress. 28
The aim of the present investigation is the synthesis of 2-[1-oxo-1-(piperidin-1-yl)propan-2-ylidene]-N-(prop-2-en-1-yl)hydrazinecarbothioamide (1-(piperidin-1-yl)propane-1,2-dione N4-allylthiosemicarbazone) (Figure 1) and its 3d metal coordination compounds, which include bidentate N-heteroaromatic bases such as 1,10-phenanthroline (1,10-Phen) and 2,2′-bipyridine (2,2′-Bpy), along with studies of the antioxidant activities.
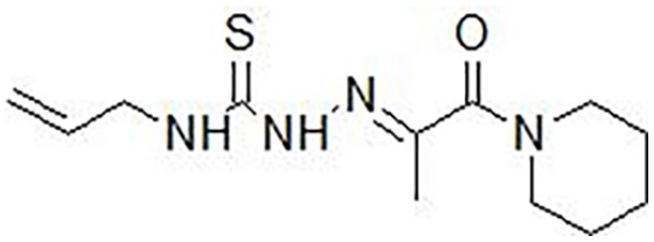
Structure of 2-[1-oxo-1-(piperidin-1-yl)propan-2-ylidene]-N-(prop-2-en-1-yl)hydrazinecarbothioamide (
Results and discussion
Synthesis and characterization
The new N4-allylthiosemicarbazone (
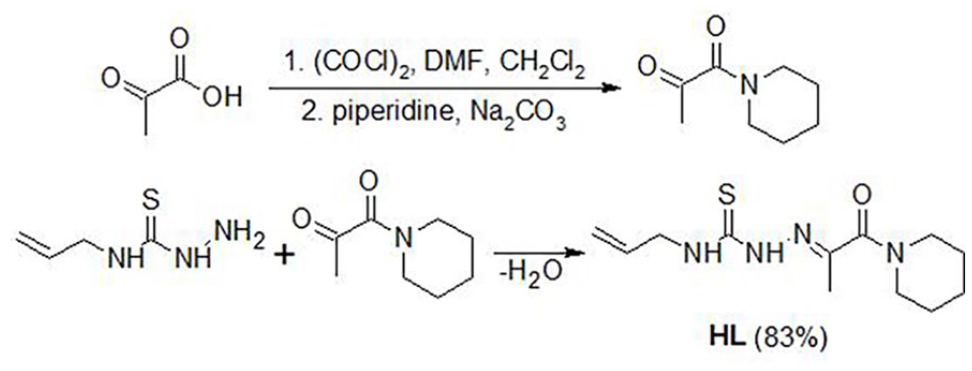
Synthesis of 2-[1-oxo-1-(piperidin-1-yl)propan-2-ylidene]-N-(prop-2-en-1-yl)hydrazinecarbothioamide (
Complex

The synthesis of coordination compounds
Spectroscopic studies
The FTIR spectra of
Selected FTIR absorption bands of

The disappearance of the ν(С=S) group indicates that the
On these bases, it can be proved that the
Structure description of HL and complex 1
X-ray single-crystal structure analysis revealed that N4-allylthiosemicarbazone
Crystal and structure refinement data for

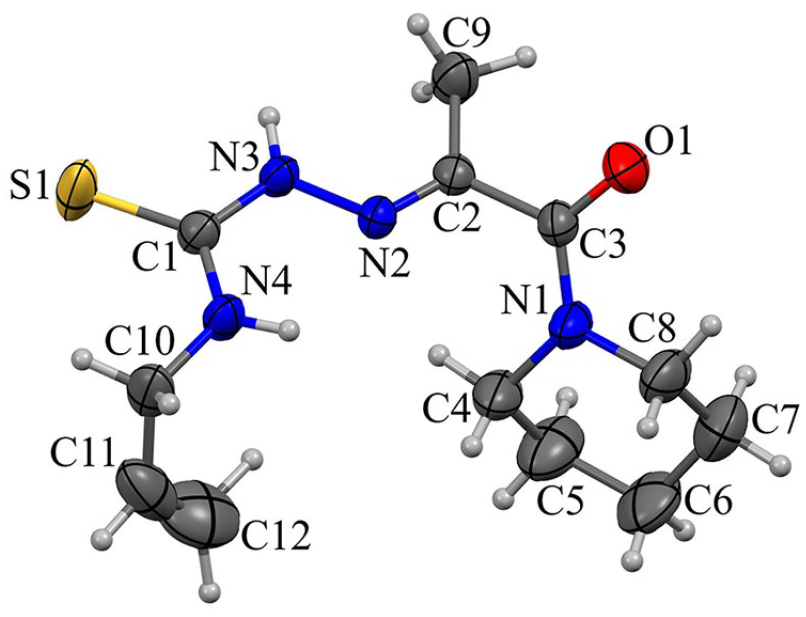
X-ray crystal structure of
Selected bond lengths and angles in

Intermolecular N–H···S and N–H···O hydrogen bonds occur between molecules in supramolecular corrugated layers parallel to the ac crystallographic plane (Table 4, Figure 3).
Hydrogen bond distances (Å) and angles (°) in


Fragment of the crystal packing of the corrugated layers parallel to ac crystallographic plane of
Complex
The Cu1 atom in complex
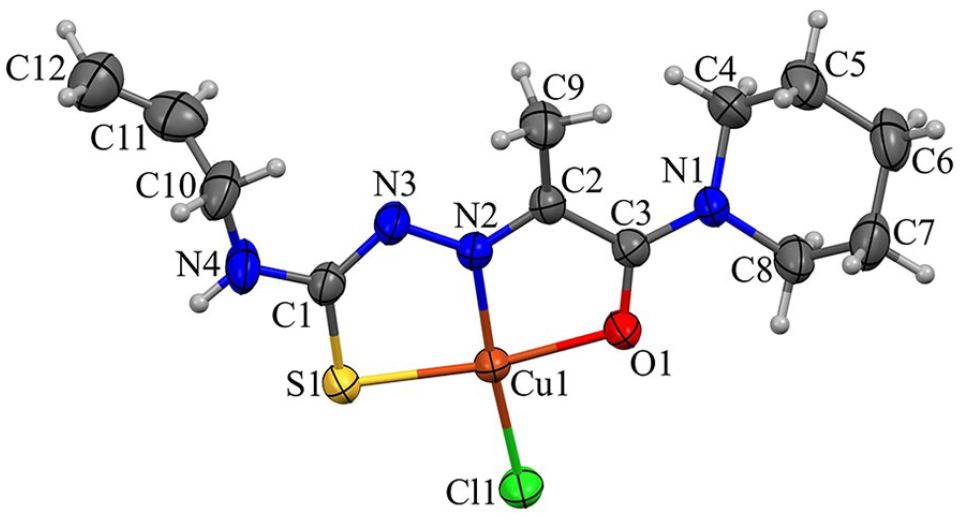
X-ray crystal structure of complex
The complexes are linked via N–H···Cl and C–H···S1 hydrogen bonds in supramolecular chains running along the crystallographic b-axis (Table 4, Figure 5). The antiparallel complexes from neighboring chains reveal stacking interactions of the metallacycles. 34 These π-π stacking interactions unite chains in supramolecular layers parallel to the (bc) plane. The interplanar separation between mean planes of overlapping fragments equals 3.421 Å, while the centroid···centroid distance is 3.547 Å and the Cu···Cu distance within stacked complexes is 4.690 Å.

The supramolecular architecture in the crystal structure of complex
Antioxidant activity
The antioxidant activity of the compounds was determined by the ABTS+• method (Table 5). The uncoordinated thiosemicarbazone
Antioxidant activity of

Among all the coordination compounds, complex
Conclusion
The new 1-(piperidin-1-yl)propane-1,2-dione N4-allylthiosemicarbazone and its six complexes have been prepared and characterized. The structures of the initial ligand
The studied complexes and ligand show antioxidant activity against ABTS+• cation radicals. The uncoordinated ligand
Experimental
Materials and methods
3-Isothiocyanatoprop-1-ene, hydrazine hydrate, pyruvic acid, oxalyl chloride, sodium carbonate, dichloromethane, dimethylformamide, piperidine, copper(II) chloride dihydrate, copper(II) nitrate trihydrate, nickel(II) nitrate hexahydrate, iron(III) chloride hexahydrate, 1,10-phenanthroline, and 2,2′-bipyridine were obtained from Sigma-Aldrich. The 1H and 13C NMR spectra were recorded on a Bruker DRX-400. Chemical shifts are measured in ppm relative to tetramethylsilane, as solvents were used CDCl3. FTIR spectra were obtained for powders on a Bruker ALPHA FTIR spectrophotometer at room temperature in the range of 4000–400 cm−1. Elemental analysis was performed similar to the literature procedure. 40 The resistance of solutions of complexes in methanol (20 °C, c 0.001 M) was measured using an R-38 rheochord bridge.
N-(Prop-2-en-1-yl)hydrazinecarbothioamide (N4-allyl-3-thiosemicarbazide) was synthesized by the reaction between 3-isothiocyanatoprop-1-ene (allyl isothiocyanate) and hydrazine hydrate. 41
1-(Piperidin-1-yl)propane-1,2-dione was synthesized by the method described in the literature, 42 with some modifications. Pyruvic acid (8.80 g and 0.100 mmol) was dissolved in CH2Cl2 (5 mL) and placed in a flat-bottomed flask. Oxalyl chloride (15.24 g and 0.120 mmol) was dissolved in CH2Cl2 (10 mL) and was added dropwise to the reaction mixture with stirring at 0 °C. Next, 3 drops of dimethylformamide were added as a catalyst, and a condenser fitted with a drying tube containing calcium chloride was attached to the flask. The reaction mixture was stirred and heated for 1.5 h to afford a yellow oil. A suspension of piperidine (8.50 g and 0.100 mmol), sodium carbonate (10.60 g and 0.100 mmol), and CH2Cl2 (10 mL) was stirred at 0 °C. 2-Oxopropanoyl chloride was added dropwise to the obtained cooled suspension, and the mixture was stirred at room temperature for 1 h. The mixture was filtered, washed with water (3 × 100 cm3), and dried in the air to afford the crude product as a mobile yellow oil.
2-[1-Oxo-1-(piperidin-1-yl)propan-2-ylidene]-N-(prop-2-en-1-yl)hydrazinecarbothioamide (1-(piperidin-1-yl)propane-1,2-dione 4-allylthiosemi-carbazone) (
The product was synthesized by the reaction between 1-(piperidin-1-yl)propane-1,2-dione (3.10 g and 20.0 mmol) and N-(prop-2-en-1-yl)hydrazinecarbothioamide (2.62 g and 20.0 mmol) in ethanol with stirring and heating. The obtained pale-yellow precipitate was filtered, washed with a small amount of ethanol, and dried in air. The solid product was crystallized from ethanol to give pale-yellow single crystals that were suitable for X-ray diffraction. The crystals were isolated by filtration. Yield: 83%; m.p. 137–139 °C. 1H NMR (Supplemental Figure S1) (400 MHz, CDCl3): δ = 8.71 (br. s, 1H, NH), 7.46 (br. s, 1H, NH), 5.89 (m, 1H, СH), 5.20 (m, 2H, СH2), 4.30 (m, 2H, NH-CH2), 3.50 (m, 2 × 2H, CH2piperidine), 2.11 (s, 3H, CH3), and 1.62 (m, 3 × 2H, CH2piperidine); 13C NMR (Supplemental Figure S2) (100 MHz, CDCl3): δ = 178.20 (C=S), 165.33 (C=O), 143.47 (C=N), 132.88 (CH=CH2), 117.37 (CH2=), 46.93 (Callyl), 45.54, 45.51, 26.02, 24.44, 20.35 (Cpiperidine), and 14.19 (CH3); FTIR: 3315, 3288 (NH), 1632 (C=O), 1620 (C=N), and 1362 (C=S) cm−1. Elemental analysis calcd (%) for C12H20N4OS: C, 53.70; H, 7.51; N, 20.88; and S, 11.95. Found: C, 53.65; H, 7.47; N, 20.80; and S, 12.00.
Synthesis of the complexes
[Cu(L)Cl] (1)
An ethanolic solution of N4-allylthiosemicarbazone
[Cu(L)NO3] (2)
An ethanolic solution of N4-allylthiosemicarbazone
[Cu(1,10-Phen)(L)]NO3 (3)
An ethanolic solution of N4-allylthiosemicarbazone
[Cu(2,2′-BPy)(L)]NO3 (4)
An ethanolic solution of N4-allylthiosemicarbazone
[Ni(HL)2](NO3)2 (5)
An ethanolic solution of N4-allylthiosemicarbazone
[Fe(L)2]Cl (6)
An ethanolic solution of N4-allylthiosemicarbazone
X-ray crystallography
The diffraction data of crystals of
Biological activity
The ABTS+• radical cation decolorization assay is a method for screening of antioxidant activity applicable to both lipophilic and hydrophilic antioxidants. 46 Standard solutions of ABTS+• radical cations and the tested substances, and the conditions for the spectrophotometric measurements and calculations of inhibition were made as described. 47 The 10 mM stock solutions of the studied compounds and trolox that was used as the reference compound were prepared by dissolving 10 μmol of the corresponding compound in dimethylsulfoxide (DMSO) 1 mL. The 1, 10, 100, and 1000 μM solutions were prepared by the dilution of stock solutions with DMSO. After that, 20 μL of each solution was transferred to a 96-well microtiter plate, and 180 μL of ABTS+• working solution was added to give solutions with final concentrations of the tested compounds of 0.1, 1, 10, and 100 μM, respectively. All tested samples were dispensed with a dispense module hybrid reader (BioTek). The decrease in absorbance at 734 nm was measured exactly after 30 min of incubation at 25 °C. DMSO was used as negative control. Blank samples were run by solvent without ABTS+•. The measurement was made by hybrid reader (Synergy H1, BioTek). All tests were performed in triplicate, and the obtained results were averaged.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231216422 – Supplemental material for A new thiosemicarbazone and its 3d metal complexes: Synthetic, structural, and antioxidant studies
Supplemental material, sj-docx-1-chl-10.1177_17475198231216422 for A new thiosemicarbazone and its 3d metal complexes: Synthetic, structural, and antioxidant studies by Ianina Graur, Tatiana Bespalova, Vasilii Graur, Victor Tsapkov, Olga Garbuz, Elena Melnic, Pavlina Bourosh and Aurelian Gulea in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was fulfilled with financial support from the National Agency for Research and Development projects 20.80009.5007.10 and 20.80009.5007.15.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
