Abstract
Two sets of novel indole-based thiosemicarbazone systems
Introduction
Indoles are important heterocyclic compounds which have been the subject of many synthetic studies.1–3 Due to the presence of the indole core in many natural products, studies have been directed towards investigations of the biological properties of natural indolic compounds, and a range of medical uses has been identified.4–7 One of the most important amino acids for the human diet, 8 L-tryptophan contains an indole nucleus. Moreover, vinblastine and vincristine are examples of indole-containing systems that show very potent anticancer activity. 9 Apart from the biological importance of indole systems, most importantly, activated indole compounds have been used in many synthetic studies as valuable intermediates.10–12 A range of reactions such as formylation or halogenation can be performed at different locations on indole systems, while the attachment of important functional groups generates valuable building blocks. These intermediates can be readily converted into other functional groups allowing the production of indole-based macrocyclic compounds.13–15
Thiosemicarbazones are important classes of Schiff base ligands due to the presence of a R1R2C=N-NH-(C=S)-NR3R4 moiety generated by the reaction between an aldehyde and the thiosemicarbazides.16–18 The conjugated N-N-S system provides an important therapeutic potential to these thiosemicarbazone-based systems, which can interact with biomolecules.19–21 Due to their biological activity, thiosemicarbazones appear in a large number of medically valuable compounds and considerable effort has been devoted towards the synthesis of these compounds. Thiosemicarbazones have also attracted the attention of researchers due to their high affinity for metal ions such as iron, zinc and copper to produce metal complexes, which are also important biological targets for interactions with nucleic acids.22–24
The design and synthesis of analogues that are structurally derived from the combination of a biologically active indole and a thiosemicarbazide has yielded new indole thiosemicarbazone systems as potential therapeutic agents.25,26 An intense literature search revealed that indole thiosemicarbazone scaffolds have been identified as promising compounds for antioxidant studies due to their ability to donate hydrogen or an electron to acceptors (i.e. 2,2-diphenyl-1-picrylhydrazyl hydrate [DPPH]) and to reduce the production of free radicals.27,28 It has also been suggested that the accumulation of the bioavailable transition metals Fe, Cu and Zn is responsible for Alzheimer’s disease (AD), since the self-aggregation of amyloid-β (Aβ) via metal peptide chelation forms senile plaques, which are deposited outside of neurons. 29 The presence of conjugated N-N-S tridentate systems on thiosemicarbazones would reduce the accumulation of these metals, and their chelation ability would be a potential therapy for the treatment of AD. 29
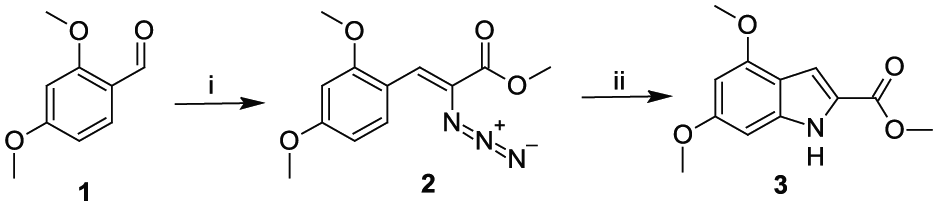
In the present work, we aimed to prepare two sets of new thiosemicarbazones with different substituents on the 4,6-dimethoxyindole nucleus. The Hemetsberger reaction was chosen as the synthetic preparation method for the designated indole compounds since it readily forms derivatives of 2-carboxyindoles from aromatic or heterocyclic aldehydes.30–32 The position 2 was chosen as the first location on the indole ring for the preparation of the first set of compounds. 4,6-Dimethoxyindole-2-carbaldehyde 33 was prepared by reduction and oxidation of methyl 4,6-dimethoxyindole-2-carboxylate, respectively. The dimethoxyindole backbone was chosen since the synthetic strategy can easily accommodate electron-donating substituents34–36 and there has been significant interest in heterocyclic aromatic systems derived from dimethoxyindoles due to their possible biological and pharmacological activities.37–39 The nucleophilic reactivity of 4,6-dimethoxyindole was reported in our previous work 40 by utilising the Vilsmeier–Haack formylation. 41 The formylation was found to occur selectively at the 7-position and the products were subsequently converted into 7-thiosemicarbazones as the second set of compounds.
Eight new 4,6-dimethoxyindole thiosemicarbazones
Results and discussion
Chemistry
Methyl 4,6-dimethoxyindole-2-carboxylate
Reagents and conditions: i: N3CH2CO2Me, Anhyd. MeOH <–10 °C, 4 h, 78%, ii: Xylene reflux, 6 h.

Reagents and conditions: i: POCl3, DMF, −10 °C overnight. a: LiAlH4/THF, −10 °C 1 h, rt 3 h b: MnO2, CH2Cl2, reflux, 24 h.

Reagents and conditions: i: EtOH, a few drops of AcOH, overnight, rt.
Preparation of methyl 4,6-dimethoxyindole-2-carboxylate 3
According to the literature, the preparation of activated indole scaffolds has been achieved by numerous methods. The Hemetsberger reaction is potentially one of the most important methods for the synthesis of indoles. In our work, methyl 4,6-dimethoxyindole-2-carboxylate was afforded via a vinyl azide, which is generated by the condensation of 2,4-dimethoxybenzaldehyde (
Preparation of indole carbaldehydes
Methyl 7-formyl-4,6-dimethoxyindole 4
The Vilsmeier–Haack formulation reaction is a very useful process for installing an aldehyde, which can be used for further synthetic transformations. In the current work, the desired formylated methyl 4,6-dimethoxyindole-2-carboxylate was synthesised by treatment of the methyl indole ester
4,6-Dimethoxyindole-2-carbaldehyde 6
The compound
The preparation of desired dimethoxyindole thiosemicarbazones
Following preparation of the targeted 4,6-dimethoxyindole 2-carbaldehyde and methyl 7-formyl-4,6-dimethoxyindole-2-carboxylate, a range of thiosemicarbazides

The 1H NMR spectrum of compound
The 1H NMR spectrum of compound
The synthetic strategy was extended to the synthesis of 4,6-dimethoxyindole-2- and -7-thiosemicarbazone systems containing methyl and ethyl substituents at R2 (Scheme 3). Hence, 7-formyl-dimethoxyindole
The 1H NMR spectrum of compound
In the 1H NMR spectrum of compound
The same synthetic route was applied to the carbaldehydes
The formation of the desired methyl 4,6-dimethoxyindole-7-thiosemicarbazone
The formation of the targeted compounds was also confirmed by 13C NMR spectroscopy, displaying the C=S carbon signals between 175.0 to 180.0 ppm. In addition, the azomethine carbon (C=N) atom resonated around 130–140 ppm. Infrared (IR) spectroscopy showed the expected absorption bands for identification of the designated compounds. The C=S stretching bands appeared around 1650–1700 cm−1, while the NH and NH2 stretches were found around 3300–3450 ppm as broad bands. The azomethine C=N stretching occurred around 1500–1550, confirming the Schiff base condensation reactions.
Biological studies
The novel methyl 4,6-dimethoxyindole-7-thiosemicarbazones
NA: not active.
Values expressed are means ± S.D. of three parallel measurements, (p< 0.05).
200 µg/mL.
Positive control.
DPPH free radical scavenging assay
Figure 1 shows the DPPH radical scavenging activity of the eight 4,6-dimethoxyindole thiosemicarbazone compounds,
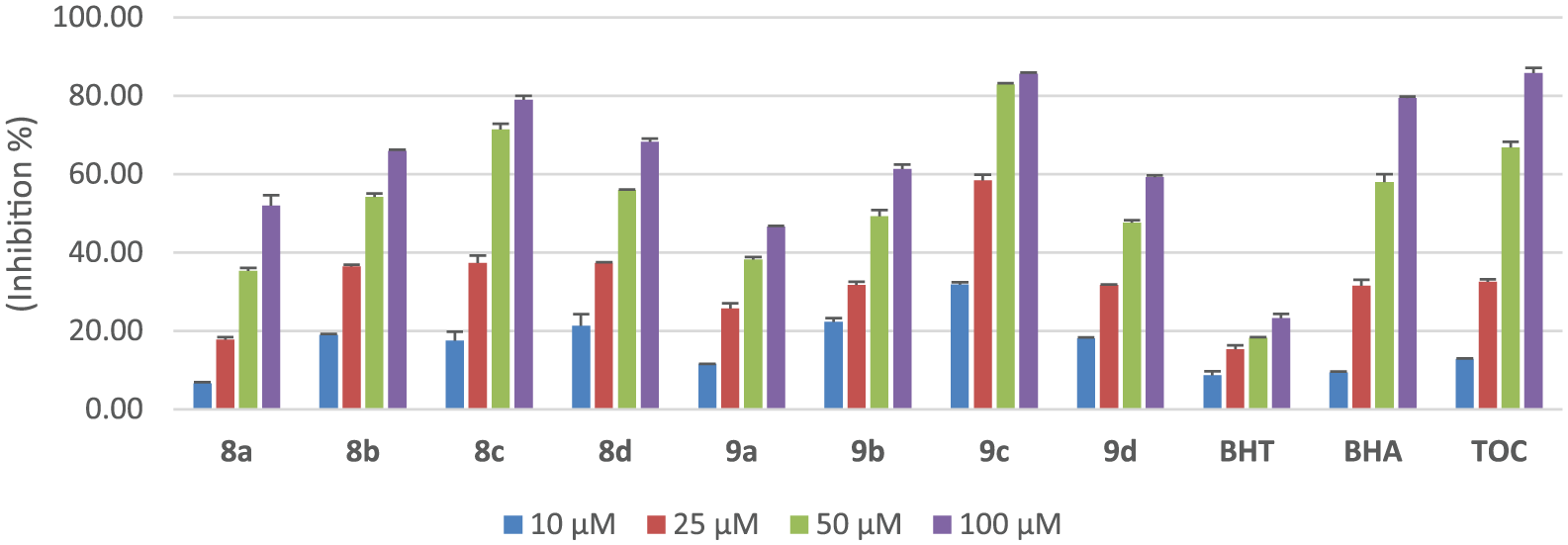
Inhibition (in %) of the DPPH free radical scavenging activity of compounds
ABTS cation radical decolorization assay
The initial cationic radical scavenging activity assay was carried out on concentrations from 10 to 100 µM and the novel 4,6-dimethoxyindole-2- and -7-thiosemicarbazones displayed promising potency and further investigations were carried out using lower concentrations (1–10 µM) and the Figure 2 shows the concentration-dependent behaviour of compounds

Inhibition (in %) of the ABTS cation radical scavenging activity of compounds
CUPRAC
Figure 3 shows the absorbance values obtained from the CUPRAC assay for the synthesised compounds

Absorbance values for CUPRAC of compounds
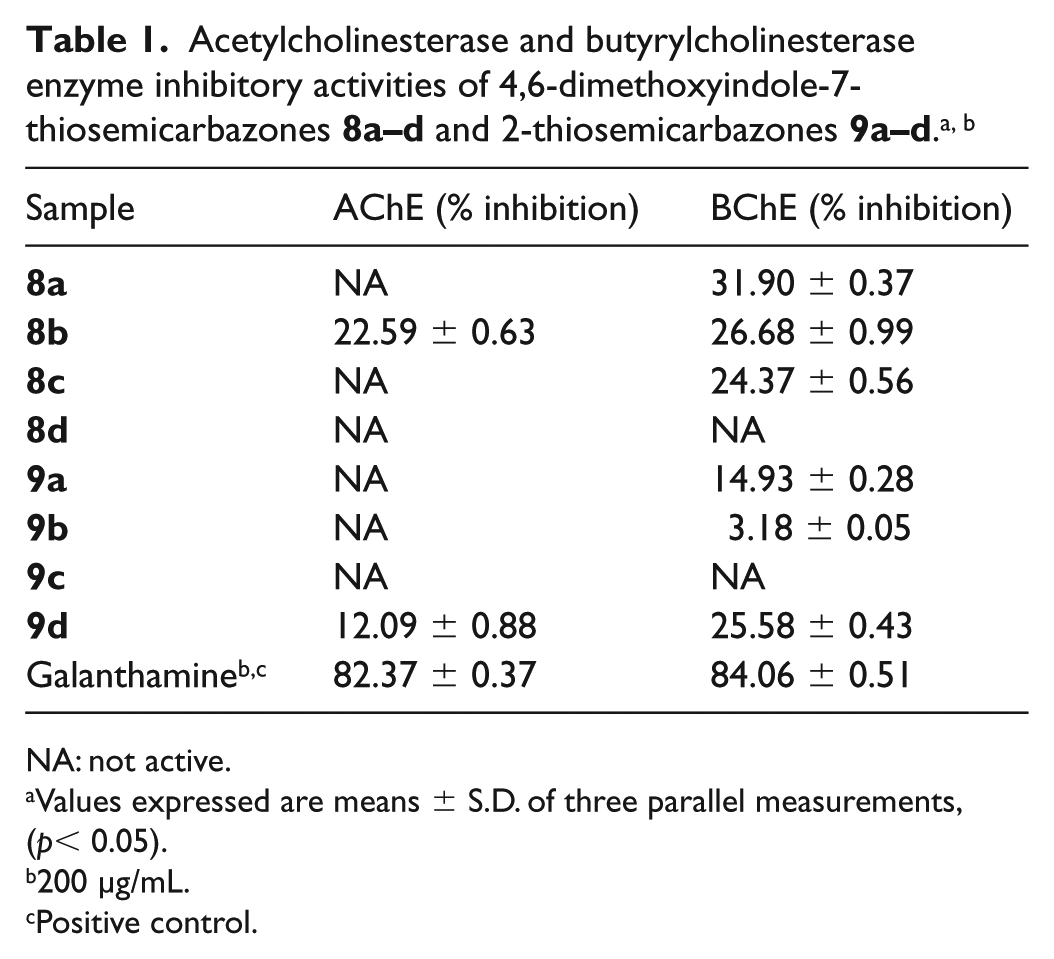
Determination of anticholinesterase activity
The ACh and BCh enzyme inhibitory activities were evaluated for determination of the therapeutic potential of the 4,6-dimethoxyindole-2- and -7-thiosemicarbazones for the treatment of AD. As expected, the positive control galanthamine showed very potent inhibition both on ACh and BCh with values of 82.37% and 84.06%, respectively. In contrast, most of the synthesised compounds demonstrated much less inhibition and no enzyme inhibitory activity for ACh. In the case of compounds
Conclusion
Indole thiosemicarbazone systems were identified as valuable synthetic intermediates for the preparation of complex structures and important ligand systems for metal complexes. Moreover, they were determined as promising targets for antioxidant and anticholinesterase studies due to their ability to donate hydrogen or electron. This work describes the synthesis of two series of indole thiosemicarbazone systems via Schiff base reactions between the corresponding carbaldehydes and thiosemicarbazides. The method involves the preparation of methyl 4,6-dimethoxyindole-2-carboxylate
Materials and methods
Chemicals and physical measurements
All commercially available reagents and the standards used for the biological assays were purchased from Sigma Aldrich and used without further purification. The synthetic procedures have been reported for the final compounds as general methods and other procedures have been given individually with appropriate references. All reactions were monitored by thin layer chromatography using Merck TLC Silica gel 60 F254. Silica gel 60 (particle size: 0.040–0.063 mm, 230–400 mesh ASTM) for column chromatography was obtained from Merck. 1H and 13C NMR spectra were recorded for all compounds either in DMSO or CDCl3 solutions on a Bruker DPX 400 MHz spectrometer at 300 K. Chemical shifts are reported in parts per million and referenced to the residual solvent peak (DMSO-d6: 1H 2.50 ppm, 13C 39.52 ppm and CDCl3: 1H 7.24 ppm, 13C 77.23 ppm). Coupling constants (J) are reported in Hertz (Hz). Standard conventions indicating multiplicity were used: m = multiplet, t = triplet, d = doublet, s = singlet, dd = doublet of doublets. Infrared spectra were recorded using a Thermo Scientific Nicolet IS10 FT-IR spectrometer between 600 and 4000 cm−1. Melting points were measured using a Mel-Temp melting point apparatus and are uncorrected high-resolution mass spectra [ESI] were recorded on an Orbitrap LTQ XL ion trap mass spectrometer (Thermo Scientific, Waltham, MA, USA) using a nanospray (nano-electrospray) ionisation source.
Biological studies
Antioxidant assays
DPPH free radical scavenging activity
The assay was performed according to the methods42,43 with minor modifications. BHT, BHA and α-TOC were used as standards for comparison.
ABTS cation radical decolorization activity
The method was followed as in the literatures43,44 with the minor changes. BHA, BHT and α-TOC were used as standards.
CUPRAC
The procedure was followed as in the literature45,46 with minor modifications. BHA, BHT and α-TOC were used as standards.
Anticholinesterase activity determination method
ACh and BCh enzyme inhibitory activities were measured by the slightly modified spectrophotometric method reported in the literature.46,47 Galanthamine was used as the standard. DMSO was used as the solvent to dissolve the samples and controls.

where A0 is the absorbance of the control and A1 is the absorbance in the presence of the sample.
Statistical analysis
The results were mean ± SD of three parallel measurements. All statistical comparisons were made by means of Student’s t test; p values< 0.05 were regarded as significant.
Chemical synthesis
Preparation of compounds 3, 4, 5 and 6
The synthetic procedures reported in the previous study
33
was followed for the preparation of methyl 4,6-dimethoxyindole-2-carboxylate
General procedure for the preparation of thiosemicarbazones
The carbaldehyde (
Methyl 7-[(E)-(2-carbamothioylhydrazinylidene)methyl]-4,6-dimethoxy-1H-indole-2-carboxylate (8a)
The title compound was synthesised following the general procedure using methyl 7-formyl-4,6-dimethoxyindole-2-carboxylate (
Methyl 4,6-dimethoxy-7-{(E)-[2-(methylcarbamothioyl)hydrazinylidene]H-indole-2-carboxylate (8b)
The title compound was synthesised following the general procedure using methyl 7-formyl-4,6-dimethoxyindole-2-carboxylate (
Methyl 7-{(E)-[2-(dimethylcarbamothioyl)hydrazinylidene]H-indole-2-carboxylate (8c)
The title compound was synthesised following the general procedure using methyl 7-formyl-4,6-dimethoxyindole-2-carboxylate (
Methyl 7-{(E)-[2-(ethylcarbamothioyl)hydrazinylidene]H-indole-2-carboxylate (8d)
The title compound was synthesised following the general procedure using methyl 7-formyl-4,6-dimethoxyindole-2-carboxylate (
(2Z)-2-[(4,6-dimethoxy-1H-indol-2-yl)methylidene]hydrazine-1-carbothioamide (9a)
The title compound was synthesised following the general procedure using 4,6-dimethoxyindole-2-carbaldehyde (
(2Z)-2-[(4,6-dimethoxy-1H-indol-2-yl)methylidene]-N-methylhydrazine-1-carbothioamide (9b)
The title compound was synthesised following the general procedure using 4,6-dimethoxyindole-2-carbaldehyde (
(2Z)-2-[(4,6-dimethoxy-1H-indol-2-yl)methylidene]-N, N-dimethylhydrazine-1-carbothioamide (9c)
The title compound was synthesised following the general procedure using 4,6-dimethoxyindole-2-carbaldehyde (
(2Z)-2-[(4,6-dimethoxy-1H-indol-2-yl)methylidene]-N-ethylhydrazine-1-carbothioamide (9d)
The title compound was synthesised following the general procedure using 4,6-dimethoxyindole-2-carbaldehyde (
Supplemental Material
supplemental_material – Supplemental material for Synthesis and characterisation of novel 4,6-dimethoxyindole-7- and -2-thiosemicarbazone derivatives: Biological evaluation as antioxidant and anticholinesterase candidates
Supplemental material, supplemental_material for Synthesis and characterisation of novel 4,6-dimethoxyindole-7- and -2-thiosemicarbazone derivatives: Biological evaluation as antioxidant and anticholinesterase candidates by Murat Bingül in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.