Abstract
In this study, a series of 4-(3-benzylbenzo[d]thiazol-2(3H)-ylidene)-cyclohexa-2,5-dien-1-one derivatives are efficiently synthesized in excellent yields by reactions of 2-chlorobenzothiazole, benzyl bromides, and phenols in acetonitrile under reflux and catalyst-free conditions in the presence of triethylamine for 2 h. The structures of the products are characterized by NMR, IR, EI-MS, and elemental analyses.
Keywords

Introduction
Multicomponent reactions (MCRs) are a type of organic reaction that consists of two or more synthetic steps that are carried out without isolation of any intermediate; therefore, they help to save time, money, energy, and raw materials.1,2 These reactions are economically and environmentally beneficial to industry and have attracted much attention. On the other hand, they are perfectly applicable to organic synthesis and agrochemistry.3,4 Benzothiazole is a bicyclic heterocyclic compound in which benzene is fused with the thiazole ring at the 4, 5-positions, containing both sulfur and nitrogen electron-rich heteroatoms in its ring.5,6 Benzothiazoles and their analogues are important building blocks and synthetic intermediates in the synthesis of new drugs, pesticides, and dyes, and are utilized in medicinal chemistry.7,8 Derivatives of benzothiazole have been reported to show a wide spectrum of pharmacological activities and agrochemical applications like anti-convulsant,
9
antiallergy,
10
antifungal,
10
anti-microbial,
11
anti-oxidant,
12
anti-inflammatory,
12
anti-diabetic,
13
neuroprotective, and many others
14
(Figure 1). On the other hand, benzothiazoles are also widely used in a variety of consumer and industrial products such as nonlinear optics (NLOs), organic light-emitting diodes (OLEDs), fluorescent probes, luminescence, chemosensors,15–17 and liquid crystals.
18
In this paper, the results of studies involving new, easy, and catalyst-free multicomponent reactions of 2-chlorobenzothiazole (
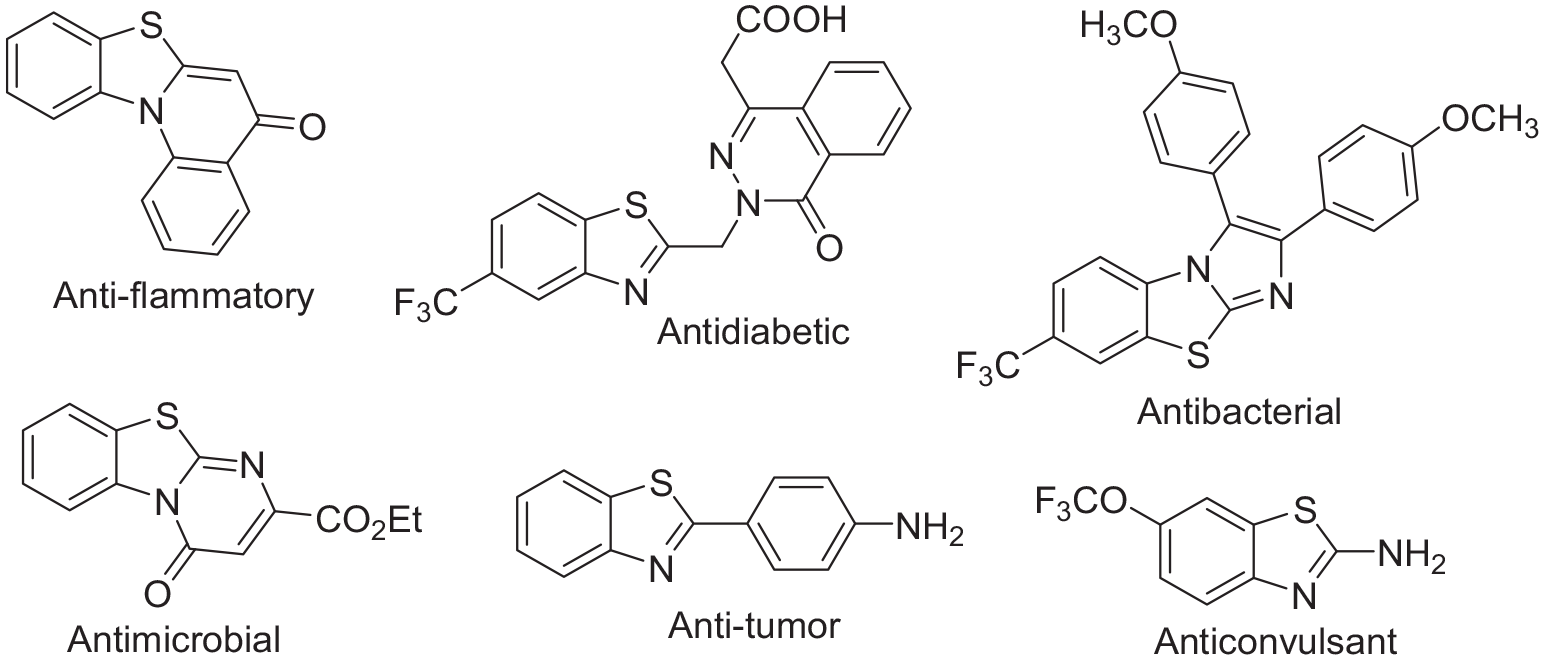
Chemical structures of some drugs containing benzothiazole nuclei.

Proposed mechanism for the preparation of compounds 4.

Chemical structures of compounds
Results and discussion
In continuation of our studies on the application of both benzothiazole and phenol in multicomponent reactions,19–22 a new, simple, and efficient method for the synthesis of 4-(3-benzylbenzo[d]thiazol-2(3H)-ylidene)-cyclohexa-2,5-dien-1-one derivatives
Conclusion
In this study, the target compounds containing both benzothiazole and phenol moieties have been synthesized with excellent yields via a convenient and efficient method, under catalyst-free conditions at reflux for 2 h. On the other hand, simple work-up, short reaction time, and easily available starting materials are the other advantages of this procedure with acetonitrile as the solvent.
Experimental
All the chemicals used in this work were purchased from Merck (Germany) and Fluka (Buchs, Switzerland) and were used without further purification. Melting points were measured on an Electrothermal 9100 apparatus. The mass spectra were recorded on a GCMS-QP5050A mass spectrometer operating at an ionization potential of 70 eV. Elemental analyses for C, H, and N were performed using a Heraeus CHN-O-Rapid analyzer. The 1H and 13C NMR spectra were recorded using a BRUKER DRX-250 AVANCE instrument with CDCl3 as the solvent and TMS as the internal standard at 250.1 and 62.9 MHz, respectively.
Preparation of compound 6a ; typical procedure
A mixture of 2-chlorobenzothiazole (
4-(3-Benzylbenzo[d]thiazol-2(3H)-ylidene)-2,6-dimethylcyclohexa-2,5-dien-1-one (4a )
Yellow powder, yield 91%, 0.31 g; m.p. 142–144 °C. IR (KBr): 1748 (C=O) cm‒1. MS (EI): m/z (%) = 345 (M+, 6), 330 (M-Me, 91), 315 (M-2Me, 84), 268 (M-Ph, 59), 254 (M-C7H7, 63), 91 (C7H7, 46). Anal. calcd for C22H19NOS (345.39): C, 76.50; H, 5.54; N, 4.05; found: C, 76.52; H, 5.59; N, 3.94. 1H NMR (250.1, CDCl3): δ 1.92 and 1.94 (6H, 2s, 2Me), 4.17 (2H, s, CH2), 6.42 (1H, s, CH=CMe), 6.43 (1H, s, CH=CMe), 6.62–7.46 (9H, m, 9ArH). 13C NMR (62.9 MHz, CDCl3): δ 15.20 and 15.21 (2Me), 51.6 (CH2), 105.6 (S-C=C), 113.7, 114.2, 116.3, 118.8, 125.2, 127.4, 128.5, 129.6, 132.6 (CH=CMe), 132.8 (CH=CMe), 143.7 (CH=CMe), 143.9 (CH=CMe), 144.1, 145.2 and 149.3 (S-C=C), 185.4 (C=O).
2,6-Dimethyl-4-(3-(2-methylbenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4b )
Yellow powder, yield 91%, 0.33 g; m.p. 135–137 °C. IR (KBr): 1749 (C=O) cm‒1. MS (EI): m/z (%) = 359 (M+, 7), 344 (M-Me, 94), 329 (M-2Me, 86), 268 (M-C7H7, 70), 254 (M-C8H9, 62), 253 (M-C7H7 and Me, 59), 105 (C8H9, 47). Anal. calcd for C23H21NOS (359.41): C, 76.86; H, 5.89; N, 3.90; found: C, 76.93; H, 5.92; N, 3.80. 1H NMR (250.1, CDCl3): δ 1.93, 1.94 and 2.12 (9H, 3s, 3Me), 4.16 (2H, s, CH2), 6.41 (1H, s, CH=CMe), 6.43 (1H, s, CH=CMe), 6.61–7.44 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 15.19 and 15.20 (2Me), 23.4 (Me), 51.6 (CH2), 105.8 (S-C=C), 113.8, 114.4, 117.2, 119.8, 121.4, 125.2, 126.3, 127.6, 128.4, 128.6, 132.7 (CH=CMe), 132.8 (CH=CMe), 143.8 (CH=CMe), 143.9 (CH=CMe), 144.7, 145.6 and 149.4 (S-C=C), 185.4 (C=O).
2,6-Dimethyl-4-(3-(2-nitrobenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4c )
Pale yellow powder, yield 90%, 0.35 g; m.p. 152–154 °C. IR (KBr): 1749 (C=O) cm‒1. MS (EI): m/z (%) = 390 (M+, 6), 375 (M-Me, 93), 268 (M-C6H4NO2, 72), 254 (M-C7H6NO2, 51), 253 (M-C6H4NO2 and Me, 59), 136 (C7H6NO2, 54). Anal. calcd for C22H18N2O3S (390.38): C, 67.69; H, 4.65; N, 7.17; found: C, 67.72; H, 4.59; N, 7.10. 1H NMR (250.1, CDCl3): δ 1.93 and 1.95 (6H, 2s, 2Me), 4.20 (2H, s, CH2), 6.43 (1H, s, CH=CMe), 6.44 (1H, s, CH=CMe), 6.60–7.46 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 15.18 and 15.21 (2Me), 52.0 (CH2), 105.7 (S-C=C), 114.7, 115.6, 117.9, 119.8, 123.6, 125.7, 127.4, 129.1, 130.3, 132.9 (CH=CMe), 133.0 (CH=CMe), 140.1, 144.0 (CH=CMe), 144.1 (CH=CMe), 146.8, 147.7 and 149.4 (S-C=C), 185.5 (C=O).
4-(3-Benzylbenzo[d]thiazol-2(3H)-ylidene)-2,6-dimethoxycyclohexa-2,5-dien-1-one(4d )
Yellow powder, yield 92%, 0.35 g; m.p. 146–148 °C. IR (KBr): 1750 (C=O) cm‒1. MS (EI): m/z (%) = 377 (M+, 8), 362 (M-Me, 95), 347 (M-2Me, 89), 346 (M-OMe, 68), 300 (M-Ph, 70), 269 (M-Ph and OMe, 49), 91 (C7H7, 57). Anal. calcd for C22H19NO3S (377.39): C, 70.02; H, 5.07; N, 3.71; found: C, 70.05; H, 5.05; N, 3.80. 1H NMR (250.1, CDCl3): δ 3.42 and 3.43 (6H, 2s, 2OMe), 4.22 (2H, s, CH2), 6.37 (1H, s, CH=COMe), 6.39 (1H, s, CH=COMe), 6.58–7.37 (9H, m, 9ArH). 13C NMR (62.9 MHz, CDCl3): δ 49.1 and 49.2 (2OMe), 51.8 (CH2), 106.4 (S-C=C), 113.9, 116.3, 117.4, 121.8, 126.2, 129.3, 130.7, 133.8 (CH=COMe), 134.1 (CH=COMe), 136.8, 143.2, 145.7, 148.6 (S-C=C), 156.4 (CH=COMe), 156.5 (CH=COMe), 186.7 (C=O).
2,6-Dimethoxy-4-(3-(2-methylbenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4e )
Golden yellow powder, yield 91%, 0.36 g; m.p. 140–142 °C. IR (KBr): 1749 (C=O) cm‒1. MS (EI): m/z (%) = 391 (M+, 9), 376 (M-Me, 94), 361 (M-2Me, 76), 360 (M-OMe, 82), 345 (M-OMe and Me, 69), 286 (M-C8H9, 60), 271 (M-C8H9 and Me, 55). Anal. calcd for C23H21NO3S (391.41): C, 70.58; H, 5.41; N, 3.58; found: C, 70.54; H, 5.47; N, 3.49. 1H NMR (250.1, CDCl3): δ 2.09 (3H, s, Me), 3.42 and 3.44 (6H, 2s, 2OMe), 4.21 (2H, s, CH2), 6.38 (1H, s, CH=COMe), 6.39 (1H, s, CH=COMe), 6.59–7.38 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 23.7 (Me), 49.1 and 49.3 (2OMe), 52.0 (CH2), 106.4 (S-C=C), 114.7, 116.8, 117.7, 121.5, 123.4, 126.5, 128.3, 129.8, 132.1, 133.9 (CH=COMe), 134.1 (CH=COMe), 136.5, 143.3, 145.9, 148.8 (S-C=C), 156.5 (CH=COMe), 156.7 (CH=COMe), 186.7 (C=O).
2,6-Dimethoxy-4-(3-(2-nitrobenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4f )
Pale yellow powder, yield 91%, 0.38 g; m.p. 157–159 °C. IR (KBr): 1750 (C=O) cm‒1. MS (EI): m/z (%) = 422 (M+, 6), 407 (M-Me, 93), 376 (M-OMe and Me, 70), 360 (M-2OMe, 54), 300 (M-C6H4NO2, 58), 224 (M-C7H6NO2 and 2OMe, 28), 136 (C7H6NO2, 47). Anal. calcd for C22H18N2O5S (422.38): C, 62.56; H, 4.29; N, 6.63; found: C, 62.61; H, 4.33; N, 6.51. 1H NMR (250.1, CDCl3): δ 3.44 and 3.46 (6H, 2s, 2OMe), 4.25 (2H, s, CH2), 6.36 (1H, s, CH=COMe), 6.38 (1H, s, CH=COMe), 6.62–7.40 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 48.9 and 49,1 (2OMe), 52.3 (CH2), 106.6 (S-C=C), 115.1, 116.6, 118.3, 121.7, 122.4, 126.5, 128.2, 130.0, 132.3, 134.2 (CH=COMe), 134.3 (CH=COMe), 136.8, 143.5, 146.1 and 148.9 (S-C=C), 156.8 (CH=COMe), 157.1 (CH=COMe), 186.9 (C=O).
4-(3-Benzylbenzo[d]thiazol-2(3H)-ylidene)-2,6-di-tert-butylcyclohexa-2,5-dien-1-one (4g )
Yellow powder, yield 91%, 0.39 g; m.p. 138–140 °C. IR (KBr): 1751 (C=O) cm‒1. MS (EI): m/z (%) = 429 (M+, 7), 414 (M-Me, 92), 372 (M-CMe3, 85), 352 (M-Ph, 77), 323 (M-C7H7 and Me, 53), 295 (M-Ph and CMe3, 50), 91 (C7H7, 37), 57 (CMe3, 48). Anal. calcd for C28H31NOS (429.55): C, 78.29; H, 7.27; N, 3.26; found: C, 78.27; H, 7.24; N, 3.18. 1H NMR (250.1, CDCl3): δ 1.14 and 1.16 (18H, 2s, 2CMe3), 4.21 (2H, s, CH2), 6.35 (1H, s, CH=CCMe3), 6.37 (1H, s, CH=CCMe3), 6.59–7.37 (9H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 27.4 and 27,5 (2CMe3), 31.7 and 31.8 (2CMe3), 51.7 (CH2), 107.3 (S-C=C), 114.4, 115.9, 117.8, 122.7, 127.1, 128.5, 132.8, 135.1 (CH=CCMe3), 135.3 (CH=CCMe3), 138.2, 141.4, 142.1, 144.3 (CH=CCMe3), 144.4 (CH=CCMe3), 148.2 (S-C=C), 185.7 (C=O).
2,6-Di-tert-butyl-4-(3-(2-methylbenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4h )
Yellow powder, yield 92%, 0.41 g; m.p. 147–149 °C. IR (KBr): 1748 (C=O) cm‒1. MS (EI): m/z (%) = 443 (M+, 8), 428 (M-Me, 90), 413 (M-2Me, 82), 386 (M-CMe3, 79), 371 (M-CMe3 and Me, 56), 338 (M-C8H9, 42), 105 (C8H9, 48), 57 (CMe3, 50). Anal. calcd for C29H33NOS (443.58): C, 78.52; H, 7.50; N, 3.16; found: C, 78.59; H, 7.45; N, 3.25. 1H NMR (250.1, CDCl3): δ 1.15 and 1.16 (18H, 2s, 2CMe3), 2.14 (3H, s, Me), 4.20 (2H, s, CH2), 6.33 (1H, s, CH=CCMe3), 6.34 (1H, s, CH=CCMe3), 6.60–7.39 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 23.6 (Me), 27.3 and 27,5 (2CMe3), 31.7 and 31.9 (2CMe3), 51.8 (CH2), 107.2 (S-C=C), 113.8, 115.6, 118.7, 123.8, 125.4, 127.1, 129.2, 132.8, 134.2, 135.0 (CH=CCMe3), 135.2 (CH=CCMe3), 138.4, 141.3, 142.0, 144.4 (CH=CCMe3), 144.6 (CH=CCMe3), 148.1 (S-C=C), 185.6 (C=O).
2,6-Di-tert-butyl-4-(3-(2-nitrobenzyl)benzo[d]thiazol-2(3H)-ylidene)cyclohexa-2,5-dien-1-one (4i )
Pale yellow powder, yield 90%, 0.43 g; m.p. 159–161 °C. IR (KBr): 1750 (C=O) cm‒1. MS (EI): m/z (%) = 474 (M+, 7), 459 (M-Me, 87), 417 (M-CMe3, 66), 360 (M-2CMe3, 40), 352 (M-C6H4NO2, 62), 337 (M-C6H4NO2 and Me, 46), 136 (C7H6NO2, 53), 57 (CMe3, 47). Anal. calcd for C28H30N2O3S (474.54): C, 70.87; H, 6.37; N, 5.90; found: C, 70.81; H, 6.41; N, 5.79. 1H NMR (250.1, CDCl3): δ 1.16 and 1.18 (18H, 2s, 2CMe3), 4.27 (2H, s, CH2), 6.35 (1H, s, CH=CCMe3), 6.37 (1H, s, CH=CCMe3), 6.58–7.40 (8H, m, 8ArH). 13C NMR (62.9 MHz, CDCl3): δ 27.7 and 27,9 (2CMe3), 31.8 and 31.9 (2CMe3), 52.4 (CH2), 107.4 (S-C=C), 115.5, 116.7, 119.6, 124.7, 126.2, 128.1, 132.8, 134.2, 135.3 (CH=CCMe3), 135.5 (CH=CCMe3), 137.3, 138.2, 141.4, 142.0, 144.2 (CH=CCMe3), 144.3 (CH=CCMe3), 148.4 (S-C=C), 185.9 (C=O).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: I express my sincere gratitude to Chabahar Maritime University’s Research Council for providing the financial support.
