Abstract
In this study, we used a synthetic aerogel based on PVA/agar/maltodextrin to remove the antibiotic ciprofloxacin (CFX) from aqueous media. Response surface method (RSM) is used to study the relationship between the factors affecting the adsorption process, thereby optimizing the adsorption efficiency, and making decisions on improving the adsorption process better sub. The research results obtained are as follows: pH2, initial CFX concentration 51 mg L−1, adsorbent dosage 0.16 g L−1, adsorption time 120 min, and temperature 51.5 °C reached the maximum adsorption capacity. The maximum additive is 38.63 mg g−1. The experimental data agree with the pseudo-quadratic kinetic model and the Langmuir and Temkin isotherm models. The composite aerogel adsorbent (AE) in this study is a potential candidate for adsorbent to remove antibiotic contamination in aqueous media.

Introduction
Humans and animals do not fully absorb antibiotic compounds, so they enter the environment through urine and feces in the primary and metabolite forms. 1 In the environment, antibiotics can be stable and long-lasting; their survival depends on their stability to light, adsorption capacity, rate of decomposition, and solubility in water. 2 Antibiotics are strongly adsorbed and tend to accumulate in soil or sediments (such as tetracyclines and fluoroquinolone) 3 and vice versa, but antibiotics with high mobility (such as sulfonamides) are less susceptible to degradation, tend to seep into groundwater, and are transported with groundwater or by drains that run into surface water. 4 Antibiotics that exist in the environment will cause serious effects such as the generation of antibiotic-resistant bacteria, strengthening the resistance of resistant microorganisms, the accumulation of antibiotics in organisms capable of effects on human health, because antibiotic-resistance genes or bacteria can be transferred from animals to humans, potentially affecting the ecological environment. 5 In nature, antibiotics are continuously introduced into the environment and the amount of antibiotics introduced into the medium is greater than their ability to degrade, leading to antibiotic persistence in the environment.6–10 Antibiotics can be removed from the environment by adsorption, biodegradation, photolysis, and hydrolysis.11–13 There are also a number of other processes such as chlorination, UV irradiation, and using ultrafiltration membranes that can also remove antibiotics. 14
In this study, we used composite aerogel material based on PVA/maltodextrin/agar in combination with montmorillonite. The combination of different substances or compounds is an effective way to synthesize aerogels with the aim of enhancing their properties through the synergistic effects of the starting substances or compounds such as mechanical strength, flexibility, and elasticity as well as porosity and transparency. In addition, when combining inorganic and organic substances, they help to improve the flexural strength of the aerogel. 15 The first studies on the synthesis of organic-inorganic aerogels were conducted by Novak et al in 1993. 16 Thanks to the properties of organic compounds, the created aerogel has more toughness, sturdiness, and higher mechanical strength. Many bioactive natural polymer composite materials have been commercialized and are being studied for biomedical applications such as bone, artificial skin, heart valve, tendon, ligament, tooth, tissue repair. mouth and wound dressing. 17
The composite aerogel material is used as an adsorbent because of its remarkable physical and chemical properties: it has a very low density, only about 3–15 mg cm−3, which reduces the load and increases the mobility of adsorbent material; has a large surface area and a hollow porosity structure, allowing the material’s very high adsorption and physisorption capacity, making the material good at adsorbing antibiotics and other toxic substances. In addition, aerogel is a non-toxic, non-flammable, and non-corrosive material, especially when made from eco-friendly materials, which helps to reduce the impact on the environment. The composite aerogel material has good mechanical properties and strength, is resistant to pressure and abrasion, and does not crack easily. The composite aerogel material can be reused after adsorbing harmful substances, reducing costs and impact on the environment. Therefore, composite aerogel materials are a good choice for use as adsorbents in applications related to water and environmental treatment, which include adsorbing antibiotics and other toxic substances.
Previous studies on using response surface software to optimize the adsorption process: Ali Fakhri and Sajjad Behrouz 17 compared the adsorption properties of MgO nanoparticles and ZnO–MgO nanocomposites to remove linezolid antibiotic from aqueous solution by response surface method (RSM). Influencing parameters such as pH, amount of adsorbent, and temperature were evaluated using Box–Behnken Design. The two models of both materials have a high level of confidence with R2 = 0.96; the predicted data are quite consistent with the experimental data. In this experiment, we also know that the condition to achieve the highest adsorption capacity (123.45 and 140.28 mg g−1 for MgO and ZnO–MgO) is 0.5 g L−1; pH 10 and 308 K. 17 Activated carbon derived from dried rice husk residue was studied to remove the antibiotic ciprofloxacin (CFX) from water in 2017 by Zhang et al. 18 Survey variables including time, concentration, content, and pH were optimized using the central design (CCD) according to the RSM response surface method with 30 experiments. The coefficient R2 used to evaluate the reliability of the model was found to be 0.9870 (consistent with the experiment). The optimal parameters for CFX adsorption were found by the regression equation with time of 306.9 min, substance content of 0.40 g L−1, concentration of 314.8 mg L−1, and pH of 7.92, and the predicted adsorption capacity is 454.68 mg g−1. 18 In 2020, to enhance the removal of erythromycin from water, Soheila Gholamiyan optimized the adsorption process of activated carbon by RSM method. She and her colleagues used the analysis of variance (ANOVA) to create 30 experiments for this model with four factors: concentration, amount of adsorbent, time, and temperature. The results obtained a good linear model with the correlation coefficient R2 = 0.9697, and the investigated variables are all significant in the established equation. In this study, the appropriate accuracy is 22,6941 and greater than 4. This proves that this model can be used to determine the maximum amount of adsorbent with higher confidence in the synthetic design center (Central Composite Design (CCD)). 19 This method was also used by Shirin Afshin to evaluate the nanocomposite parameters of activated carbon from filamentous algae to effectively remove cephalexin from water. Various parameters in the equation such as time, pH, concentration, content, and effects of cations and anions that can interfere with the adsorption of cephalexin are evaluated on three levels. The proposed quadratic model is the best model for the adsorption process (R2 = 0.99094 and R2 adj = 0.9991). According to the results, the best parameters such as cephalexin concentration, content, time, and pH value were 28.16 mg L−1, 2 g L−1, 30.04 min, and 3.02 respectively. 20 Soheila Gholamiyan’s study also showed a high degree of agreement (R2 = 0.9697) when optimizing the erythromycin adsorption process of magnetic activated carbon using the RSM model. The results show that the treatment process is optimized at the initial concentration of 65 mg/L, the amount of adsorbent of 1.55 g/L, the time is 76.25 minutes, temperature35C, the treatment efficiency of 95.125%. 21
Results and discussion
Structural properties of materials
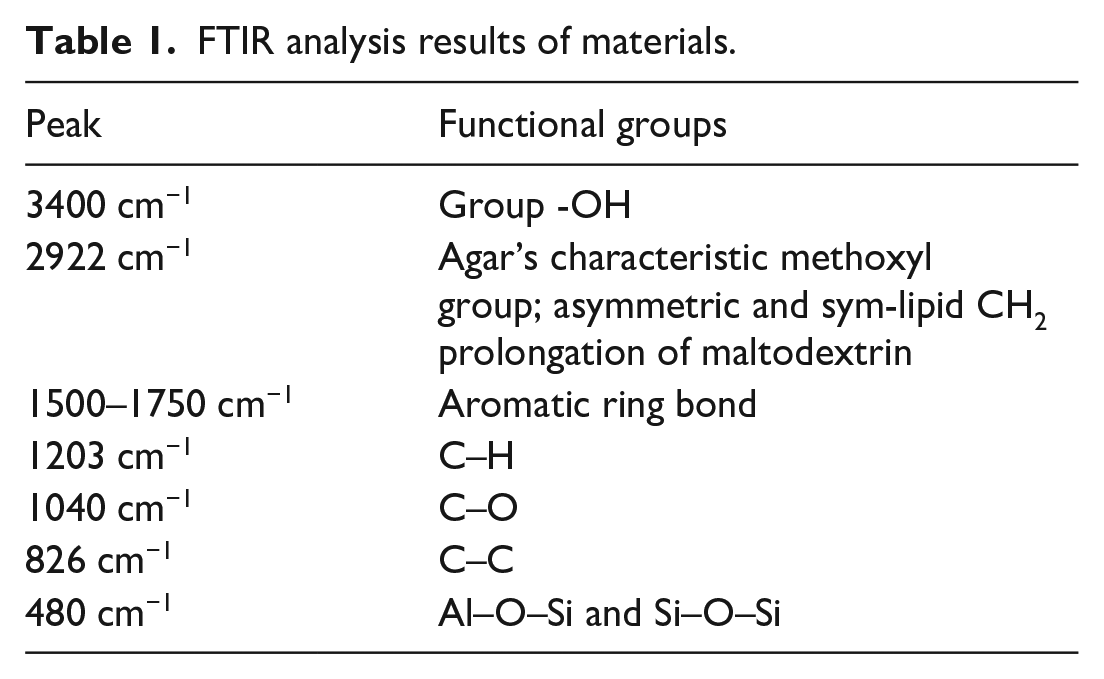
Analysis of the infrared spectrum of materials
The FTIR analysis results in Figure 1 shows the functional groups in the AE material structure. The characteristic peaks of the materials are shown in Table 1. Comparing AE1 and AE2 samples, there is a clear difference between the signals of the spectral peaks: the AE2 sample loses signal at 1730 cm−1, the tip at 1120 cm−1 appears clearly in the AE2 sample as oscillation, stretching C–O–C. In AE2 sample, the absorption band at about 3600 cm−1 is due to the stretching oscillation of the structural OH groups of montmorillonites; bands corresponding to AlAlOH and AlMgOH bending vibrations are observed at 916 cm−1 first. A complex band at 1040 cm−1 is related to the stretching vibrations of the Si–O group, while the bands at 480 cm−1 are due to the Al–O–Si and Si–O–Si bending vibrations, respectively. Noses below 500 cm−1 are typical for quartz.

FTIR infrared spectrum of materials.
FTIR analysis results of materials.
X-ray diffraction analysis
X-ray diffraction analysis of AE materials is observed in Figure 2. For both AE substrates, the broad peak at 2θ = 20º characterizes the crystalline structure of the matrix.22,23 There is also the appearance of 2θ = 12.2°, 25°, 35.1°, 61.9° which is characteristic for the structure of the components present in the material. 24

X-ray diffraction pattern of materials.
Morphological analysis of the material surface
Figure 3 shows the morphology of the AE material. The SEM image results show that the material has pores, which is consistent with the morphological structure of the aerogel material. Compared with AE1 material, AE2 material has more porosity, so in this study, we chose AE2 material as the adsorbent.

Surface morphology analysis (SEM) of (A) AE1; (B) AE2 materials.
Evaluation of factors affecting the adsorption process of materials
Effect of adsorption time
One of the important physical factors that directly affect the adsorption of materials is time. In this experiment, the solution will be shaken continuously to increase the mass transfer coefficient to optimize the adsorption capacity of the material. Figure 4 shows that the material reached equilibrium at 180 min (16.75 mg g−1) and reached the highest value of adsorption capacity at 120 min (17.9 mg g−1). After about 150 min, the material began to stabilize and gradually reached equilibrium; the adsorption capacity increased and decreased insignificantly. Phase from 0 to 120 minutes, the material takes place very quickly. The material then tends to desorb until equilibrium is reached. In Yuan Zhuang’s study on CFX adsorption of soy protein porous graphene (GS) aerogel, the initial concentration was 20 mg L−1 at 25 °C. The adsorption capacity increased continuously for the first 60 min and peaked at 22 mg g−1. 25
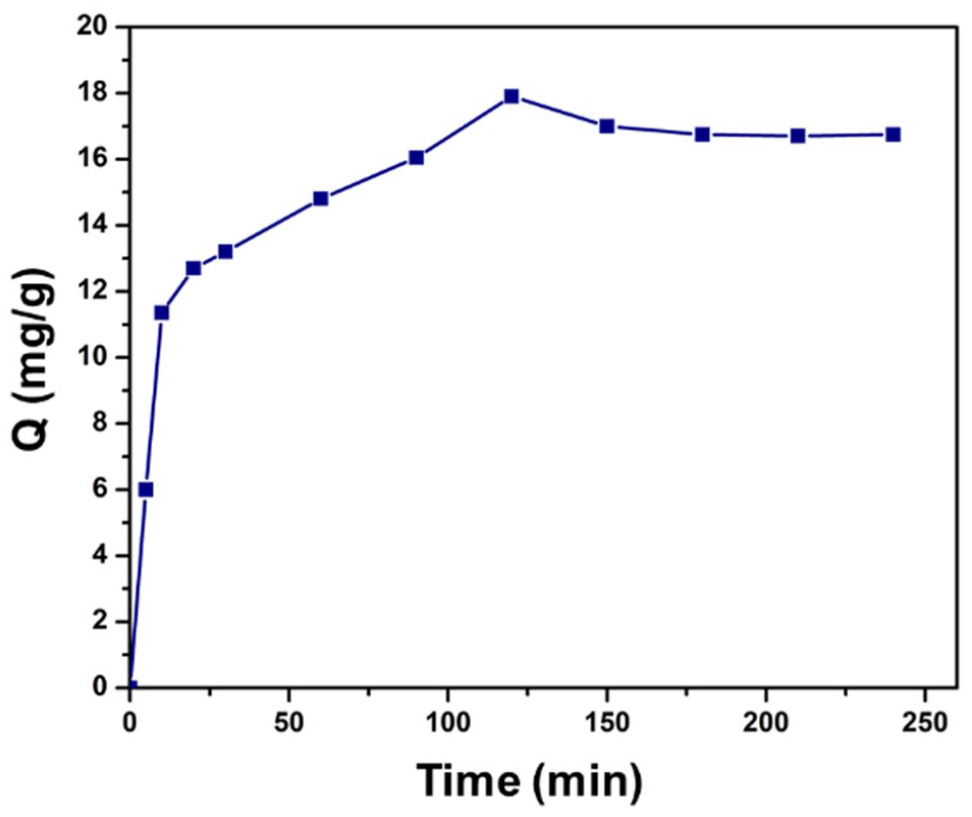
Effect of time.
Effect of pH
The pH value is one of the important factors determining the adsorption capacity of a material because it affects the molecular form of CFX in the solution. In this experiment, we will investigate the effect of pH on the CFX adsorption capacity of the materials in the pH range from 2 to 10. From Figure 5, it can also be easily seen that the adsorbent is better in the form of an acidic solution. The adsorption capacity peaked at pH 2 (35.7 mg g−1), and at pH 10, no adsorption seemed to take place. Therefore, we choose pH 2 as the best value to conduct further experiments. The adsorption capacity has a decreasing trend from pH 2-6 due to the difference in charge of CFX at different pH levels. The main principle is based on the electrostatic interaction between adsorbent and adsorbate. In 2016, Yu Fei investigated the effect of pH from 2 to 12 of GO-SA-A material on CFX adsorption at initial concentration of 50 mg L−1, temperature 25 °C. The adsorption capacity also peaked at pH 2 and decreased gradually with higher pH. 26
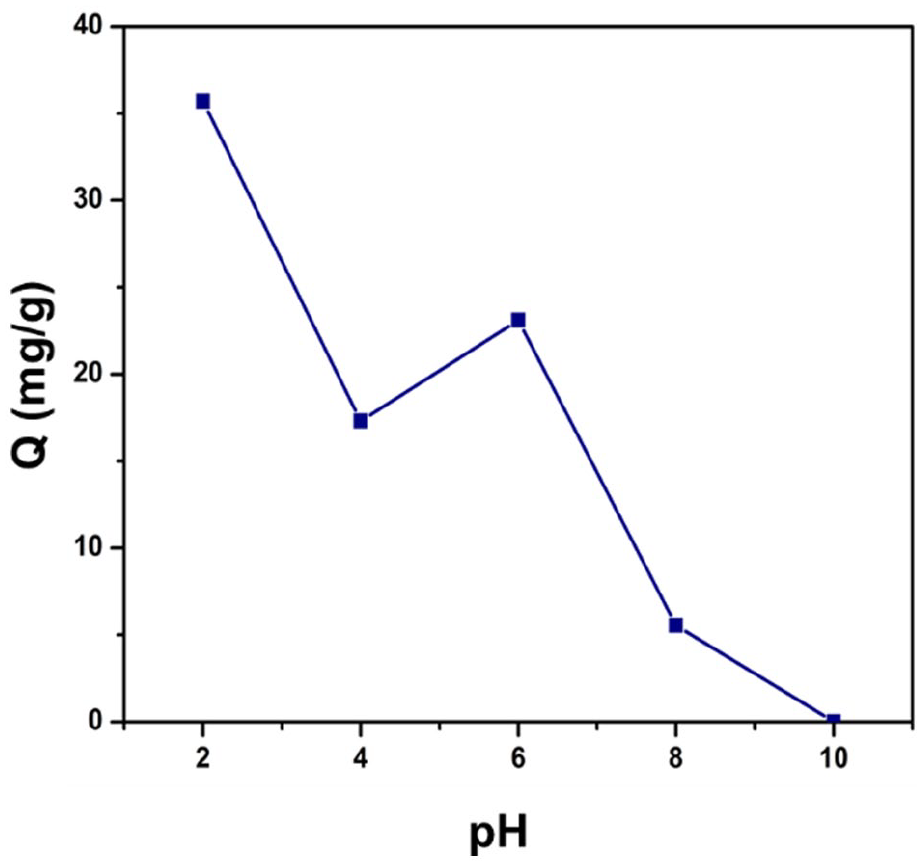
Effect of pH.
Effect of the amount of adsorbent
The effect of adsorbent dosage on the removal of antibiotics is shown in Figure 6. It was observed that the maximum adsorption capacity of CFX was achieved at the dose of 0.15 g L−1 (corresponding to 0.003 g of material with 20 mL of antibiotic solution) with a capacity of 14.9 mg g−1. In the first stage, the adsorption capacity increased rapidly as the amount of adsorbent increased. The adsorption capacity is proportional to the adsorbent dose, possibly due to the increased surface area of the adsorbent and the presence of more effective sites. After that, when increasing the dose, the adsorption capacity began to decrease sharply. This can be explained by the presence of too much absorbent in the solution which will interfere with the contact between the CFX molecules on the surface of the adsorbent and in the solution. 27 Therefore, optimal adsorption is achieved when the adsorbent concentration is 0.15 mg L−1. A study on CoFe-MOF aerogel by Tran Thi Kim Ngan et al. also performed an experiment on the effect of adsorbent content: when increasing the content from 0.002 to 0.04 g L−1, the adsorption capacity also tends to decrease. 28

Effect of amount of adsorbent.
Effect of adsorbent concentration
The initial concentration of antibiotics in wastewater is also an important factor to be investigated. In this experiment, we will investigate the effect of initial concentration on the absorbance of the material performed in the range of 10–80 ppm. Figure 7 shows the maximum adsorption capacity at an initial concentration of 40 ppm with an adsorption capacity of 22 mg g−1. The increase of the initial concentration makes the adsorption process strong because it increases the interaction of antibiotic molecules into the vacant positions on the surface of the material. But if the increase continues like this, the adsorption efficiency of the material will reach saturation and the adsorption capacity will also decrease gradually due to the repulsion between the antibiotic molecules on the surface of the material and the molecules. The antibiotic in the solution is produced and interferes with the adsorption process. A study by Meltem Okan and colleagues on the antibiotic adsorption capacity of carbon aerogel materials showed that as the concentration of the substance increases, the adsorption capacity increases steadily, then reaches a maximum value, and then decreases to equilibrium. 29

Effect of concentration.
Research on adsorption isotherm models
The CFX adsorption isotherm models on experimental data of AE materials are converted into various forms based on the adsorption isotherm models: Langmuir, Freundlich, Temkin, and D-R.
As can be seen in Figure 8, the four models fit well with the experimental data and the calculated R2 is greater than 0.9 as described in Table 2. However, based on the value of R2, the correlations of the models appear in the following order: D-R < Freundlich < Temkin < Langmuir. Therefore, the adsorption occurs on the surface according to the mechanism of the Langmuir model. Besides, the experimental data show that it is consistent with the Temkin model.

Adsorption isotherm model.
Adsorption isotherm constant.
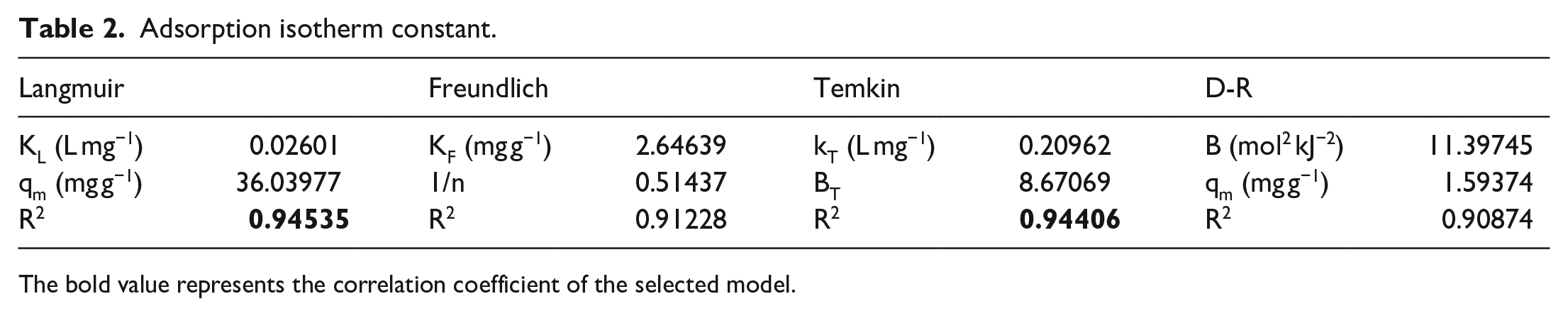
The bold value represents the correlation coefficient of the selected model.
Studying adsorption kinetic models
The adsorption kinetics of composite aerogels (AE) was studied using four models including PFO, PSO, Elovich, and Bangham. The kinetic constants are also shown in Table 2 and Figure 9.

Adsorption kinetic model.
Based on Table 3, the correlation coefficient (R2) for all adsorption kinetic models is very high (R2 = 0.97), showing good statistical compatibility between the survey data. However, the second-order kinetics model is the most compatible adsorption kinetic model among the four given models with the highest correlation coefficient (R2 = 0.97). In general, the above results show the high compatibility and superiority of this model for the CFX autoclaving of the materials.
Adsorption kinetic constants.
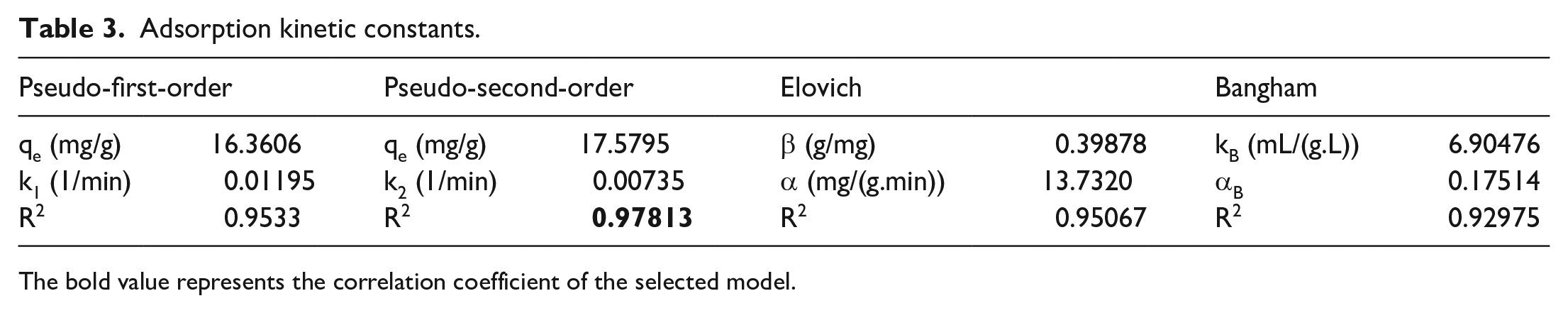
The bold value represents the correlation coefficient of the selected model.
Optimization of the adsorption process by response surface model
In this study, to optimize the treatment of the antibiotic ciprofloxacin (CFX) in aqueous media by aerogel composite materials, our research team used the reactive surface method (RSM). All 17 experiments for each antibiotic were designed and statistically processed based on response surface method. 5 Specifically, three variables including initial antibiotic concentration, adsorbent mass, and pH value were investigated at different value ranges in Table 4, and experimental and predicted values are presented in Table 5.
Survey list of experimental variables.

Experimental value, prediction of adsorption capacity, and adsorption efficiency of AE composite aerogel with CFX antibiotic.

CFX: ciprofloxacin.
ANOVA was calculated using Design-Expert software (version 11, State Ease, Minneapolis, USA). ANOVA of the quadratic linear regression model is used to analyze the influence of input and output variables as well as the correlation of response functions and independent variables. From the RSM model, the recorded results include the model’s quadratic equation, residual value plots, line and 3D inter-factor plots, and variance analysis table. From the practical experiments, the residual value graphs are shown in Figure 10. It can be seen that the experiments are independently distributed, and the quadratic equation is used to calculate the variance values.

Residual value chart of aerogel composites AE.
Evaluation results from DX11 software for quadratic regression equation showing the relationship between response value and independent variables

To build and evaluate the model’s compatibility, the ANOVA presents the coefficients including sum of squares, degrees of freedom, mean squared, and F and P values of the model (Table 6). The analytical results in Table 6 show that the obtained quadratic model is significant at the 95% confidence level because the P value (Prop > F) of the model is less than 0.05.
ANOVA values of the AE composite aerogel model for CFX antibiotics.

ANOVA: analysis of variance; SD: standard deviation; CFX: ciprofloxacin.
S is significant (P < 0.05), N is not significant (P > 0.05).
In the CFX antibiotic treatment model, most of the model’s parameters have statistical significance at the 95% confidence level (P < 0.05). The accuracy of the CFX rejection model is 21.67, and this ratio is also greater than 4.0. The Local Outlier Factor (LOF) value for the CFX treatment model is also not statistically significant (F value = 6.43, P = 0.05). Therefore, the model is statistically significant and can be used to evaluate the influence of three factors (first concentration of antibiotic, amount of adsorbent, and temperature of medium) on the ability to treat antibiotic properties of aerogel composites AE.
Figure 11 shows the three-dimensional response surface of the CFX antibiotic showing the effect of two factors (the other being in the center) on the response factor. Factors (head concentration, amount of substance, temperature) have been shown to affect the CFX adsorption capacity. In other words, for CFX, the adsorption capacity can be improved by increasing the concentration (30–70 mg L−1), reducing the amount of substance (0.2–0.1 g L−1), and increasing the temperature value (30–70 mg L−1, 70 °C) and is clearly shown in the projection in Figure 11. In summary, it can be confirmed that the proposed models can be used to find the optimal conditions for CFX adsorption.

CFX processing optimization model (A–C).

3D model projection of the process of optimizing CFX antibiotic adsorption capacity of AE materials.
By applying the response surface method, several optimal conditions have been proposed for the CFX treatment (Table 7). To check the accuracy and reliability of the obtained model, a validation test is performed under optimal conditions. The results were consistent with the predicted value, showing the high reliability of the proposed model.
Optimization model test.
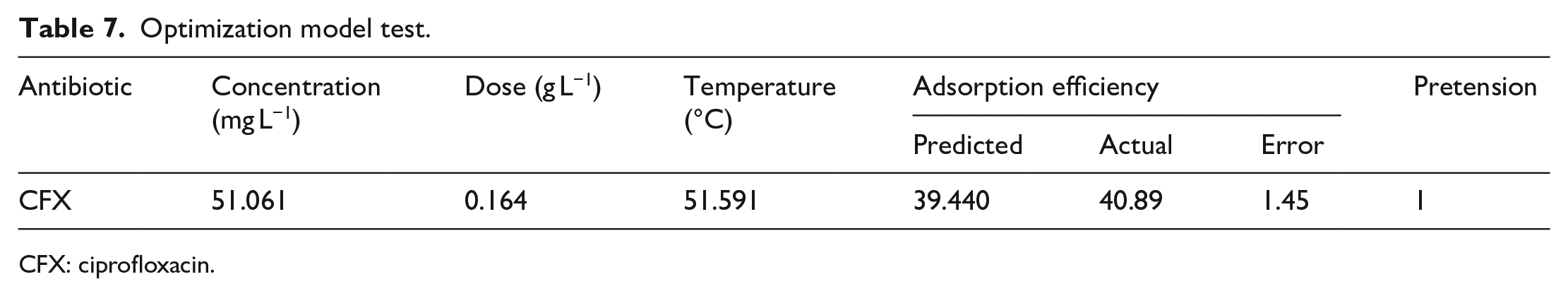
CFX: ciprofloxacin.
Compare with another research
To compare the treatment efficiency of the antibiotic CFX in water, Table 8 summarizes the research results of different materials in terms of maximum adsorption capacity. Compared with other studies, the maximum adsorption capacity in this study is relatively high, proving that aerogel composite materials can be a promising material for the adsorption and removal of antibiotics in the environment polluted water field.
Comparison of the adsorption capacity of CFX with other adsorbents.

CFX: ciprofloxacin.
Conclusion
The survey results on the CFX antibiotic adsorption process showed that the highest adsorption capacity was 36.04 mg g−1 in the following conditions: pH2, initial CFX concentration 50 mg L−1, adsorbent dosage 0.15 g L−1, adsorption time 120 min, and temperature 50 °C. The experimental data are consistent with the pseudo-quadratic kinetic model and the Langmuir and Temkin isotherm model. Applying RSM surface response software to optimize the adsorption process achieved the following results: pH2, initial CFX concentration 51 mg L−1, adsorbent dosage 0.16 g L−1, adsorption time after 120 min, and the temperature of 51.5 °C achieved the maximum adsorption capacity of 38.63 mg g−1.
Experimental
Composite aerogels (AE) based on PVA/maltodextrin/agar were synthesized: first, we stirred 3% PVA, 2% agar, 2% maltodextrin until the solution was clear. Then, pour all the above solutions into the same cup, continue to stir. Add 0.6% glycerol to the mixture. Next, the sample was poured into a 96-well mold and refrigerated overnight. The end of the sample area is dried to increase flowers. The temperature change during the synthesis of materials is modeled as AE1 (60) and AE2 (80), respectively. The material samples obtained after the synthesis process were evaluated for structural characteristics of the materials such as XRD, FTIR, and SEM.
The factors affecting the adsorption process: concentration, time, solution pH and volume on the adsorption efficiency of ciprofloxacin antibiotic (CFX) were performed in this study. Weigh 0.001 g of material into 10 mL of antibiotic-forming solution at different concentrations from 20 to 160 mg/L, time from 10 to 400 minutes, dye concentration from 20 to 160 mg/L, solution pH (2, 4, 6, 8, 10), the volume of raw materials (0.05; 0.1; 0.2; 0.3 g/L). Stir at constant temperature and 200 rpm. Sample centrifugation to remove solids from solution using a centrifuge (5000 rpm, 10 min). Measure residual antibiotic concentration by UV-Vis. The expected product will be a highly efficient antibiotic adsorption process. The experimental data of the study were evaluated to be consistent with the kinetic and isothermal models of adsorption.
The interaction between the main factors and the optimal conditions was tested by Design Expert software. This method helps us to understand the influence of different process parameters on the adsorption process to find the optimal adsorption conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by a grant from the Science and Technology Development Foundation of Nguyen Tat Thanh University, Ho Chi Minh City, Viet Nam.
