Abstract
Microporous ZSM-5, hierarchical ZSM-5, and hierarchical ZSM-11 zeolites with different crystalline sizes were prepared to determine the relations of mesoporosity with catalytic performance in methanol-to-propene reaction. The physicochemical properties were investigated by X-ray diffraction, Fourier-transform infrared spectroscopy, scanning electron microscopy, Temperature-Programmed Desorption, and sorption techniques. It was shown that the low pore tortuosity, hierarchical structure, and the reduction of the crystal size all can shorten the diffusion path and reduce the diffusion resistance, leading to the increase of propylene yield. By comparison, the propene yield increase to the increment of mesoporous volume due to the tortuosity is more effective than other aspects. For silica–alumina zeolites, the effective way to reduce the diffusion resistance and increase the selectivity of propylene is to lower the pore tortuosity degree, and then introduce the mesoporous structure and reduce the grain size.
Introduction
Propene serves as a key component in the petrochemical industry. The high propylene-to-ethylene (P/E) ratio is the most distinguishable feature for methanol-to-propene (MTP) process that enables the high propylene yield in the recirculation process. In the case of MTP process on the preferred catalyst ZSM-5, low yield of propene and short regeneration cycle still limit its further application in the fixed-bed process. The propene selectivity can be improved through suppressing the aromatization ability of higher olefins and the production of ethylene in aromatic-based cycle.
Several strategies targeted to solving these problems toward the catalyst have been attempted, including improving the diffusivity of intermediate species and products as well as properly tailoring the Brønsted acid sites.1,2 The intracrystalline and intercrystalline diffusion capabilities of zeolite are important factors to control the adsorption kinetics and product shape selectivity. The diffusion performance can be improved by reducing the crystal size, synthesizing multi-channel molecular sieve, and adopting molecular sieve with suitable topological structure.3,4 Nano-zeolites have a larger outer surface, more exposed unit cells, and higher surface energy, which can shorten the diffusion path and the residence time of products in the tunnel. 5 However, nano-scale zeolites are easy to agglomerate and are relatively difficult to regenerate, making it difficult to apply in industrial applications. Hierarchical zeolites not only have strong acidity and hydrothermal stability of microporous molecular sieves, but also have better diffusion properties than conventional molecular sieves due to the introduction of mesopores. 6 Methods to achieve multi-stage molecular sieve pores include post-processing the synthesized molecular sieves and adding mesoporous templates in the synthesis of zeolite.7,8 Removal of the inert template through hard template-directed synthesis by combustion or dissolution leads to isolation of large zeolite single crystals with a mesopore system, simultaneously the high price and complicated preparation process limits its industrial applications. 9 It is more advantageous to obtain a molecular sieve with nanometer and mesoporous structure directly by adjusting the crystallization conditions and the type of structure directing agent in one-step crystallization.
In the present work, microporous ZSM-5, hierarchical ZSM-5, and hierarchical ZSM-11 zeolites with different crystalline sizes were synthesized. The main purpose is to get a better insight into the influence of mesoporosity directly formed and channel intersections on diffusion and catalytic performance of the zeolite catalysts in MTP reaction through comparison of the structural and catalytic properties of samples.
Results and discussions
Catalyst characterization
The X-ray diffraction (XRD) patterns shown in Figure 1 indicate that the H-11, H-5, and M-5 zeolites are obtained with high crystallinity and pure phase. As shown in Figure 1, the M-5 and H-5 samples exhibited representative XRD patterns of a fully crystalline MFI structure that showed five characteristic diffraction peaks indexed to the [501], [051], [151], [303], and [133] crystal planes, while H-11 zeolite only exhibited [501] and [303] two peaks with MEL framework structure. At the 2θ range of 45.0°–45.7°, the tetragonal ZSM-11 has only one symmetrical diffraction peak corresponding to the [1000] crystal plane, while there are also diffraction peaks on the [0100] crystal plane, 10 for the orthorhombic ZSM-5. The analysis shows that the half-peak width of hierarchical materials is slightly higher than that of microporous materials, which may be induced by the high crystallinity and similar crystal size.

XRD patterns of M-5, H-5, and H-11 zeolites.
The introduction of mesopores affects the rate of crystal formation, leading to changes in surface morphology. Representative scanning electron microscopy (SEM) images of the samples are shown in Figure 2. It can be seen that the crystal grains of each sample are uniform and well dispersed with particle sizes of 5 μm. M-5 zeolite presents a cubic morphology, the outer surface of which is relatively smooth. The spindle-shaped crystals units can be seen in the H-5 zeolite. Higher magnification (Figure 2(b)) reveals clearly that each crystal grain is composed of nanorods inserted in different directions, and these nanorod-like structures are all crystals with good crystallization. The H-11 zeolite has a typical spherical structure, and nanorod-inserted structures also appear on the crystal surface.

SEM images of (a) M-5, (b) H-5, and (c) H-11 zeolites.
In order to verify that the pore structure properties of the synthesized sample are consistent with expectations, the N2 adsorption and desorption curves and pore structure data are shown in Figure 3 and Table 1, respectively. As shown, the commercial M-5 sample shows a type I isotherm corresponding to the mostly microporous structure, whereas the adsorption/desorption isotherms of H-5 and H-11 exhibited a type-IV argon adsorption isotherm with an additional capillary condensation step in the mesopores inducing obvious hysteresis loops at relative pressures (P/P0) >0.4, which is typical for hierarchical materials with both micro- and mesoporosity. 11 At the same time, there are no obvious difference in the Brunauer Emmett Teller (BET) surface area, micropore volume, and specific surface area of the three samples. The external surface area and mesopore volume of H-5 and H-11 zeolites are higher than those of M-5 molecular sieve (57 m2/g and 0.064 cm3/g), indicating that the formation of mesopores is not at the expense of micropores, and the mesopores in the hierarchical zeolite may only be intercrystalline pores. In addition, the external surface area and mesoporous volume of H-11 sample are higher than those of H-5 zeolite, which may be due to the different pore structure and the larger cavity (180–230 Å3) of ZSM-11 zeolite.

Nitrogen adsorption/desorption isotherms of M-5, H-5, and H-11 zeolites.
Textural characteristics and chemical composition of M-5, H-5, and H-11 zeolites.

SBET: specific surface area calculated by Brunauer Emmett Teller (BET) method; SExt and SMicro: external and micropore surface area determined by t-plot method, respectively; VT: total pore volume at p/p0 = 0.990; Vmicro: micropore volume studied by t-plot method.
The textural properties and surface morphology exhibited that the nanorod microcrystals are the source of the micropores, and the gap between the nanocrystals during the insertion process is the source of mesopores of the H-11 and H-5 zeolites. In addition, the zeolite also differs greatly in channel pattern and channel diameter. ZSM-5 is composed of straight channels (0.51 nm × 0.55 nm) and intersecting sinusoidal channels (0.53 nm × 0.56 nm), the windows of which are both 10-membered rings. However, ZSM-11 contains intersecting straight channels (0.53 nm × 0.54 nm), with the same 10-membered ring elliptical openings. Despite their similar channel diameters, HZSM-11 has a less complex channel structure than HZSM-5.
The conversion of methanol to light olefins is an acid-catalyzed process. In order to understand the catalytic performance of the prepared zeolites, NH3-TPD and Py-FTIR were used to determine the acid properties of the three zeolites (Table 2 and Figure 4). The temperature range for calculating the amount of weak acid is 100–300 °C and for strong acid is 300–550 °C. 12 The weak acid sites usually assigned as silanol groups at the external surface or at lattice defects and OH groups bonded to the extra-framework aluminum species. The strong acid sites that are important for the catalysis were generally resulted from the framework tetrahedral aluminum species. From the NH3-TPD results, the amount of the internal strong acid sites on the three zeolite catalysts is very close. Thus, the diffusion of intermediates due to strong acid sites will not be significantly changed. In addition, the acid concentration of the H-5 zeolite, especially the B acid amount is lower than that of the M-5 zeolite, and the B acid amount of the H-11 sample is slightly lower than that of the H-5 zeolite, which is mainly due to the difference in the distribution of aluminum atoms in the framework of the zeolite. 5
Acid properties of M-5, H-5, and H-11 zeolites.

Area under the desorption peak curve in the range 100–300 °C represents the amount of weak acid, while that above 300 °C represents the amount of strong acid.

Py-FTIR spectra of M-5, H-5, and H-11 zeolites.
Catalytic performance
In order to understand the role of mesoporosity in the catalytic performance, the commercial M-5 and the mesoporous H-5 and H-11 samples were comparatively investigated under the same reaction conditions. Figure 5 illustrates the methanol conversion and the yield of main products with reaction time. At the initial stage, each sample has a high methanol conversion activity, and methanol can be completely converted. Compared with M-5 catalyst, on which the methanol conversion drops abruptly after on stream for 12 h, the mesoporous catalysts (H-5 and H-11) exhibit similar stability. This indicates that the carbon deposition precursors of microporous zeolite tend to be generated in the micropores, and the internal surface carbon is formed by polymerization reaction, 8 leading to a fast carbon deposition rate. However, for the mesoporous zeolite with larger outer surface, the short diffusion path is conducive to the migration of the carbon deposition precursor to the outer surface, and the outer surface carbon cannot completely affect the contact between the reactant and the active sites. 13 Moreover, the low acid concentration on H-5 and H-11 zeolites is also beneficial to the weak polymerization, aromatization reactions, and low deactivation rate.

Methanol conversion and product distribution versus time over M-5, H-5, and H-11 zeolites.
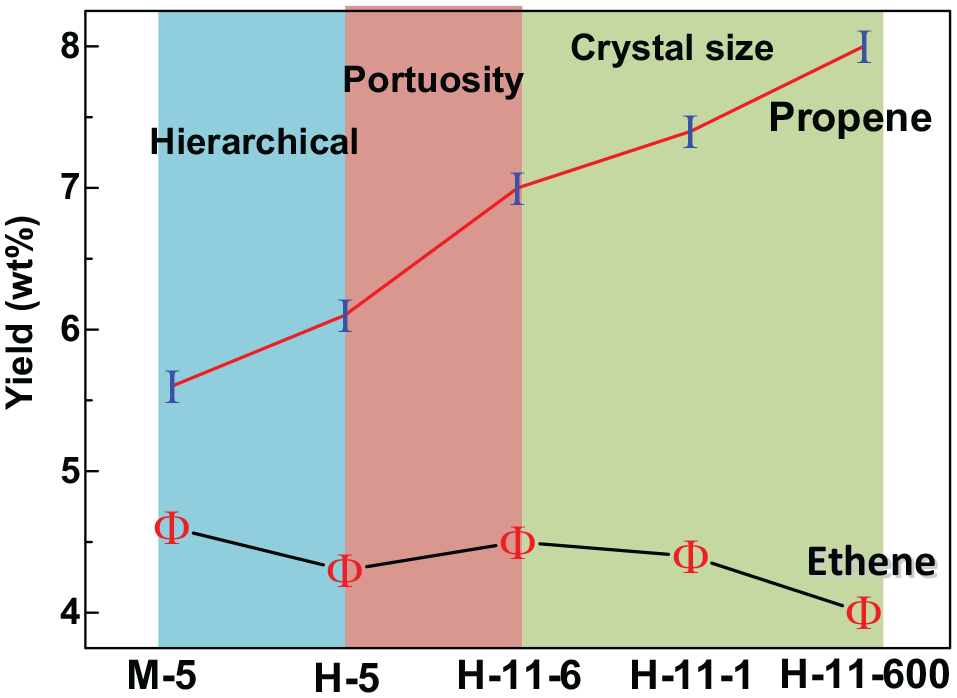
Notably, the propylene yield (6.1 wt%) of H-5 catalyst is higher than that of M-5 (5.6 wt%), while the ethylene yield is slightly lower. The ratio of propylene yield to the increment of mesoporous volume due to the hierarchical structure is 7.9 wt%/(cm3/g). The ratio of ethylene yield decrease to the increment of mesoporous volume due to the hierarchical structure is 8.9 wt%/(cm3/g). The increment of P/E ratio can be mainly attributed to the increase in mesoporosity of the H-5 catalyst resulting from induction of intergranular mesopores, which facilitates the molecular transport in the MTP reaction exceedingly. In addition, the weak acid sites density of H-5 zeolite inhibited the further hydrogen transfer reactions of alkenes. From the reaction mechanism, there are two reaction paths of aromatic-based cycle and olefin-based cycle on ZSM series zeolites. 14 The smaller diffusion resistance and weaker acid sites density weaken the formation of poly-methyl-benzene in the aromatic hydrocarbon cycle to a certain extent, and the aromatic content in gasoline decreases accordingly (Table 3). The decline of hydrogen transfer activity of olefin increases the probability of olefin methylation-cracking reaction, leading to the increase of the yield of high-carbon olefins in propylene, butylene, and gasoline.
Catalytic performance of M-5, H-5, and H-11 zeolites at 2-h time on stream.

P/E: propylene to ethylene.
Except for directly introducing nanostructures and mesopores in the zeolite synthesis process, reducing the distortion of the pores can also significantly reduce the diffusion resistance. Zhang et al. 15 found that the diffusion rate of straight pores of zeolite was significantly faster than that of curved pores. ZSM-5 zeolite contains two intersecting pore systems, among which the 10-membered ring pores parallel to the a-axis are sinusoidal, and the 10-membered ring pores parallel to the b-axis are linear. The channel structure of ZSM-11 is more regular, on which the straight channels of 10-membered ring are parallel to the a-axis and b-axis. At the same time, the intersection of the sinusoidal channel and the straight channel of ZSM-5 is orthogonal. There is only one kind of crossing cavity. Except for the same crossing point as ZSM-5, there is another kind of intersection with more space (30% more than the former) for ZSM-11 zeolite. The average pore diameters of ZSM-11 and ZSM-5 are basically the same, but the difference between the acidity and acid distribution caused by the difference in pore structure, the “cavity” effect, and the uneven configuration of aluminum atoms in the zeolite lead to the diffusion and the differences in the catalytic reactions, 16 which showed that it should be more suitable for the process of high-selectivity methanol to propylene.

The topologies of ZSM-5 (MFI) and ZSM-11 (MEL) zeolites. 15
Compared with H-5 zeolite, H-11 zeolite shows a slow deactivation rate and a high ratio of propylene to ethylene. After 2 h on stream, methanol can still be completely converted, and the propylene yield can reach 7.0 wt%. In view of the similar crystal size, surface area, and acid properties of the two zeolites, the lower distortion of the pore structure is the main cause of the reaction performance. The channel system formed by the intersection of straight channels has low diffusion resistance, which is beneficial to the contact between methanol and active sites, and the product is more easily diffused out of the channels, effectively inhibiting the occurrence of secondary reactions. In addition, the larger cavity volume of the ZSM-11 zeolite is more conducive to the generation of tri-tetra-methylbenzene, which is the intermediate species required for the formation of propylene and butylene in the hydrocarbon pool mechanism, thereby increasing the propylene yield. The lower yield of aromatics and ethylene in the product further supports the above speculation. The ratio of propylene yield to the increment of mesoporous volume due to the hierarchical structure is 21.4 wt%/(cm3/g), while the ethylene yield is increased slightly.
Mesoporosity from crystalline size
In order to effectively utilize the special morphology and mesoporous structure of the nanorods-inserted morphology and further study the role of the mesoporous porosity from different crystal sizes, H-11 zeolite with different crystalline sizes was synthesized. The samples are named H-11-6, H-11-1, and H-11-600 according to the different crystal grain sizes.
The SEM images of various zeolites (Figure 7) show that the crystal sizes of the three ZSM-11 zeolites are 6 μm, 1 μm, and 600 nm, respectively, and each sample has obvious nanorod insertion morphology. The first two zeolites have large crystal grains and uniform crystals. There is a certain agglomeration between the crystals, and the microcrystals that make up a single particle are also reduced from nanorods to nanocrystals for H-11-600 sample. However, unlike the solid structure of large particles, the particles show obvious intercrystalline mesopores, and this gap penetrates the entire microsphere. It can be concluded that the reduction of the crystal size not only shortens the diffusion path of the zeolite, but also increases the number of intercrystalline voids by the loose arrangement, which is more conducive to the diffusion of molecules.

SEM images of H-11 zeolites with different crystalline sizes.
In order to further confirm the changes in the mesoporous properties caused by the reduction of the crystal size, the samples were characterized by N2 adsorption and desorption. It can be seen from the textural properties data (Table 4) that with the decrease of the crystal size, the outer surface area and mesopore volume of the zeolite increase significantly, while the micropore surface area and micropore volume have no obvious changes. Moreover, it is generally believed that the reduction of the crystal size will shorten the length of the micropore channels, and expose more unit cells in the pores, thereby generating more accessible active sites. 17 The NH3-TPD results show that the amount of acid, especially B acid, increases with the decrease of crystal size.
Textural and acid properties of H-11 zeolites with different crystalline sizes.

Table 5 lists the product distribution in the MTP reaction on ZSM-11 zeolite with different crystalline sizes. In order to weaken the influence of the reaction induction period and carbon deposition, the product with a reaction time of 10 min is selected in this section.
Product distribution of H-11 zeolites with different crystalline sizes in methanol conversion.

PE: propylene to ethylene.
It is generally believed that ethylene is mainly produced through aromatic-based cycle, and its methylation ability is much lower than that of propylene. 18 Propylene is formed from the cracking reaction of higher olefins in the olefin-based cycle, and the dealkylation reaction in the aromatic hydrocarbon-based cycle. 19 The crystal size can adjust the ratio of the two cycles. Rownaghi et al. 20 also found that the ethylene yield of nano-scale zeolite is lower than that of traditional large-grain ZSM-5 zeolite.
Combined with the reaction results, it can be seen that with the decrease of crystal size, the diffusion path is short, and the low-carbon olefins are easily diffused out of the pores, which reduces the occurrence of secondary reactions, and is beneficial to the production of low-carbon olefins and the propene. Moreover, the probability of olefin-based cycle is higher than that of aromatic-based cycle, leading to a higher propene-to-ethene ratio. However, the increase of the crystal size prolongs the residence time of polymethyl-benzene in the pores, and is more conducive to the transfer of aromatic hydrocarbon groups, 4 and the production of ethene. The propene yield increases with the increment of mesoporous volume due to the crystalline size. The ethene yield decreases with the increment of mesoporous volume due to the crystalline size. Therefore, it can be concluded that for silica–alumina zeolites, the effective way to reduce the diffusion resistance and increase the selectivity of propene is to lower the pore tortuosity degree. On this basis, the mesoporous structure should be introduced and the grain size should be further reduced (Scheme 1).

The effect of mesoporosity from different forms in MTP.
Conclusion
The nanocrystals, hierarchical pores, and low pore tortuosity can shorten the diffusion path and reduce the diffusion resistance. It facilitates the migration of coke precursors to the outer surface and improves the stability of the zeolite. Moreover, it is beneficial to the timely diffusion of product olefins out of the pores, reduces the probability of hydrogen transfer reactions, and promotes the generation of low-carbon olefins. Among them, the channel tortuosity has a more obvious influence on the diffusion, which is more conducive to the generation of propene.
Experimental
Catalyst preparation
Hierarchical ZSM-5 and hierarchical ZSM-11 zeolite samples were prepared basically according to the method described in the literature. 10 The molar composition of the hierarchical ZSM-11 zeolite mixture was 9.0 Na2O:1.0 Al2O3:65 SiO2:0.53 (TBA)2O:1300 H2O. The whole gel mixture was transferred into a Teflon-lined stainless-steel autoclave. The crystallization reaction was first carried out at 90 °C for 24 h and then was rapidly transferred to another oven preheated to 170 °C for another 24 h. The synthesis of hierarchical ZSM-5 zeolite follows the same procedure as above, only the template is TPABr. The product was filtered, washed, dried at 110 °C overnight and then calcined at 550 °C for 3 h to remove the template. The parent zeolites were turned into the H-form by three consecutive ion exchanges in 1 M NH4NO3 solution with a solution/zeolite ratio of 10 mL/g at 80 °C for 2 h. Microporous ZSM-5 sample was purchased from Hebei Chemical Technology Limited Corporation. The preparation method of hierarchical ZSM-11 with different crystallite sizes followed the patent. 21
Catalyst characterization
Bulk crystalline phases of the catalysts were determined by XRD on Philips X’Pert PRO MPD diffractometer (PANalytical Company, the Netherlands) using Cu Kα radiation (40 kV, 40 mA) in the range between 3° and 75° with a step of 0.02°.
Morphology and structure of the solids were investigated with SEM S-4800 (Hitachi Company, Japan) and high-resolution transmission electron microscopy JEM-2100UHR (JEOL, Japan) after being sonified in ethanol.
Temperature-programmed desorption of ammonia (NH3-TPD) measurements were performed to study the surface acidity (concentration and strength of acid sites) on the catalyst using TP-5079. X-ray fluorescence spectroscopy (Axios) was used to analyze the composition of the samples.
Textural parameters of the samples were determined by nitrogen adsorption isotherms which were recorded at 196 °C using Autosorb iQ apparatus (Quantachrome, America). Prior to the measurement, the samples were outgassed under vacuum at 200 °C for 16 h. Specific surface area measurement was obtained by the BET equation for the adsorption data in a relative pressure range p/p0 from 0.05 to 0.30. The total volume (Vtotal) was determined from the nitrogen adsorbed volume at P/P0 = 0.990. Both the micropore surface area (Smicro) and the micropore volume (Vmicro) were calculated using the t-plot method. The external surface area (Sext) was obtained by the calculated total data minus the corresponding micropore data. The mesopore volume (Vmeso) was carried out for the adsorption branch of the isotherm using the Barrett-Joyner-Halenda (BJH) modes.
Catalytic testing
The catalytic testing was carried out at 450 °C in a fixed-bed microreactor under atmospheric pressure. In a typical experiment, 2 g catalyst with particle size of 0.250–0.425 mm was packed between two layers of quartz wool in the reactor. The reaction feed, a pure methanol solution, was introduced into the system by a HPLC infusion pump at a fixed rate of 0.164 mL/min. The weight hourly space velocity (WHSV) for methanol was 5.53 h−1 with flowing N2 gas (21 mL/min). The inlet flow rate was adjusted using a mass flow controller.
The gaseous products were analyzed using a Bruker 450-GC gas chromatograph (GC) with a thermal conductivity detector (TCD) to analyze content of hydrogen, nitrogen and carbon oxide, and a flame ionization detector (FID) column to determine the composition of hydrocarbons. Liquid products were analyzed on Agilent 6820 GC equipped with HP-INNOWax capillary column (30 m × 0.32 mm × 0.25 μm) and an FID using ethanol as an internal standard. The composition of gasoline was analyzed using PerkinElmer Clarus 580 GC with TCD and FID columns. Liquid products were analyzed on Agilent 6820 GC equipped with HP-INNOWax capillary column (30 m × 0.32 mm × 0.25 μm) and an FID using ethanol as an internal standard.
The conversion of methanol and product yield could be calculated with equations (1) and (2), respectively


Superscript i refers to the components at the inlet of reactor, and superscript o refers to the components at the reactor outlet.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (No. 22202026), the Scientific and Technological Research Program of Chongqing (KJQN202001504), and Natural Science Foundation of Chongqing (cstc2019jcyj-msxmX0359).
