Abstract
Three new complexes, {[Ni2(3-PCA)2(DMAP)4(H2O)2]·2H2O}2, [Cu2(3-PCA)2(4-PP)2·2H2O]n, and [Cu2(4-PCA)2(2,2′-bpy)2·2H2O]n, are synthesized by the solvothermal method with pyrazole-3-carboxylic acid or pyrazole-4-carboxylic acid as the main ligands and 2,2′-bipyridine, 4-dimethylaminopyridine, or 4-phenylpyridine as auxiliary ligands. The crystal structures of the three complexes are determined by single-crystal X-ray diffraction, powder X-ray diffraction, thermogravimetric analysis, and Fourier transform infrared analysis. The magnetic detection proves that the three complexes are all antiferromagnetic.
Keywords
Three new complexes, {[Ni2(3-PCA)2(DMAP)4(H2O)2]·2H2O}2, [Cu2(3-PCA)2(4-PP)2·2H2O]n, and [Cu2(4-PCA)2(2,2′-bpy)2·2H2O]n, are synthesized by the solvothermal method with pyrazole-3-carboxylic acid or pyrazole-4-carboxylic acid as the main ligands and 2,2′-bipyridine, 4-dimethylaminopyridine, or 4-phenylpyridine as auxiliary ligands.

Introduction
Coordination compounds are highly valued by the scientific community because of their various spatial structures and excellent properties. 1 They not only play an important role in promoting the development of inorganic and organic chemistry, but also have important research value and significance in scientific fields such as materials science and bio-pharmacology.2–4 Complexes have the characteristics of self-assembly, and they can form multifunctional materials with different sizes and different characteristics. Coordination compounds having infinite structures have been intensively studied, with significant progress having been made in the fields of materials science and coordination chemistry.5–11
It is well known that many factors such as synthetic methods, central metals, ligands, and pH can influence the final structures of complexes.12,13 Heterocyclic carboxylic acids are important intermediates in the synthesis of nitrogen-containing biologically active compounds, pharmaceuticals, and agrochemicals.
14
Pyrazole carboxylic acids can coordinate with metals via nitrogen and oxygen atoms. They play a crucial role in modern coordination chemistry due to their stability, accessibility, and versatile coordination modes, making them important ligands for complexes with interesting magnetic, sorption or luminescent properties.15–17 Meanwhile, Jiang reported two pyrazole-3-carboxylic acid (3-PCA) complexes and they exhibit excellent heterogeneous catalytic performance. One complex is a binuclear copper structure of 3-PCA without any auxiliary ligands, the other compound is a mononuclear manganese complex of 3-PCA with 2,2′-bipyridine auxiliary ligand.
18
Here, three new metal-organic carboxylic acid complexes based on pyrazole-carboxylic-formic acid (Scheme 1), {[Ni2(3-PCA)2(DMAP)4(H2O)2]·2H2O}2
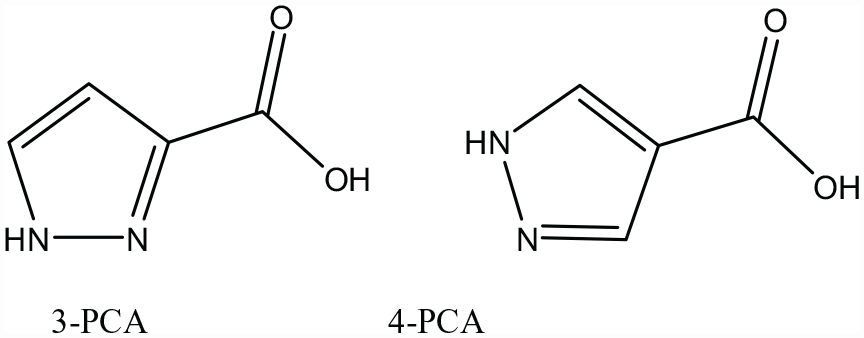
The structures of 3-PCA and 4-PCA.
Results and discussion
Synthesis of compounds 1–3
Compounds
Description of the structure of 1
The X-ray crystallographic data show that complex

(a) Coordination environment of the Ni(II) ions in
Selected bond lengths (Å) and angles (°) for complex

Symmetry codes: A: −x+2, −y+1, −z.
The hydrogen bond lengths (Å) and angles (°) of complex

Symmetry codes: B: −x+1, −y+1, −z+1; C: x−1, y, z; D: −x+1, −y+1, −z.
Description of the structure of 2
The X-ray crystallographic data reveal that the minimum structure unit of complex

(a) Coordination environment of the Cu(II) ions in
Selected bond lengths (Å) and angles (°) for complex

Symmetry codes: A: x+1/2, −y+3/2, z; B: −x, −y+1, −z+1.
Description of the structure of 3
The X-ray crystallographic data show that the molecular structure of complex

(a) Coordination environment of the Cu(II) ions in
Selected bond lengths (Å) and angles (°) for complex

Symmetry codes: A: −x, −y, z; B: x+1/2, −y+1/2, −z+1.

IR spectra of complexes
IR spectra
IR spectroscopy is an excellent approach to characterize and study crystallization. The IR spectra of complexes
Thermal analyses
Thermogravimetric analyses were carried out for complexes

TGA curves of complexes
For complex
For complex
Powder X-ray diffraction analysis
The purities of the synthesized complexes were determined by powder X-ray diffraction (PXRD). Cu-Kα was chosen as a monochromatic light diffraction source, the scanning range is 5–50°, and diffraction data are collected. The theoretical diffraction peaks were calculated using Mercury software, and the actual diffraction peaks obtained experimentally were plotted and compared. It was found that the actual diffraction peaks were basically consistent with the simulated diffraction peaks (Figure 6), indicating that the synthesized complexes are pure samples.

PXRD patterns of complexes
Hirshfeld surface analysis for complex 1
Hirshfeld surface (HS) analysis is a useful tool for the in-depth visualization of aspects of crystal packing and supramolecular arrangements in crystal lattices. The color mapping of functions describing specific properties of the HS allows for intuitive recognition and quantitative analysis of interactions between molecules.19–22 For a deeper understanding of the molecular structure of complex
The results of the HS analysis of complex

Hirshfeld surface mapped with dnorm for complex

Fingerprint plots for the H···H (a), C···H (b), O···H (c), and N···H (d) contacts of complex
Magnetic properties
The temperature dependences of the magnetic susceptibilities for the three complexes were measured in the 2–300 K temperature range under an applied field of 1000 Oe. The χM, χMT versus T plots are illustrated in Figure 9. For complex
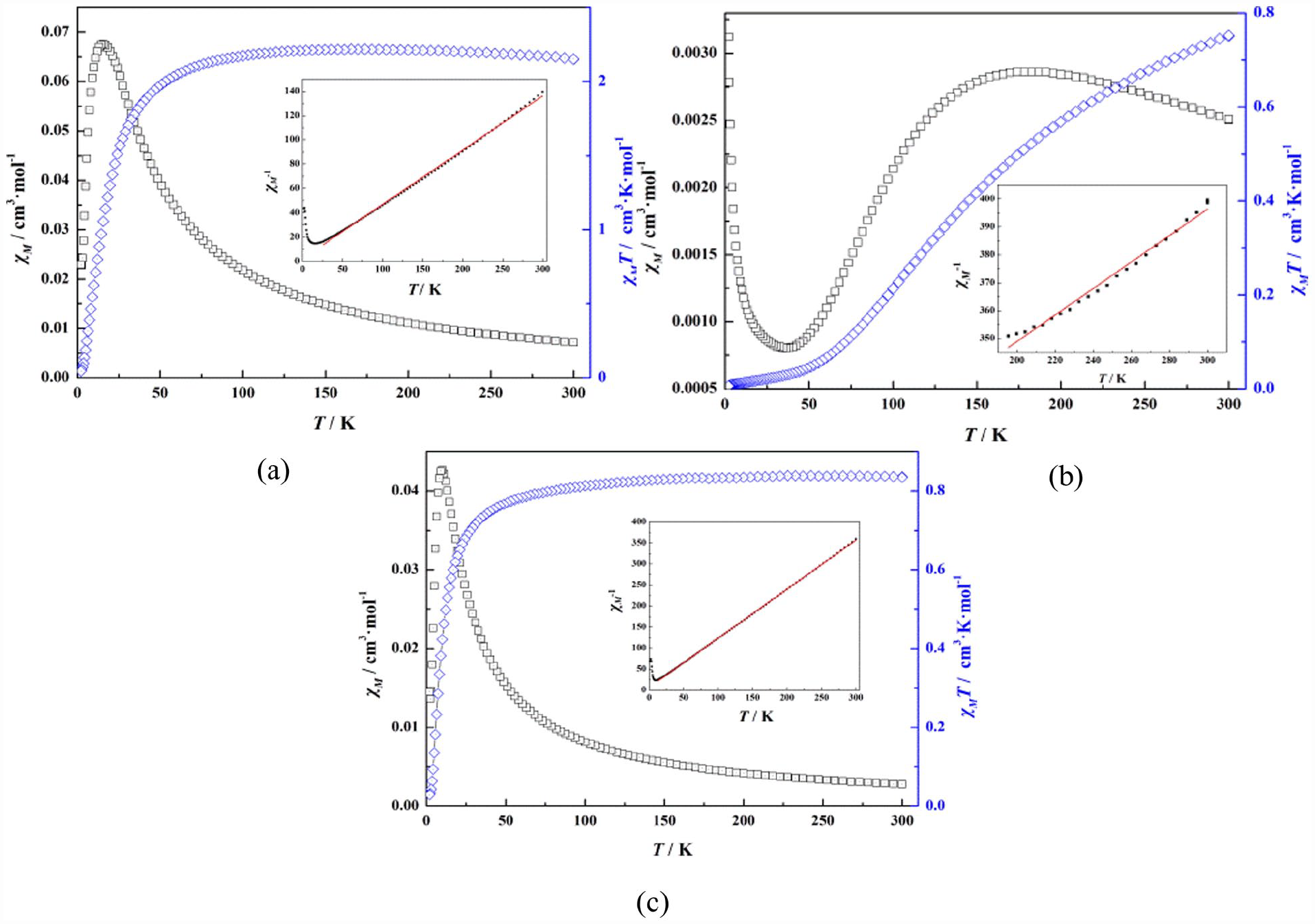
Temperature dependence of χm, χmT, and χm−1 for complexes
For complex
For complex
Conclusion
In summary, three new complexes, {[Ni2(3-PCA)2(DMAP)4(H2O)2]•2H2O}2
Experimental
Instruments and reagents
All commercial chemicals and solvents were used without purification. The structures of the complexes were also been confirmed by single-crystal X-ray (Agilent G8910A CCD) diffraction analyses. IR spectra were recorded on a PE Spectrum One FT-IR Spectrometer (KBr pellet). PXRD data were obtained on a Bruker D8 Advance X-ray diffractometer. The thermogravimetric measurements were performed with an SDTQ 600 apparatus. Magnetic measurements were recorded on an MPMS SQUID magnetometer.
Preparation of the complexes
{[Ni2(3-PCA)2(DMAP)4(H2O)2]•2H2O}2 (1)
A mixture of Ni(SO4)2·6H2O (0.0526 g, 0.2 mmol), 3-PCA (0.0244 g, 0.2 mmol), DMAP (0.0610 g, 0.5 mmol), CH3OH (5 mL), H2O (10 mL), and triethylamine (40 μL) was stirred at room temperature for 10 min, and then sealed in a 25-mL Teflon-lined stainless-steel container. The mixture was heated at 110 °C for 72 h. After slowly cooling to room temperature, the product was washed with ethanol/distilled water several times and filtered to afford blue needle-like crystals. Yield: 53% based on Ni. anal. calcd. for C36H48N12Ni2O6·2(H2O): C, 49.58; H, 5.85; N, 17.19. Found (%): C, 48.44; H, 5.69; N, 17.12. IR (KBr, cm−1): 3321 (br), 1626 (s), 1562 (m), 1537 (w), 1447 (w), 1380 (s), 1228 (s), 1138 (m), 1063 (m), 1010 (s), 786 (m), 530 (w).
[Cu2(3-PCA)2(4-PP)2·2H2O]n (2)
A mixture of Cu(NO3)2·3H2O (0.0483 g, 0.2 mmol), 3-PCA (0.0244 g, 0.2 mmol), 4-PP (0.0621 g, 0.4 mmol), CH3OH (5 mL), and H2O (10 mL) was stirred at room temperature for 10 min, and then sealed in a 25-mL Teflon-lined stainless-steel container. The mixture was heated at 90 °C for 72 h. After slowly cooling to room temperature, the product was washed with ethanol/distilled water several times and filtered to afford blue needle-like crystals. Yield: 61% based on Cu. anal. calcd. for (C30H22Cu2N6O4)·2(H2O): C, 51.95; H, 3.78; N, 12.12. Found (%): C, 50.74; H, 3.71; N, 12.62. IR (KBr, cm−1): 3466 (br), 1586 (s), 1515 (w), 1459 (w), 1368 (s), 1273 (w), 1224 (m), 1146 (m), 1073 (m), 1034 (w), 824 (w), 768 (m), 693 (w).
[Cu2(4-PCA)2(2,2′-bpy)2·2H2O]n (3)
A mixture of CuSO4·5H2O (0.0749 g, 0.3 mmol), 4-PCA (0.0244 g, 0.2 mmol), 4-PP (0. 0621 g, 0.5 mmol), CH3OH (5 mL), and H2O (10 mL) was stirred at room temperature for 10 min, and then sealed in a 25-mL Teflon-lined stainless-steel container. The mixture was heated at 90 °C for 72 h. After slowly cooling to room temperature, the product was washed with ethanol/distilled water several times and filtered to afford blue needle-like crystals. Yield: 53% based on Ni. anal. calcd. for (C28H18Cu2N8O3)·2(H2O)
X-ray structure determination
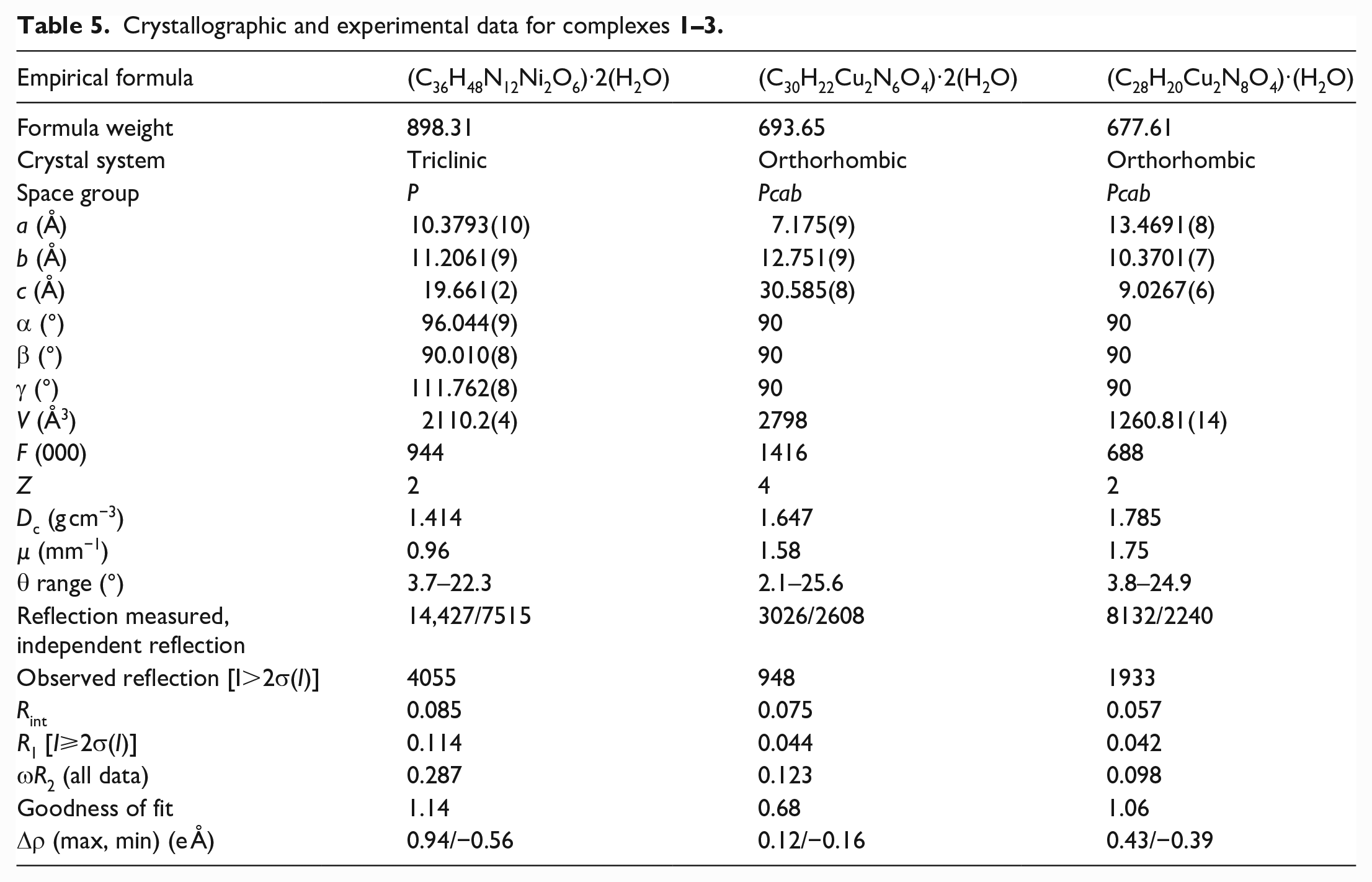
Single-crystal X-ray diffraction analyses of the complexes were carried out on an Agilent Technologies G8910A CCD diffractometer equipped with MoKα radiation (λ = 0.71073 Å) using an ω-scan mode at 293 K. The υ-scan technique was employed to measure intensities. The SADABS program was used to correct the Lorentz and Polarization (LP) factors and semi-empirical absorption of the data. 29 For the initial data, the direct method of the SHELXS program 30 was used to solve the positions of all non-hydrogen atoms and the theoretical hydrogenation method was used to hydrotreat all hydrogen atoms. The analytical data were refined and improved by the least squares method of the SHELXS program. All data analysis and refinement processes were completed using Olex2.31,32 Crystal data as well as details of the data corrections and refinements for the three complexes are summarized in Table 5.
Crystallographic and experimental data for complexes
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nature Science Foundation of Guangxi Province of China (no. 2017GXNSFAA198268).
