Abstract
A coordination compound constructed from a nitronyl nitroxide biradical NITPh(3-NIT) and CuII(hfac)2(H2O)2 building blocks [NITPh(3-NIT) = 1,3-bis(1′-oxyl-3′-oxido-4′,4′, 5′, 5′-tetramethyl-4,5-dihydro-1H-imidazol-2-y1)-benzene, hfac = hexafluoroacetylacetonato] is successfully synthesized. The crystal structure is determined by X-ray single-crystal diffraction. The asymmetric complex {[(NITPh(3-NIT)]Cu(hfac)2} consists of one Cu(II) ion and two >N–O• groups and adopts a distorted triangular bipyramid with a penta-coordinated central copper(II) atom and three hfac oxygen atoms at the base and a >N–O• oxygen atom and one hfac oxygen atom at the apices. Intramolecular O. . .O bonding and π–π stacking interactions are observed between molecules. A magnetic susceptibility study of the coordination compound shows antiferromagnetic interactions between Cu(II) ions and >N–O• groups and very weak ferromagnetic interactions between Cu(II) ions and the free >N–O• group through O. . .O bonding between the nitroxide group oxygen atom and the oxygen atom of hfac.

Introduction
Cu(hfac)2 is widely used to prepare molecular magnets (hfac = hexafluoroacetylacetonate). On one hand, it is a strong acceptor readily forming complexes with a coordinated nitroxyl group because of the presence of the strong electron-withdrawing hfac co-ligands. On the other hand, the interactions of heterospin systems with nitroxides yield magnetic properties that are convenient for subsequent theoretical analysis. In recent years, multifunctional derivatives of stable nitroxides have been used for syntheses of heterospin systems that are increasingly complicated with three-dimensional structures and heterospin exchange clusters with different structures.1–3 Hence, favorable opportunities are available for sequential definition of magnetostructural correlations through modification of the paramagnetic ligand molecule, which can change the structure and the magnetic behavior of the exchange cluster.4–6
The use of organic monoradicals is well documented, with many interesting examples of the creation of new molecular magnetic compounds in the literature. Such complexes display several architectures and different behaviors.7–14 Organic biradicals have also attracted considerable interest as building blocks for molecular magnetic materials. 15 This is because it is possible to vary the ground spin state by choosing an appropriate conjugated spacer between groups with unpaired spin. The m-phenylene ring is one of the most frequently used spacers due to its non-disjointed connectivity which induces an S = 1 spin ground state for many biradicals.16–22 Also, heteroatom substituents or the inclusion of functional group substituents on this ring allows modification of the overall magnetic properties.23,24 In addition, much attention has been devoted to nitronyl nitroxide (NIT) biradicals in the field of molecular magnetism. They represent useful building blocks for heteromolecular complexes in the so-called metal-radical approach. Hence, many possibilities remain unexplored for coordination using these spin carriers to tune structural and magnetic behavior by using different spacer groups to connect the radical units. 25
Inspired by these ideas, we synthesized an NIT biradical (Scheme 1), which can provide an efficient route to magnetic spin exchange. In this contribution, we report the crystal structure and magnetic properties of {[(NITPh(3-NIT)]Cu(hfac)2} obtained from NIT biradicals and CuII(hfac)2 building blocks.
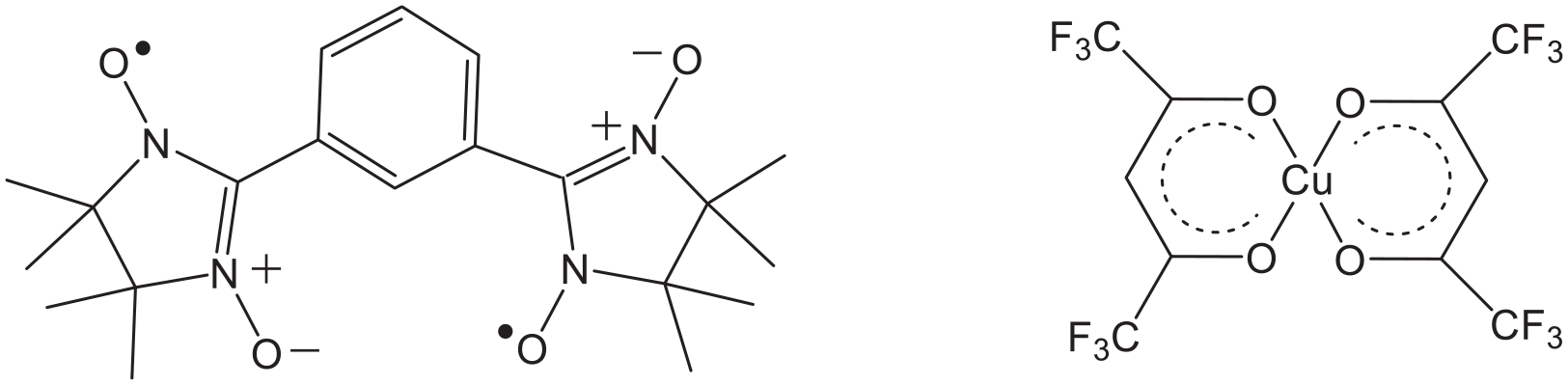
Organic biradicals NITPh(3-NIT) and inorganic complexes: CuII(hfac)2.
Results and discussion
Preparation of the biradical NITPh(3-NIT)
The biradical NITPh(3-NIT) was prepared as reported previously with only minor modifications;26,27 the synthetic route is shown in Scheme 2.

Synthesis of the NITPh(3-NIT) biradical.
The preparation of the {[NITPh(3-NIT)]Cu(hfac)2} complex is described in the experimental.
Crystal structure of the {[NITPh(3-NIT)]Cu(hfac)2} complex
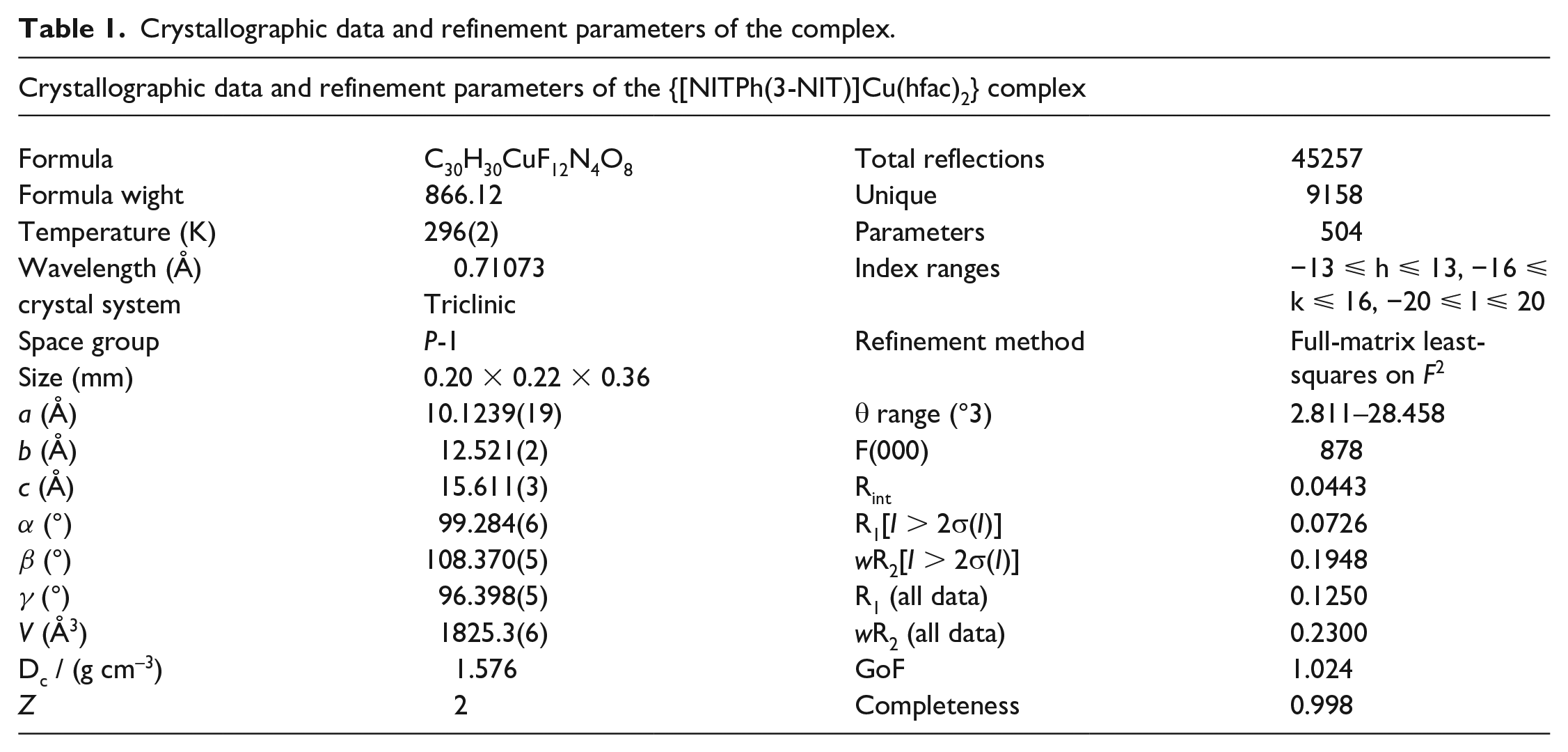
The molecular structure of {[NITPh(3-NIT)]Cu(hfac)2} was successfully determined by means of X-ray crystallographic analysis. The crystal and refinement data are summarized in Table 1. Table 2 shows selected crystallographic data. {[NITPh(3-NIT)]Cu(hfac)2} crystallizes in the triclinic space group P-1 with Z = 2. The molecular structure is shown in Figure 1(a), revealing that the complex forms a centrosymmetric dimer. The asymmetric unit of the complex consists of one crystallographic-dependent copper center coordinated to one NITPh(3-NIT) biradical ligand. The CuII atom is penta-coordinated by one radical and two hfac ligands, leading to a compressed triangular bipyramid with three hfac oxygen atoms (O5, O7, O8) at the base and with N–O (O1) and an hfac oxygen atom (O6) at the apex as shown in Figure 1(b). The packing arrangement of the {[NITPh(3-NIT)]Cu(hfac)2} complex shows that the nearest intermolecular metal–metal contacts give a Cu. . .Cu distance of 7.610(1) Å, while the intercell Cu. . .Cu distance is 10.272(2) Å.
Crystallographic data and refinement parameters of the complex.
Selected bond lengths (Å) and angles (°) for {[NITPh(3-NIT)]Cu(hfac)2} at 296 K.

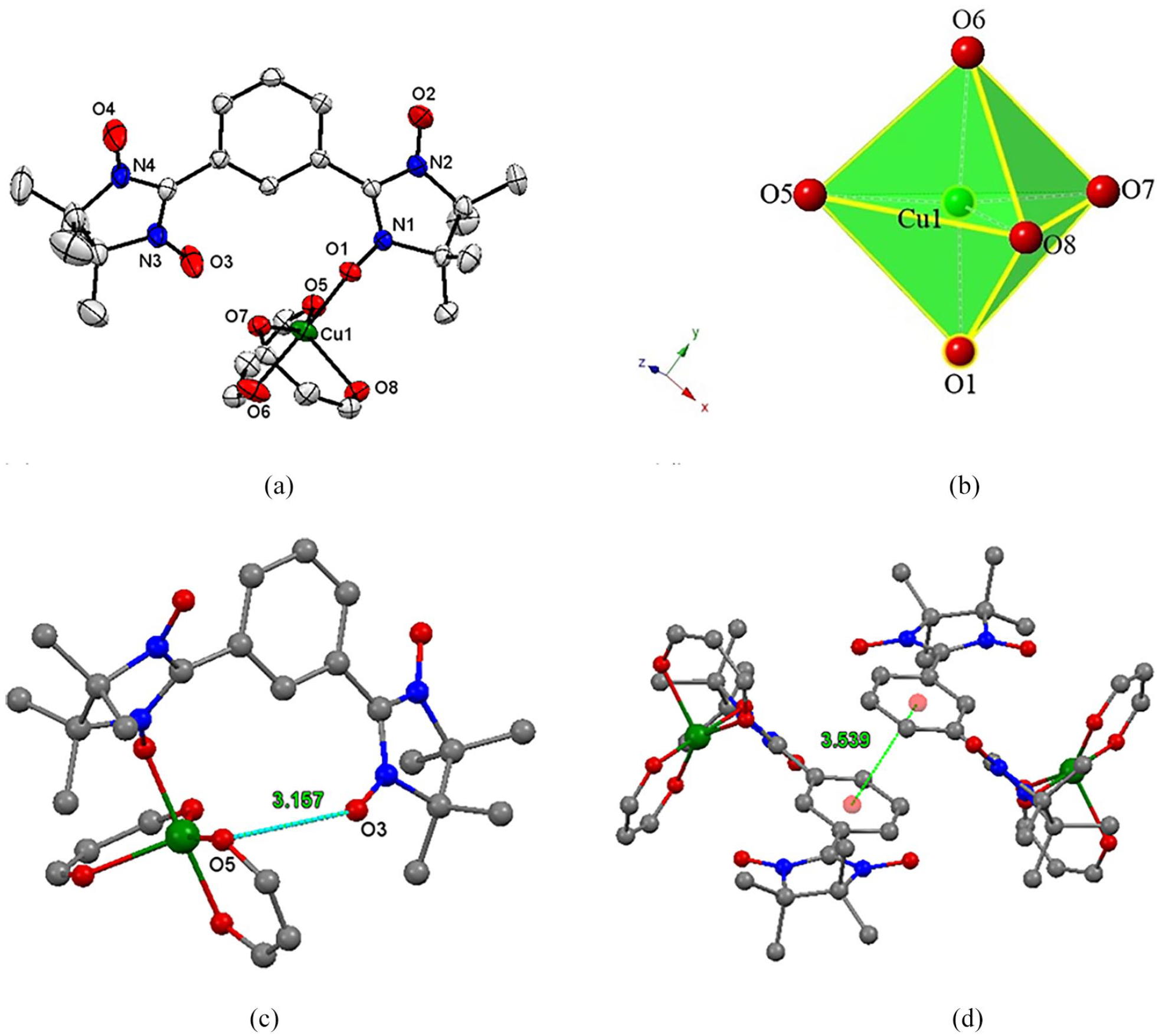
(a) Molecular structure of the asymmetric unit of {[(NITPh(3-NIT)]Cu(hfac)2} with ellipsoids at 50% probability level, (b) the bipyramid structure with a penta-coordinated central copper(II) atom, (c) intramolecular O. . .O contacts, and (d) π. . .π stacking interactions. Fluorine and hydrogen atoms are omitted for clarity.
The apical Cu–O1 and Cu–O6 bond lengths are 1.937(2) and 1.960(3) Å, respectively, which are typical for copper(II)–nitroxide equatorial coordinated bonds (1.93–2.03) Å, while the equatorial Cu–O(hfac) bonds, Cu–O5, Cu–O7 and Cu–O8, are 1.920(3) Å, 2.001(3) Å, and 2.157(3) Å, respectively. The bond angles of N1–O1–Cu and O1–Cu–O6 are 125.2(2)° and 175.4(1)°, respectively. In the biradical, the O1–N1–C8–N2–O2 and O3–N3–C7–N4–O4 angles are almost planar, and the conjugation plane O1–N1–C8–N2–O2 and the benzene plane have a dihedral angle of 41.44°. The plane of the benzene ring makes an angle of about 20.71° with the O3–N3–C7–N4–O4 plane. In addition, there is one slightly longer intramolecular O. . .O interaction (O3⋯O5 = 3.157(6) Å) (Figure 1(c)) and two intermolecular F. . .F interactions (F9. . .F11 = 2.972(6) Å and F3. . .F12 = 3.08(1) Å), which contribute to the layered structure as shown in Figure 2. Moreover, interlayer π···π stacking interactions with a distance of 3.539 Å make the adjacent coordination layers more stable (Figure 1(d)).

Crystal packing of the {[(NITPh(3-NIT)]Cu(hfac)2} complex showing the F. . .F (blue) intramolecular short contacts. Hydrogen atoms are omitted for clarity.
Magnetic properties
The temperature-dependent magnetic susceptibilities of the complex were measured in the temperature range of 2–300 K in an external magnetic field of 20 kOe, and the data were corrected for the diamagnetism of the components.
The magnetic data for the {[(NITPh(3-NIT)]Cu(hfac)2} complex are shown in the plots in Figure 3 as χm versus T and χmT versus T. For χmT (square), at room temperature, the value is 0.378 cm3 K mol−1, which is equal to the theoretical value for an uncoupled system of one radical (S = 1/2, χmT = 0.375 cm3 K mol−1), assuming g = 2, indicating the existence of strong antiferromagnetic interactions between copper and the coordinating N–O• group. At a lower temperature, χmT increased to reach a value of 0.419 cm3 K mol−1, which is greater than the spin value for S = 1/2 with g = 2. The Curie and Weiss constants were 0.378 cm3 K mol−1 and 3.01 K, respectively, from the Curie–Weiss fit of the reciprocal susceptibility versus the temperature curve for the complex (Figure 3, red solid line). The ferromagnetic interactions among the spin carriers are weak. In fact, the intramolecular distances between the nitroxide group oxygen atom and the CuII center are large, being 7.610 Å. A possible path way for the magnetic interactions would be through intramolecular O. . .O bonding between the nitroxide (O3) and the hfac oxygen atom (O5). A careful analysis of this intramolecular contact showed that the distance between the magnetic centers was shorter than the intramolecular distance (O3. . .Cu = 3.477 Å).

Temperature dependence of χm and χmT versus T for the {[NITPh(3-NIT)]Cu(hfac)2} complex. The solid lines show the best fit.
In order to account for the magnetic behavior of the complex, a model with three exchange coupling constants was used (Figure 3), where J1, J2, and J3 represent the intermolecular magnetic interactions between radical–radical, metal–radical and metal–uncoordinated radical, respectively. The magnetic data are the result of the three interactions, but the magnetic interaction between the metal and uncoordinated radical, J3, made the most important contribution. It is important to stress that a simplified model was used to describe the magnetic behavior. The blue solid line in Figure 3 shows the best fit achieved with grad = 2.00 (fixed) and J = 3.47 cm−1. These values indicate that the magnetic coupling between the Cu(II) atom and the uncoordinated nitroxide radical is weakly ferromagnetic.
The field dependence of the magnetization has been determined at 2 K in the 0–50 kOe field range (Figure 4). Upon increasing the applied field, M increases up to 0.96 NµB at 50 kOe, which is near saturation.
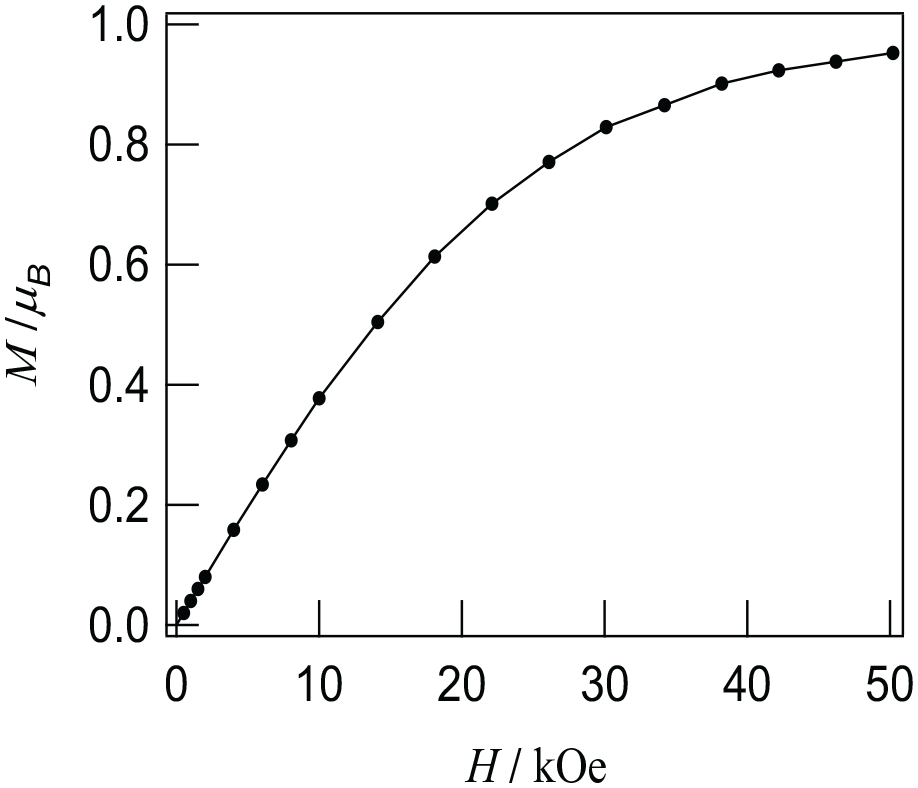
Isothermal magnetization at 2 K of {[NITPh(3-NIT)]Cu(hfac)2}.
Conclusion
A centrosymmetric {[(NITPh(3-NIT)]Cu(hfac)2} complex based on copper(II) and NIT biradicals has successfully been prepared. The single-crystal X-ray structure shows that one of the >N–O• groups is linked to the Cu(II) ion to form a distorted triangular bipyramid with a penta-coordinated central copper(II) atom. The magnetic measurements indicate that the complex exhibits antiferromagnetic interactions between Cu(II) ions and the linking >N–O• group, but weak ferromagnetic interactions with the free >N–O• group through the O. . .O contacts. The magnetostructural correlations are explained based on the crystal structure.
Experimental
All reagents were obtained from commercial sources and used without further purification.
Preparation of Cu(hfac)2·2H2O
Hfac (1 g, 7.1 mmol) was added to a suspension of Cu(CH3COO)2·H2O (0.71 g, 3.55 mmol) in H2O (17 mL) at room temperature and then NaHCO3 (0.071 g, 0.8 mmol) was added with stirring. After 10 min, a light blue precipitate was obtained, which was washed three times with n-hexane. The light blue solid was dried at 50 °C for 3 days to afford Cu(hfac)2·2H2O (0.74 g). FTIR (KBr, cm−1): 3396(s), 1647(s), 1551(s), 1493(s), 1216(s), 1157(s), 808(s), 769(w), 745(w), 669(s), 591(s).
Preparation of {[NITPh(3-NIT)]Cu(hfac)2}
Cu(hfac)2(H2O)2 (1 mmol, 51.5 mg) was dissolved in 10 mL of hot n-heptane and then the solution was cooled to about 50 °C. NITPh(3-NIT) (1 mmol, 39.0 mg), dissolved in 2 mL of CHCl3, was added with constant stirring. The mixed solution was stirred for ca. 3 min and then cooled to room temperature. The filtrate was kept under an N2 stream at room temperature until a solid appeared. A few drops of CHCl3 were added until the solid disappeared and then the flask was sealed and placed in a dark room. After 5–6 days, block-shaped, dark blue crystals were obtained. Yield: 72%. Anal. calcd for C30H30F12CuN4O8: C, 41.57; H, 3.46; N, 6.47; found: C, 41.74; H, 3.69; N, 6.32. FTIR (KBr, cm−1): 1662(s), 1554(w), 1531(s), 1477(s), 1358(w), 1223(s), 1147(s), 1087(s), 866(s), 800(w), 661(s), 604(s).
General characterization
Infrared spectra were recorded on a JASCO FT/IR-660 PLUS spectrometer by transmission through KBr pellets in the range of 400–4000 cm−1. Elemental analyses for C, H, and N were recorded using a PerkinElmer series II CHNS/O Analyzer 2400. The magnetization measurements on the crystalline solids were carried out using Quantum Design MPMS-5S and MPMS-2 SQUID magnetometers. The magnetic field was varied from −50 to 50 kOe and the temperature from 2–300 K. The data were corrected for the sample diamagnetism using Pascal’s constants.28,29
Single-crystal X-ray diffraction studies
The X-ray diffraction studies were performed on for the selected single crystals individually mounted on glass fibers at different temperatures using Bruker SMART-APEX II diffractometer with a CCD, D8-QUEST, and a CMOS area detector. Graphite-monochromated MoKα (λ = 0.71073 Å) radiation was employed. Data reduction was performed using SAINT and the intensities were corrected for absorption by SADABS.30,31 The structures were solved by direct methods and refined by full-matrix least-squares against F 2 using ShelXL. 32 Part of the hydrogen atoms was located in Fourier difference maps and those not found were added at theoretical positions using the riding model. The details can be obtained from the cif files deposited at the Cambridge Crystallographic Data Centre.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Doctoral Scientific Research Foundation of Yulin University (No. 18GK24), the Science and Technology Program of Shaanxi Province (No. 2017GY-131), a Project of Production and Study of Yulin City (No. 2016CXY-01), and the National Nature Science Foundation of China (No. 21968036).
