Abstract
Allyl alcohol and water can form the lowest boiling point azeotrope, but it is very hard to acquire high-purity allyl alcohol by normal distillation methods. Herein, a separation and purification protocol is developed for perfectly separating an azeotropic mixture of allyl alcohol and water using the salting-out method, in which three potassium phosphate salts, K5P3O10, K3PO4, and K4P2O7, are systematically investigated as salting-out agents, and finally a product consisting of > 99% allyl alcohol is obtained. A thermodynamic study demonstrates that this process involves endothermy and increment entropy. There is a good correlation between the solubility of allyl alcohol and the molar concentration of salt (mol per 1 kg water), and the solubility of the allyl alcohol in the organic phase or the water phase can be forecast using the mass percentage of salts. This work provides a new methodology for the efficient separation of an azeotropic mixture of allyl alcohol and water.
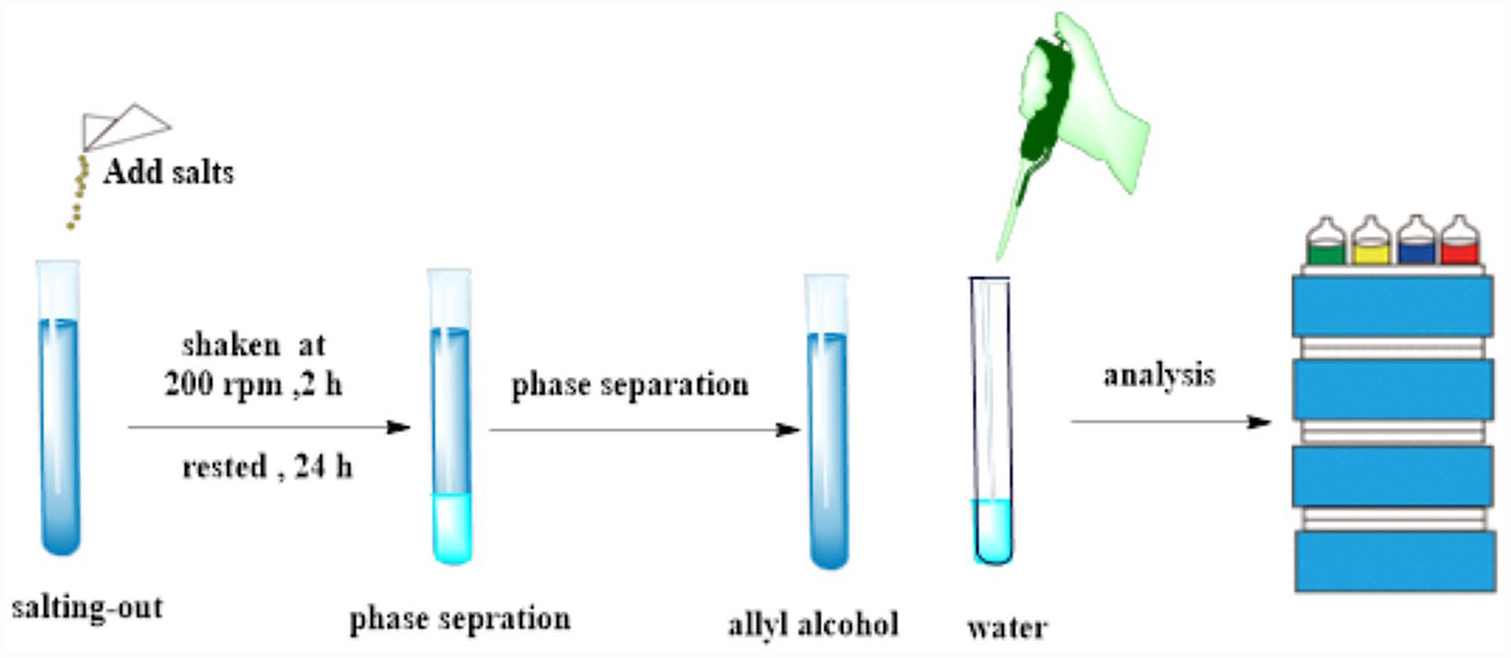
Introduction
Allyl alcohol contains C=C and C–OH groups, and a series of allyl alcohol derivatives can be easily obtained by substitution, oxidation, addition, rearrangement, and polymerization reactions.1–4 Allyl alcohol-based chemicals have numerous applications, for example, fragrances, pharmaceuticals, food formulations, lenses, coupling agents, plasticizers, crosslinking agents, and paint additives. The wide application of allyl alcohol has created a huge market demand.5–7 In industry, the hydrolysis of allyl acetate is often used to prepare allyl alcohol, resulting in a product mixture consisting of allyl alcohol, water, minute quantities of allyl aldehyde, and acetic acid.8,9 Allyl alcohol (b.p. 96.9 °C) and water have very similar boiling points and can form an allyl alcohol (72 wt%)–water (28 wt%) azeotrope. 10 It is almost impossible to produce allyl alcohol with a purity higher than 72% using conventional distillation methods.11,12 Special distillation methods are proposed for separating azeotropes, for example, extractive, pressure swing, azeotropic and reactive distillation,13,14 and distillation itself requires high energy consumption. 15 Consequently, it is not an ideal method to separate allyl alcohol and water.
Previous work has reported the use of vapor–liquid equilibrium (VLE) for the efficient separation of allyl alcohol and water.11,12,16,17 The VLE data for systems consisting of allyl alcohol + water, allyl alcohol + water + calcium nitrate, allyl alcohol + water + calcium chloride, and allyl alcohol + water + magnesium nitrate were measured at a pressure of 101.3 kPa. 12 Salting-out technologies have many advantages, such as energy saving, simple operation, and no need for extractants.18–20 Three high-solubility salting-out potassium salt-based agents (K2CO3, K3PO4, and K4P2O7) showed outstanding salting-out effects in a 1,3-propanediol solution system.21,22 K2HPO4 has been widely studied as a promising salting-out agent, which can be recovered (acetone + butanol + ethanol) from the pre-fractionation tower.23–25 The salting-out effects of four high-solubility salts (K4P2O7·3H2O, K2HPO4·3H2O, K2CO3, and K3PO4·3H2O) on 2,3-butanediol from various aqueous solutions were studied at 298.15 K.26,27
In this work, a separation and purification protocol has been developed for the highly efficient separation of allyl alcohol from its azeotrope with water using the salting-out method (Scheme 1). In addition, the salting-out effects of three high-solubility salts (K5P3O10, K3PO4, and K4P2O7) on the separation of an allyl alcohol from azeotrope (allyl alcohol + water) were investigated in detail. As a result, a product > 99% allyl alcohol was obtained. A thermodynamic study demonstrated that this process involved endothermy and increment entropy. There is a good correlation between the solubility of allyl alcohol and the molar concentration of salt (mol per 1 kg water), and the solubility of the allyl alcohol in the organic phase or the water phase can be forecast using the mass percentage of salts.

Process for the separation of allyl alcohol from its azeotrope with water.
Results and discussion
The liquid–liquid equilibria
Allyl alcohol and water are miscible, and no interface was observed. Thus, for their separation, the salting-out technology was applied, and three potassium phosphate salts, K5P3O10, K3PO4 and K4P2O7, were used to obtain high-purity allyl alcohol. Salts having a mass percentage greater than 10% induced liquid–liquid phase splits owing to the salting-out effect. The salting-out procedure is often interrelated to the cations and anions produced by the dissociation of inorganic salts. Water is largely attracted by anions and cations to form a hydration shell. Large amounts of K5P3O10, K3PO4, and K4P2O7 were ionized in the aqueous solution to produce cations and anions that repelled allyl alcohol, thereby enriching the organic phase. Hence, this is necessary to increase the mass percentage of salt to enrich the total ions. Although the phase separation was induced by relatively low salt contents, the separation efficiency was not significant, as shown in Figures 1 and 2. The mass percentage of allyl alcohol in the organic phase was relatively low and the mass percentage of water in the organic phase was relatively high. However, enhancing the mass percentage of salts increased the allyl alcohol content in the organic phase and reduced the water content in the organic phase. When the mass percentage of K4P2O7 was increased from 10% to 60%, the mass percentage of allyl alcohol in the organic phase rose from 87.1% to > 99%. When 60% K5P3O10, 70% K3PO4, and 60% K4P2O7 were added to the allyl alcohol solution, the allyl alcohol solution reached its saturation point. When the mass percentage of salts continued to rise, crystals were observed at the bottom of the vials. The results indicate that K4P2O7 produced the strongest hydrophilic interaction compared with the other salts. This may be because K4P2O7 has a strong hydrophilic effect and hydrolyzable hydroxide ions, 28 and thus K4P2O7 shows an excellent salting-out effect for biobutanol. 29 In this work, K5P3O10, K3PO4, and K4P2O7, as salting-out agents, were developed for perfectly separating an azeotropic mixture of allyl alcohol and water through using the salting-out method, and finally a product consisting of > 99% allyl alcohol was obtained.

Mass fraction of allyl alcohol in the organic phase plotted for different salting-out agents versus the mass fraction of salts at room temperature.

Mass fraction of water in the organic phase plotted for different salting-out agents versus the mass fraction of salts at room temperature.
The recovery efficiency of allyl alcohol
The ultimate goal of salting-out is to recover allyl alcohol. According to our experiment, the recovery efficiency of allyl alcohol (R) can be calculated as follows
where m represents the mass of allyl alcohol before the salting-out experiment, m2 represents the mass of the organic phase, w22 represents the mass percentage of allyl alcohol in the organic phase, and m and m2 are determined by a gravimetric analysis.
As shown in Figure 3, there were significant improvements in the recovery of allyl alcohol after increasing the mass percentage of K5P3O10, K3PO4, and K4P2O7. The order of the recovery was as follows: R(K4P2O7) > R(K3PO4) > R(K5P3O10) at the same salt mass percentage. After the addition of 60% K4P2O7, the recovery of allyl alcohol was 99.7%, which was much higher than the results processed by 60% K5P3O10 and 70% K3PO4 at room temperature. Thus, K4P2O7 has a better separation effect than the other two salts.

Recovery of allyl alcohol with different salting-out agents versus the different mass fraction of salts.
The thermodynamic parameters of salting-out
The salting-out temperature of the allyl alcohol system effects the distribution coefficient. The relationship between the distribution coefficient and temperature is as follows
where K represents the distribution coefficient, T represents the absolute scale of temperature, R represents the universal gas constant, and ΔH0 represents the molar enthalpy change, and
where Corg represents the concentration of allyl alcohol in the organic phase, and Caq represents the concentration of allyl alcohol in the water phase.
When the type of salt was changed and the other conditions were fixed, various distribution coefficient values were calculated at different temperatures. The relationships between lnK and 1/(R × T) on a salt basis for the water + allyl alcohol system are shown in Figure 4. When the mass percentage of K5P3O10 was 20%, then ΔH = 10.3 kJ·mol−1, when the mass percentage of K3PO4 was 20%, ΔH = 11.6 kJ·mol−1, and when the mass percentage of K4P2O7 was 20%, ΔH = 15.1 kJ·mol−1.

Relationship between lnK and 1/(R × T) on a salt-free basis for water + allyl alcohol system with ■ 20% K5P3O10, ● 20% K3PO4, and▲ 20% K4P2O7.
In accordance with equation (2),30,31 if salting-out is endothermic, then the distribution coefficient K increases along with temperature and vice versa. Thus, the salting-out reactions of K5P3O10, K3PO4, and K4P2O7 were endothermic. ΔG and ΔS can be calculated using the following two formulae
in which ΔG represents the Gibbs energy change and ΔS represents the entropy change.
As shown in Table 1, as the temperature increased, ΔG decreased and was less than zero; consequently, the salting-out of allyl alcohol was a spontaneous and entropy-increasing process.
The thermodynamic parameters of salting-out at different temperatures.

The solubility correlation
All the experimental results showed that the salting-out efficiency from the allyl alcohol aqueous solution was relevant to the type of salt and the mass percentage of salts. The process decreased the solubility of allyl alcohol in the water phase and enriched the allyl alcohol in the organic phase as the mass percentage of salts increased. The dilute aqueous solution became a supersaturated solution after a large number of water molecules had been captured by salt ions. The Setschenow equation shows that the solubility of organic compounds in salt solution has a good linear relationship with salt concentration. 32 Thus, the solubility of allyl alcohol can be correlated with the mass percentage of salts. It was therefore necessary to find a method to evaluate the salting-out effects of K4P2O7, K3PO4, and K5P3O10 on the salting-out of allyl alcohol + water solutions.
The solubility of allyl alcohol in the water phase calculated in units of g per 100 g water is defined as follows

in which w21 represents the mass percentage of allyl alcohol in the water phase, and w11 represents the mass percentage of water in the water phase.
The measurement of the salt concentration in the water phase is provided as follows

where b represents the molarity of salts in units of mol per 1 kg water, M represents the molar mass (for K5P3O10, M = 448.4 g mol−1, for K3PO4, M = 212.3 g mol−1, for K4P2O7, M = 330.3 g mol−1), w31 represents the mass percentage of salts in the water phase, and w11 represents the mass percentage of water in the water phase.
To fully understand the salting-out process of allyl alcohol, the relationship between the natural logarithm of the solubility of allyl alcohol in the aqueous phase and the molar concentration of salt was plotted (Figure 5). The two factors showed a linear relationship. Thus, it was concluded that the solubility of allyl alcohol in the water phase was greatly influenced via the molality of the salt. The solubility of allyl alcohol in the water phase decreased as the molality of the salt increased. However, in the cases of K4P2O7, K3PO4, and K5P3O10, the solubility of allyl alcohol changed greatly because different anions (P3O105−, PO43−, and P2O74−, respectively) had obvious negative effects on the salting-out of alcohol. 33 The fitting empirical constants of the linear regression are displayed in Table 2.

Plot of ln(S21) against the molality of the salt.
The constants α and β and the coefficient of determination, R2.

The linear regression of the data obtained from a particular allyl alcohol aqueous solution and a particular salting-out agent (K4P2O7, K3PO4, or K5P3O10) was determined using the following equation

The slope of α(K4P2O7) < α(K3PO4) < α(K5P3O10) which indicated that the solubility reduction of allyl alcohol in the water phase caused via 1 mole of K4P2O7 was greater than that caused by 1 mole of K3PO4 or K5P3O10. Therefore, the salting-out effects of the three salting-out agents on an allyl alcohol–water solution were as follows: K4P2O7 > K3PO4 > K5P3O10, which was also verified from the recovery ratios of allyl alcohol. Improving the amount of salt strengthened the hydration power of the salt in the allyl alcohol aqueous solution, with more water molecules being attracted from the organic phase to the water phase. Therefore, the solubility of allyl alcohol in the water phase was significantly reduced, and the solubility in the organic phase was significantly increased. The solubility of allyl alcohol in the organic phase is shown below
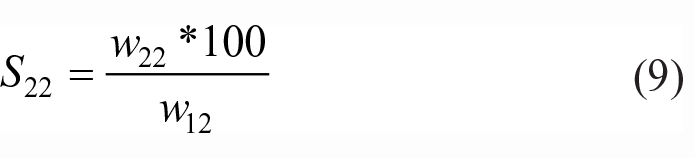
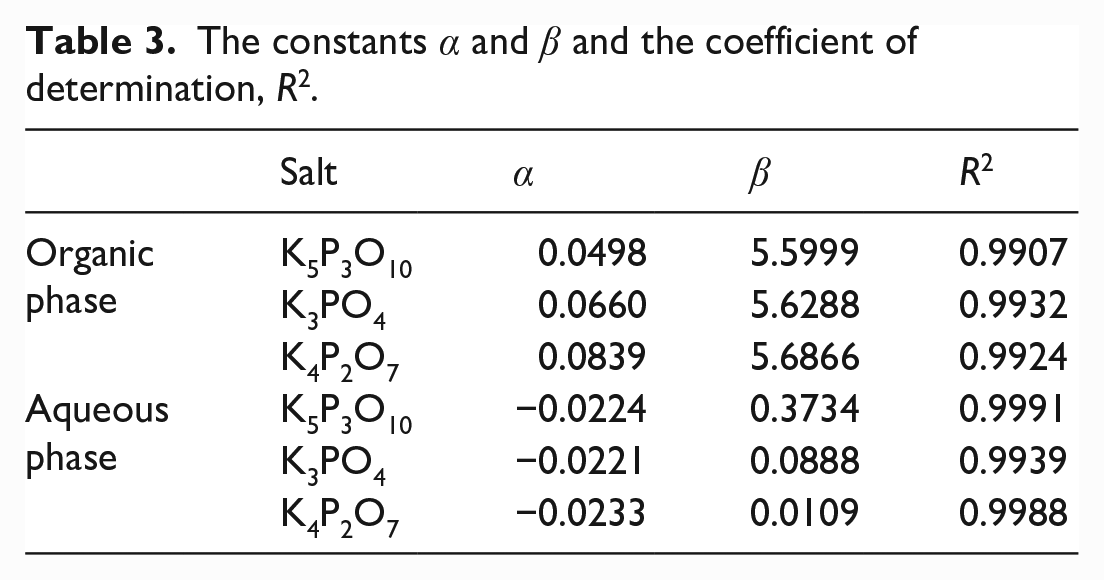
The solubility of allyl alcohol in the organic phase increased and the solubility of allyl alcohol in the water phase decreased as the mass percentage of salt increased. The ln(S) value and mass percentage of salts showed a good linear relationship. The constants α and β and the correlation coefficient, R2 values, are shown in Table 3.
The constants α and β and the coefficient of determination, R2.
Thus, the solubility of allyl alcohol in the organic phase or the water phase can be projected by the mass percentage of salts, as follows

Once the mass percentage of a specific salt is known, the solubility of allyl alcohol in both phases can be calculated.
The K4P2O7 residue in the organic phase
The mass percentage of salts in the organic phase is a key factor for assessing the energy-saving efficiency. If a salt dissolves in the organic phase, then it leads to the challenge of salt separation. As shown in Figure 6, the K4P2O7 content in the organic phase decreased when more K4P2O7 was added into the (allyl alcohol + water + salt) system. In addition, the mass percentage of 60% K4P2O7 in the organic phase was less than 120 ppm, which may be because the high mass percentage of allyl alcohol repelled the ions. The residue of K4P2O7 in the organic phase (less than 120 ppm) did not necessitate subsequent distillation. The trace of salt in the organic phase will not negatively affect the industrial applications of allyl alcohol.

Mass fraction of K4P2O7 in the organic phase plotted against different mass fractions of salts.
Conclusion
A separation and purification protocol has been developed for the efficient separation of the azeotropic mixture of allyl alcohol + water using the salting-out method and a greater than 99% allyl alcohol content was obtained after the salting-out process. The recovery ratio of allyl alcohol using the three tested potassium phosphate salts can be ordered as follows: K5P3O10 < K3PO4 < K4P2O7 at the same salt mass percentage. Hence, K4P2O7 is the best salt to separate allyl alcohol compared with K3PO4 and K5P3O10. When the mass percentage of K4P2O7 was 60%, allyl alcohol was separated with the purity of 99%. A thermodynamic study demonstrated that the process involved endothermy and increment entropy. A new equation for predicting the solubility of the allyl alcohol in both phases using the mass percentage of salts has been constructed. This work provides a new methodology for the efficient separation of an azeotropic mixture of allyl alcohol + water.
Experimental section
Materials
The specific information for the reagents, including the supplier, purity, and analysis approach for purity, is given in Table 4. Potassium pyrophosphate, potassium tripolyphosphate, and potassium phosphate were vacuum-desiccated for at least 24 h.
Specification of the chemicals.

High-performance liquid chromatography.
Karl Fischer titration.
Salting-out experiments
The salting-out experiments were performed in gas airtight vials with sealing lids at 25 °C. Each vial was charged with 72 wt% allyl alcohol aqueous solution (10 mL) and a quantitative amount of salting-out agent, and then shaken at 25 °C at 200 rpm for 2 h, mixtures were allowed to stand for 24 h to reach complete phase separation. All the salting-out experiments were repeated two times.
Analytical approaches
The content of allyl alcohol in the salting-out experiment was ascertained through high-performance liquid chromatography (HPLC, Shimadzu, LC-20AD), and analytical methods were performed as described in the literature. 34 The salt ion contents were determined through inductively coupled plasma atomic emission spectrometry (Agilent 725, ICP-AES). The water content was determined through Karl Fischer titration (831 KF coulometer, Metrohm, Switzerland).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was jointly funded by the National Natural Science Foundation of China (Grant No. 52203110), the Doctoral Research Initiation Fund of Shanxi University of Chinese Medicine (Grant Nos. 2020BKS05 and 2020BK14), the Shanxi Provincial Department of Education Fund Project (Grant No. 2021L365), the Shanxi Provincial Science and Technology Department Fund Project (Grant No. 202203021212338), the Shanxi Provincial Science and Technology Innovation Talent Team (Grant No. 2022040510010), the Scientific and Technological Innovation Team of Shanxi University of Chinese Medicine (Grant No. 2022TD2010), and the Leading Team of Medical Science and Technology, Shanxi Province (Grant No. 2020TD05).





