Abstract
The essential factors that affect the interfacial mass transfer rate of crown ether–ionic liquid systems are studied by examining the physicochemical properties of mixtures of ionic liquids with benzo-15-crown-5. In the present work, the 1-alkyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)imides ionic liquids ([C2MIm][NTf2], [C3MIm][NTf2], [C4MIm][NTf2], and [C5MIm][NTf2]) are adopted as the solvent and benzo-15-crown-5 is used as the solute. A series of binary mixtures of the ionic liquid and benzo-15-crown-5, with different molar fractions of ionic liquids, is formulated by the weight method. The viscosity and density are determined for four binary mixtures of ionic liquid and benzo-15-crown-5 at atmospheric pressure in a temperature range of 298.15 to 343.15 K. The values obtained for viscosity and density are fitted with empirical equations, and the energy barrier, a-constant, and the isobaric thermal expansion coefficient are all calculated. Interactions between the ionic liquid and the solute benzo-15-crown-5 are analyzed, and the above properties are discussed by comparison with systems in which different solutes are present in the same ionic liquid. It is found that interactions between the ionic liquid and benzo-15-crown-5 in the mixtures are more intense than in mixed systems composed of ionic liquids and other solutes.

Introduction
The utilization of nonvolatile ionic liquids (ILs) as extraction solvents in separation processes represents an alternative to conventional volatile, flammable, and toxic organic reagents to prevent the escape of volatile organic compounds (VOCs). In particular, the sector concerning the separation of metallic elements has attracted extensive attention from researchers.1–6 Industrial applications of pure ILs and their binary or ternary mixtures require highly sophisticated data on physicochemical properties such as density and viscosity. These properties are used to calculate process parameters, to design equipment, and to optimize processes to understand the interfacial mass and momentum transfer processes in biphasic systems and to develop corresponding predictive models utilizing state equations.7–9
A major contribution of ILs in the separation and extraction of metal ions is as diluents or co-extractants to assist with chelating agents (e.g. crown ethers, 10 cryptands, 11 etc.) performing extraction. Crown ethers (CEs) are macrocyclic polyethers containing multiple coordination atoms that interact with metal ions matched in size to form stable complexes. 12 Crown ether–ionic liquid extraction systems are of significant interest due to their high selectivity in the extraction and separation of metal ions. As the viscosity of ILs is one to three orders of magnitude higher than those of traditional organic reagents, researchers often prefer to add traditional organic reagents to ILs in order to reduce their viscosity. The viscosity and density properties of mixtures of ILs with conventional organic reagents have been investigated extensively. For example, [C6MIm][NTf2] with 1-octene, 13 [CnMIm][NTf2] with 1-propanol, 14 [C4MIm][NTf2] with tetrahydrofuran (THF) 15 and anisole, 16 and [C4MIm][OAc]) with dimethyl sulfoxide (DMSO), N,N-dimethylacetamide (DMA), and N,N-dimethylformamide (DMF). 17 However, the physicochemical properties of binary mixtures formed by ionic liquids with macromolecular solutes have not been widely reported.
The application of ILs with low viscosity is preferable in separation processes. In the present article, the selected 1-alkyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)imide IL is one of the most commonly utilized extraction solvents for the separation process. In this way, [NTf2−] is a weakly coordinated negative ion that stimulates the complexation of other ligands with metal ions. At the same time, the hydrophobicity and low viscosity of [NTf2−] facilitate the separation of substances. 18 The cavity size of benzo-15-crown-5 (B15C5) is close to the ionic diameter of Li+, which makes it a promising coordination medium for lithium, and it has been widely used for the separation and extraction of lithium isotopes.19–22 Besides, B15C5 is soluble in many organic solvents but is insoluble in water. In addition, B15C5 has the advantages of low cost and a simple synthetic process, thus making it more suitable for use in industry. The physicochemical properties of binary mixtures of ILs mixed with ethers that are liquid at ambient temperatures, such as the cyclic ethers tetrahydrofuran and 1,4-dioxane, have been previously reported. 15 Herein, we report for the first time the physicochemical properties and derivative properties of mixtures of liquid ILs and solid cyclic ethers over a certain temperature range.
The objectives of this work are to investigate the fundamental physicochemical properties of IL–crown ether systems, to provide a basis for the chemical applications of the systems, and to contribute to the physicochemical database of ILs and mixtures. In this report, mixtures of ILs and B15C5 with different molar fractions of ILs were formulated. The viscosities and densities of binary mixtures in the temperature range from 298.15 to 343.15 K at ambient pressure were determined using a viscosity densitometer. Limited by the solubility of B15C5 in ionic liquids, only the basic properties were determined for IL molar fractions in the range of 0.5 to 1. Fitting of the dependence of viscosity and density on the temperature was carried out according to empirical equations, and the energy barriers, a-constants, and the isobaric thermal expansion coefficients were calculated.
Results and discussion
The viscosities and densities of four pure ILs over different ranges have been reported previously by many researchers. For comparison with the present work, the literature experimental data on the viscosities and densities of [CnMIm][NTf2] (n = 2, 3, 4, 5) have been collected and the corresponding relative deviations are presented in Table S1 (see the Supporting Information). It should be noted that the experimental data presented in this work are relatively close to those published in the literature. However, the relative deviations of the viscosity values of [C4MIm][NTf2] at 333.15, 338.15, and 343.15 K reported in Liu et al. 23 are more extensive than those in this work, which are −4.86%, −5.56%, and −5.54%, respectively. Such high deviations may be related to the purity of the IL sample (e.g. water content), and deficiencies in the experimental method. In addition, there are few reports on physicochemical studies for [C5MIm][NTf2], and therefore, the research in this paper can make up for some deficiencies in this regard.
Viscosity and derivative properties
Viscosity
Table 1 shows the viscosity values for pure ILs and the binary mixtures of the ILs + B15C5 at atmospheric pressure (P = 0.1 MPa), and over the temperature range of 298.15–343.15 K. Figure 1 illustrates the viscosities of the four pure ILs [CnMIm][NTf2] (n = 2, 3, 4, 5) with different carbon chain lengths as a function of temperature. As can be seen, as the length of the carbon chain of the substituent on the imidazole group increases at the same temperature, so does the viscosity of the respective IL. This is attributed to the fact that as the length of the cationic chain increases, so does the size of the IL molecules, which in turn leads to an increase in viscosity. 24 Figure 2 indicates the variation of the viscosity as a function of the number of cationic side chain alkyl carbon atoms (N) at 298.15 K. The results show a subtle but clearly visible odd-even effect for the viscosity as the number of alkyl side chain carbon atoms increases. An odd-even effect in asymmetric ionic liquids (from [C2MIm][NTf2] to [C6MIm][NTf2]) was also observed by Rocha et al. 25 In addition, the temperature greatly affects the viscosity, with the viscosity of the four ionic liquids decreasing by 15.433%, 18.115%, 18.586%, and 19.594%, respectively, as the temperature increases from 298.15 to 303.15 K. At higher temperatures, the viscosity changes more slowly with temperature. This is because higher temperatures accelerate the migration of ions, thereby reducing the aggregation of molecules and hence the viscosity. 26 In addition, increasing the temperature leads to a reduction in the strength of intermolecular interactions, which reduces the viscosity.
Experimental values of viscosity, η (mPa·s), for four ILs + B15C5 as a function of mole fraction from 298.15 to 343.15 K at atmospheric pressure (P = 0.1 MPa).

B15C5: benzo-15-crown-5; ILs: ionic liquids.
Standard uncertainties: u(T) = 0.02 K, u(p) = 5 kPa, u(x) = 0.0002, ur(η) = 0.02.
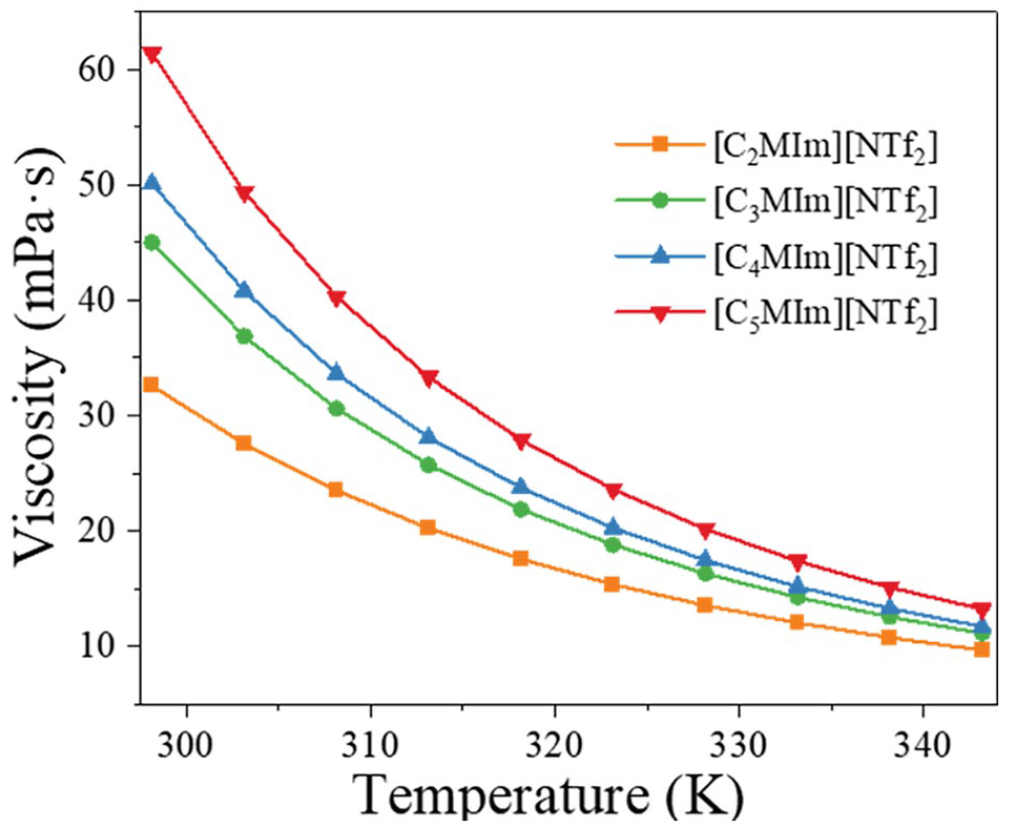
Viscosity as a function of temperature for the pure ILs [C2MIm][NTf2], [C3MIm][NTf2], [C4MIm][NTf2], and [C5MIm][NTf2].

Viscosity as a function of the number of carbon atoms (N) in the IL cationic side chain substituents at 298.15 K.
The Eyring kinetic equation (1)27,28 and the Vogel–Fulcher–Tammann (VFT) equation (2)28–32 are regularly used to characterize the dependence of viscosity on temperature

where η∞ is the viscosity at infinite temperature, R is the gas constant (8.3144621 J·K−1·mol−1), Ea is the activation energy of the solution and is related to the temperature, T is the absolute temperature in Kelvin, and η is the dynamic viscosity
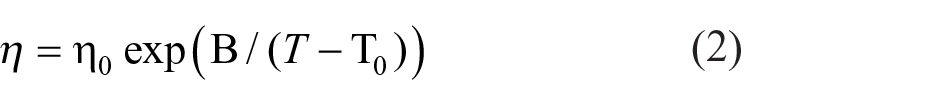
where η0, B, and T0 are the fitted parameters.
The values of the parameters Ea, η∞, and the correlation coefficient R2 fitted by equation (1) are listed in Table S2 (see the Supporting Information), while the parameters η0, B, T0, and the correlation coefficient R2 are provided by equation (2) are listed in Table S3. The values for R2 for the fitting using the VFT equation are all higher than 0.99999, which indicates that the VFT equation is more adequate for the viscosity of ILs + B15C5.
Viscosity depicts the intrinsic resistance of a fluid against shear stress, and the energy barrier E of the fluid versus shear stress can be appraised as a function of viscosity and temperature (equation (3))25,29

where R is the gas constant and B and T0 are the fitted parameters of equation (2).
The values of E for the ILs + B15C5 at a temperature of 298.15 K are listed in Table S3 (see the Supporting Information). As can be seen from the results, the value of E follows the order [C2MIm][NTf2] < [C3MIm][NTf2] < [C4MIm][NTf2] < [C5MIm][NTf2]. The increase in E value with increasing side chain alkyl chain length is consistent with the increase in the energy barrier caused by the increase in shear stress, which is attributed to the van der Waals interaction forces being progressively stronger than the electrostatic interactions. This is consistent with the observations reported by Martins et al. 29 and Ding et al. 33
a-constant
Figure 3 shows the viscosities of the four ILs + B15C5 mixtures versus the molar fraction of the solute B15C5 at 298.15 K. As can be seen from the plots, there is a striking increase on increasing the molar fraction of B15C5. The organic solutes seem to exert a remarkably similar influence on the viscosity of the ILs. The trends in the viscosity of the mixtures can all be described using a facile exponential equation. However, the addition of the solute B15C5 to the ILs causes a dramatic increase in the viscosity of the mixture, in contrast to the addition of generally conventional organic solutes that decrease the viscosity of the mixture substantially, such as acetonitrile, dichloromethane, 2-butanone, N,N-dimethylformamide, with an increasing molar fraction of organic solutes decreasing the viscosity of the [BMIm][BF4] mixtures. 34 According to Wang et al., 34 Seddon et al. 35 when the addition of the solute reduces the viscosity of the mixture, it can be described by equation (4)


Viscosity as a function of the molar fraction xB15C5 for four ILs + B15C5: [C2MIm][NTf2] + B15C5, [C3MIm][NTf2] + B15C5, [C4MIm][NTf2] + B15C5, and [C5MIm][NTf2] + B15C5.
where ηmix and η IL are the viscosities of the mixture and the pure IL (mPa·s), respectively, a is a constant depending on the nature of IL, and xs is the molar fraction of the solute.
In the present work, however, the addition of a solute resulted in an increase in the viscosity of the mixtures, thus fitting the viscosity of the mixtures with equation (5)

where xBC is the molar fraction of B15C5. The linear equation (6) is obtained by taking the natural logarithm of both sides of equation (5)

The a-constant and correlation coefficients (R2) obtained from the fitting are listed in Table S4 (see the Supporting Information). From the results, it can be seen that the R2 values are all higher than 0.99, and it would appear that the equation is more descriptive of the viscosity for mixtures of ILs + B15C5 than for mixtures of conventional organic solvents with ILs, a significant reason for this being due to the volatility of the conventional solutes. The a-constant values increase with a rise in temperature and an increase in the length of the alkyl chain. The viscosity of the reaction mixtures can therefore be predicted from the concentration of the dissolved species, irrespective of their polarity.
The values of the a-constant are in the order: [C2MIm][NTf2] + B15C5 < [C3MIm][NTf2] + B15C5 < [C4MIm][NTf2] + B15C5 < [C5MIm][NTf2] + B15C5. This order illustrates that the longer the molecular chain of the imidazole-based cation, the more it will affect the viscosity of the IL systems. The sequence is, nevertheless, quite a departure from that of the other [CnMIm][NTf2] ILs + organic reagents. We speculate that this is attributable to the fact that both B15C5 and the ILs are large molecules, and that as the molecular size of the IL increases, the magnitude of their intermolecular interactions with B15C5 changes, with progressively stronger dipole interactions and decreased electrostatic interactions. Thus, the larger changes in viscosity are more often attributed to the contribution of ILs with increased cationic groups. In this work, the a-constant values of [C2MIm][NTf2] + B15C5, [C3MIm][NTf2] + B15C5, [C4MIm][NTf2] + B15C5, and [C5MIm][NTf2] + B15C5 are 0.264, 0.289, 0.309, and 0.340, respectively, at 298.15 K. At the same temperature, according to Liu et al., 23 the a-constant values of [C2MIm][NTf2], and [C4MIm][NTf2] with propylene carbonate (PC) binary systems are 0.529 and 0.441, respectively. According to François et al., 36 the a-constant values for the binary mixtures of [C2MIm][NTf2] with acetonitrile (ACN), methanol (MeOH), trifluoroethanol (TFE), and dimethylformamide (DMF) are 0.342, 0.411, 0.413, and 0.367, respectively, and the a-constant values for the binary mixtures of [C4MIm][NTf2] with ACN, MeOH, TFE, and DMF are 0.314, 0.385, 0.363, and 0.342, respectively. The a-constant values of [C2MIm][NTf2] + B15C5 and [C4MIm][NTf2] + B15C5 are smaller than their a-constant values with general organic reagents. This indicates that B15C5 affects the viscosity of the ILs to a greater extent than general organic reagents. Only B15C5 causes the viscosity to increase, while general organic reagents decrease the viscosity.
Density and derivative properties
Density
The density values for different molar fractions of ILs + B15C5 at different temperatures are listed in Table 2. Figure 4(a) shows the density versus temperature plots for the four pure ILs at atmospheric pressure. The densities all decrease as the temperature increases. Also at the same temperature, it decreases as the alkyl chain increases in length. Similarly, the cation size impacts the density: the larger the cation, the lower the density. 37 As the temperature increases from 298.15 to 343.15 K, all four pure ILs decrease in density by only 3%, demonstrating that the temperature is not the main factor affecting the IL density. According to Chernikova et al., 37 the density of ILs is strongly dominated by the nature of their anions. Figure 4(b) presents the density values of the binary mixtures at 298.15 K as a function of the molar fraction of the ILs. The densities of the binary mixtures decrease as the concentration of the crown ether increases and reduce linearly as the molar fraction of B15C5 increases. Mixtures of the ionic liquids [C2MIm][C2SO4] or [C2MIm][CH3CO2] with the inorganic salt [NH4][SCN] have been reported in Pereiro et al. 38 The density measurements of [NH4][C2MIm][SCN][C2SO4] also decreased in an approximately linear fashion with increasing [NH4][SCN] concentration until the solubility limit peaked. Typically, the dependence between density and temperature can be correlated either linearly (equation (7)) or by a second-order polynomial (equation (8)) 39


Experimental values of density, ρ (g·cm−3), for the four ILs + B15C5 as a function of the mole fraction from 298.15 to 343.15 K at atmospheric pressure (P = 0.1 MPa).

B15C5: benzo-15-crown-5; ILs: ionic liquids.
Standard uncertainties: u(T) = 0.02 K, u(p) = 5 kPa, u(x) = 0.0002, u(ρ) = 0.00020 g·cm−3.
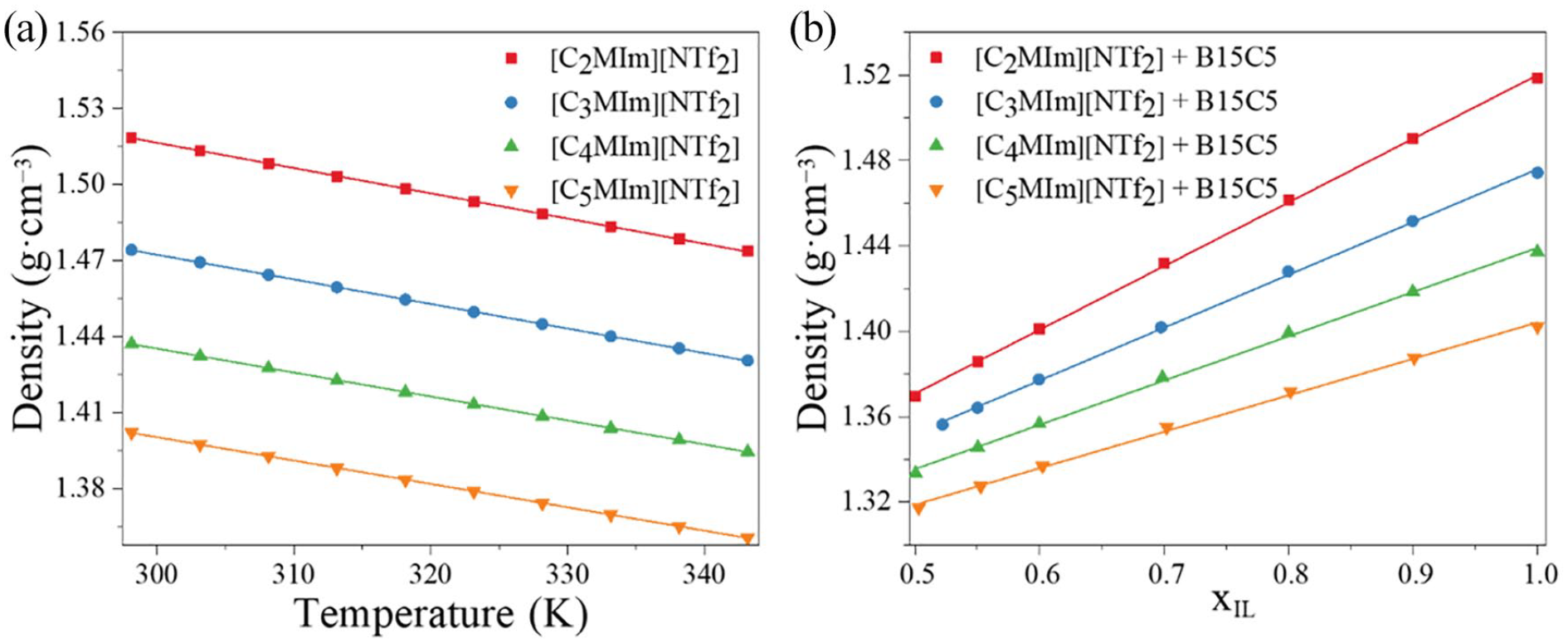
(a) Density as a function of temperature for the different ILs with [C2MIm][NTf2], [C3MIm][NTf2], [C4MIm][NTf2], and [C5MIm][NTf2]. (b) Density as a function of the molar fraction of the IL (xIL) for four ILs + B15C5: [C2MIm][NTf2] + B15C5, [C3MIm][NTf2] + B15C5, [C4MIm][NTf2] + B15C5, and [C5MIm][NTf2] + B15C5.
where a (g·cm−3), b and b1 (g·cm−3·K−1), and b2 (g·cm−3·K−2) are fitted parameters, and T is the absolute temperature in Kelvin. The values of a, b, b1, and b2, and the correlation coefficients R2 are summarized in Tables S5 and S6 (see the Supporting Information). As can be seen from the results, both equations are a better fit for the density data, but equation (8) is much more adaptable.
The isobaric thermal expansion coefficient
The isobaric thermal expansion coefficient (αP) is an essential parameter for characterizing the properties of a material, which takes into account the change in volume with temperature. It can be calculated according to equation (9)25,29,40

where ρ is the density in kg·m−3 and T is the absolute temperature in Kelvin.
The isobaric thermal expansion coefficients for the ILs + B15C5 are presented in Table S7 (see the Supporting Information), which increase with increasing temperature. In comparison, the addition of B15C5 causes a slight decrease in the thermal expansion coefficient, but there is no specific trend in effect. Also, the length of the carbon chain of the alkyl group does not have a significant impact on the thermal expansion coefficient. Despite the increased alkyl chain from [C1MIm][NTf2] to [C16MIm][NTf2], αP only increased from 6.6 × 10−4 K−1 to 7.0 × 10−4 K−1, with a very nearly similar thermal expansion coefficient. 25
Rocha et al. 25 report values for the thermal expansion coefficients of [CnMIm][NTf2] (n = 2, 3, 4, 5) at 323.15 K, and atmospheric pressure, which were compared with the present work and exhibited relative deviations of −0.31%, −0.22%, 0.27%, and 0.25%, respectively. Liu et al. 28 also report the thermal expansion coefficients for the pure ILs [C2MIm][NTf2] and [C4MIm][NTf2], with relative deviations of −0.31% and −0.48%, respectively, from the present work.
Conclusion
The viscosities and densities of the ILs [CnMIm][NTf2] (n = 2, 3, 4, 5) with B15C5 were determined at atmospheric pressure over a temperature range of 298.15 to 343.15 K. The values of the viscosity and density were fitted using empirical equations, and we found that the VFT equation and the second-order polynomial were more appropriate to describe the relationship between the viscosity and temperature, and the density and temperature, respectively. The energy barrier, a-constant values, and the isobaric thermal expansion coefficient have been calculated and discussed. The results indicate that the introduction of B15C5 has a greater effect on the thermodynamic properties of the ILs. Also, according to the a-constant values, the effect of B15C5 on ILs is manifested to a greater extent than the effect of general organic solutes on ILs.
Experimental section
Materials
All chemicals utilized in the present work along with their sources are listed in Table S8 (see the Supporting Information). [C2MIm][NTf2], [C3MIm][NTf2], and [C5MIm][NTf2] were purchased from the Lanzhou Institute of Chemical Physics, China. [C4MIm][NTf2] was purchased from Zhejiang Lande Energy Technology Co., Ltd, China. B15C5 was purchased from Qingdao Bakwal Technology Co., Ltd, China. All chemicals were 99% mass fraction purity. The water content of the ILs was determined using a Mettler Toledo V20 Volumetric KF Titrator, and the values were less than 0.1% for multiple measurements.
Preparation of binary mixtures
The samples were prepared by weighing on a Mettler Toledo XS204 microanalytical balance with an accuracy of 0.0001 g. Adequate amounts of the crown ether and the ionic liquid were weighed and taken separately in a dried and otherwise completely clean vial, to avoid moisture absorption. The standard uncertainty of the molar fraction, considering impurities, was estimated to be 0.0002. Since the dissolution of the crown ether in the ILs was extremely sluggish, the mixture was gently heated. After the crown ether had completely dissolved in the IL, the binary mixture was again mixed thoroughly. The solution was then cooled to room temperature and left to stand in a desiccator for 3 h to ascertain that no solids had precipitated out of solution before the viscosity and density were measured.
Viscosity and density measurements
The viscosities and densities of all the samples were measured at atmospheric pressure (P = 0.1 MPa) using a rotating-cylinder viscometer (Anton Paar, Stabinger SVM 3001) that was equipped with a built-in densimeter in the temperature range from 298.15 to 343.15 K. The measurable temperature range of the apparatus is from −60°C to 135°C, and its temperature control reproducibility is less than 0.005°C. The uncertainty of the viscosity and density are within 0.10% and 0.00020 g·cm−3, respectively, according to the manufacturer. Although 1-alkyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)imides are hydrophobic ionic liquids, significant effects of moisture absorption on the samples are avoided as much as possible during the experiments.17,39,41 The mixtures were stored in airtight glass-stoppered bottles to minimize moisture absorption. The samples were transferred to a gastight syringe under a dry nitrogen atmosphere and injected into the instruments without contact with moisture.
Supplemental Material
sj-docx-1-chl-10.1177_17475198231156358 – Supplemental material for A study on the viscosity, density, and derivative properties of 1-alkyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)imides with benzo-15-crown-5 binary mixtures
Supplemental material, sj-docx-1-chl-10.1177_17475198231156358 for A study on the viscosity, density, and derivative properties of 1-alkyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)imides with benzo-15-crown-5 binary mixtures by Ju Tian, Qi Tang, Yongshen Zhang, Yuzhen Shu, Lihua Zhang and Weiming Zheng in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the President’s Foundation of the China Institute of Atomic Energy (No. YZ222305000103).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
