Abstract
A polymerizable difunctional photoinitiator 2-methylene-succinic acid bis-{2-[4-(2-hydroxy-2-methylpropionyl)phenoxy]ethyl} ester (IAHHMP) based on the commercial photoinitiator 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methylpropanone (HHMP) and a biorenewable itaconic acid is synthesized by esterification. The structure is confirmed by ultraviolet spectroscopy, Fourier transform infrared spectroscopy, nuclear magnetic resonance spectroscopy (1H NMR, 13C NMR) and thermogravimetric analysis. The photopolymerization behaviour of the photoinitiator is investigated using photo-differential scanning calorimetry and compared with that of two commercial photoinitiators, HHMP (or photoinitiator 2959) and 1-hydroxycyclohexyl phenyl ketone (or photoinitiator 184). The results show that IAHHMP has a strong UV absorption capacity at 245~300 nm and can initiate polymerization of monomers containing a double bond. The relative migration of IAHHMP is less than that of the systems containing an HHMP or 1-hydroxycyclohexyl phenyl ketone photoinitiator. Therefore, IAHHMP is expected to have potential applications in more environmentally friendly materials, such as in food and medical packaging.
By esterification, a polymerizable difunctional photoinitiator 2-methylene-succinic acid bis-{2-[4-(2-hydroxy-2-methyl-propionyl)- phenoxy]-ethyl} ester (IAHHMP) based on the commercial photoinitiator 2-hydroxy-1-[4-(2-hydroxyethoxy)phenyl]-2-methylpropanone (HHMP) and a biorenewable itaconic acid (IA) is synthesized. The relative migration of IAHHMP is less than that of the systems containing a commercial HHMP or 1-hydroxycyclohexyl phenyl ketone (HCPK) photoinitiator.

UV curing technology has attracted increasing interest for its rapid curing, low energy consumption, environmentally friendly nature and ability to be used in coatings,1,2 inks, 3 adhesives, 4 microelectronic devices,5,6 biomedical applications, 7 3D printing, 8 and so on. A photoinitiator is an important part of a UV-curing system. Common low-molecular-weight photoinitiators include α-hydroxyketones, benzophenone derivatives, and thioxanthones, such as 1-hydroxycyclohexyl phenyl ketone (HCPK), which is one of the most widely used photoinitiators, benzophenone, and 2-isopropylthioxanthone. 9 However, these photoinitiators often have disadvantages in terms of migration, toxicity, and odour.3,10–11 This has limited their applications in food and drug packaging.12,13 Hence, the development of new photoinitiators continues to be of interest to research workers. 14
In our previous study, we synthesized tri- and tetra-functionalized photoinitiators with larger moleculer weights that improve their initiating activities and decrease their migration after UV irradiation.15,16
For UV-curable systems, many bio-based materials has been used as subsititutes for mineral resources,17–20 Itaconic acid, as a renewable chemical, is obtained from natural source such as carbohydrates, molasses and hydrolysed starch, or through a fermentation process.21,22 Because itaconic acid contains a polymerizable vinylidene group, 23 it is expected that itaconic-acid-modified photoinitiators will possess the properties of polymerizability and low migration.
In this paper, a direct esterification method is used to react itaconic acid with an α-hydroxyketone photoinitiator to prepare a bifunctional IAHHMP polymerizable photoinitiator. The UV absorption capacity, high temperature resistance, migration ability and photopolymerization ability of IAHHMP, HHMP, HCPK and other α-hydroxyketone photoinitiators are discussed and compared. The structure of and synthetic route towards IAHHMP are shown in Scheme 1.

Synthetic route to IAHHMP.
Results and discussion
UV spectral analysis
The UV absorption peaks of the HCPK, HHMP and IAHHMP photoinitiators in the range of 200–400 nm were measured at 25 °C using acetonitrile as a reference solution. The structures and ultraviolet absorption spectra of these three compounds are shown in Figure 1.

Structures and UV-Vis spectra of HCPK, HHMP and IAHHMP.
Figure 1 shows that the maximum absorption peak of the HCPK photoinitiator is at 246 nm, the maximum absorption peak of HHMP is at 274 nm, and the maximum absorption peak of IAHHMP is at 273 nm. Table 1 shows the maximum absorption wavelengths of HCPK, HHMP and IAHHMP. The corresponding maximum molar extinction coefficients are also shown. The maximum ultraviolet absorption wavelengths of HHMP and IAHHMP are not very different. Compared with the HCPK photoinitiator, IAHHMP has a longer maximum ultraviolet absorption wavelength, which may be because the molecular structure of the IAHHMP photoinitiator contains more carbonyl (C=O) and other chromophoric groups.
The maximum absorption wavelengths and maximum molar extinction coefficients of HCPK, HHMP and IAHHMP.

Solvent: acetonitrile.
FT-IR spectral analysis
It can be seen from Figure 2 in the infrared spectrum of IAHHMP is a symmetric stretching vibration absorption peak of OH at 3440 cm-1 with a wide absorption band due to the association of molecules. Peaks at 962 cm-1 and 839 cm-1 are characteristic absorption peaks caused by the contraction vibration of C=C double bonds. Peak 2937 cm-1 is the asymmetric contraction vibration absorption peak of -CH3. Peak 1417.8 cm-1 is the absorption peak caused by the contraction vibration of benzene ring. The C=O characteristic absorption peak of saturated ester group will be generated near 1723.7 cm-1; the characteristic absorption peak of the carboxylic acid has completely disappeared. The peak at 1579 cm-1 is the symmetric stretching vibration absorption peak of the C=O. These results are consistent with expectations.

FT-IR spectra of HHMP and IAHHMP.
Thermogravimetric analysis
A TG-209 thermogravimetric analyser was used to study the thermal stability of the HCPK, HHMP and IAHHMP photoinitiators. The temperature range of the instrument is 70 °C to 500 °C, and the heating rate is 10 °C min; the instrument collects data every 0.5 s for 20 min, and the atmosphere is nitrogen.
It can be seen from Figure 3 that the HCPK photoinitiator experienced a weight loss of 5% at 71 °C, and the final weight loss temperature was 178 °C, HHMP experienced a weight loss of 5% at 187 °C and finally stopped losing weight at 410 °C. IAHHMP experienced a weight loss of 5% at 202 °C and finally stopped losing weight at 300 °C. Degradation of the product can be divided into three stages. In the first stage at T < 178 °C, the moisture contained in the product is lost at approximately 100 °C, and the HCPK photoinitiator is degraded. The second stage is at 178 °C< T < 290 °C and mainly consists of degradation of HHMP by volatilization. The third stage is at 290 °C < T < 410 °C and mainly consists of degradation of IAHHMP by volatilization. It can be concluded that the thermal stability of the IAHHMP photoinitiator is significantly higher than that of the HCPK photoinitiator, which greatly reduces its volatility.

TG spectra of HCPK, HHMP and IAHHMP.
Photopolymerization kinetics
At 25 °C under a N2 atmosphere, an empty aluminum crucible was used as a reference sample, with a light intensity of 20 mW/cm2 and a light duration of 40 s. A total of 0.006 mol of HCPK photoinitiator, 0.006 mol of HHMP photoinitiator and 0.006 mol of IAHHMP photoinitiator were used to initiate the polymerization of 2 g of TMPTA monomer. The relationship between the reaction rate of TMPTA monomer polymerization and the photopolymerization conversion rates of the HCPK, HHMP and IAHHMP photoinitiators were studied. The results are shown in Figures 4 and 5.

Rate curves of the HCPK, HHMP and IAHHMP initiated polymerization reactions of TMPTA.

Conversion via the HCPK, HHMP and IAHHMP initiated polymerization of TMPTA.
It can be seen from Figures 4 and 5 that the maximum reaction rate of the TMPTA polymerization initiated by the HCPK photoinitiator was 0.032 s-1, and the final double bond conversion rate was 34.8%. The maximum reaction rate when HHMP initiated the TMPTA polymerization was 0.0396 s-1, and the final double bond conversion rate was 42.8%. The maximum reaction rate of IAHHMP-initiated TMPTA polymerization was 0.0388 s-1, and the final double bond conversion rate was 41.7%. By comparison, the photoinitiation activity of HHMP is slightly higher than that of IAHHMP, which is mainly because IAHHMP contains two ester groups in its molecular structure, resulting in a decrease in the photoinitiation activity. The photoinitiation activity of IAHHMP is higher than that of HCPK, mainly because IAHHMP has a higher active radical concentration than HCPK after photolysis, which makes the IAHHMP-initiated TMPTA polymerization rate higher.
Properties of the UV-cured films
The preparation of the coating is described the experimental section, and its properties are shown in Table 2. As the amount of IAHHMP increases, the photocuring rate of the coating also gradually increases. Considering the light-curing efficiency and economic factors, when the amount of IAHHMP is 5%, the photoinitiation effect is better. It can be seen from Table 3 that the photocuring rate of IAHHMP is greater than that of the commercially available HCPK photoinitiator, and the film has high hardness after coating, better adhesion, and excellent acid and alkali resistance.
The curing time of the IAHHMP coating with different contents.

Film properties.

Extraction analysis experiment
The migration of photolysis fragments of the photoinitiator in the cured films can be analysed by ultraviolet absorption of the photoinitiator extracted from the polymer, that is, the amount of residual photoinitiator in the solution can be determined by the relationship between the absorbance and the concentration at different wavelengths. 24 The UV absorption spectra of the HCPK, HHMP and IAHHMP photoinitiator extracts are shown in Figure 6. Table 4 lists the mobilities R of IAHHMP relative to HCPK and HHMP.

UV-Vis absorption spectra of the extraction solutions of HCPK, HHMP and IAHHMP.
The migration of IAHHMP and HHMP relative to HCPK.

It can be seen from Figure 6 that the absorption peaks of the HCPK, HHMP and IAHHMP photoinitiators at the maximum wavelength have not disappeared, indicating that the photoinitiator remains in the cured film. At the same time, it can be seen from Table 4 that under the same experimental conditions, the migration of the IAHHMP system is only 25.96% that of the HHMP system, and the migration of the IAHHMP system is only 11.90% that of the HCPK system. This demonstrates that the IAHHMP polymerizable photoinitiator containing a double bond is effective in immobilizing the coating in the cured film, thereby greatly reducing the biotoxicity of the cured film.
Conclusion
A polymerizable photoinitiator, IAHHMP, has successfully been synthesized by direct esterification, and its structure has been studied by UV-visible, FTIR and 1H NMR spectroxomy and by TG. The UV-visible spectrum and photo-DSC spectrum showed that IAHHMP has strong UV absorption ability and can initiate photopolymerization. Thermogravimetric analysis showed that the thermo stability of IAHHMP is better than that of HHMP and HCPK. This is because itaconic acid is introduced. In addition, the compatibility between the photoinitiator and the monomer or resin is increased The extraction experiments and UV-visible spectrum prove that migration of the IAHHMP system is lower than those of the HHMP or HCPK systems. Excellent high-temperature resistance and low migration make the IAHHMP polymerizable photoinitiator more environmentally friendly. It has the potential to be applied to UV photopolymerization systems of various food or biomedical packaging materials.
Experimental
Instruments and reagents
A Magna380 type Fourier infrared spectrometer (KBr tablet, Nicolet, USA), a T6 ultraviolet spectrometer (acetonitrile solvent, Beijing Pu Analysis General Instrument Co., Ltd.), a Bruker 400 MHz NMR spectrometer (CDCl3 solvent, TMS internal standard, Bruker, Germany), a TG-209 Thermogravimetric Analyzer (Netzsch, Germany), a photo-differential scanning calorimeter (Perkin Elmer, USA), and a UVA-T-400W portable UV-curing light (Dongguan Gu Dejia Machinery Equipment Co., Ltd.) were used.
Itaconic acid and NaCl (Aladdin Reagent Co. Ltd.), AR; methylene chloride, methanesulfonic acid, N, N-dimethylformamide (DMF), hydroquinone, petroleum ether, and ethyl acetate (Xiqi Chemical Co. Ltd.), AR; acetonitrile (National Pharmaceutical Group Chemical Reagent Co. Ltd.), AR; and 2-hydroxy-2-methyl-p-hydroxyethyl ether phenylacetone (HHMP, or photoinitiator 2959), 1-hydroxycyclohexyl phenyl ketone (HCPK, or photoinitiator 184), and trimethypropane triacrylate (TMPTA monomer) (Guangzhou Sunnysale New Material Technology Co. Ltd.), industrial grade, were used. All reagents were used without purification.
Experimental methods
A 250 mL three-necked flask connected to a condenser, a water separator, and a thermometer were used after drying. Next, 0.5 g (0.0038 mol) of itaconic acid, 0.05 g of hydroquinone, and 60 mL of DMF were added to the flask under a N2 atmosphere. After the itaconic acid had completely dissolved, 1.75 g (0.0078 mol) of HHMP was added, and after stirring to achieve a uniform mixture, 0.05 g of the catalyst methanesulfonic acid were added. The temperature was increased to 105°C and heating was continued for 6 h. After the reaction was complete, the crude product was dissolved in 40 mL of dichloromethane and washed with brine (60 mL) and deionized water(3*60 mL). The combined organic phase was dried, concentrated and further purified by column chromatography. The eluent was a mixed solvent of petroleum ether and ethyl acetate (2:1v:v). The product (1.41 g, (68%). 1H NMR (CDCl3, 400 MHz), δ: 8.05 (m, 4H, CH, benzene ring), 6.94 (m, 4H, CH, benzene ring), 6.38 (s, 1H, =CH), 5.80 (s, 1H, =CH), 4.60–4.46 (m, 2H, CH2), 4.30–4.24 (m, 2H, CH2), 4.21–4.12 (m, 2H, CH2), 4.03–3.94 (m, 2H, CH2), 2.86 (s, 2H, CH2), 1.61 (s, 12H, CH3), 13C NMR (CDCl3, 101 MHz), δ: 200.2, 160.55, 132.30, 126.32, 114.03, 77.22, 76.90, 76.58, 69.31, 65.62, 61.69, 61.07, 36.31, 28.55, IR (KBr),: 3080, 1723, 1643, 1417, 1308, 958, 670cm-1. ESI-MS: m/z, calcd for C29H34O10 542.57, found 542.0.
Preparation of the coating
In the dark, three small beakers were used. To the first beaker, 60% B-605 polyurethane resin, 36% TMPTA monomer, and 4% IAHHMP photoinitiator were added. In the second beaker, 60% B-605 polyurethane resin, 36% TMPTA monomer, and 4% HHMP photoinitiator were added. In the third beaker, 60% B-605 polyurethane resin, 36% TMPTA monomer, and 4% HCPK photoinitiator were added. The mixtures were stirred well, allowed to stand for 10 min and then applied as a film onto a glass slide. The thickness of the film was approximately 50 μm. In air, the film was exposed to a high-pressure mercury lamp at a distance of 5.5 cm and a power of 400 W. Curing was performed at a dominant wavelength of 365 nm.
Coating performance test
The degree of curing of the photoinitiated polymerized monomer coatings of IAHHMP was investigated by gel conversion. The coating, which solidified after exposure to light, was immersed in acetone for 30 min, and the cured resin and the monomer dissolved in the acetone solution. The quality of the coating after drying was compared with the quality before exposure of the coating to light, and the coating properties were calculated. The conversion rate of the layer is the degree of curing of the coating. The surface drying time of the coating was tested by the finger touch method. A pencil hardness test was conducted according to the ASTM D3363-05 standard. The adhesion was referred to ASTMD3002 (0B-5B)
IAHHMP migration
A TMPTA solution of 1% photoinitiator (IAHHMP, HHMP, HCPK) was prepared. 24 Each solution was coated on a glass slide with a thickness of approximately 80 μm. The films were exposed to air in a portable curing machine with a lamp width of 5.5 cm and a power of 400 W for 3 min. The cured film was then cut, with each piece weighing 0.075 g, and extracted with 20 mL of acetonitrile at room temperature for 3 d. Finally, equal amounts of extract was examined by a UV absorption test. The relative mobility R of IAHHMP relative to the HHMP photoinitiator and the HCPK photoinitiator can be obtained from formulae (1), (2) and (3):


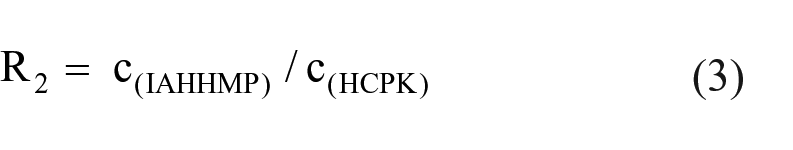
In the formulae, c is the concentration of the photoinitiator in the extract, mol/L; A is the absorbance; ε is the molar extinction coefficient, L / (mol • cm); b is the sample cell thickness, cm; c (IAHHMP) is the concentration of IAHHMP in the extract; c(HHMP) is the concentration of the HHMP photoinitiator in the extract; c(HCPK) is the concentration of the HCPK photoinitiator in the extract; and R is the relative mobility of IAHHMP.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors appreciate the financial support provided by the National Natural Scientific Foundation of China (21364008), the Natural Science and Technology Foundation of Jiangxi Education Department (DA201702347), and the Graduate Innovation Funds of Nanchang Hangkong University (YC2017011)
