Abstract
Oxidative coupling of 2-methylquinolines with quinoxalines via a TBHP (t-butyl hydroperoxide) and TFA (trifluoroacetic acid) mediated Minisci reaction affords quinolin-2-yl(quinoxalin-2-yl)methanone derivatives. This approach provides a simple and efficient method to construct various acyl derivatives in moderate to good yields (45%–92%) without any metal catalyst. Mechanistically, homolytic aromatic substitution (HAS) of acyl radicals mediated by TBHP is crucial for the construction of the quinolin-2-yl(quinoxalin-2-yl)methanone products.
Keywords

Introduction
Acyl N-heterocycles, for example, the quinolin-2-yl(quinoxalin-2-yl)methanones reported in this article, play key roles in pharmaceuticals1–3 and possess different biological activities such as antimicrobial, 4 antipsychotic, 5 antithrombotic, 6 antitussive, 7 and antihpyerglycemic. 8 There have been numerous reports on the preparation of electron-deficient heterocycles, but examples on their functionalization by oxidative coupling are rare. Compared to electron-rich heterocycles,9–13 the acylation of electron-deficient heterocycles is more difficult. It is worth mentioning that the Minisci reaction and aromatic homolytic substitution can overcome efficiently difficulties via nucleophilic acyl radical addition to protonated electron-deficient heterocycles.14,15 Recently, several researchers reported the preparation of acyl N-heterocycles using the Minisci reaction. Yuan et al. 16 reported that 3-acylated quinoxalin-2(1H)-ones could be obtained by oxidative coupling of quinoxalin-2(1H)-ones with diverse aldehydes. Meanwhile, Siddaraju et al. 17 reported that acylations of isoquinoline, quinoline, and quinoxaline derivatives could be accomplished by employing cross dehydrogenative coupling reactions with aldehydes using a substoichiometric amount of TBAB (tetrabutylammonium bromide) and K2S2O8 as the oxidant. Zhang et al. 18 showed that isoquinolin-1-yl(p-tolyl)methanones could be prepared by the reaction of isoquinoline with 4-methylbenzaldehyde catalyzed by TBHP under irradiation with blue LEDs. Furthermore, Rahim et al. 19 reported that I2/DMSO could promote the reactions of 2-methylquinolines with benzothiazoles to form 2-heteroaryl benzothiazoles under acid- and peroxide-free conditions. Also, Chen et al. 20 showed that TBHP/TFA could promote the reaction of isoquinolines with benzaldehydes to give the corresponding acyl derivatives. All these methods used aldehydes as acyl radical donors. However, Ali et al. 21 reported that acyl radicals derived from methylbenzenes could be utilized for the direct acylation of N-heterocycles promoted by AlCl3 at 95 oC under N2. Inspired by these examples, we envisaged that 2-methylquinolines could be used as acyl radical donors under oxidative conditions for the acylation of quinoxalines via intermolecular homolytic aromatic substitution of acyl radicals, and our studies proved this to be the case. Therefore, in this paper, we disclose the first oxidative coupling of 2-methylquinolines with quinoxalines via TBHP (t-butyl hydroperoxide) and TFA (trifluoroacetic acid) mediated Minisci reactions to prepare quinolin-2-yl(quinoxalin-2-yl)methanones without any metal catalyst. Homolytic aromatic substitution of acyl radicals mediated by TBHP is crucial in the mechanism of this process for the construction of acyl derivatives.
Results and discussion
Quinoxaline (
Optimization of the reaction conditions for the synthesis of

Reaction conditions:
TBHP in decane (5–6 M).
Isolated yield.
TBHP (3.0 equiv).
TBHP (4.0 equiv).
TBHP (5.0 equiv).
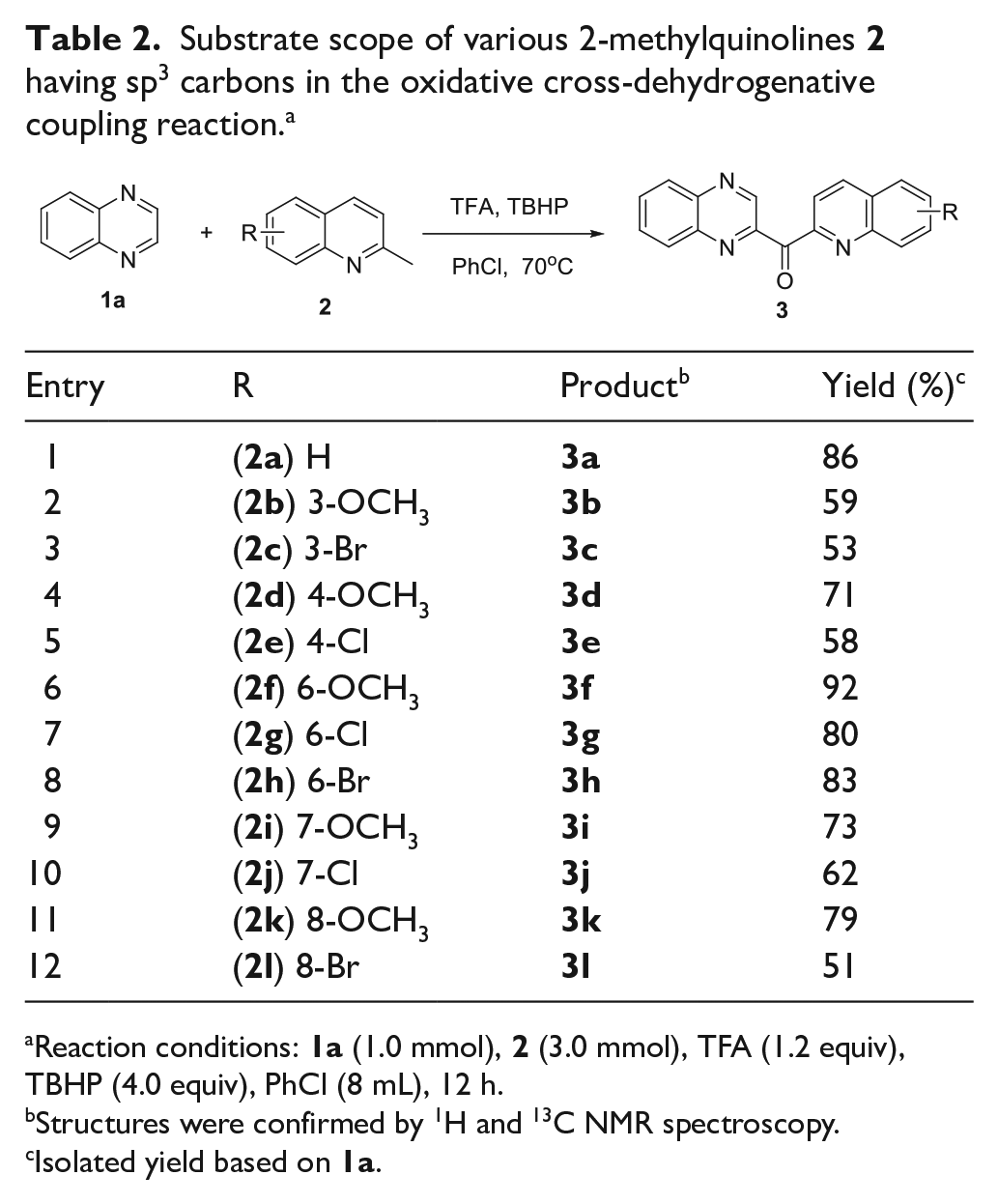
With optimized conditions in hand, the substrate scope was next investigated. 2-Methylquinolines
Reaction conditions:
Structures were confirmed by 1H and 13C NMR spectroscopy.
Isolated yield based on
Subsequently, we set out to examine the substrate scope of quinoxalines
Substrate scope of various quinoxalines

Reaction conditions:
Structures were confirmed by 1H and 13C NMR spectroscopy.
Isolated yield based on
Ratio was determined by 1H NMR (600 MHz, CDCl3) analysis.
Finally, 2-methylpyridine (

2-Methylpyridine (
To further explore the mechanism of the oxidative coupling of 2-methylquinolines with quinoxalines, several control experiments were carried out. First, when adding the radical scavenger TEMPO (1.0 equiv) to the standard reaction, product

Control experiments.
Based on these observations and related references,22,23 a plausible mechanism for the cross-dehydrogenative coupling process is proposed in Scheme 3. TFA can coordinate with quinoxaline leading to the protonated intermediate (
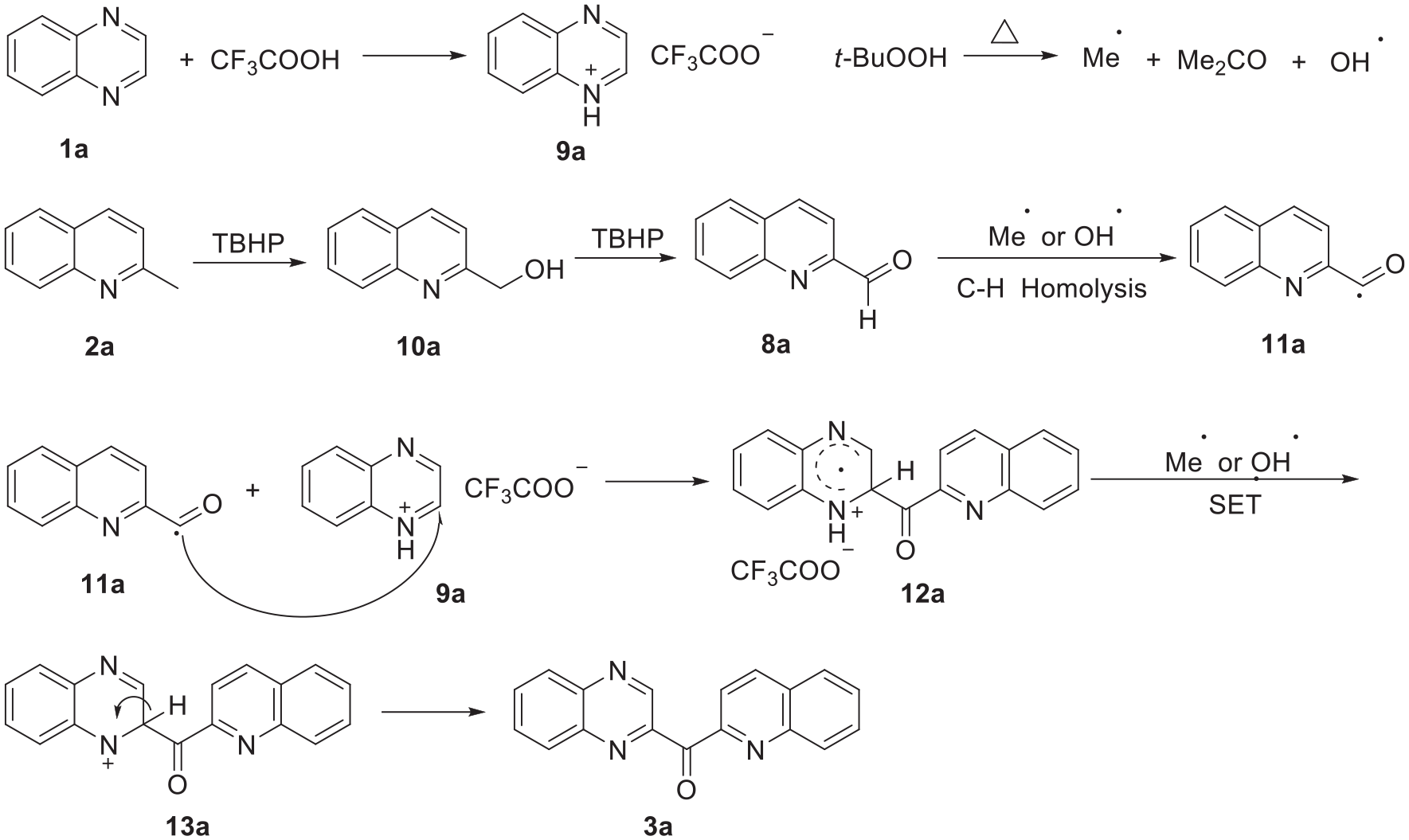
A plausible mechanism for the formation of quinolin-2-yl(quinoxalin-2-yl)methanones.
Conclusion
In conclusion, we have developed a procedure for the oxidative coupling of 2-methylquinolines with quinoxaline via a TBHP/TFA-mediated Minisci reaction for the synthesis of quinolin-2-yl(quinoxalin-2-yl)methanones without any metal catalyst. This works shows that 2-methylquinolines can be used as acyl radical donors under oxidative conditions and be utilized for the acylation of quinoxalines via intermolecular homolytic aromatic substitution of acyl radicals. The reaction temperature is 70 oC, below the alarm temperature 75 oC and autoignition temperature 88 oC of TBHP, which shows that the reaction condition is relatively safe. This approach encompasses relatively mild reaction conditions and affords moderate to good yields.
Experimental
Melting points were measured on a Büchi B-540 capillary melting point apparatus and are uncorrected. Mass spectra (ESI-MS) were recorded on a Thermo Finnigan LCQ-Advantage spectrometer. High-resolution mass spectrometry (ESI-HRMS) was performed using an Agilent 6210 TOF instrument. 1H NMR and 13C NMR spectra were recorded on a Bruker-600 spectrometer (600 MHz), δ in parts per million, J in Hertz, and using TMS as the internal standard. Signal multiplicities are assigned as singlet (s), doublet (d), multiplet (m). All analytical reagents were commercially available and were used directly without further purification.
Synthesis of quinolin-2-yl(quinoxalin-2-yl)methanones (
3a
selected as an example); tipical procedure
A mixture of quinoxaline (
Quinolin-2-yl(quinoxalin-2-yl)methanone (
(3-Methoxyquinolin-2-yl)(quinoxalin-2-yl)methanone (
(3-Bromoquinolin-2-yl)(quinoxalin-2-yl)methanone
(4-Methoxyquinolin-2-yl)(quinoxalin-2-yl)methanone (
(4-Chloroquinolin-2-yl)(quinoxalin-2-yl)methanone (
(6-Methoxyquinolin-2-yl)(quinoxalin-2-yl)methanone (
(6-Chloroquinolin-2-yl)(quinoxalin-2-yl)methanone (
(6-Bromoquinolin-2-yl)(quinoxalin-2-yl)methanone (
(7-Methoxyquinolin-2-yl)(quinoxalin-2-yl)methanone (
(7-Chloroquinolin-2-yl)(quinoxalin-2-yl)methanone (
(8-Methoxyquinolin-2-yl)(quinoxalin-2-yl)methanone (
(8-Bromoquinolin-2-yl)(quinoxalin-2-yl)methanone (
(3-Methylquinoxalin-2-yl)(quinolin-2-yl)methanone (
(3-Chloroquinoxalin-2-yl)(quinolin-2-yl)methanone (
(6-Methylquinoxalin-2-yl)(quinolin-2-yl)methanone (
(6-Chloroquinoxalin-2-yl)(quinolin-2-yl)methanone (
Pyridin-2-yl(quinoxalin-2-yl)methanone (
Diquinolin-2-ylmethanone (
Quinoline-2-carbaldehyde (
Supplemental Material
sj-doc-1-chl-10.1177_17475198221126026 – Supplemental material for Homolytic aromatic substitution of acyl radicals: Oxidative coupling of 2-methylquinolines with quinoxalines
Supplemental material, sj-doc-1-chl-10.1177_17475198221126026 for Homolytic aromatic substitution of acyl radicals: Oxidative coupling of 2-methylquinolines with quinoxalines by Xue Li, Xian Hao Wu, Jia Feng Yu, Yuan Yuan Zhang, Zhong Ke Hou and Yuan Yuan Xie in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to the Natural Science Foundation of Jiangxi Province (20212BAB206073) for financial support.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.