Abstract
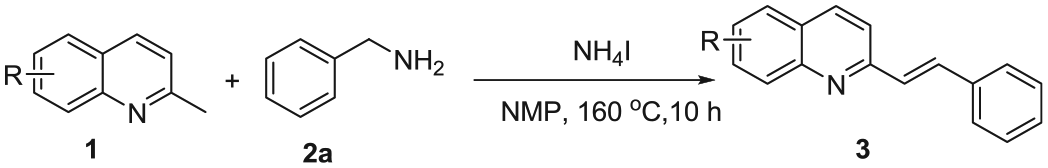
Without any metal catalyst, a simple and efficient method for the synthesis of E-2-styrylquinolines through sp3 C-H cross-dehydrogenative coupling of benzylamines with 2-methylquinolines mediated by NH4I under air is successfully developed. The oxidative olefination proceeded through deamination and sp3 C–H bond activation. A plausible mechanism is proposed for the construction of E-2-styrylquinolines.
Keywords

Introduction
On account of their unique structures and reactivities, quinoline and its derivatives are widely utilized to prepare various biologically important compounds.1,2 Among quinoline derivatives, E-2-styrylquinolines are typical bioactive compounds acting as potent HIV-1 integrase inhibitors,3–5 leukotriene receptor antagonists,6,7 and antiallergic drugs. 8 The wide applications of E-2-styrylquinolines have attracted considerable attention in synthetic chemistry. In the past 10 years, widely used methods for the synthesis of E-2-styrylquinolines have mainly involved the reactions of 2-methylquinolines with benzyl alcohols,9–12 benzyl amines,9,13–15 aldehydes,16–19 and N-benzylidene-4-methylbenzenesulfonamides 20 (Scheme 1). In 2011, Qian et al. 20 disclosed that Fe(OAc)2 promoted the reactions of 2-methylquinolines and N-benzylidene-4-methylbenzenesulfonamides by removing a molecule of p-toluenesulfonamide to give E-2-styrylquinolines. In 2019, Liang et al. 16 reported that various 2-alkenylquinolines could be produced from 2-methylquinolines and aldehydes under the synergistic organocatalysis of 1,3-dimethylbarbituric acid/HOAc for 24 h. In 2020, Susanta et al. 9 reported that E-2-styrylquinolines could be prepared from 2-methylquinolines by reactions with primary alcohols or primary amines using the NaCl/TBHP oxidative system. In the same year, Zhang et al. 10 showed that reactions of 2-methylquinolines and primary alcohols catalyzed by MnO2 in the presence of KOH eliminated molecule of H2O and were transformed into E-2-styrylquinolines. Nevertheless, these methods suffer from environmental and economic concerns as they utilize strong oxidants and transition-metal catalysts, which impede the applicability of these methods. To avoid these drawbacks, we successfully developed a method that involves deamination and sp3 C-H cross-dehydrogenative coupling of 2-methylquinolines and benzylamines to give various E-2-styrylquinolines mediated by NH4I in moderate to good yields (30%–93%) with NMP (N-methyl-2-pyrrolidone) as the solvent, without any other metal catalyst at 160 °C for 10 h (Scheme 1).
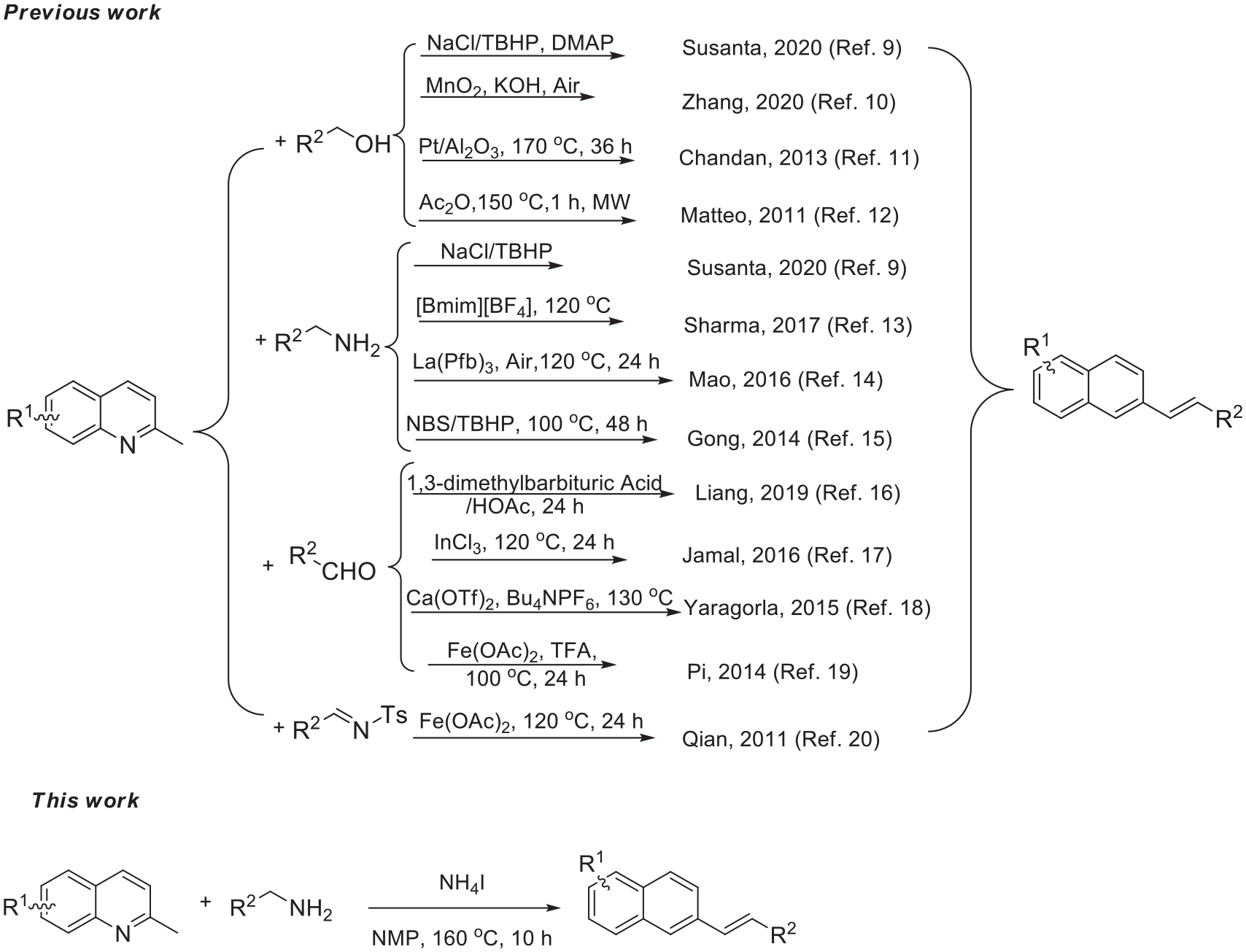
Methods for the preparation of E-2-styrylquinolines.
Results and discussion
The 2-Methylquinoline (
Optimization of the reaction conditions for the synthesis of

NBS: N-bromosuccinimide; NIS: N-iodosuccinimide; TBAI: tetra-n-butylammonium iodide; DMSO: dimethyl sulfoxide.
Reaction conditions:
Isolated yield.
Reaction time: 14 h.
Reaction time: 18 h.
Reaction time: 24 h.
Under N2.
Under O2.
With optimized conditions in hand, we set out to explore the substrate scope of various quinolines having sp3 carbons for oxidative cross-dehydrogenative coupling reactions. The target products
Substrate scope of various quinolines having sp3 carbons in the oxidative cross-dehydrogenative coupling reaction. a
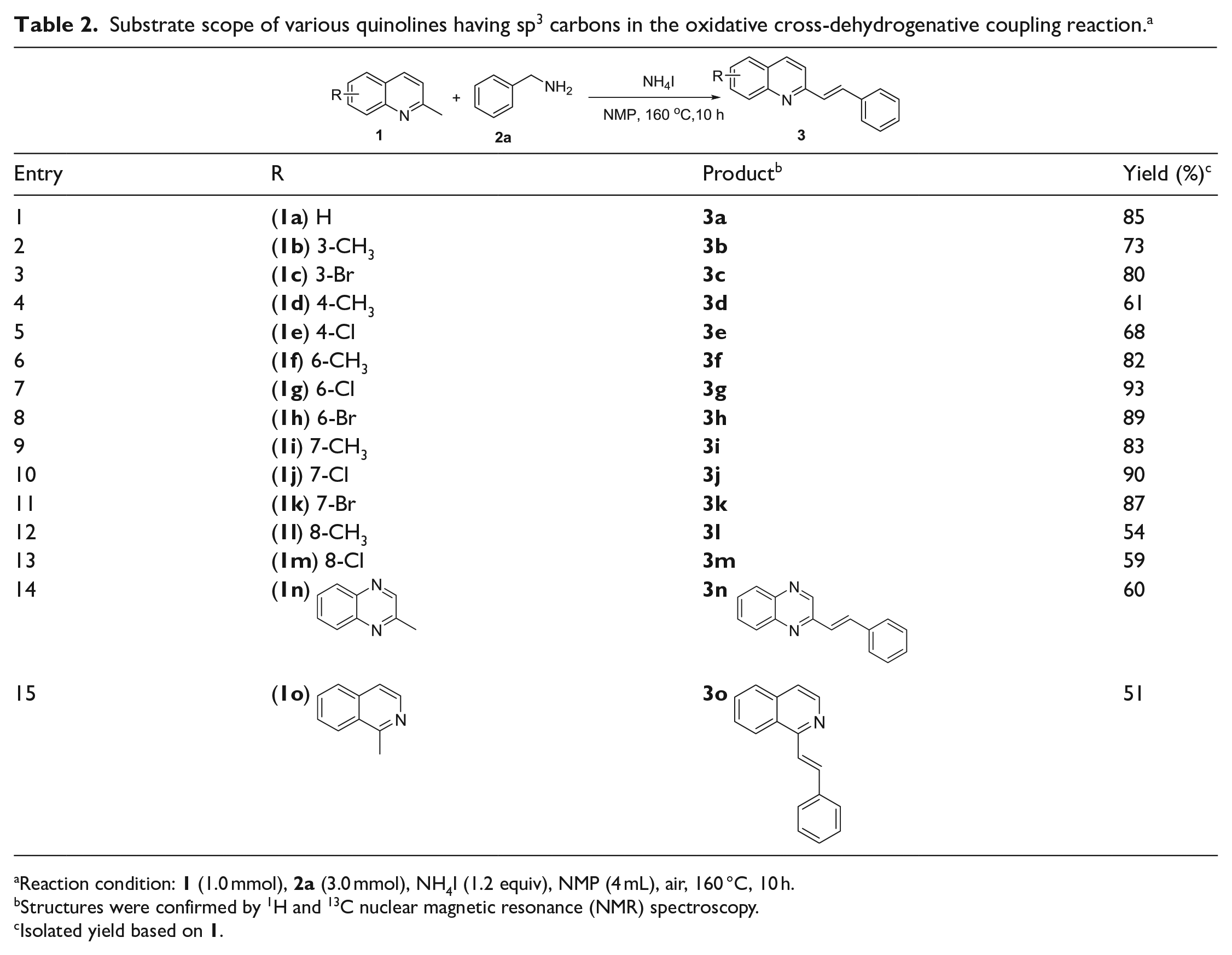
Reaction condition:
Structures were confirmed by 1H and 13C nuclear magnetic resonance (NMR) spectroscopy.
Isolated yield based on
Subsequently, we set out to examine the substrate scope of various methanamines
Substrate scope of various primary amines. a
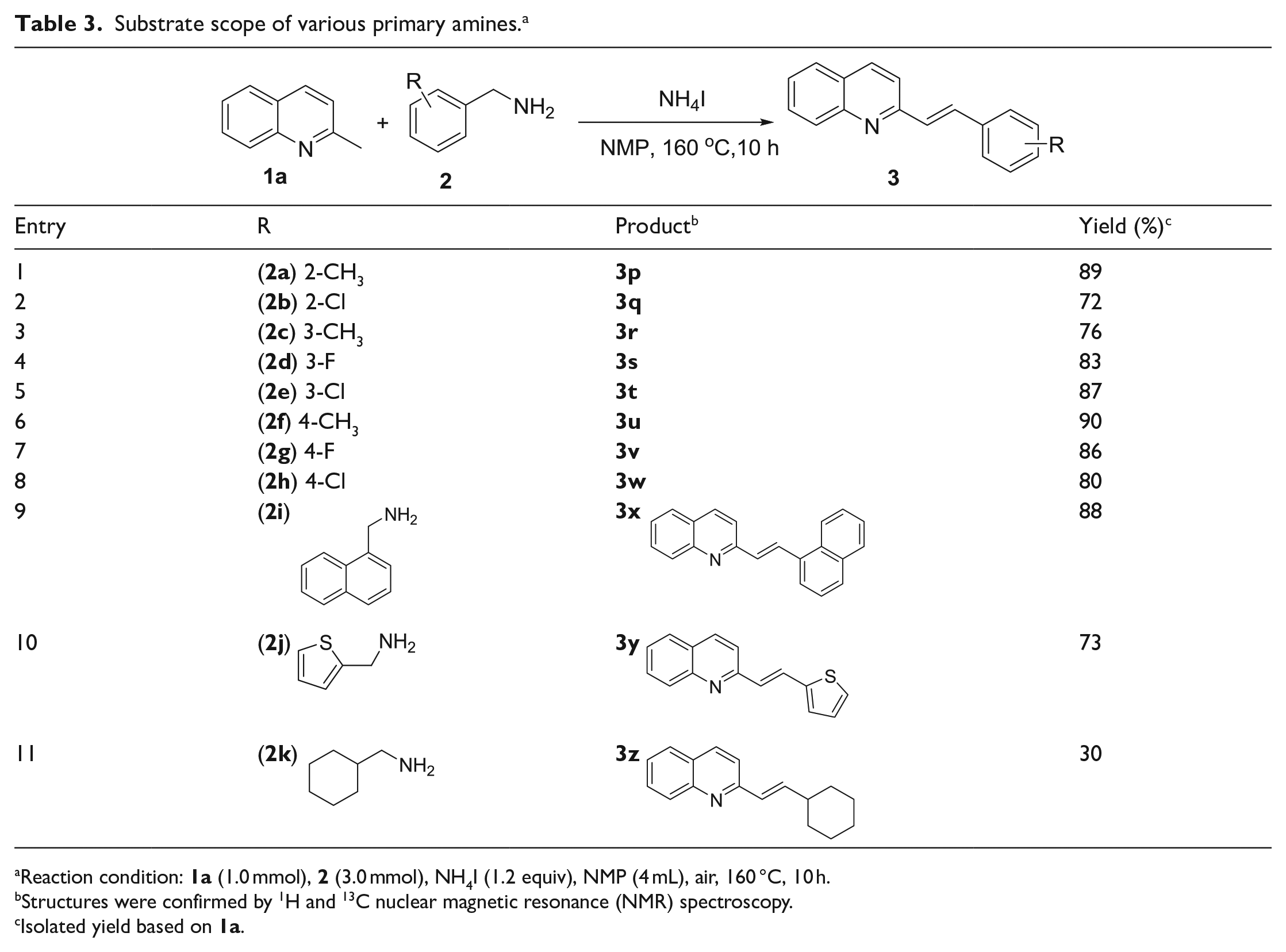
Reaction condition:
Structures were confirmed by 1H and 13C nuclear magnetic resonance (NMR) spectroscopy.
Isolated yield based on
To further explore the mechanism of oxidative olefination for the construction of E-2-styrylquinolines, several control experiments were carried out. First, when adding the radical scavenger TEMPO (1.0 equiv) to the standard reaction,

Control experiments.
Based on these observations and related references,13,15 a plausible mechanism is proposed in Scheme 3. Initially, NH4I is oxidized to a highly active “I+” species in air. Second, the benzylamine is transformed into a phenylmethanimine by elimination under the influence of “I+.” Finally, phenylmethanimine is attacked by the 2-methylquinoline and is transformed into the corresponding olefination product via elimination of a molecule of NH3.

A plausible mechanism.
Conclusion
In summary, we have developed an efficient approach for the synthesis of a variety of E-2-styrylquinolines through sp3 C-H cross-dehydrogenative coupling of benzylamines with 2-methylquinolines promoted by NH4I under air without any metal catalyst. The approach provides relatively mild reaction conditions, moderate to good yields, and encompasses a broad substrate scope. A plausible mechanism has been proposed for the oxidative olefination through deamination.
Experimental
Infrared spectra were determined on a Nicolet Avatar-370 spectrometer in KBr (ν in cm–1). Melting points were measured on a Büchi B-540 capillary melting point apparatus and are uncorrected. Mass spectra (ESI-MS) were recorded on a Thermo Finnigan LCQ-Advantage spectrometer. High-resolution mass spectra (ESI-HRMS) were obtained using an Agilent 6210 TOF instrument. 1H NMR and 13C NMR spectra were recorded on a Varian Mercury Plus-400 spectrometer (400 and 100 MHz), δ in parts per million, J in Hertz, using TMS as the internal standard. Signal multiplicities are assigned as singlet (s), doublet (d), multiplet (m). All analytical reagents were commercially available and were used directly without further purification.
Synthesis of E-2-styrylquinolines (3a selected as an example); general procedure
A mixture of 2-methylquinoline (
2-[(1E)-2-phenylethenyl]quinoline (3a )
Pale yellow solid; 85%, 0.20 g; m.p. 91–92 °C (Lit. 21 91–93 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.14–8.06 (m, 2H), 7.76 (d, J = 8.2 Hz, 1H), 7.74–7.64 (m, 5H), 7.55–7.46 (m, 1H), 7.43–7.38 (m, 3H), 7.34–7.30 (m, 1H). 13C NMR (100 MHz, CDCl3): δ = 156.4, 148.2, 136.6, 136.3, 134.5, 129.7, 129.3, 129.0, 128.9, 128.6, 127.6, 127.5, 127.2, 126.3, 118.9. MS (ESI): m/z (%) = 232.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C17H14N: 232.1126, found: 232.1133.
3-Methyl-2-[(1E)-2-phenylethenyl]quinoline (3b )
Pale yellow solid; 73%, 0.18 g; m.p. 98–100 °C (Lit.21 98–100 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.09 (d, J = 8.4 Hz, 1H), 8.02 (d, J = 15.6 Hz, 1H), 7.88 (s, 1H), 7.72–7.63 (m, 4H), 7.51 (d, J = 15.6 Hz, 1H), 7.47–7.39 (m, 3H), 7.35–7.32 (m, 1H), 2.32 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 154.9, 146.9, 137.1, 136.2, 135.6, 129.4, 129.1, 128.7, 128.6, 128.5, 127.8, 127.5, 126.7, 125.8, 124.3, 19.6. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1290.
3-Bromo-2-[(1E)-2-phenylethenyl]quinoline (3c )
Pale yellow solid; 80%, 0.25 g; m.p. 103–105 °C (Lit.21 103–105 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.37 (s, 1H), 8.08–8.03 (m, 2H), 7.80 (d, J = 15.6 Hz, 1H), 7.73–7.67 (m, 4H), 7.50 (t, J = 7.3 Hz, 1H), 7.45–7.40 (m, 2H), 7.37–7.32 (m, 1H). 13C NMR (100 MHz, CDCl3): δ = 152.9, 147.0, 139.1, 136.9, 136.6, 130.1, 129.2, 128.9, 128.6, 128.4, 127.7, 126.8, 126.5, 124.8, 118.3. MS (ESI): m/z (%) = 310.0 ([M]+, 51), 312.0([M]+, 49). HRMS (ESI): m/z [M]+ calcd for C17H1379BrN: 310.0231, found: 310.0239; C17H1381BrN: 312.0211, found: 312.0218.
4-Methyl-2-[(1E)-2-phenylethenyl]quinoline
15
(3d )
Pale yellow solid; 61%, 0.15 g; m.p. 122–124 °C (Lit. 15 122–123 °C). 1H NMR (400 MHz, CDCl3): δ = 8.04 (d, J = 8.0 Hz, 1H), 7.75 (d, J = 8.0 Hz, 1H), 7.56–7.47 (m, 4H), 7.37–7.32 (m, 2H), 7.28–7.24 (m, 3H), 7.22–7.17 (m, 1H), 2.61(s, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.0, 148.2, 144.3, 136.8, 134.2, 129.8, 129.5, 129.2, 128.8, 128.5, 127.5, 127.3, 125.9, 123.7, 119.8, 19.2. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1291.
4-Chloro-2-[(1E)-2-phenylethenyl]quinoline (3e )
Pale yellow solid; 68%, 0.18 g; m.p. 109–111 °C (Lit. 21 110–111 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.19–8.16 (m, 1H), 8.09 (d, J = 8.4 Hz, 1H), 7.77–7.72 (m, 2H), 7.69–7.63 (m, 3H), 7.60–7.56 (m, 1H), 7.43–7.37 (m, 2H), 7.36–7.31 (m, 2H). 13C NMR (100 MHz, CDCl3): δ = 156.1, 149.2, 142.7, 136.2, 135.3, 130.5, 129.5, 128.9, 128.7, 127.9, 127.3, 127.0, 125.3, 124.0, 119.0. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0743; C17H1337ClN: 268.0707, found: 268.0716.
6-Methyl-2-[(1E)-2-phenylethenyl]quinoline (3f )
Pale yellow solid; 82%, 0.20 g; m.p. 141–143 °C (Lit. 21 142–144°C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.11 (d, J = 8.4 Hz, 1H), 7.97 (d, J = 8.8 Hz, 1H), 7.67–7.62 (m, 4H), 7.55–7.53 (m, 2H), 7.43–7.38 (m, 3H), 7.34–7.31 (m, 1H), 2.49 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 155.3, 146.2, 136.9, 136.2, 135.7, 133.9, 132.0, 129.2, 128.8, 128.6, 128.5, 127.3, 127.2, 126.4, 118.9, 21.7. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1290.
6-Chloro-2-[(1E)-2-phenylethenyl]quinoline (3g )
White solid; 93%, 0.25 g; m.p. 147–149 °C (Lit. 21 148–149°C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.09 (d, J = 8.8 Hz, 1H), 8.03 (d, J = 9.0 Hz, 1H), 7.80–7.78 (m, 1H), 7.72–7.61 (m, 5H), 7.43–7.31 (m, 4H). 13C NMR (100 MHz, CDCl3): δ = 156.4, 146.3, 136.6, 135.5, 134.8, 131.7, 130.7, 130.5, 128.9, 128.6, 128.5, 127.8, 127.3, 126.2, 120.0. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0744; C17H1337ClN: 268.0707, found: 268.0715.
6-Bromo-2-[(1E)-2-phenylethenyl]quinoline (3h )
Pale yellow solid; 89%, 0.28 g; m.p. 164–166 °C (Lit. 21 164–166°C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.11 (d, J = 8.6 Hz, 1H), 8.05–7.98 (m, 2H), 7.71–7.68 (m, 1H), 7.69–7.61 (m, 4H),7.42–7.31 (m, 4H). 13C NMR (100 MHz, CDCl3): δ = 156.6, 147.0, 136.6, 135.5, 135.0, 133.0, 130.8, 129.5, 128.9, 128.8, 128.5, 128.4, 127.3, 120.5, 119.5. MS (ESI): m/z (%) = 310.0 ([M]+, 51), 312.0 ([M]+, 49). HRMS (ESI): m/z [M]+ calcd for C17H1379BrN: 310.0231, found: 310.0238; C17H1381BrN: 312.0211, found: 312.0219.
7-Methyl-2-[(1E)-2-phenylethenyl]quinoline (3i )
Pale yellow solid; 83%, 0.20 g; m.p. 106–108 °C (Lit. 21 106–108 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.11 (d, J = 8.6 Hz, 1H), 7.89–7.86 (m, 1H), 7.66–7.59 (m, 5H), 7.41–7.35 (m, 3H), 7.32–7.29 (m, 2H), 2.34 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.2, 148.6, 140.0, 136.2, 136.0, 134.1, 129.1, 128.8, 128.5, 128.4, 128.2, 127.3, 127.1, 125.5, 118.0, 21.9. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1291.
7-Chloro-2-[(1E)-2-phenylethenyl]quinoline (3j )
Pale yellow solid; 90%, 0.24 g; m.p. 118–120 °C (Lit. 21 118–119°C). 21 1 H NMR (400 MHz, CDCl3): δ = 7.98–7.97 (m, 2H), 7.82 (d, J = 8.2 Hz, 1H), 7.80–7.74 (m, 5H), 7.50–7.47 (m, 1H), 7.40–7.37 (m, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.7, 148.5, 136.3, 136.0, 135.6, 135.0, 128.9, 128.7, 128.6, 128.4, 128.1, 127.4, 127.0, 125.6, 119.3. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0742; C17H1337ClN: 268.0707, found: 268.0717.
7-Bromo-2-[(1E)-2-phenylethenyl]quinoline (3k )
Pale yellow solid; 87%, 0.27 g; m.p. 127–129 °C (Lit. 14 127–129°C). 14 1 H NMR (400 MHz, CDCl3): δ = 8.22 (d, J = 1.6 Hz, 1H), 8.08 (d, J = 8.8 Hz, 1H), 7.79–7.74 (m, 5H), 7.65–7.62 (m, 1H), 7.41–7.32 (m, 4H). 13C NMR (100 MHz, CDCl3): δ = 156.8, 148.7, 136.4, 136.2, 135.2, 131.7, 129.6, 128.9, 128.8, 128.7, 128.4, 127.5, 125.8, 123.8, 119.6. MS (ESI): m/z (%) = 310.0 ([M]+, 51), 312.0 ([M]+, 49). HRMS (ESI): m/z [M]+ calcd for C17H1379BrN: 310.0231, found: 310.0238; C17H1381BrN: 312.0211, found: 312.0219.
8-Methyl-2-[(1E)-2-phenylethenyl]quinoline (3l )
Pale yellow solid; 54%, 0.13 g; m.p. 72–73 °C (Lit. 21 72°C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.05 (d, J = 8.6 Hz, 1H), 7.76 (d, J = 16.2 Hz, 1H), 7.68–7.62 (m, 4H), 7.56 (d, J = 6.7 Hz, 1H), 7.43–7.31 (m, 5H), 2.76 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 154.7, 147.2, 137.1, 136.7, 136.3, 133.7, 129.7, 129.4, 128.7, 128.4, 127.2, 127.1, 125.8, 125.3, 119.2, 18.4. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1289.
8-Chloro-2-[(1E)-2-phenylethenyl]quinoline (3m )
Pale yellow solid; 59%, 0.16 g; m.p. 88–90 °C (Lit. 16 88–90 °C). 16 1 H NMR (400 MHz, CDCl3): δ = 8.11 (d, J = 8.3 Hz, 1H), 7.80–7.75 (m, 2H), 7.64–7.68 (m, 4H), 7.47–7.31 (m, 5H). 13C NMR (100 MHz, CDCl3): δ = 156.8, 144.3, 136.6, 136.4, 135.2, 133.1, 129.7, 128.8, 128.7, 128.4, 127.3, 127.1, 126.4, 125.8, 119.6. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0745; C17H1337ClN: 268.0707, found: 268.0715.
2-[(1E)-2-phenylethenyl]quinoxaline (3n )
Pale yellow solid; 60%, 0.14 g; m.p. 101–103 °C (Lit. 21 101–103 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 9.01 (s, 1H), 8.08 (d, J = 7.9 Hz, 2H), 7.88 (d, J = 16.3 Hz, 1H), 7.75–7.62 (m, 4H), 7.44–7.33 (m, 4H). 13C NMR (100 MHz, CDCl3): δ = 150.4, 144.7, 142.4, 141.6, 136.6, 135.9, 130.0, 129.4, 129.3, 129.2, 129.1, 128.9, 127.4, 125.1. MS (ESI): m/z (%) = 233.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 233.1079, found: 233.1086.
1-[(1E)-2-phenylethenyl]isoquinoline (3o )
Pale yellow solid; 51%, 0.12 g; m.p. 97–99 °C (Lit. 21 97–98 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.58 (d, J = 5.6 Hz, 1H), 8.36 (d, J = 8.5 Hz, 1H), 8.01 (d, J = 1.2 Hz, 2H), 7.83 (d, J = 7.8 Hz, 1H), 7.72–7.66 (m, 3H), 7.65–7.62 (m, 1H), 7.57 (d, J = 5.5 Hz, 1H), 7.44–7.41 (m, 2H), 7.35–7.32 (m, 1H). 13C NMR (100 MHz, CDCl3): δ = 154.6, 142.7, 136.9, 136.7, 135.8, 129.9, 128.7, 128.6, 127.4, 127.3, 127.1, 126.7, 124.5, 122.8, 119.7. MS (ESI): m/z (%) = 232.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C17H14N: 232.1126, found: 232.1134.
2-[(1E)-2-(2-Methyl)-phenylethenyl]quinoline (3p )
Pale yellow solid; 89%, 0.22 g; m.p. 69–71 °C (Lit. 22 69–71 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.30 (d, J = 8.4 Hz, 1H), 8.06–8.01 (m, 2H), 7.98–7.92 (m, 2H), 7.83–7.77 (m, 2H), 7.60–7.57 (m, 1H), 7.37 (d, J = 16.2 Hz, 1H), 7.27–7.21 (m, 3H), 2.48 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.4, 148.2, 136.9, 136.8, 135.2, 131.9, 131.0, 130.4, 130.3, 129.1, 129.0, 128.2, 127.6, 126.9, 126.7, 126.2, 120.5, 20.0. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1291.
2-[(1E)-2-(2-Chloro)-phenylethenyl]quinoline (3q )
Pale yellow solid; 72%, 0.19 g; m.p. 78–80 °C (Lit.22 78–80 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.35 (d, J = 8.4 Hz, 1H), 8.11 (d, J = 16.2 Hz, 1H), 8.06–7.98 (m, 2H), 7.95 (d, J = 8.0 Hz, 1H), 7.83–7.76 (m, 2H), 7.60–7.53 (m, 3H), 7.44–7.37 (m, 2H). 13C NMR (100 MHz, CDCl3): δ = 155.6, 148.2, 137.3, 134.2, 133.4, 132.0, 130.5, 130.4, 130.3, 129.4, 129.2, 128.3, 128.1, 127.8, 127.7, 126.9, 121.0. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0744; C17H1337ClN: 268.0707, found: 268.0715.
2-[(1E)-2-(3-Methyl)-phenylethenyl]quinoline (3r )
Pale yellow solid; 76%, 0.19 g; m.p. 68–69 °C (Lit. 22 68–69 °C). 22 1 H NMR (400 MHz, CDCl3): δ = 8.33 (d, J = 8.4 Hz, 1H), 8.10 (d, J = 16.2 Hz, 1H), 8.04–7.95 (m, 3H), 7.83 (d, J = 8.4 Hz, 1H), 7.81–7.78 (m, 1H), 7.40–7.33 (m, 3H), 7.24–7.16 (m, 2H), 2.34 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.4, 148.2, 138.3, 136.9, 135.6, 132.1, 131.2, 130.5, 130.3, 129.2, 129.0, 128.3, 127.5, 126.7, 126.5, 126.2, 120.7, 21.1. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1292.
2-[(1E)-2-(3-Fluoro)-phenylethenyl]quinoline (3s )
Yellow solid; 83%, 0.21 g; m.p. 91–93 °C (Lit. 16 91–93 °C). 16 1 H NMR (400 MHz, CDCl3): δ = 8.12 (d, J = 8.4 Hz, 1H), 8.07–48.0 (m, 1H), 7.72–7.67 (m, 2H), 7.58–7.53 (m, 2H), 7.52–7.46 (m, 1H), 7.41–317 (m, 4H), 7.01 (t, J = 8.0 Hz, 1H). 13C NMR (100 MHz, CDCl3): δ = 163.1 (d, J = 243 Hz), 155.2, 148.1, 138.8 (d, J = 8 Hz), 136.3, 132.9 (d, J = 3 Hz), 130.2, 130.0, 129.6, 129.1, 127.4, 127.3, 126.1, 123.1 (d, J = 3 Hz), 119.3, 115.2 (d, J = 21 Hz), 113.3 (d, J = 22 Hz). MS (ESI): m/z (%) = 250.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C17H13FN: 250.1032, found: 250.1039.
2-[(1E)-2-(3-Chloro)-phenylethenyl]quinoline (3t )
Yellow solid; 87%, 0.23 g; m.p. 90–92 °C (Lit. 21 90–91 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.16 (d, J = 8.5 Hz, 1H), 8.08 (d, J = 8.4 Hz, 1H), 7.79–37.7 (m, 2H), 7.64–7.61 (m, 3H), 7.53–7.49 (m, 2H), 7.38 (d, J = 16.2 Hz, 1H), 7.34–7.29 (m, 2H). 13C NMR (100 MHz, CDCl3): δ = 155.6, 148.2, 138.4, 136.5, 134.6, 132.8, 130.2, 130.1, 129.8, 129.3, 128.4, 127.5, 127.3, 127.0, 126.4, 125.4, 119.6. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0742; C17H1337ClN: 268.0707, found: 268.0715.
2-[(1E)-2-(4-Methyl)-phenylethenyl]quinoline (3u )
Yellow solid; 90%, 0.22 g; m.p. 110–112 °C (Lit. 16 110–112 °C). 16 1 H NMR (400 MHz, CDCl3): δ = 8.08 (m, 2H), 7.77–7.66 (m, 4H), 7.55 (d, J = 8.2 Hz, 2H), 7.46 (t, J = 7.4 Hz, 1H), 7.38 (d, J = 16.2 Hz, 1H), 7.22 (d, J = 7.9 Hz, 2H), 2.35 (s, 3H). 13C NMR (100 MHz, CDCl3): δ = 156.3, 148.2, 138.6, 136.3, 134.5, 133.6, 129.6, 129.3, 129.0, 127.9, 127.5, 127.2, 127.1, 126.2, 119.1, 21.2. MS (ESI): m/z (%) = 246.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C18H16N: 246.1283, found: 246.1292.
2-[(1E)-2-(4-Fluoro)-phenylethenyl]quinoline (3v )
Yellow solid; 86%, 0.21 g; m.p. 120–122 °C (Lit. 16 120–122 °C). 21 1 H NMR (400 MHz, CDCl3): δ = 8.10–8.06 (m, 2H), 7.78 (d, J = 8.2 Hz, 1H), 7.74–637 (m, 5H), 7.56–7.49 (m, 1H), 7.32 (d, J = 16.2 Hz, 1H), 7.13–77.0 (m, 2H). 13C NMR (100 MHz, CDCl3): δ = 163.0 (d, J = 246 Hz), 155.9, 148.3, 136.3, 133.4, 132.8 (d, J = 3 Hz), 129.8, 129.2, 128.9, 128.6 (d, J = 3 Hz), 127.6, 127.3, 126.4, 119.6, 115.9 (d, J = 22 Hz). MS (ESI): m/z (%) = 250.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C17H13FN: 250.1032, found: 250.1040.
2-[(1E)-2-(4-Chloro)-phenylethenyl]quinoline (3w )
Yellow solid; 80%, 0.21 g; m.p. 141–143 °C (Lit. 16 141–142 °C). 16 1 H NMR (400 MHz, CDCl3): δ = 8.09 (d, J = 8.4 Hz, 2H), 7.77 (d, J = 8.0 Hz, 1H), 7.72–7.68 (m, 1H), 7.65–87.5 (m, 2H), 7.55–67.4 (m, 3H), 7.36–17.3 (m, 3H). 13C NMR (100 MHz, CDCl3): δ = 155.7, 148.3, 136.3, 135.1, 134.2, 132.8, 129.8, 129.5, 129.1, 128.9, 128.4, 127.5, 127.4, 126.2, 119.2. MS (ESI): m/z (%) = 266.1 ([M]+, 75), 268.1 ([M]+, 25). HRMS (ESI): m/z [M]+ calcd for C17H1335ClN: 266.0736, found: 266.0742; C17H1337ClN: 268.0707, found: 268.0715.
2-[(1E)-2-(2-(Naphthalen-1-yl))vinyl]quinoline (3x )
Yellow solid; 88%, 0.25 g; m.p. 104–106 °C (Lit. 22 105–106 °C). 17 1 H NMR (400 MHz, CDCl3): δ = 8.50 (d, J = 16.2 Hz, 1H), 8.32 (d, J = 8.4 Hz, 1H), 8.12 (d, J = 8.4 Hz, 2H), 7.86–7.71 (m, 6H), 7.56–7.45 (m, 5H). 13C NMR (100 MHz, CDCl3): δ = 156.2, 148.3, 136.2, 134.1, 133.7, 131.6, 131.5, 131.3, 129.6, 129.3, 128.8, 128.6, 127.5, 127.4, 126.3, 126.2, 125.8, 125.7, 124.1, 123.8, 119.5. MS (ESI): m/z (%) = 282.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C21H16N: 282.1283, found: 282.1292.
2-[(1E)-2-(2-(Thiophen-2-yl))vinyl]quinoline (3y )
Yellow solid; 73%, 0.17 g; m.p. 89–91 °C (Lit. 17 89–91 °C). 17 1 H NMR (400 MHz, CDCl3): δ = 8.07–8.00 (m, 2H), 7.84 (d, J = 16.2 Hz, 1H), 7.77–7.72 (m, 2H), 7.56 (d, J = 8.4 Hz, 1H), 7.50–7.47 (m, 1H), 7.30–7.19 (m, 3H), 7.05–7.02 (m, 1H). 13C NMR (100 MHz, CDCl3): δ = 155.6, 155.5, 148.2, 142.1, 136.2, 129.8, 129.1, 128.2, 128.0, 127.7, 127.4, 127.2, 126.0, 125.9, 119.2. MS (ESI): m/z (%) = 238.1 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C15H12NS: 238.0690, found: 238.0697.
2-[(1E)-2-(2-Cyclohexylvinyl)]quinoline (3z )
Pale yellow oil; 30%, 0.07 g. 17 1H NMR (400 MHz, CDCl3): δ = 8.07–8.02 (m, 2H), 7.76–7.72 (m, 1H), 7.68–7.63 (m, 1H), 7.54 (d, J = 8.4 Hz, 1H), 7.47–7.43 (m, 1H), 6.80–6.76 (m, 1H), 6.66 (d, J = 16.2 Hz, 1H), 2.30–2.15 (m, 1H), 1.77–1.64 (m, 4H), 1.33–1.19 (m, 6H). 13C NMR (100 MHz, CDCl3): δ = 156.6, 148.2, 143.5, 136.2, 129.4, 129.1, 128.5, 127.4, 127.2, 125.8, 118.6, 41.0, 32.3, 26.1, 26.0. MS (ESI): m/z (%) = 238.2 ([M]+, 100). HRMS (ESI): m/z [M]+ calcd for C17H20N: 238.1596, found: 238.1604.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to the Traditional Chinese Medicine Research Projects of Health and Family Planning Commission of Jiangxi Province (No. 2019A228 and 2020B0388) for financial support.