Abstract
New edaravone derivatives containing a benzylpiperazine moiety are designed and synthesized. The structures are characterized by 1H NMR, 13C NMR, and high-resolution mass spectrometry. The potential neuroprotective activities of the target compounds are evaluated in differentiated rat pheochromocytoma cells (PC12 cells) and in mice subjected to acute cerebral ischemia. Most of the target compounds showed neuroprotective activities both in vivo and in vitro, especially 1-(4-(4-fluorobenzyl) piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone and 1-(4-(4-nitrobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone, which displayed significant protective effects on cell viability against damage caused by H2O2, and remarkably prolonged the survival time of mice subjected to acute cerebral ischemia and decreased the mortality rate at all doses. These compounds represent lead compounds for the further discovery of neuroprotective agents for treating cerebral ischemic stroke. Molecular docking studies and basic structure–activity relationships are also presented.
New edaravone derivatives containing a benzylpiperazine moiety are designed and synthesized.
Introduction
Ischemic stroke, the most common cerebrovascular disorder, accounts for more than 80% of all strokes and is one of the leading causes of morbidity and mortality worldwide.1,2 Currently, the treatment of ischemic stroke is accomplished using for thrombolytic therapy in general, which can lead to secondary brain injury, namely, cerebral ischemia–reperfusion injury.3–5 Oxidative stress is considered as the most important reason among a series of mechanisms related to the pathogenesis of cerebral ischemia–reperfusion injury. Oxidative stress can accelerate the generation of reactive oxygen species (ROS), which significantly exacerbate ischemic brain damage. 6 However, overproduction of ROS cannot be neutralized under ischemic conditions because the antioxidant defense system in ischemic tissues is interrupted. 7 Hence, exogenous supplementation of antioxidants with ROS scavenging activity would be a potential therapy for cerebral ischemia–reperfusion injury.
Edaravone (EDA, 3-methyl-1-phenyl-2-pyrazolin-5-one), an acid pyrazoline compound, is a recently developed neuroprotective drug that has been successfully used for treating acute stroke caused by cerebral thrombosis and embolism. 8 EDA displays potent antioxidant properties, and consequently, it might be useful in managing other pathological processes involving oxidative stress.9–11
Moreover, there is increasing interest in the synthesis of EDA derivatives designed to potentiate the free-radical scavenging activity of this compound. Some of these derivatives have been reported to present other beneficial properties, such as antioxidant activity, vasodilator activity, anticerebral ischemia activity and antimyocardial ischemia activity.12–14
The piperazine nucleus is capable of binding to multiple receptors with high affinity. Piperazine derivatives are widely applied in a number of different therapeutic areas, including antitumor, antifungal, antidepressant, and antiviral.15–17 The piperazine scaffold occupies an important position in the field of medicinal and pesticide chemistry (Figure 1).18,19 Our previous studies discovered that the benzylpiperazine scaffold often affords neuroprotective activity. 20 Given that a number of drug molecules containing piperazine exhibit improved efficacy in several aspects, especially for vasodilation, antihypertension, increasing cerebral blood flow, and neuroprotection,21–23 we therefore designed and synthesized a new class of compounds obtained by joining EDA through an acetyl linker with benzhydryl piperazines. The obtained edaravane derivatives were evaluated for their antioxidant activities in vitro and anticerebral ischemia activities in vivo. In addition, molecular modeling was employed to further investigate the mechanisms underlying their activities.

Examples of benzylpiperazine-based drug molecules.
Results and discussion
Synthetic chemistry
The EDA derivatives

Reagents and conditions: (i) ethyl o, EtOH, reflux, 8 h. (ii) 40% HBr, AcOH, reflux, 10 h. (iii) NBS, AIBN, CCl4, reflux, 1 h. (iv) N-Boc-piperazine, K2CO3, CH2Cl2, reflux, 10 h; then CH2Cl2, TFA, r.t., 2 h. (v) chloroacetyl chloride, Et3N, THF, r.t., 6 h. (vi) K2CO3, MeCN, reflux, 10 h.
In vivo anti-ischemic activity
First, the target compounds were screened in mice that had undergone bilateral common carotid artery occlusion to study the effects of the prepared compounds on acute cerebral ischemia. As shown in Table 1, most of the target compounds were active in mice intraperitoneally (i.p.) at different doses (200, 100, 50, 25 mg kg−1), among which compounds
In vivo anti-ischemic effects of compounds

NS: normal saline.
p < 0.05 versus NS.
In vitro antioxidant activity related to the structure
In order to study the potential antioxidant activity of the title compounds, another screening was performed to investigate neuroprotection on impairment induced by H2O2 in PC12 cells, as evaluated by the MTT assay. We damaged PC12 cells with 10 mM H2O2 and assessed the protective actions of 12 EDA derivatives. We found that most of the new compounds exhibited moderate to good protection against H2O2-induced cell damage (Table 2).
In vitro neuroprotective effects of compounds

p < 0.05 versus H2O2-treated group.
As shown in Table 2, compounds
The derivatives were substituted with different functional groups to study the influence of the substituent effect on the biological activity and find new compounds with better neuroprotective activity. The substituents were considered and selected based on lipophilic and electronic attributes, for example, lipophilic electron–withdrawing groups (NO2, CN, F, Cl, and Br), hydrophilic electron–donating groups (OCH2CH3), and lipophilic electron–releasing groups (COOCH3 and naphthyl). The general structure–activity relationship (SAR) of these compounds showed that compounds with a 4-substituent on the benzylpiperazine moiety were more active than those with a 2-substitutent (cell protection:
Molecular docking
The nuclear factor erythroid 2-related factor 2 (Nrf2) is a principal regulator of the cellular defense system against oxidative stress, promoting the transcription of an expansive set of antioxidants and cytoprotective enzymes.24,25 Under normal conditions, Nrf2 is kept at low concentrations by the cytosolic repressor protein Kelch-like ECH-associated protein 1 (Keap1). By contrast, under pathological conditions, Nrf2 is released from Keap1.26–28
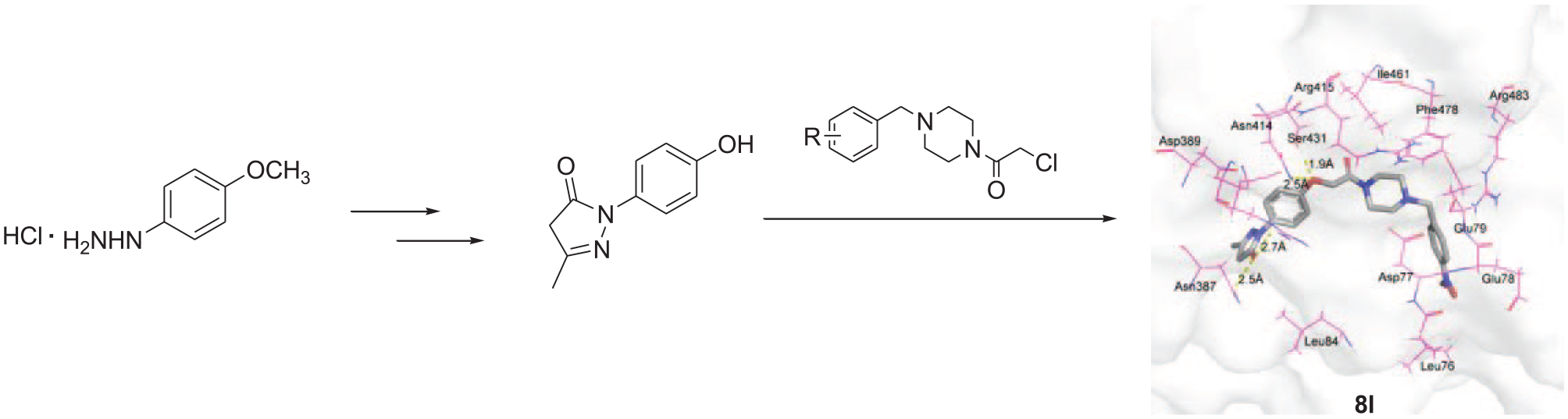
In order to further evaluate these promising compounds and to guide further structure–activity relationship (SAR) studies, the interaction effects of the electron-releasing naphthyl-substituted compound

A 3D plan view of the

A 3D plan view of the
As shown in Figure 2, the oxygen atom of the carbonyl moiety of
The above research suggests that compounds
Conclusion
In summary, 12 novel EDA derivatives
Experimental
Materials and apparatus
All chemicals, reagents, and solvents were commercially available and used without further purification. Melting points were determined on an electrothermal digital apparatus model WRR-401 (Shanghai, China) without correction. The 1H and 13C NMR spectra were recorded on a Bruker ACF-300 MHz instrument (Bruker, Billerica, MA, USA) with CDCl3 as the solvent and tetramethylsilane as an internal standard (chemical shifts are expressed as δ values, and J in hertz). High-resolution mass spectrometry (HRMS) was performed with a MALDI Micro MX mass spectrometer (Waters, Milford, MA, USA) with electrospray ionization (ESI). The measurements were performed using either positive or negative ionization modes. Fetal bovine serum (FBS) was obtained from HyClone (Logan, UT, USA). Horse serum (HS), penicillin, and streptomycin were obtained from Gibco BRL (Div. of Invitrogen, Gaithersburg, MD, USA). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) was purchased from Sigma Chemical Co. (St. Louis, MO, USA).
1-(4-Hydroxyphenyl)-3-methyl-1H-pyrazol-5(4H)-one (3 )
A mixture of 4-methoxyphenylhydrazine hydrochloride (
Substituted benzyl bromides 5a–l ; general procedure
A mixture of the corresponding commercially available substituted toluene
Substituted N-benzylpiperazines 6a–l ; general procedure
To a stirred solution of N-Boc-piperazine (2 mmol) in CH2Cl2 (20 mL) were sequentially added the substituted benzyl bromide
Substituted 1-(4-benzylpiperazin-1-yl)-2-chloroethanones 7a–l ; general procedure
A mixture of the corresponding substituted benzylpiperazine
EDA derivatives 8a–l ; general procedure
A mixture of the corresponding substituted 1-(4-benzylpiperazin-1-yl)-2-chloroethanone
4-((4-(2-(4-(5-Hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)acetyl)piperazin-1-yl)methyl)-N,N-dimethylbenzenesulfonamide (
Methyl 4-((4-(2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)acetyl)piperazin-1-yl)methyl)benzoate (
2-(4-(5-Hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)-1-(4-(naphthalen-1-ylmethyl)piperazin-1-yl)ethanone (
1-(4-(4-Fluorobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(2-Fluorobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(4-Iodobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(2-Bromobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
4-((4-(2-(4-(5-Hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)acetyl)piperazin-1-yl)methyl)benzonitrile (
1-(4-(4-Bromobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(4-Chlorobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(2-Ethoxybenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
1-(4-(4-Nitrobenzyl)piperazin-1-yl)-2-(4-(5-hydroxy-3-methyl-1H-pyrazol-1-yl)phenoxy)ethanone (
In vitro antioxidant activity assays
The in vitro antioxidant activities of target compounds
PC12 cells were inoculated in a 96-well microplate (105 cells/well in 100 µL medium) for 24 h. After washing with phosphate-buffered saline (PBS), the PC12 cells were incubated with H2O2 (10 mM), or H2O2 (10 mM) with compounds
In vivo anti-ischemic activity assays
The in vivo anti-ischemic activity was tested using bilateral common carotid artery occlusion.41,42 Kunming mice of both sexes were randomly divided into groups (10 mice per group). The title compounds
Molecular modeling
The molecular docking procedure was performed within SYBYLX 1.3 software. The crystal structure of Keap1-Nrf2 (PDB ID: 1X2R) was obtained from the RCSB Protein Data Bank. Keap1-Nrf2 was modified using the Prepare Protein Structure Tool. The designed ligands were constructed and energy minimized using the Tripos force field. The convergence accuracy and the maximum number of iteration steps were 1000 and 0.005 kcal mol−1 Å, respectively. The Keap1 protein was defined as a receptor. Nrf2 was removed, and its site sphere was selected as a reference for inserting
Supplemental Material
sj-docx-1-chl-10.1177_17475198221116827 – Supplemental material for The design and synthesis of benzylpiperazine-based edaravone derivatives and their neuroprotective activities
Supplemental material, sj-docx-1-chl-10.1177_17475198221116827 for The design and synthesis of benzylpiperazine-based edaravone derivatives and their neuroprotective activities by Mengjie Gao, Shuangyan Ma, Tong Xu, Nan Jiang, Yi Xu, Yan Zhong and Bin Wu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
