Abstract
The reaction of RCH2X with [PtMe2(DPA)],
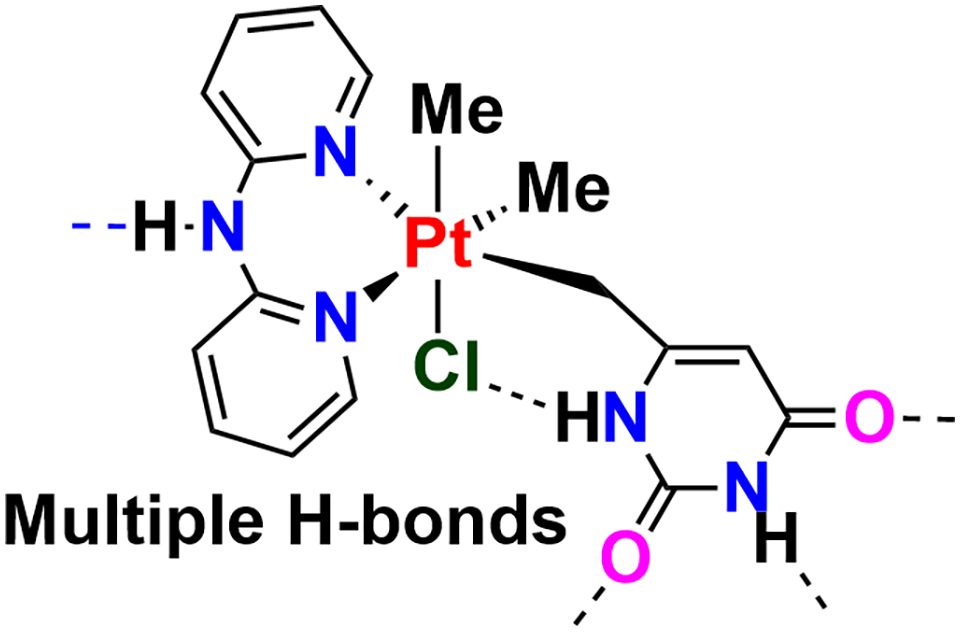
Introduction
The fields of supramolecular chemistry and organometallic chemistry continue to flourish, and there is increasing interest in the interface of these areas, since the products have potential applications in molecular materials, polymers, catalysis or pharmaceuticals.1–7 For example, there are now many platinum complexes that contain substituents with hydrogen bonding properties that can participate in supramolecular self-assembly.8–15 The incorporation of nucleobase units in platinum complexes has been a particular area of interest because both have important applications in cancer therapy, either separately or in combination. For example, a combination of 5-fluorouracil and cisplatin is effective against several tumour types.16–18
A versatile route to organoplatinum(IV) complexes is by oxidative addition of alkyl halides to electron-rich platinum(II) complexes. These reactions typically occur by the polar SN2 mechanism, leading to trans oxidative addition.19,20 The reactions are tolerant of many functional groups. Specifically, hydrogen bonding groups, including carboxylic acids and the nucleobase uracil can be incorporated in this way (Scheme 1).21–24 Complexes

The synthesis and supramolecular self-assembly of some organoplatinum(IV) complexes with hydrogen bonding substituents.
This paper describes related chemistry using the chelate ligand di-2-pyridylamine [HN(2-C5H4N)2, DPA] in place of the 4,4′-di-t-butyl-2,2′-bipyridine (bubipy) ligand of Scheme 1. 21 DPA has an NH group and so there is potential for more extensive hydrogen bonding and self-assembly. 25 Surprisingly, a difference in the stereochemistry of oxidative addition was observed, and the structure of a syndiotactic supramolecular polymer is described. We note the related expertise of Alwyn Davies in the self-association of organometallic compounds.26,27
Results and discussion
The new organoplatinum complexes are shown in Scheme 2. The complex [PtMe2(DPA)] (DPA = 2,2′-dipyridylamine),

The synthesis of organoplatinum(IV) complexes with hydrogen bonding groups.
We were not able to grow good single crystals of the carboxylic acid derivatives
The 1H NMR spectrum of complex
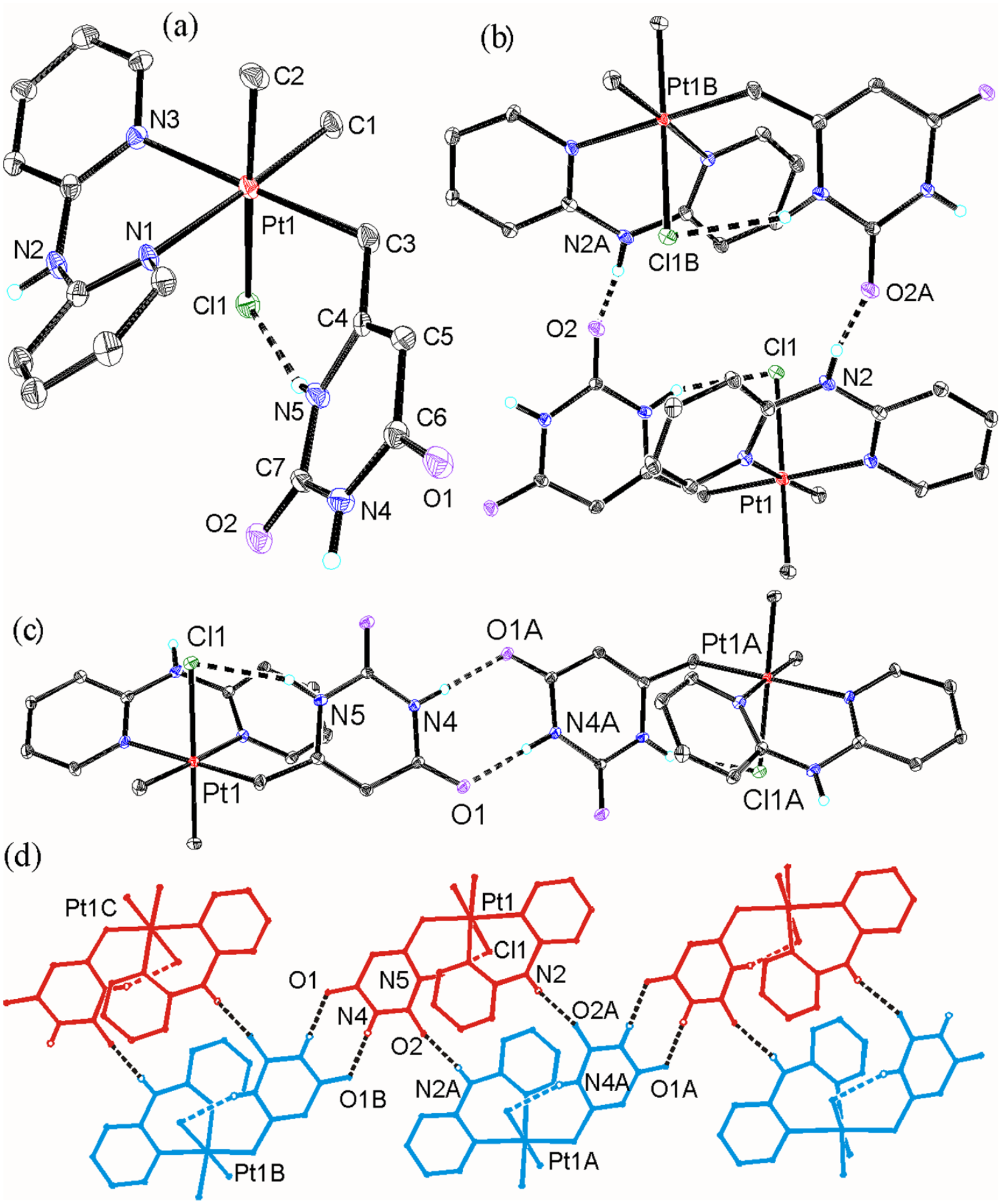
Views of the structure of complex 2a; (a) the molecular structure and intramolecular hydrogen bond [graph set S11(6), N(5)..Cl(1) 3.125(5) Å]; (b) intermolecular hydrogen bonds [graph set R22(20), N(2)..O(2B) = O(2)..N(2B) = 2.865(6) Å, symmetry equivalent atoms x,y,z; 2-x, -y,1-z]; (c) intermolecular hydrogen bonds [graph set R22(8), N(4)..O(1A) = O(1)..N(4A) = 2.789(7) Å, symmetry equivalent atoms x,y,z; 2-x, -1-y, -z]; (d) part of the supramolecular polymer formed by intermolecular hydrogen bonding (opposite enantiomers shown as red and blue).
To gain further insight into the molecular stereochemistry of the complexes, DFT calculations were carried out for both products of cis and trans oxidative addition (see experimental for details).
29
The calculated structures and relative energies are shown in Figure 2. The calculations successfully predict the greater relative stability of

Calculated structures of trans and cis isomers of the complexes and their relative energies: (a) complex
Conclusion
The prediction that the platinum(IV) complexes with DPA ligand might give different structures compared to the bubipy complexes of Scheme 1 has been upheld (Scheme 2). The observation of much faster cis-trans isomerism of the DPA complexes was not predicted but can be rationalized according to Scheme 3. The easy flexing of the 6-membered Pt(DPA) ring can allow the NH group to hydrogen bond to the leaving halide ion in the first step of the SN2 oxidative addition to give 5-coordinate intermediate

A likely mechanism of cis-trans isomerization for the DPA complexes.
The most interesting case was the oxidative addition of 6-(chloromethyl)uracil to give the chiral complex
Experimental
The complex [Pt2Me4(µ-SMe2)2] was prepared according to the literature method.36,37 NMR spectra were recorded by using a Varian Mercury 400 NMR or Varian Inova 600 NMR spectrometer. 1H chemical shifts are reported relative to TMS and assignments were aided by 1H-1H NOESY and 1H-1H gCOSY experiments. The NMR labels are shown in Chart 1. IR spectra were recorded using a Bruker Vector spectrometer by ATR. X-ray data were collected at 150 K using a Nonius Kappa CCD diffractometer with graphite-monochromated Mo-Κα radiation (λ = 0.71073 Å). A suitable crystal of

NMR labels.
[PtMe2(DPA)] (DPA = 2,2′-dipyridylamine), 1
To a stirred solution of [Pt2(SMe2)2Me4] (1.50 g, 2.61 mmol) in diethyl ether (30 mL) was added 2,2′-dipyridylamine (1.25 g, 7.34 mmol) and the mixture was stirred for 3 h. The yellow precipitate which formed was separated by filtration, washed with ether (2 × 5 mL) and dried under vacuum. Yield 1.37 g, 61%. NMR in acetone-d6: δ(1H) = 0.58(s, 6H, 2J(PtH) = 86 Hz, PtMe), 6.94 (dd, 1H, 3J(H6H5) = 6 Hz, 3J(H5H4) = 8 Hz, H5), 7.17 (d, 2H, 3J(H4H3) = 8 Hz, H3), 7.82 (t, 2H, 3J(H4H3) = 3J(H4H5) = 8 Hz, H4), 8.59 (d, 2H, 3J(H6H5) = 6 Hz, H6), 9.28 (s, 1H, NH7). Anal. calcd for C12H15N3Pt: C 36.4, H 3.8, N 10.6. Found: C 36.2, H 3.6, N 10.3%.
[PtClMe2(CH2C4H3N4O2)(DPA)], 2a
To a solution of [PtMe2(DPA)],
NMR in DMSO-d6: δ(1H) = 0.84 (s, 3H, 2J(PtH) = 73 Hz, PtMe), 0.87 (s, 3H, 2J(PtH) =68 Hz, PtMe), 2.53 (d, 1H, 2J(HH) = 10 Hz, 2J(PtH) = 82 Hz, CHa), 3.11 (d, 1H, 2J(HH) = 10 Hz, 2J(PtH) = 114 Hz, CHb), 5.19 (s, 1H, uracil CH); 7.16 (dd, 1H, 3J(H6aH5a) = 6 Hz, 3J(H5aH4a) = 7 Hz, H5a); 7.18 (dd, 1H, 3J(H6bH5b) = 6 Hz, 3J(H5bH4b) = 7 Hz, H5b); 7.29 (d, 1H, 3J(H4bH3b) = 8 Hz, H3b); 7.30 (d, 1H, 3J(H4aH3a) = 8 Hz, H3a); 7.92 (m, 2H, H4a, H4b); 8.25 (d, 1H, 3J(H6bH5b) = 6 Hz, H6b); 8.40 (d, 1H, 3J(H6aH5a) = 6 Hz, H6a), 9.84 (s, 1H, uracil NH1); 10.66 (s, 1H, uracil NH3; 10.71 (s, 1H, NH7). IR (cm–1): 1715, 1612 [ν(C=O)], 3100 [br, ν(NH)]. Anal. calcd for C17H20ClN5O2Pt: C 36.7, H 3.6, N 12.6. Found: C 37.0, H 3.6, N 12.3%.
[PtBrMe2(CH2CO2H)(DPA)], 3a
To a solution of complex
[PtBrMe2(4-CH2C6H4CO2H)(DPA)], 4a and 4b
This was prepared similarly from complex
Supplemental Material
sj-cif-2-chl-10.1177_17475198221114610 – Supplemental material for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents
Supplemental material, sj-cif-2-chl-10.1177_17475198221114610 for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents by Michael J Nieradko and Richard J Puddephatt in Journal of Chemical Research
Supplemental Material
sj-docx-1-chl-10.1177_17475198221114610 – Supplemental material for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents
Supplemental material, sj-docx-1-chl-10.1177_17475198221114610 for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents by Michael J Nieradko and Richard J Puddephatt in Journal of Chemical Research
Supplemental Material
sj-xyz-3-chl-10.1177_17475198221114610 – Supplemental material for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents
Supplemental material, sj-xyz-3-chl-10.1177_17475198221114610 for Supramolecular chemistry of organoplatinum(IV) complexes: A syndiotactic polymer with uracil substituents by Michael J Nieradko and Richard J Puddephatt in Journal of Chemical Research
Footnotes
Acknowledgements
The authors thank Dr PD Boyle and MC Jennings for assistance with X-ray structure determination, and Dr M. Rashidi for experimental expertise.
Authors’ note
Dedicated to Alwyn Davies for his outstanding research and mentorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Supporting information
Calculated atomic coordinates from the DFT calculations. Details of the X-ray structure determination are given in the cif file and have been deposited at the Cambridge Crystallographic Data Centre (CCDC 2168906).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
