Abstract
Selenium compounds have a checkered history. Originally considered to be highly toxic, the tide turned in the 20th century when selenium was discovered to be an essential trace element; indeed, selenium is the least abundant element on Earth to have a well-defined biological role. Despite this new-found importance, organoselenium compounds were largely curiosities because methods for their synthesis were cumbersome and unpleasant and often involved toxic reagents and/or hazardous procedures. This paper describes how work carried out in collaboration with Alwyn Davies in the late 1980s, aimed at acquiring Electron Spin Responance (ESR) spectra of selenophene radical anions and cations, led to the development of free-radical methods for the synthesis of numerous selenium-containing heterocycles, many of which showed interesting and useful biological properties. This journey ends with the development of selenium-containing carbohydrates (selenosugars) that exhibit unique skin-repair properties and the establishment of Seleno Therapeutics as a vehicle for the commercialization of these selenosugars.

Introduction
Selenium chemistry has been my companion throughout my professional career, from the early days as a postdoctoral researcher, through various incarnations of my research group, and finally to a more-corporate focus on selenium-based therapies. To have predicted the outcome of this journey back when it began would have been impossible because, without serendipity, good fortune, and a background in free-radical chemistry, this journey would never have begun.
Serendipity first appears in this story in the form of Alwyn Davies, to whom this paper is dedicated and with whom I had the pleasure of working as a Ramsay Memorial Research Fellow at University College London during the late 1980s. At that time we were interested in acquiring Electron Spin Responance (ESR) spectra of the radical cations and anions of various heterocycles, and my focus turned to the 2,5-diphenylchalcophenes

Preparation of 2,5-diphenylchalcophenes
Radical anion

ESR spectra of (a) the 2,5-diphenylselenophene and (b) the 2,5-diphenyltellurophene radical anion (
We next turned our attention to other selenophenes and tellurophenes, which is where the story gets messy and complicated. Not only are smaller selenophenes and tellurophenes more difficult to make, but the chemistry to make them proved to be extremely unpleasant. For example, the very malodorous parent selenophene (

Early syntheses of selenophenes.
Faced with choices that were unpleasant, dangerous, or simply too complicated, I turned my attention to developing a new method for the synthesis of selenium-containing heterocycles.
I was fortunate enough to have apprenticed with Athel Beckwith at the Australian National University during the free-radical Renaissance period, 6 which was beginning to wane toward the end of the 1980s, having contributed to our understanding of intramolecular free-radical addition reactions to C–C double and triple bonds.6,7 Knowing that selenium is an important antioxidant element, 8 and with the knowledge of some basic rate data for intermolecular homolytic substitution (SH2) reactions at selenium, 9 I began to plan a new route to selenocycles that was both efficient and avoided the unpleasant drawbacks of the abovementioned existing methods; this strategy laid the foundations for a fruitful career, both academically and (later) commercially.
This short review paper briefly describes the development of intramolecular homolytic substitution (SHi) chemistry at selenium, how it led to the synthesis of numerous biologically relevant selenium-containing molecules, and how a chance discussion with Michael Davies (then at the Heart Research Institute in Sydney) led to several patentable inventions and the establishment of Seleno Therapeutics as a vehicle for the commercialization of small selenium-containing biologically active molecules.
Developing intramolecular homolytic substitution chemistry at selenium
A few sulfur-containing rings had been prepared using homolytic substitution chemistry by 1990; notably, Kampmeier and Evans
10
and Tundo and colleagues
11
reported the preparation of dibenzothiophene (

Some pre-1990 examples of intramolecular homolytic substitution at sulfur.
Armed with little more than a few guesstimates for rate constants and plenty of enthusiasm, in 1991 Sutej synthesized Barton ester precursor

First examples of SHi chemistry at selenium.

Determining the first rate constant for SHi at selenium.
As I wrote some 10 years ago: “There was a time when free radicals were scorned by organic chemists and when practically every organic textbook written contained a statement that free radicals were incapable of an independent existence.” 6 We now appreciate that the enlightenment provided by the mechanistic and kinetic understanding forged during the free-radical Renaissance period transformed free-radical C–C bond-forming chemistry into a powerful synthetic tool that now rivals its ionic siblings. Because free-radical reactions are mostly under kinetic control, rate constants for intramolecular homolytic substitution chemistry are needed to judiciously design the correct reaction conditions to afford the required outcome. For example, Scheme 5 shows that the choice of precursor is important; while the iodide leads to cyclization, the bromide does not because tributyltin radicals react about 1000 times more slowly with aryl bromides than aryl iodides. 19
Rate data for intramolecular homolytic substitution at chalcogen
Newcomb provides an excellent account for the need for “radical clocks” in the Encyclopedia of Radicals in Chemistry, Biology and Materials published in 2012 and provides a comprehensive list of rate data.
20
With similar intentions, my group also dedicated themselves over many years to creating a rate scale for SHi chemistry at chalcogen, sometimes in collaboration with others. While discussing the details of these endeavors falls outside of the scope of this paper, there are a few points worth noting. The rate constants for intramolecular homolytic substitution at chalcogen depend on the chalcogen, the nature of the carbon-centered radical, and (surprisingly) the presence or absence of a lone pair of electrons on the chalcogen. The rate scale spans almost 46 orders of magnitude, from the ring-closure at methyl sulfone

Kinetic scale for intramolecular homolytic substitution reactions at chalcogens.
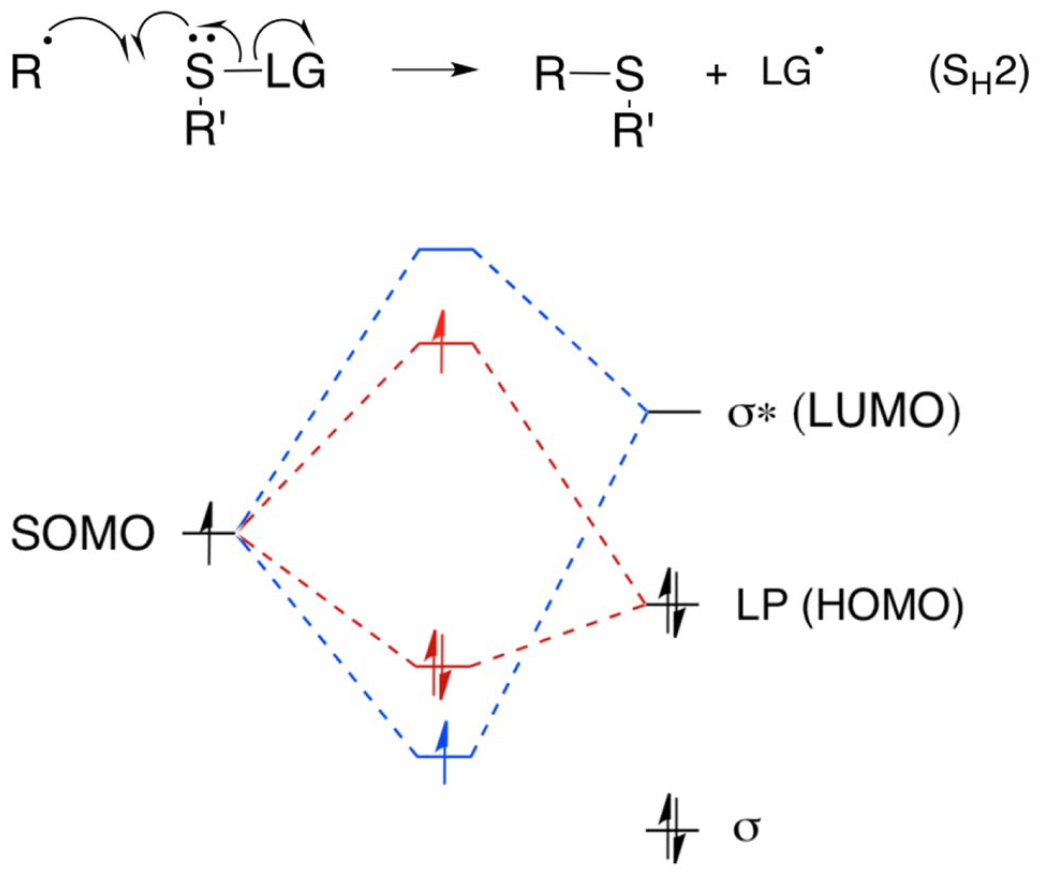
Dual orbital interactions involved in the homolytic substitution reaction of a carbon radical at sulfur, which is assisted by the lone pair of electrons.
Applications to the syntheses of biologically active molecules
Our foray into biologically active compounds began quite serendipitously over a bet at a conference, where it was suggested that nitrogen-centered radicals would fail to undergo SHi at selenium. As shown in Scheme 4, nitrogen-centered radicals react to produce selenazoles and benzoselenazol-3(2H)-one analogues in moderate-to-good yields (so I won the bet). To be fair, we were unable to get aminyl radicals to cyclize at selenium, most likely because they are better leaving groups than benzyl (which I concede might have been the intention of the bet).
While we did not intend to prepare any biologically active compounds, 2-phenyl-1,2-benzoselenazol-3(2H)-one piqued my interest as I had not expected it to be (somewhat) famous. Indeed, as more commonly known, Ebselen (Scheme 4) is a powerful antioxidant and glutathione peroxidase mimic 32 that, despite a flurry of activity in the 1990s, has not yet made it to market, but I note that it has been examined for the treatment of chemotherapy-induced hearing loss in cancer patients, 33 and that there is renewed interest in its ability to covalently inhibit the SARS-CoV-2 (COVID-19) papain-like protease. 34
Numerous selenium-containing pharmaceutically relevant compounds were synthesized in my research group from the late 1990s onwards (Figure 4). Examples include selenotocopherol analogue

Pharmaceutically relevant compound synthesized from the late 1990s.
Is it worth mentioning here that commercializing selenium-containing compounds can be particularly difficult. Selenium has been unfairly (blanket) labeled as “toxic,” often by individuals who don’t really understand the importance of molecular structure. This prejudice can be traced as far back as the voyages of Marco Polo, 45 whose horses suffered from equine alkali disease (blind staggers) 46 after ingesting grass high in inorganic selenium in western China; similar equine responses to vegetation growing in selenium-rich soil were reported to the United States Congress in 1857 47 and by numerous farmers in Nebraska, South Dakota, and other regions over the years. 48 Despite being well recognized as an essential trace element since 1957, 49 selenium remains a tainted element, with reports of human death by misadventure 50 and even murder 51 through selenium administration unhelpful to its reputation. In addition, regulations that place blanket bans on selenium-containing compounds, irrespective of toxicity considerations, as is the case in the European Union for cosmetics, 52 provide additional stumbling blocks.
The need for potent water-soluble antioxidants
A wide range of oxidants are produced biologically, including those generated by the inflammatory enzyme myeloperoxidase (i.e. hypochlorous acid, HOCl; hypobromous acid, HOBr; hypothiocyanous acid, HOSCN), peroxynitrous acid (ONOOH), peroxides (H2O2, ROOH), and singlet oxygen (
1
O2)). Significant amounts of data acquired over the years indicate that these oxidants can damage human cells, tissue, and plasma proteins; consequently, there is a need for powerful antioxidants capable of protecting even the most vulnerable amino acid residues (i.e. Cys, Met) against oxidative damage. The chemistry of these biological oxidants has occupied the interests of Mike Davies (University of Copenhagen) for many years.
53
At a fortuitous meeting, we discussed the benefits of selenium-containing compounds as antioxidants and lamented the fact that most organic selenides are water (and therefore plasma) insoluble and not particularly effective at protecting biological materials against oxidative damage. After some consideration, we hypothesized that selenium-containing carbohydrates should be infinitely water soluble and therefore address the abovementioned issues. Unfortunately, we were unable to deprotect
Inspired by the work of Pinto,
54
we prepared a series of selenosugar derivatives and examined their antioxidant capacities. For example, 1,5-anhydro-5-seleno-
Despite this success, 1,5-anhydro-5-selenohexitols

Syntheses of 1,4-anhydroseleno-
Not unexpectedly, the prepared selenosugars proved to be potent antioxidants, with second-order rate constants of about 108 M−1 s−1,56 sufficient to protect even the most vulnerable protein residues from oxidative damage associated with the action of the human heme enzyme myeloperoxidase; indeed, they exhibit among the highest rate constants for reactions with HOCl and HOBr and are capable of protecting biomolecules against oxidative damage. Further details can be found in a recent perspective article. 53 With these excellent data in hand, we sought patent protection for our selenosugars. 57
Among the prepared selenosugars, one (
Concluding remarks
While Plato famously said (in the guise of Socrates and Adeimantus in the Republic): “our need will be the real creator” (or “necessity is the mother of invention”), I strongly believe that serendipity plays an equally important role in the creation of new ideas. In this short review paper, I show how both necessity and serendipity, which began with Alwyn Davies and a simple ESR spectrum, the need to prepare novel molecules, and my unforeseen discovery of the biological importance of selenium, transformed a largely obscure research field (selenium-containing heterocycles) into a thriving discipline that produced a number of key outcomes of strong relevance to the healthcare sector. I am profoundly grateful for the introduction to the field afforded to me by Alwyn and sincerely hope that the chemistry that evolved over the ensuing decades will provide significant global health benefits.
Footnotes
Author’s note
Dedicated to Professor Alwyn G Davies on the occasion of his 95th birthday with the sincere congratulations that such a good innings deserves.
Declaration of conflicting interests
The author is a financial shareholder in Seleno Therapeutics and a named co-inventor on several patents of relevance to the work presented in this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the Australian Research Council over many years is gratefully acknowledged.
