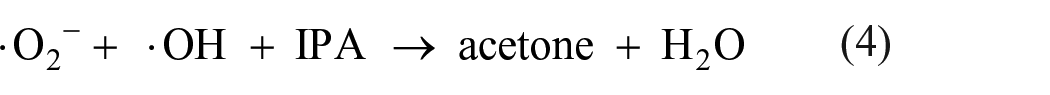Abstract
ZnFe2O4 is an environmentally friendly semiconductor material which has potential applications in catalytic organic pollutant degradation. Here, we demonstrate the synthesis of ZnFe2O4 and its photothermal catalytic application. The obtained ZnFe2O4 prepared by using the sol-gel method has a large specific surface area which reaches 56.4 m2/g. Electron paramagnetic resonance, X-ray photoelectron spectra, and photoabsorption results indicate that ZnFe2O4 has plentiful oxygen vacancies. When evaluated by the degradation of the gaseous organic pollutant isopropanol, oxygen-vacancy-rich ZnFe2O4 presents a high and stable thermal catalytic performance, while driven by high-intensity light. In addition, a distinct improvement is observed. Moreover, a synergic photothermal catalytic mechanism of isopropanol degradation on ZnFe2O4 is proposed.

Introduction
Catalytic technology has significant prospects in environmental purification, especially for the degradation of toxic organic chemicals. Recently, much attention has been paid to catalytic technology, including photocatalysis, 1 thermal catalysis, 2 and photo electrocatalytic degradation. 3 Among them, photocatalytic pollutant removal, which utilizes photo-generated electrons and holes to degrade organic pollutants at room temperature, is a hot research field and has been studied intensively. 4 Semiconductor photocatalysts have stable chemical properties, are low-cost, and have high recycling rates; however, researchers still face challenges that need to be solved. One challenge is due to the rapid combination of photo-generated electrons and holes in a redox reaction, the quantum efficiencies of most photocatalysts are low. 5 Another challenge is that during the process of catalytic reaction, refractory intermediates are sometimes deposited, which hinders the adsorption of reactants on photocatalytically active sites. 6
As is known, when the catalyst is irradiated by light, some light energy is converted into photo-generated electron-holes, and other light energy transfers into heat to increase the temperature of samples.7,8 The high temperature, that is, thermal energy, usually cannot be utilized for photocatalysts to produce photo-generated photons and holes for photocatalytic reactions. To make better use of solar energy, a feasible method is combining photocatalysis and low temperature thermal catalysis, which shows some advantages: photothermal catalysis can maximize the utilization of solar energy and thermal energy, and it has excellent durability for thermal catalytic oxidation with the low energy consumption of photocatalytic oxidation. 9 Moreover, a synergic photothermal catalyst can generate hot carriers to take part in redox reactions or convert photon energy into heat to accelerate the reaction rate.10–12 Although photothermal synergic catalysis is an effective strategy, reported photothermal catalysts are rare.
ZnFe2O4 is a type of ferrite with a spinel structure and is also known as a soft magnetic material, an antibacterial agent and as a photocatalyst. 13 It is cheap, non-toxic, environmentally friendly, and has high chemical stability. ZnFe2O4 has a narrow bandgap of about 1.9 eV, which means that it can utilize sunlight energy efficiently. Li et al. 14 have prepared ZnFe2O4 nanotube arrays to degrade 4-chlorophenol in solution, with the photocatalytic degradation rate of 4-chlorophenol being 100%. Dhiman et al. 15 studied the effects of different ZnFe2O4 nanostructures, with the results showing that nanorods have the best photocatalytic activity for dye degradation. Wu et al. 16 successfully prepared well-organized porous hierarchical ZnFe2O4 nanostructures and studied their solar light-driven photocatalytic performance for methylene blue (MB) degradation. Oliveira et al. 17 synthesized ZnFe2O4 photocatalysts by the combustion reaction in solution. ZnFe2O4 exhibited good photocatalytic response to the degradation of the dyes at 400 °C, and photoinduced electron-hole pairs can react directly with the dye as well as interact with O2 and H2O forming oxidizing radicals during the catalytic process.
Until now, many researchers have focused on the photocatalytic performance of ZnFe2O4; however, only a few reports have considered the photothermal catalytic properties of ZnFe2O4. It has been reported that oxygen vacancies can significantly improve the thermal catalytic performance of a catalyst, 18 because oxygen vacancies can change the phase of the material (such as electrical conductivity and material energy band structure) and the surface (surface composition, molecular adsorption, etc.) properties, which increases the thermal catalytic performance greatly.19,20 Therefore, endowing ZnFe2O4 with thermal catalytic properties by introducing oxygen vacancies should improve the performance of catalyst when driven by solar light.
In this work, we demonstrate the synthesis of ZnFe2O4 and its performance in photothermal catalytic degradation of the gaseous organic pollutant isopropanol (IPA). A large specific surface area and oxygen-vacancy-rich ZnFe2O4 with photothermal catalytic performance has been obtained. Also, defective ZnFe2O4 exhibits a synergic improvement of the photothermal catalytic degradation of IPA. We believe this study will promote applications of ZnFe2O4 in air purification.
Results and discussion
Thermogravimetry–differential scanning calorimetry analysis
The conversion of precursors of ZnFe2O4 was investigated by thermogravimetry–differential scanning calorimetry (TG-DSC). As shown in Figure 1, from 23 °C to 200 °C, the rate of weight loss is about 11.15%, which is due to water release from the hybrid of organic and inorganic materials. 21 From 200 °C to 400 °C, a rapid weight loss of up to 50.91% might be due to decomposition of the organic component of polyethylene glycol and the detachment of neutral water molecules existing in the crystal material lattice. 22 An obvious and intense exothermic peak from 300 °C to 400 °C corresponds to the decomposition of the organic component of polyethylene glycol in the DSC curve. 23 From 400 °C to 1000 °C, the weight loss rate is only 4.06%, indicating that the precursor had completely been converted into highly stable and crystalline ZnFe2O4. The optimum temperature for the synthesis of spinel ZnFe2O4 was 400 °C. 24

TG-DSC of ZnFe2O4 precursors in an air atmosphere.
X-ray diffraction analysis
The obtained ZnFe2O4 powder samples were characterized by X-ray diffraction (XRD; Figure 2). All diffraction patterns could be indexed to cubic spinel ZnFe2O4 (JCPDS, No. 22-1012).25,26 When the calcination temperature reached 400 °C, no impurity appeared because a high calcination temperature is beneficial to the diffusion of ions and accelerates the growth of ZnFe2O4 crystals. 27 According to Scherrer’s formula, calculated from the half-width of the (311) peak, the average crystal size is 18.3, 22.1, 51.2, and 85.3 nm for ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700, respectively. A large average crystal size indicates that the crystals of ZnFe2O4 grow well and that less boundaries or defects exist.

XRD patterns of ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700.
Scanning electron microscopy analysis
The morphology of the sample was observed by scanning electron microscopy (SEM). As shown in Figure 3, ZnFe2O4-400 is an agglomeration of irregular particles. There are many pores between the particles. With an increase in calcination temperature, the grain size of the nanoparticles becomes larger, which is consistent with the results of XRD. When the calcination temperature exceeds 700 °C, the grains reach about 100 nm. Figure 4 shows the element mapping and confirms the existence of Zn, Fe, and O and they are well-dispersed in ZnFe2O4.

SEM images of (a) ZnFe2O4-400, (b) ZnFe2O4-500, (c) ZnFe2O4-600, and (d) ZnFe2O4-700.
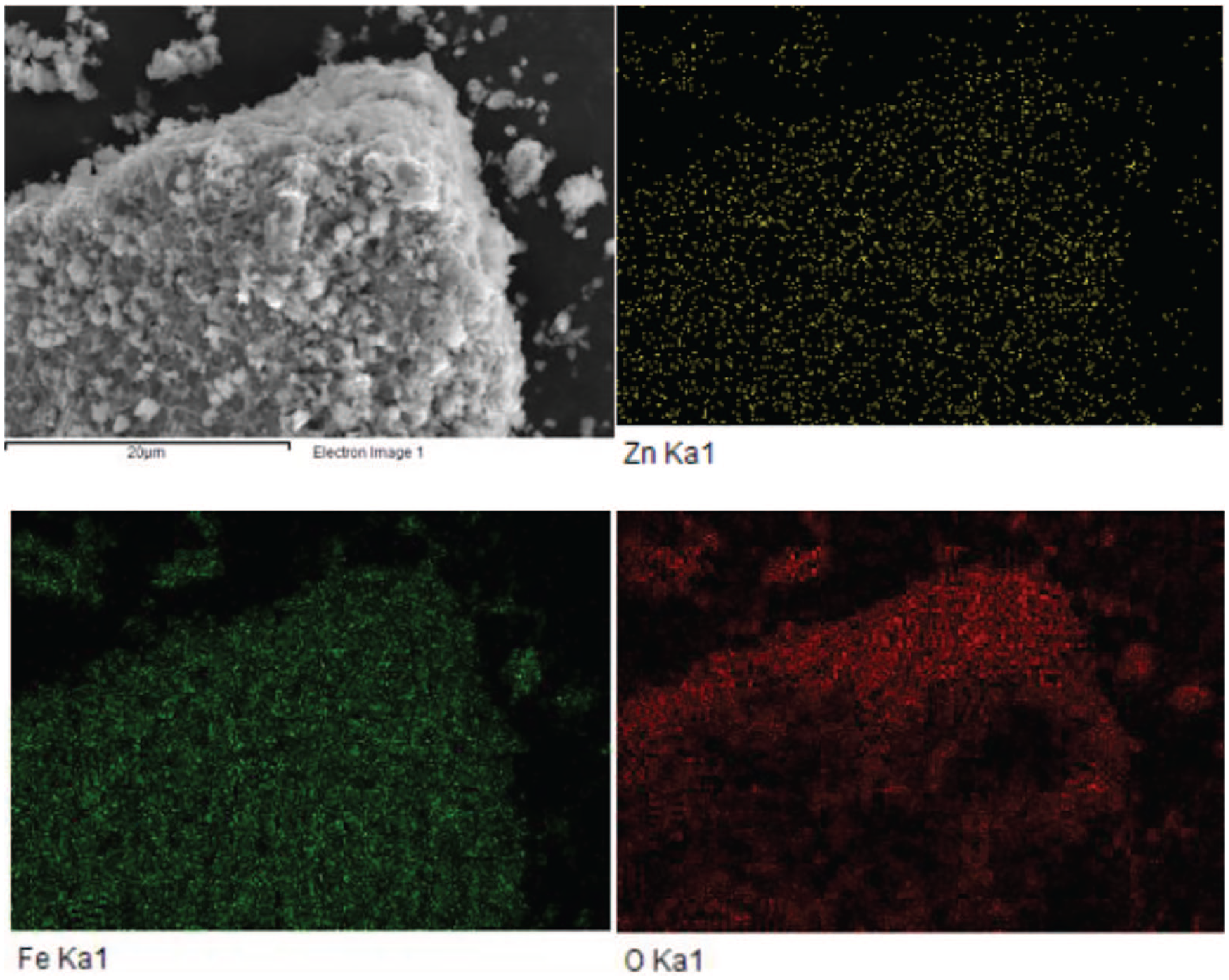
Element mapping of ZnFe2O4-400.
X-ray photoelectron spectra analysis
To study the chemical composition and element valence of ZnFe2O4, X-ray photoelectron spectra (XPS) analysis was performed. The survey spectra shown in Figure 5(a) suggest ZnFe2O4 is mainly composed of Zn, Fe, and O elements. 28 Figure 5(b) shows the O 1s spectrum which can be divided into three peaks located at 529.6, 530.3, and 531.56 eV, respectively. The peak at 529.6 eV corresponds to the lattice oxygen O2− in ZnFe2O4, including Zn-O and Fe-O, and the peak at 530.3 eV is from oxygen vacancies. 29 The peak at 531.56 eV is adsorbed oxygen from the water molecules.30–32 The formation mechanism of oxygen vacancies has been reported in several literatures. By using solution combustion synthesis with nitrate precursors, oxygen vacancy generation occurs because the reaction conditions result in oxygen being expelled from the reaction vessel. 33 By comparing three peaks of oxygen element, we found that the oxygen vacancies are appreciable. In Figure 5(c), the Zn high-resolution XPS spectrum, the peaks at 1020.92 and 1043.9 eV can be assigned to Zn 2p3/2 and Zn 2p1/2, confirming the presence of Zn2+.34,35 From Figure 5(d), the peak at 724.8 eV can be assigned to Fe 2p 1/2, and the peak of Fe 2p 3/2 can be divided into two peaks at 711.17 and 713.7 eV, while those at 711.17 and 713.7 eV corresponding to the existence of Fe3+.29,36 Based on the above analyses, it can be seem that there are numerous oxygen vacancies existing on the surface of the obtained ZnFe2O4.

(a) XPS survey spectra, and (b) O 1s, (c) Zn 2p, and (d) Fe 2p spectra of ZnFe2O4.
N2 adsorption and desorption analysis
The specific surface areas and pore volumes of the ZnFe2O4 samples were studied. As shown in Figure 6, there is a hysteresis loop constructed by type-IV isotherms, implying that a porous structure exists.37,38 The pore size distribution is shown in the inset in Figure 6. All the samples present a wide pore size distribution, indicating that the pores are mainly formed by the agglomeration of nanoparticles. The specific surface areas of ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700 are 56.4, 33.2, 14.4, and 8.2 m2g−1, respectively. With an increase in calcination temperature, the specific surface area of samples decreased; this phenomenon is caused by crystal growth leading to a reduction of the porosity.

The N2 adsorption–desorption isotherms and pore size distribution of the ZnFe2O4 samples.
Ultraviolet-visible diffuse reflectance spectra and electron paramagnetic resonance analysis
The ultraviolet (UV)-visible diffuse reflectance spectra (DRS; Figure 7) show that the obtained ZnFe2O4 samples have a broad spectrum of light absorption until to 650 nm. With an increase in calcination temperature, the absorption edge of ZnFe2O4 blueshifts due to a decrease in the number of oxygen vacancies.
39
Furthermore, the oxygen vacancies are also determined from the electron paramagnetic resonance (EPR) spectra, EPR was carried out at ambient temperature to evaluate the defects in samples, and the EPR spectra are shown in Figure 8. According to the formula

UV-vis diffuse reflectance spectra of ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700.

Ambient temperature EPR spectra for ZnFe2O4-400 and ZnFe2O4-600.
Thermal and photothermal catalytic performance
The catalytic performance under irradiation of Xe light was evaluated. As shown in Figure 9, the conversion rate increases with the time elapsed. After 120 min of the catalytic reaction, the conversion rates of IPA were 85.45%, 81.39%, 32.77%, and 4.98% for ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700, respectively. Because the light irradiation not only generated electron-hole pairs and plenty of heat, under our experimental conditions, the temperature of the samples under 300 W Xe light irradiation can reach 140 °C. In order to investigate the influence of thermal energy on the catalytic performance, the thermal catalytic capacity of ZnFe2O4 was studied by placing the catalytic reactor in a 140 °C oven without light irradiation. To eliminate the possibility of IPA self-degradation, a blank experiment without ZnFe2O4 was carried out. No degradation of IPA occurred spontaneously at 140 °C, so, it is believed that the degradation of IPA occurs due to the catalyst. As shown in Figure 10, over a 120 min catalytic reaction, the conversion rate of IPA tends to saturation and are 30.45%, 26.67%, 22.34%, and 1.33% for ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700, respectively. Compared with the thermal performance, the photothermal catalytic activity increases greatly, suggesting a synergistic enhancement of light and heat.

The conversion rate of IPA under visible light irradiation time elapsed.

The conversion rate of IPA at 140 °C.
In both photothermal and thermal catalytic reactions, ZnFe2O4-400 exhibits the highest activity among all the samples, which are 23.42 and 17.16 times higher than ZnFe2O4-700, respectively, and the catalytic activities reduced on raising the of calcination temperature. According to the specific surface area results, EPR studies, and UV-vis analysis, the higher calcination temperature can reduce the specific surface area and oxygen vacancy concentration. As is known, a large specific surface area can provide more active sites for catalytic reactions, and the surface oxygen vacancies also play a vital role in absorbing and activating O2 during the degradation of gaseous organic chemicals. 42
Thermal and photothermal catalytic mechanisms
On the defective surface, the thermal catalytic oxidation of the IPA is believed to conform to the Langmuir–Hinshelwood mechanism.43,44 The oxygen vacancies can act as the active sites for absorbing O2 to form active oxygen species
The role of oxygen vacancy in catalysis
Since ZnFe2O4 is a typical n-type semiconductor, the formation of oxygen vacancies causes the redistribution of excess electrons in its adjacent atoms, which is to produce an electron donor level below the ZnFe2O4 conduction band. The density functional theory (DFT) method was utilized to calculate the total density of states, observed from Figure 11(b), compared to ZnFe2O4 without oxygen vacancies; an electron donor level exists between the conduction band and the valence band for ZnFe2O4 with oxygen vacancies. These electron donor levels increase with an increase in the number of oxygen vacancies and sometimes overlap with the conducting band if the oxygen vacancy concentration is high enough.20,39,48 The electron donor level plays a vital role in reducing the bandgap width and the energy for electron transitions, indicating that the improved photoabsorption of ZnFe2O4 may be attributed to the existence of oxygen vacancies. 49

The density of states (DOS) of (a) ZnFe2O4 and (b) ZnFe2O4 with oxygen vacancies.
Moreover, oxygen vacancies can absorb oxygen molecules from air to form active oxygen, which is related to the thermal catalytic activity. The more oxygen on the surface of the sample, the higher the thermal catalytic activity. Therefore, in our experiment, the ZnFe2O4 oxygen vacancies exhibit high thermal catalytic activity.
Conclusion
ZnFe2O4 with oxygen vacancies are obtained via a soft-chemistry method. The obtained ZnFe2O4 samples exhibit high performance in the photothermal catalytic degradation of the gaseous organic pollutant IPA. ZnFe2O4-400 has a larger specific surface area and numerous oxygen vacancies, which are the reasons for its high photothermal catalytic performance. It combines the advantages of photocatalysis and thermal catalysis. In the process of photothermal catalytic degradation, oxygen vacancies on the surface of ZnFe2O4 facilitate the reaction being carried out as well as electron and hole separation. This study shows ZnFe2O4 is a potential material for organic matter degradation.
Experimental details
The synthesis of ZnFe2O4 powder
Ferric nitrate nonahydrate (4.03 g) and zinc acetate dihydrate (1.1 g) were dissolved in deionized water (100 mL) and stirred for 0.5 h. The resulting solution was treated with polyethylene glycol (2 g) and stirred for another 0.5 h. The solution was placed in an oven at 120 °C for 24 h. Finally, the obtained dry gel was calcined at 400 °C, 500 °C, 600 °C, and 700 °C for 2 h each with a heating rate of 3 °C/min in a muffle furnace. According to the different calcination temperatures, the obtained samples were named ZnFe2O4-400, ZnFe2O4-500, ZnFe2O4-600, and ZnFe2O4-700.
Characterization
X-ray diffraction (XRD) patterns of the powders were recorded at room temperature using a Bruker D8 Advance X-ray diffractometer. A scanning electron microscope (SEM) was used to characterize the morphology of the obtained products (S4800, Hitachi). EPR spectroscopy was accomplished using a CW-EPR Bruker ElEXSYS spectrometer. The UV-vis diffuse reflection spectra were obtained for dry-pressed disk samples using a Scan UV-vis spectrophotometer (UV-vis DRS UV-2450, Shimadzu). N2 adsorption was measured at 77 K on an Autosorb-1 (Quantachrome Instruments). Thermogravimetry-differential scanning calorimetry (TG-DSC) was carried out (Netzsch STA449) in a flow of air (20 mL/min) at a heating rate of 10 °C/min. X-ray photoelectron spectroscopy (XPS) measurements were performed on a Kratos XSAM 800 X-ray photoelectron spectroscope equipped with Mg-Kα radiation source operated at 200 W and all the binding energies were calibrated as the (C 1s) peak at 284.6 eV.
Measurements of thermal and photothermal catalytic activity
The thermal catalytic activity was measured by the oxidation of IPA in a closed system equipped with a quartz window. In brief, 0.2 g of powdered ZnFe2O4 was placed on a 2 × 2 cm2 glass sheet, and the glass with the sample was placed in a 325 cm3 glass reactor. Then 5 μL of liquid IPA was injected into the reactor through a gastight port. Finally, the reaction was placed in a dark oven at a temperature of at 140 °C. The generated acetone and IPA were detected by gas chromatography with FID detectors (GC1690, JieDao Tech. Ltd.). For the photothermal catalytic test, all conditions were the same as those of the thermal catalytic test, but the only difference was that the reactor was placed irradiation with a 300 W Xe arc lamp. The surface temperature of the sample was tested using a thermocouple.
Theoretical calculation
The electronic structure of the material was calculated by using the Vienna ab initio Simulation Package (VASP).50,51 ZnFe2O4 was calculated based on DFT. The projector-augmented wave method (PAW) was used in the calculations. The exchange-correlation energy used the generalized gradient approximation (GGA) of the Perdew–Burke–Ernzerhof (PBE) functional. The plane wave cutoff was set to 600 eV, and the Gaussian smearing width was limited to 0.05 eV. The 5.0 × 5.0 × 5.0 Monkhorst–Pack scheme was used for sampling of the Brillouin zone. ZnFe2O4 nanostructures were optimized until the Hellmann–Feynman forces were less than 0.01 eV/Å and the energy cutoff was converged within 1.0 × 10−5 eV. Spin polarization was applied in the calculations.
Footnotes
Data availability statements
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (21773099) and the Natural Science Foundation of Liaoning Province (20180550097). Xiaoxing Fan is indebted to the support from the Program of Liaoning Key Laboratory of Semiconductor Light Emitting and Photocatalytic Materials.