Abstract
In this article, the electronic, optical, and charge transfer properties of a Schiff base ligand prepared using 5-bromo-2-hydroxybenzaldehyde and ethyl 6-acetyl-2-amino-4,5,6,7-tetrahydrothieno[2,3-
Keywords

Introduction
Schiff base ligands have interesting electrochemical properties since they possess electron-donor or electron-attracting groups. In recent years, Schiff base metal complexes have received significant attention. Transition-metal complexes, especially Schiff base complexes, have emerged as promising candidates for the design and development of nonlinear optical (NLO) materials because of their efficient charge transfer behavior and ability to tailor metal-organic-ligand interactions.1,2 Transition-metal complexes are used as organic light-emitting diodes (OLEDs) because of their optoelectronic properties.3–7 Schiff bases and their transition metal complexes are also used as photometric materials in optical computers and display systems. 8 In particular, Ni(II), Pt(II), and Zn(II) complexes of Schiff bases have found widespread use in organic optoelectronics because of their NLO properties.9,10 In addition, metal complexes are used as hole-transport materials in solar cells.11–13 Both optical and hole-transport properties are important factors in OLEDs because the formation and propagation of light is directly influenced by the movement of electrons and holes in the material. 14
In recent studies, materials with high charge transfer rates have been designed for better OLED performance in the visible region.15–18 Also, theoretical calculations have become important in terms of the electronic, optical, and structural properties of complexes in comparison with experimental results.19–22
The ligand and its metal complexes that we use in this study have not been reported in the literature. In this study, the electronic and optical properties of the newly synthesized ligand and its metal complexes, which are organic semiconductors in various solvent environments (dichloromethane (DCM), dimethylformamide (DMF), chloroform and ethanol), have been investigated along with their suitability for optoelectronic applications. Both experimental techniques and theoretical methods such as density functional theory (DFT) are used to obtain the results. First, the theoretically most stable structure of the ligand and its Fe(II), Ni(II), and Zn(II) complexes was identified. The ultraviolet-visible (UV-Vis) spectra, frontier molecular orbitals (HOMO and LUMO), and the energy difference between these orbitals (
Results and discussion
UV spectra analysis in different solvents
UV-Vis spectroscopy is an important technique in terms of determining electronic, optical, solvatochromic, and charge transfer properties of compounds dissolved in various solvent environments. For the ligand, Fe(II), Ni(II), and Zn(II) complexes (Scheme 1), electronic absorption spectra were obtained in different solvent environments and are presented in Figure 1. The ligand and its metal complexes exhibited small and large peaks in the near ultraviolet (NUV), UV-A, UV-B, and visible regions in different solvents. The ligand exhibited maximum peaks in DCM and chloroform at 403 nm. In Figure 1, it can be seen that the Fe(II) complex exhibited maximum peaks at 403 nm in DCM and chloroform. Also, the Fe(II) complex did not show strong absorbance peaks in DMF and ethanol. The Ni(II) complex in DCM and chloroform gave maximum peaks at 404 nm, and in ethanol and DMF gave maximum peaks at 501 nm. The Zn(II) complex gave maximum peaks at 402 and 404 nm in DCM and chloroform, respectively, and in DMF gave a maximum peak at 339 nm. The Zn(II) complex did not exhibit strong absorbance peaks in ethanol. A solvent with a large dielectric constant leads to a greater stabilization energy for polar species. 24 The stabilizing effects of DMF (ε = 36.7) and ethanol (ε = 24.5) were greater than those of DCM (ε = 8.93) and chloroform (ε = 4.81). As a result, the absorbance values in DMF and ethanol were higher than those in DCM and chloroform. As can be seen from Figure 1, the wavelengths greater than 500–600 nm remained constant at the lowest values and in the visible region of the strongest peaks. In addition, the title compounds gave the same wavelength peaks in DCM and chloroform, while in DMF and ethanol, they gave different wavelength peaks.

Suggested structure of the ligand and its Fe(II), Ni(II), and Zn(II) complexes.

The absorbance spectra of the ligand and its Fe(II), Ni(II), and Zn(II) complexes in DCM, DMF, chloroform, and ethanol.
The experimental absorbance values of the ligand and complexes in chloroform gave peaks at almost the same maxima (see Figure 1). Theoretical absorbance calculations for all solvents used in the experimental measurements were made using the TD-DFT method and are presented in Figure 2. It can be seen that the absorbance values of the studied compounds did not change much with the solvent, similar to the experimental results. The ligand produced the maximum absorbance peak at approximately 411 nm, the Fe(II) complex at 535 and 541 nm, the Ni(II) complex at 393 and 493 nm, and the Zn(II) complex at 470 nm. In addition, it is seen from theoretical results that the ligand and its metal complexes give many peaks in the UV-Vis region.

The theoretical absorbance spectra of the ligand and its Fe(II), Ni(II), and Zn(II) complexes in ethanol.
The optical parameters of the ligand and its complexes in different solvents
We next investigated optical parameters such as the optical band gap (
The correlation between the
Figure 3 gives the experimental Tauc plots of the ligand and its metal complexes in DCM, DMF, chloroform, and ethanol. The
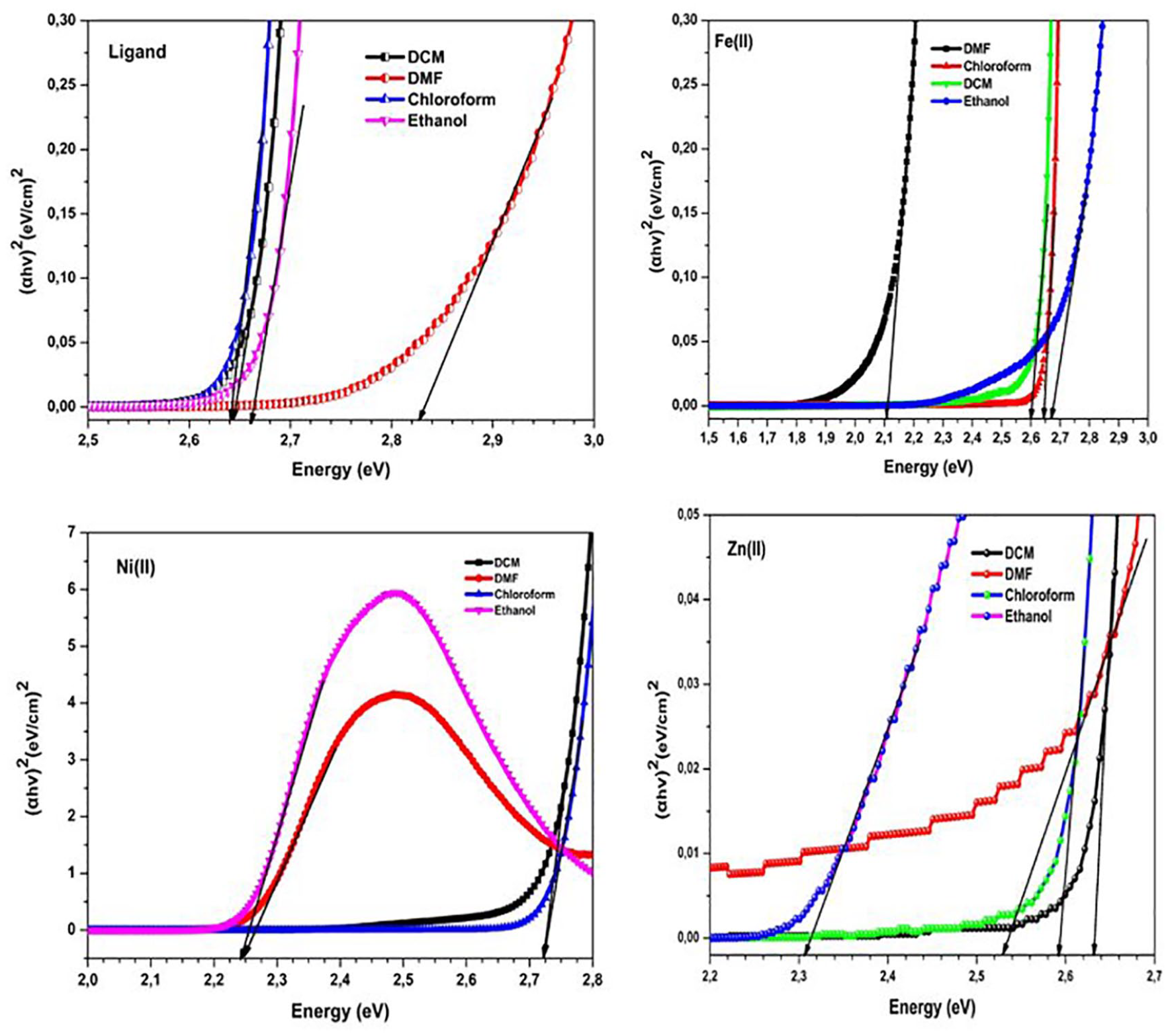
The (
The molecular orbitals (HOMO, LUMO) that play an important role in the transfer of electrical, optical, and molecular charge of a molecule were calculated using the TD-DFT/UB3LYP/LANL2DZ basis set for all the studied solvents and the results are presented in Table 1. The electrical band gap values shown in Table 1 define the energy difference between the HOMO and LUMO levels, and the optical band gap describes the excitation energy for transitions between vertical bands. 26 HOMO’s high energy value indicates that it is prone to donating electrons. Likewise, the low value of LUMO indicates its greater interest in accepting electrons.27,28 Therefore, the Ni(II) complex has the highest potential for electron donation in DCM and chloroform. The electrical band gap having values greater than the optical band gap is an expected result in charge transfer materials. 29 In addition, the LUMO energy level of the Ni(II) complex has the greatest value compared to the ligand and other complexes (see Table 1). This showed that Ni(II) had better electron-transfer properties. Similarly, Table 1 shows that the Fe(II) complex had the highest HOMO energy level indicating that the Fe(II) complex is the best transporting of holes.
The calculated energies values of the ligand and its Fe(II), Ni(II), and Zn(II) complexes.
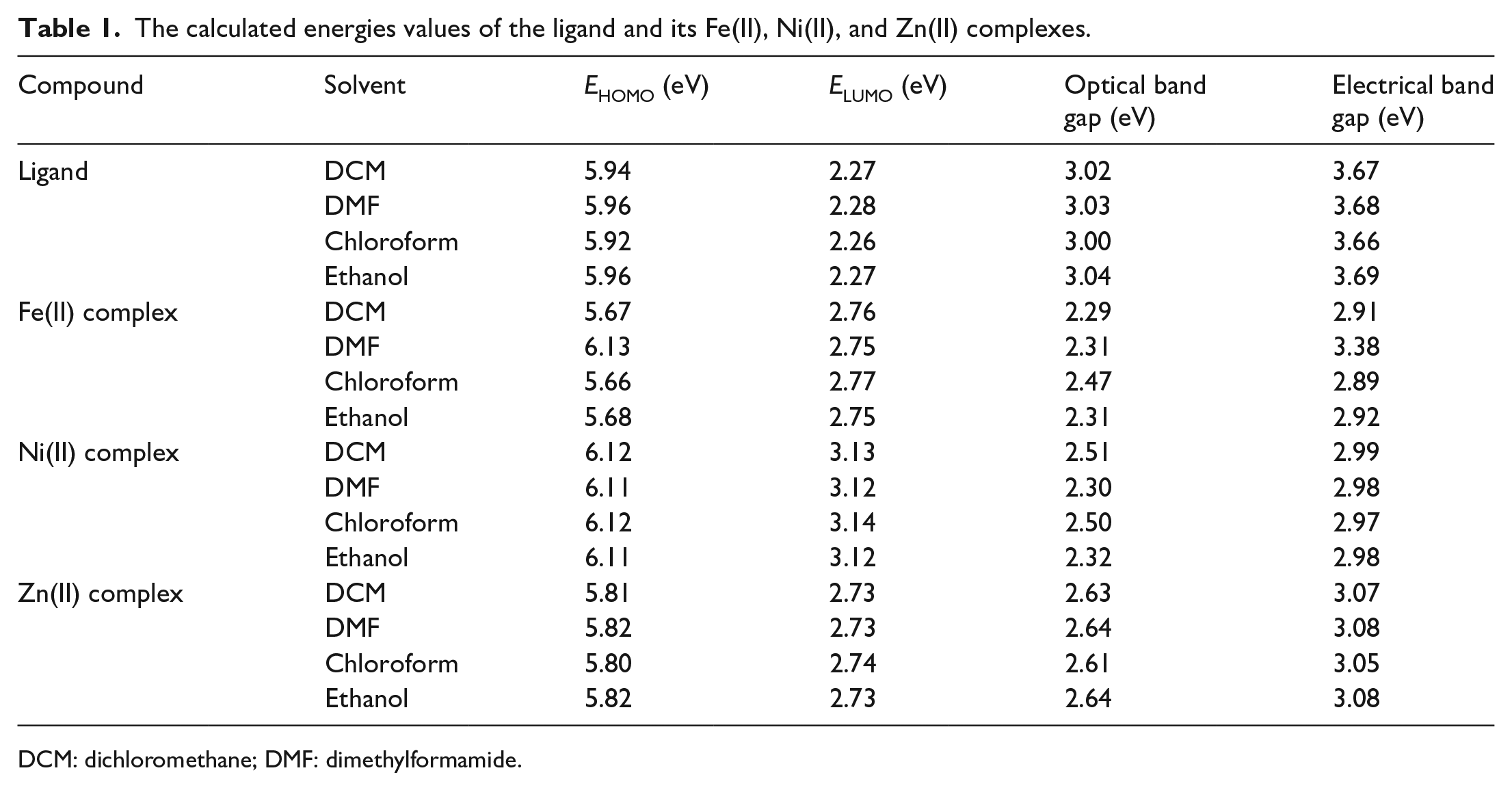
DCM: dichloromethane; DMF: dimethylformamide.
We calculated the refractive indices of the ligand and its metal complexes using equation 1 with the reflectance (
Refractive indices (

DCM: dichloromethane; DMF: dimethylformamide.

NLO analysis
NLO materials are widely utilized in areas such as laser technology, optical switching, photonic devices, optical sensors, screens, and data storage.30–32 Useful and reliable results are obtained in the search of new nonlinear materials through quantum computing approaches. In this respect, DFT-based methods are the best in terms of computation time and accuracy.
33
To investigate the electrical and optical responses of the ligand and its metal complexes, electrical dipole moment (µ), hyperpolarizability (




The calculated values are tabulated as Supplementary Information in Tables SI 3–6. For a molecule to be considered a good NLO material, the
Charge transport properties and reorganization energy values (λ)
The calculated reorganization energies (
The calculated reorganization energies (

Conclusion
In this work, the electronic, optical, and charge transfer properties of the synthesized compounds were explored using theoretical methods and compared with experimental data. The UV spectra of the ligand and the Fe(II), Ni(II), and Zn(II) complexes were studied in DCM, DMF, chloroform, and ethanol. The effects of these different solvents on the optical properties of the ligand and its Fe(II), Ni(II), and Zn(II) complexes have been studied in detail. From their optical band gap values and NLO properties in all the solvents used, it is apparent that the compounds studied herein are potential candidate compounds for OLEDs and NLO materials. Moreover, from the calculated reorganization energy, it can be seen that the ligand and Fe(II) complex are hole-transfer materials, while the Ni(II) and Zn(II) complexes are electron-transfer materials.
Experimental and theoretical details
All chemicals and solvents used for the synthesis and the measurements were of analytical reagent grade. The optical measurements were recorded using a Shimadzu UV-1800 Spectrophotometer in different solvents (DCM, DMF, chloroform, and ethanol) and at different molarities using the solution technique. Fourier transform infrared spectra were recorded on a Perkin Elmer 65 Spectrum One spectrometer in the range 400–4000 cm−1 as KBr disks. The Schiff base ligand and its metal complexes were prepared according to the method described in the literature. 28
All calculations were performed using the Gaussian 09 program
37
by employing DFT calculations based on the B3LYP change correlation, which is functional on the mixed basis of 6-311++G(d,p)+LanL2DZ.
38
The molecular structures of the Fe(II), Ni(II), and Zn(II) complexes were confirmed by elemental analysis. The complexes exist in octahedral geometry. The structures of the ligand and its metal complexes were optimized [DFT/B3LYP/6-311++G(d,p)] and are presented in Figure 4. The electronic, molecular orbital energies (HOMO and LUMO),
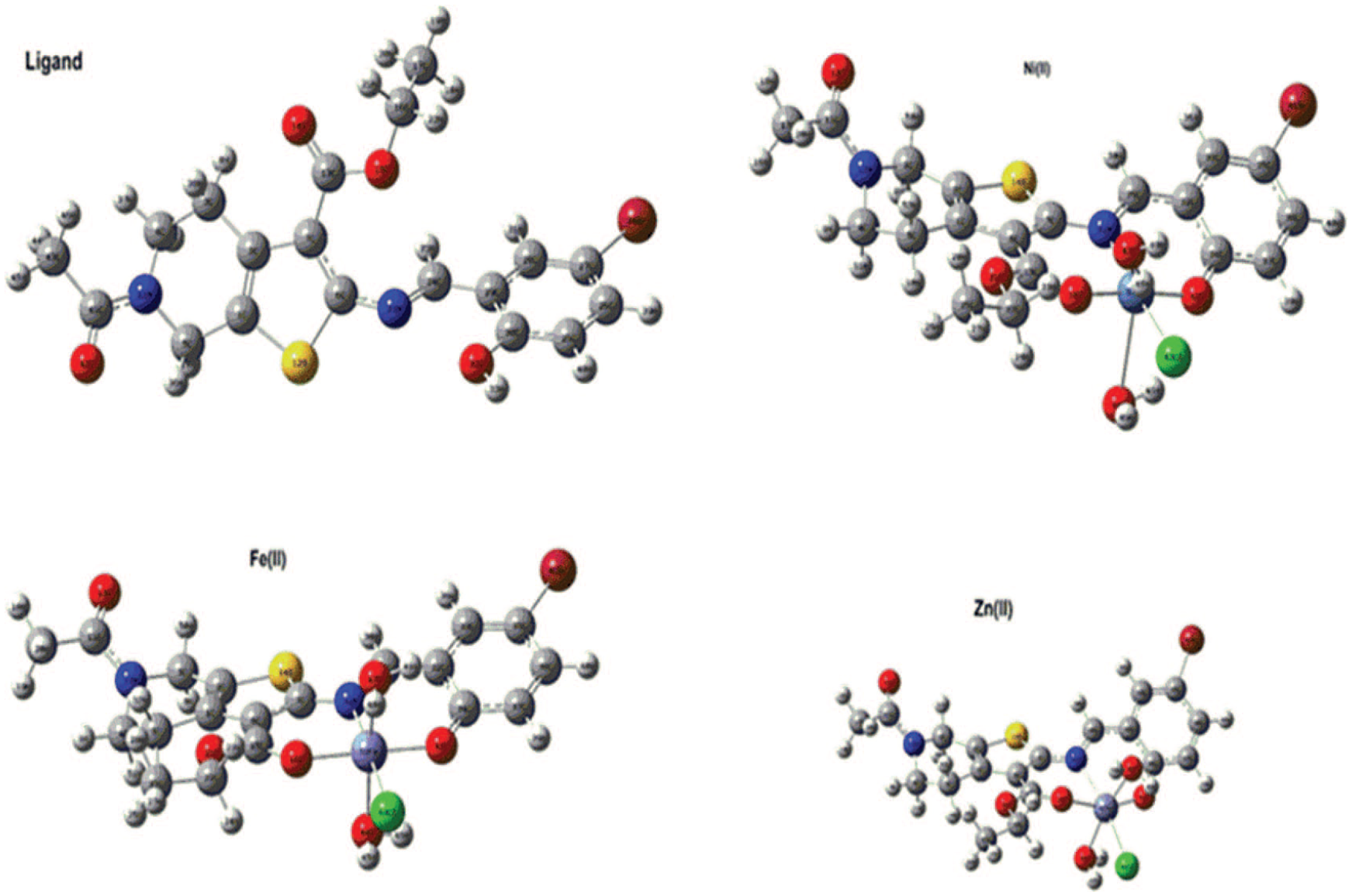
The optimized ground state structures of the ligand and the Fe(II), Ni(II), and Zn(II) complexes.
The reorganization energy consists of two parts: internal and outer contributions. The internal reorganization energy results from molecular geometric relaxation, while the outer reorganization energy is caused by the polarization of the surrounding compounds.
32
External organization energy is neglected by researchers because it is calculated as being too small for semiconductors.33,35 Here, we took into account the internal energy using the adiabatic potential energy surface method.33–36 The reorganization energy for holes (
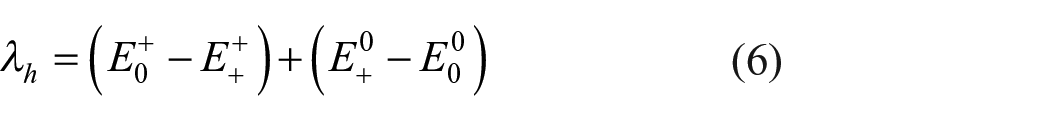
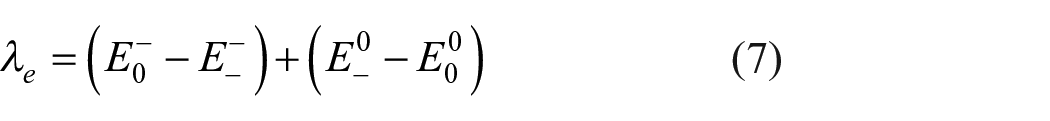
where
Supplemental Material
sj-pdf-1-chl-10.1177_1747519821995424 – Supplemental material for Solvent effects on the electronic and optical properties of Ni(II), Zn(II), and Fe(II) complexes of a Schiff base derived from 5-bromo-2-hydroxybenzaldehyde
Supplemental material, sj-pdf-1-chl-10.1177_1747519821995424 for Solvent effects on the electronic and optical properties of Ni(II), Zn(II), and Fe(II) complexes of a Schiff base derived from 5-bromo-2-hydroxybenzaldehyde by Kenan Buldurun, Emine Tanış, Nevin Turan, Naki Çolak and Nevin Çankaya in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
