Abstract
New aldehyde- and halogen- (Cl, Br, I) substituted double-armed benzo-15-crown-5 derivatives are synthesized by the reactions of 4′,5′-bis(bromomethyl)benzo-15-crown-5 with 5-chlorosalicylaldehyde, 5-bromosalicylaldehyde, or 5-iodosalicylaldehyde. The sodium and potassium complexes are obtained by reaction of crown ether with sodium perchlorate and potassium iodide, respectively. Novel Schiff base compounds containing three groups of benzo-15-crown-5 are obtained from the condensation of aldehydes with 4′-aminobenzo-15-crown-5. The structures of all compounds are elucidated by elemental analysis, 1H, 13C NMR, IR, and mass spectra. The antifungal and antibacterial effects of the synthesized ligands are evaluated against pathogenic microorganisms and show varying degrees of inhibitory effects against the growth of different pathogenic strains.
Graphical abstract
Keywords
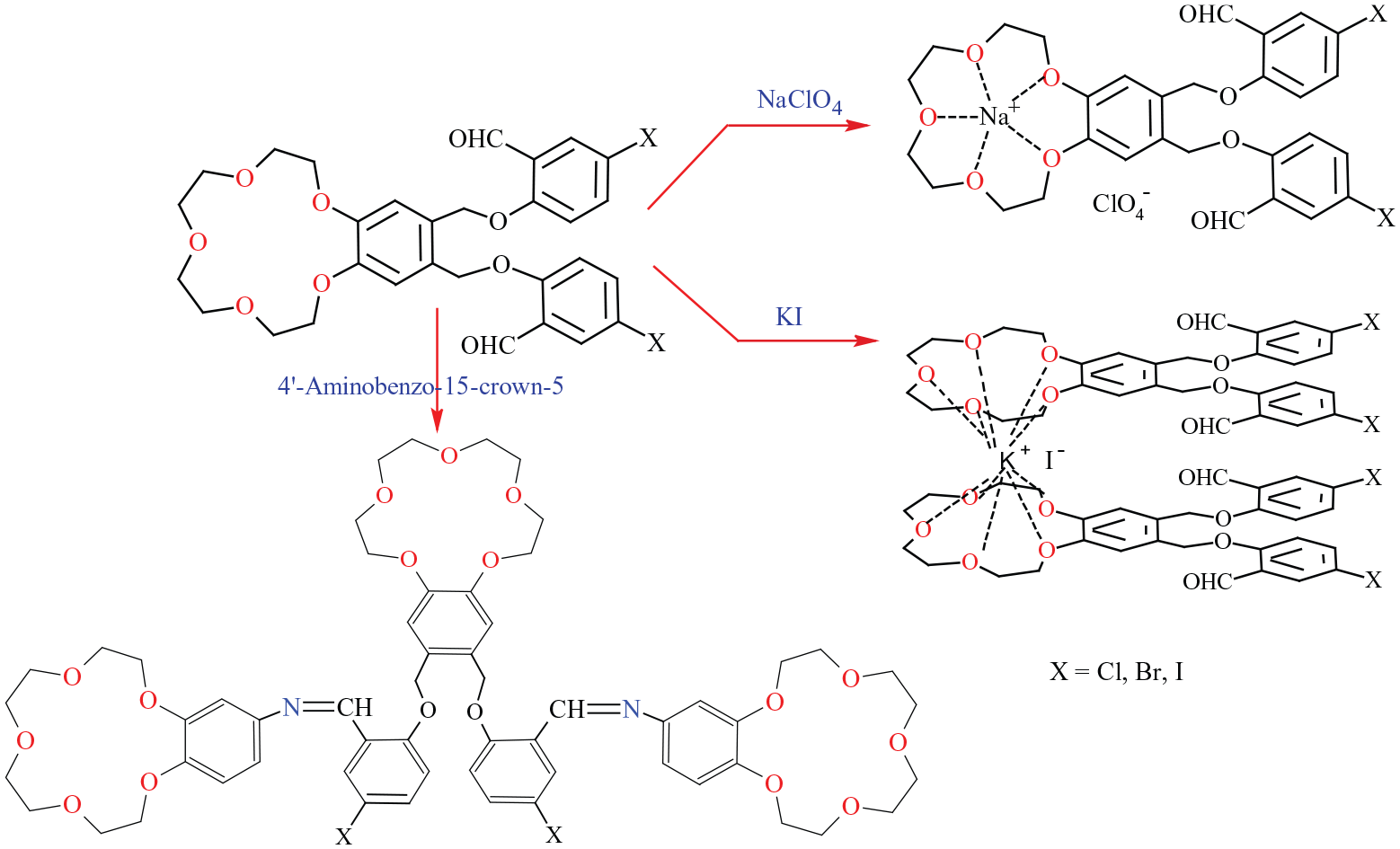
Introduction
Crown ethers and their complexes have been the subject of significant interest for more than 50 years. Numerous types of macrocyclic ligands and their complexes have been synthesized, including crown ethers.1–6 Structurally, crown ethers are cyclic structures having a cavity that allows them to form stable complexes with metal ions.7–11 In particular, bis-crown ethers have superior selectivity for metal cations. Bis-crown compounds form intramolecular sandwich-type complexes with large metal cations (such as Cs+, Sr2+, Tl+, Ba2+).2,3,12 The flexible linker between both crown ether moieties provides better extraction efficiency, that is, good coordination ability, which increases the selectivity for the cations in solution. There are also examples of substituted crown ethers having different selectivity centers.6,13–15 The crown ether center can generate stable complexes with alkali metal and alkaline earth metal cations, while the substituted side center can form transition metal complexes.12,16–23 Their complexing properties make crown ethers especially interesting and useful compounds in biological research and in chemistry.24–31 The ion complex–carrying ability of crown ethers results in them resembling natural ionophores (such as gramicidin). In this context, similar to natural ionophores, their ability to transport ions through lipid membranes by channeling has enabled the use of crown ethers in various biological applications. 27 In addition, bis-crown ether Schiff base derivatives and their sodium complexes have been shown to exhibit antibacterial activity against gram-positive and gram-negative bacteria. 32
Numerous examples can be found in the literature with bis-crown ether ligands and complexes, but up to now there have only been a few examples reported of tris-crown ethers.4,5 This prompted us to synthesize new ionophoric receptors containing more than one crown ether unit in their structure. Herein, we have designed and synthesized double-armed crown ethers possessing dialdehydes
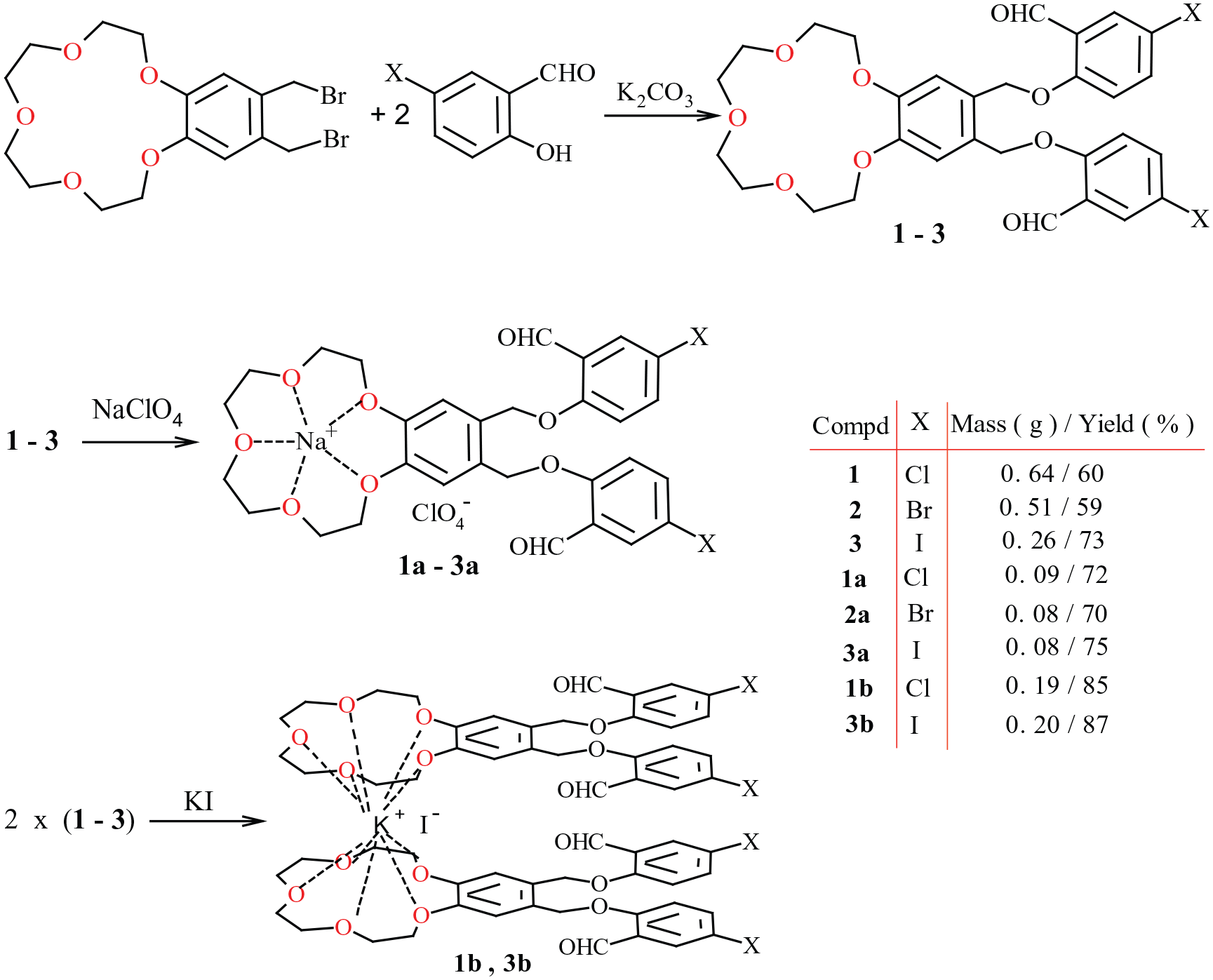
Synthesis of new crown ether aldehydes (
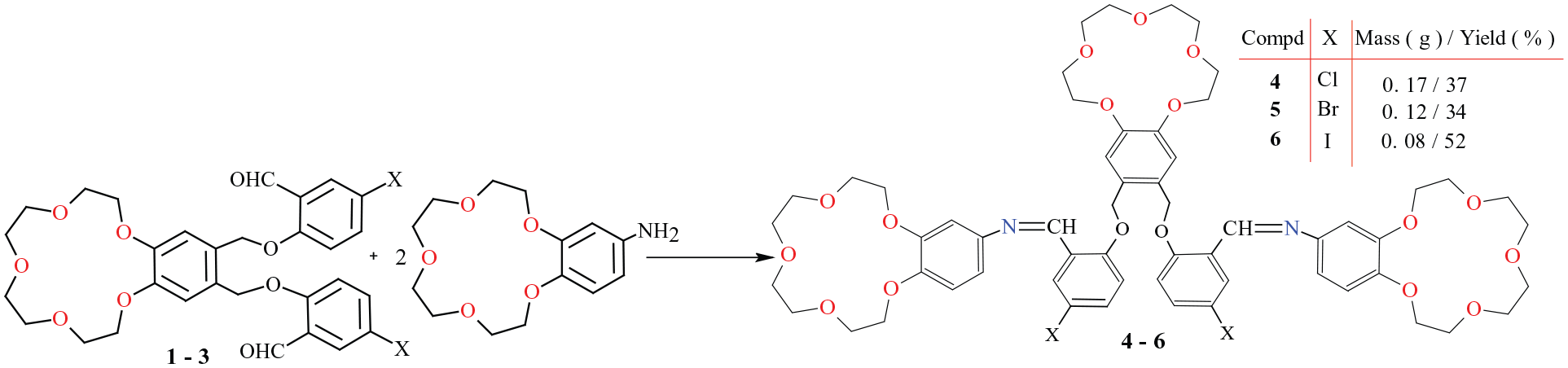
Synthesis of tris-crown ether Schiff bases (
Results and discussion
Syntheses and structural characterizations
New substituted crown ethers
The novel Schiff bases
The sodium
The structures of the solid crown ether ligands
Fourier-transform infrared spectroscopy spectra
The Fourier-transform infrared spectroscopy (FTIR) spectra of all the compounds displayed strong stretching bands between 1016 and 1145 cm−1 attributed to the (C–O–C) bond of the crown ether ring. The spectra of the crown aldehydes
1H and 13C NMR spectra
As expected, the structures of compounds

Numbering of the crown ether carbons and protons.
In the 1H NMR spectra of crown ether compounds

1H NMR spectral changes of the crown ether (–OCH2–CH2O–) and aromatic protons of free ligand
The 1H NMR spectra of tritopic Schiff bases

1H NMR spectrum of compound
In 13C NMR, the signals of the carbonyl carbons (C-15) in compounds
Due to the additional crown ring for compounds
In the 13C NMR spectra of the complexes
Mass spectra
The mass spectra of the synthesized crown aldehydes
The mass spectral data of crown ether aldehydes

The mass spectra of the synthesized alkali metal complexes
The mass spectral data of the sodium and potassium complexes

Antimicrobial activity
All the newly synthesized compounds

Biological activity of compounds
Salmonella typhi H, Sh. dysenteriae type 2 (Sh. dys. type 2), and S. epidermis are pathogenic microorganisms, and these microorganisms are gaining resistance to conventional antibiotics day by day.
37
Therefore, there is a need for new antibiotics effective against these pathogenic microorganisms. Compound
It is known that C. albicans showing pathogenicity in human fungal infections causes significant morbidity and mortality in immunocompromised patients (AIDS, cancer chemotherapy, organ, or bone transplantation).
38
All the synthesized compounds
Conclusion
In conclusion, a series of new crown ether compounds containing halogen (Cl, Br, or I), aldehyde, and imine groups have been synthesized. Aldehyde crown ethers
Experiment
Reagents and equipments
The starting materials, tetraethylene glycol dichloride, benzo-15-crown-5, and 4′,5′-bis(bromomethyl)benzo-15-crown-5 were prepared according to the literature.39–41 4′-Aminobenzo-15-crown-5, 5-chlorosalicylaldehyde, 5-bromosalicylaldehyde, and 5-iodosalicylaldehyde were purchased commercially from Aldrich and used without any further purification. All the solvents (dimethylformamide (DMF), THF, and EtOH) were purified and dried by standard procedures. The perchlorate salts of sodium complexes with organic ligands are potentially explosive and strong oxidizing, which means they help other materials burn. Due to the increased explosion potential, extreme caution should be exercised when working with perchlorate salts. The measurement of melting points was determined on a Gallenkamp melting point apparatus. The IR spectra were recorded using a Shimadzu Infinity model FTIR spectrometer equipped with an attenuated total reflectance (ATR) attachment. 1H and 13C NMR spectra were recorded with a Varian Mercury, high-performance Digital FT-NMR (400 MHz) spectrometer (chemical shift values are expressed in ppm, SiMe4 as an internal standard). Mass spectra were obtained using a Waters 2695 Alliance Micromass ZQ LC/MS spectrometer. The content of carbon, hydrogen, and nitrogen in all compounds was determined on a LECO CHNS-932 elemental analyzer.
Detection of antimicrobial activity
Staphylococcus aureus ATCC25923, Staphylococcus epidermis ATCC12228, Micrococcus luteus ATCC9341, Bacillus cereus RSKK-863, Listeria monocytogenes 4b ATCC19115, Salmonella typhi H NCTC901.8394, Enterobacter aerogenes sp., Escherichia coli ATCC1280, Sh. dysenteriae type 2 sp. were used as pathogenic bacterial cultures and C. albicans Y-1200-NIH was used a fungal agent. The antimicrobial activities of the synthesized compounds
The synthesized new compounds were dissolved (103 μM) in DMF (DMF was used as a solvent because it had no antimicrobial activity against any of the tested organisms). 1% (v/v) of the 24-h broth culture (pathogenic microorganisms) containing 106 CFU/mL was placed in sterile Petri dishes. Mueller-Hinton Agar (MHA) (15 mL), which was then maintained at 45 °C, was poured into a Petri dish and allowed to solidify slowly. The 6-mm-diameter wells were then carefully punched using a sterile cork borer and filled fully with the synthesized compounds. The plates were incubated for 24 h at 37°C. At the end of this period, the mean value obtained for the two wells was used to calculate the zone of growth inhibition of each compound. Pathogenic bacterial cultures and yeast were investigated for resistance to five antibiotics (ampicillin, nystatin, kanamycin, sulphamethoxazole, and amoxycillin) produced by Oxoid Ltd., Basingstoke, UK.
Synthesis of compounds
General method for the preparation of crown ether aldehydes 1–3
To the corresponding salicylaldehyde (5-chlorosalicylaldehyde, 5-bromosalicylaldehyde, or 5-iodosalicylaldehyde) (3.5 mmol) dissolved in DMF (20 mL), K2CO3 (3.5 mmol) was added, and the reaction mixture was left to stir overnight at room temperature. While stirring, a solution of 4′,5′-bis(bromomethyl)benzo-15-crown-5 (1.75 mmol) in DMF (20 mL) was added dropwise and then the reaction was left to stir for 36 h at 110°C under reflux. The consumption of the starting compounds was monitored by thin-layer chromatography (TLC) (silica, eluent; THF). Water was then added until the reaction solution became cloudy. The resulting precipitate was filtered off. The crude or pure product was recrystallized from THF/methanol (10:1).
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-chlorobenzaldehyde)
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-bromobenzaldehyde) 2: M.p.: 151 °C; IR (KBr) cm−1: 2937 (C–H)aliph, 1680 (C=O), 1589 (C=C), 1273–1234 (C–O–C)arom, 1122, 1064 (C–O–C)aliph; 1H NMR (400 MHz, DMSO-d6): δ = 3.61–4.09 (8H, m, –OCH2CH2O–), 5.31 (2H, s, Ph–CH2O–), 7.32 (1H, s, H-6), 7.37 (1H, d, J = 8.9 Hz, H-14), 7.71 (1H, d, J = 2.8 Hz, H-11), 7.79 (1H, dd, J = 8.9 Hz, J = 2.8 Hz, H-13), 10.12 (1H, s, CHO); 13C NMR (400 MHz, DMSO-d6): δ = 68.7 (C-8), 69.0, 69.1, 70.0, 70.7 (C-1-4), 113.0 (C-6), 115.7 (C-12), 117.2 (C-14), 126.4 (C-10), 127.6 (C-7), 130.7 (C-13), 138.7 (C-11), 148.6 (C-5), 160.0 (C-9), 188.3 (C-15); Anal. calcd for C30H30Br2O9 (694.36): C, 51.89; H, 4.35; found: C, 51.52; H, 4.10.
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-iodobenzaldehyde) 3: M.p.: 156 °C; IR (KBr) cm−1: 2921 (C–H)aliph, 1676 (C=O), 1583 (C=C), 1271–1234 (C–O–C)arom, 1118, 1045 (C–O–C)aliph; 1H NMR (400 MHz, CDCl3): δ = 3.75–4.16 (8H, m, –OCH2CH2O–), 5.15 (2H, s, Ph–CH2O–), 6.79 (1H, d, J = 8.8 Hz, H-14), 7.00 (1H, s, H-6), 7.77 (1H, dd, J = 8.8 Hz, J = 2.7 Hz, H-13), 8.08 (1H, d, J = 2.7 Hz, H-11), 10.26 (1H, s, –CHO); 13C NMR (400 MHz, CDCl3): δ = 68.6 (C-8), 69.2, 69.4, 70.4, 71.1 (C-1-4), 83.7 (C-12), 110.0 (C-10), 115.3 (C-14), 115.4 (C-6), 126.7 (C-7), 137.7 (C-11), 144.2 (C-13), 149.4 (C-5), 160.1 (C-9), 187.7 (C-15); Anal. calcd for C30H30I2O9 (788.36): C, 45.71; H, 3.84; found: C, 45.34; H, 3.74.
General method for the preparation of sodium complexes 1a–3a
To a stirred solution of crown ether aldehydes (1–
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-chlorobenzaldehyde) sodium complex (
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-bromobenzaldehyde) sodium complex (
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-iodobenzaldehyde) sodium complex (
General method for the preparation of potassium complexes 1b and 3b
Crown ether aldehyde
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-chlorobenzaldehyde) potassium complex (
2,2′-[2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis(methyleneoxy)]bis(5-iodobenzaldehyde) potassium complex (
General method for the preparation of Schiff bases 4–6
To a solution of the corresponding crown ether aldehyde
N,N′-{2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzo-pentaoxacyclopentadecine-15,16-diylbis[methyleneoxy(5-chloro-2,1-phenylene)(Z)methylylidene]}bis(2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecin-15-amine)
N,N′-{2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine-15,16-diylbis[methyleneoxy(5-bromo-2,1-phenylene)(Z)methylylidene]}bis(2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecin-15-amine)
N,N′-{2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benz-opentaoxacyclopentadecine-15,16-diylbis[methyleneoxy(5-iodo-2,1-phenylene)(Z)methylylidene]}bis(2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecin-15-amine)
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially assisted by the Scientific and Technical Research Council of Turkey (TUBITAK), grant no. TBAG 210T122, and Ankara University, grant no. 17B0430004.
