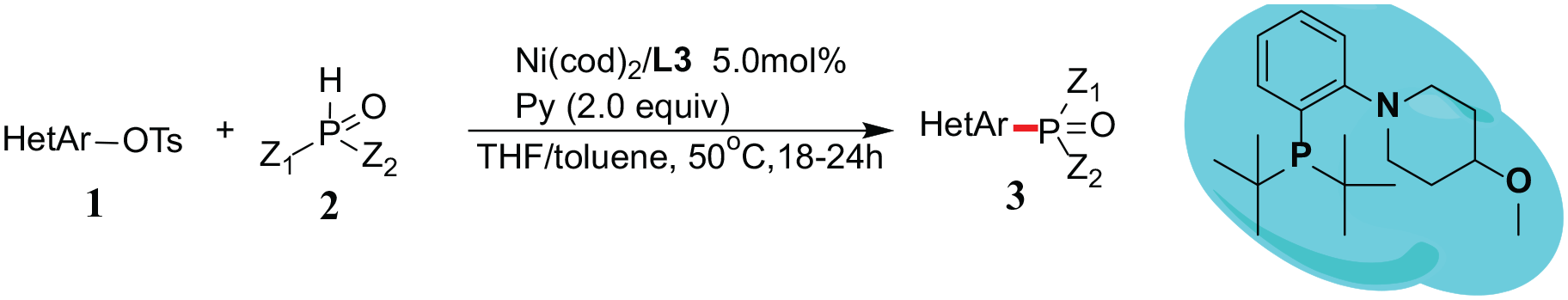Abstract
A novel and convenient approach to the synthesis of various tertiary phosphine oxides via nickel-catalyzed cross-coupling of (het)aromatic tosylates with secondary phosphine oxides is developed. The reaction employs cheap nickel as the catalyst, 1-(2-(di-tert-butylphosphanyl)phenyl)-4-methoxypiperidine

Introduction
The development of efficient methods for preparing aryl phosphorus compounds is of great importance because of their wide application in medicinal chemistry, materials chemistry, and organic synthesis,1,2 and their arylphosphine derivatives play an important role in organometallic catalysis3,4 and organocatalysis.5,6 Following on from the pioneering work of the Hirao group in this area, 7 after further development and modification, researchers have developed the palladium-, nickel-, and copper-catalyzed processes for the construction of C(sp)2-P bonds (Scheme 1, a).8–10 Recently, oxidative phosphonylation of arylboronic acids catalyzed by palladium or nickel has become feasible (Scheme 1, b).11,12 Diaryliodonium salts, 13 as important and valuable electrophilic arylation reagents, have attracted significant attention in recent years due to their high reactivity and nontoxicity and they have been found to serve as potential arylating agents for phosphorus nucleophiles (Scheme 1, c). The last two decades have witnessed the rapid development of bismuth chemistry. Organobismuth compounds are nontoxic and are easily available building blocks among the heavy nonradioactive main group elements.14,15 Recently, Zhao and co-workers described the first examples of reactions of triarylbismuths with P(O)-H compounds under Pd(0) catalysis (Scheme 1, c). 16
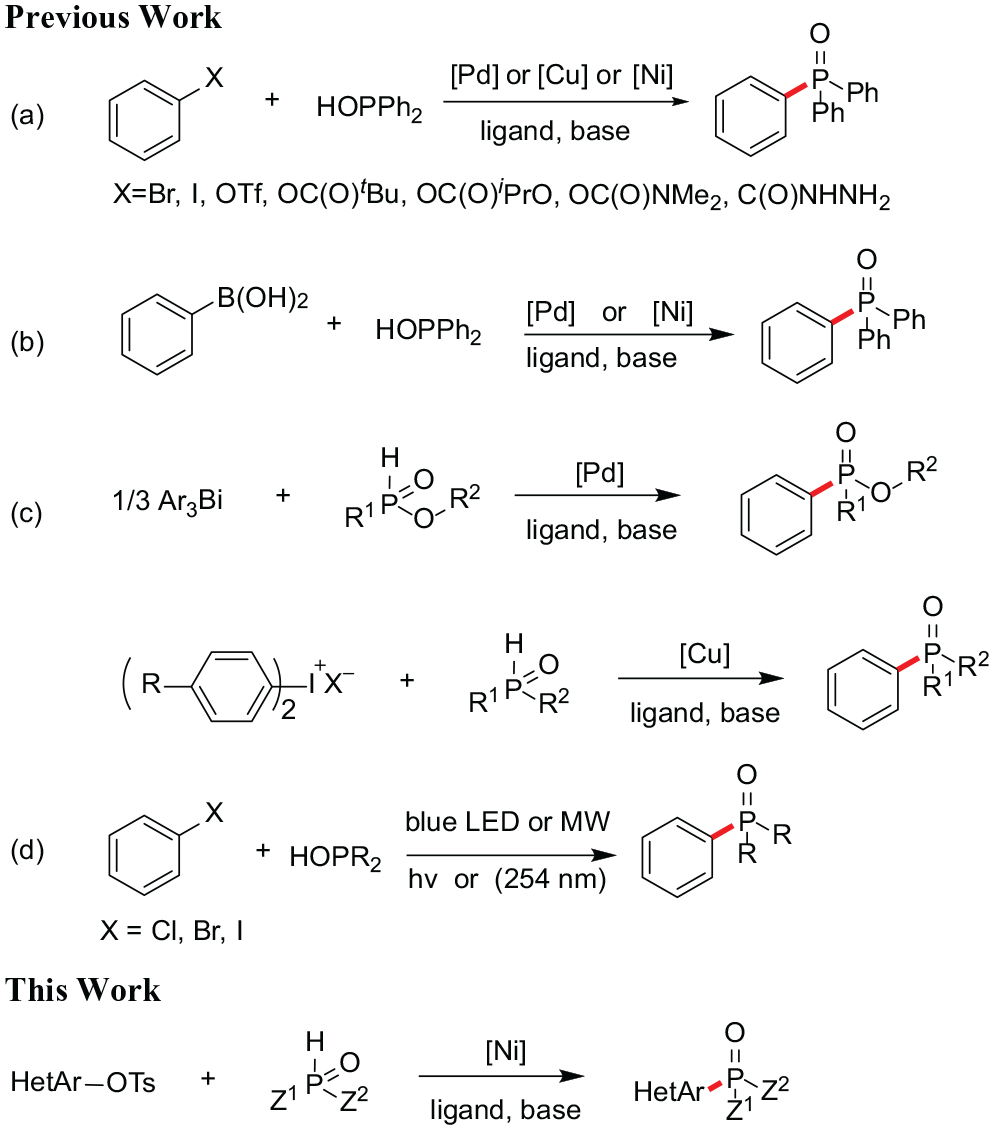
Methods for the synthesis of tertiary phosphine oxides.
Furthermore, microwave-(MW) and visible-light-irradiated P-C coupling reactions have also been developed (Scheme 1, d).17–19 To date, very few examples of nickel-catalyzed phosphinations of aryl mesylates and tosylates have been reported. In 2012, Zhang’s group reported the first Ni-catalyzed P-arylation using aryl mesylates and tosylates as coupling partners in this field, greatly expanding the scope of transition-metal-catalyzed C-P couplings.
20
Yu’s group has reported a novel phosphorylation of alkenyl and aryl C–O bonds via photoredox/nickel dual catalysis.
21
A variety of easily available and inexpensive sulfonates could be transformed into alkenyl phosphonates and aryl phosphine oxides with high selectivity and efficiency under mild reaction conditions. Very recently, Li reported the nickel-catalyzed phosphorylation of aryl tosylates; however, the use of an appropriate phosphine ligand was crucial for this reaction.
22
Ten significant ligands were then evaluated for their efficacy in this C–P bond coupling reaction, with the bidentate phosphine ligand 2-(di-tert-butylphosphino)-4-methoxyl-N,N-dimethylaniline
Optimization of the reaction conditions. a

Unless otherwise stated, the reaction conditions are as follows: Heteroaromatic tosylate
Stirred at 80 °C.
Yield of isolated product.
[Ni]/ligand (3.0%).
No reaction.
No base was used.
Stirred at 50 °C.
Py (2.0 equiv.).
Py (2.0 equiv.), reaction time was 18 h.
Results and discussion
Initially, when
We, in Li’s report, have described recently the application of L1 as a catalyst ligand for the cross-coupling of aryltosylate and dialkyl phosphite, despite the reactivity is modest in our system, L1 was shown to be among the most active ligands in their report. Encouraged by the catalytic abilities of L1, and the needs of our current research, we have designed several new ligands. Next, a subsequent screening of the role of different ligands to gain more insight and to find a more active and robust system was undertaken (Table 1, entries 1 and 10–17). Notably, in the course of these investigations, most of the ligands examined gave fairly poor results, with only bidentate ligand
Thus, the optimized experimental conditions are as follows: Ni(cod)2/
Cross-coupling of various heteroaromatic tosylates with disubstituted phosphine oxide. a

Reaction conditions: heteroaromatic tosylate
Yield of the isolated product.
Py (3.5 equiv.).
However, secondary phosphine oxides with strong electron-withdrawing groups, such as fluoro, resulted in a lower yield. These results indicate that the yields were dependent primarily on the electronic properties of the secondary phosphine oxides (Table 2, entry 11). Secondary phosphine oxides with sterically demanding o-substituted aromatic rings afforded inferior yields, illustrating that steric hindrance has an influence on this coupling reaction (Table 2, entry 9).
We also tested our cross-coupling procedure for the assembly of a bis(heterocyclic) product. A 2-hydroxybenzothiazolyl tosylate readily underwent coupling with dithiophenylphosphine oxide to provide a bis(heterocyclic) compound (Table 2, entry 13). Other hydrogen phosphoryl compounds were also applicable in this transformation. In addition to diarylphosphine oxides
Conclusion
In summary, we have developed a novel nickel-catalyzed carbon-phosphine cross-coupling protocol from a wide range of (het)aromatic tosylates and different secondary phosphine oxides. Notably, the process is simple and proceeds under mild reaction conditions. Moreover, the process is generally cheaper overall because more accessible (het)aryl phenol derivatives are used to form the C-P bond. These advantages should help this C-P bond-forming method to find broad application in both complex molecule synthesis and for the preparation of P-chiral organophosphorus compounds.
Experimental
General
Ni(cod)2 (98%), Ni(acac)2 (>98%), NiCl2(PCy3)2 (>98%), NiCl2(dppe) (98%), NiCl2(dcype) (>98%), NiCl2(dppf) (99%), NiCl2(PPh3)2 (>98%), Pd2dba3, Pd(OAc)2, and DiPPF were purchased from Aldrich. Ni(OAc)2·4H2O, NiCl2, racemic-2,2’-Bis(diphenylphosphino)-1,1’-binaphthyl (BINAP), and Tetramethylethylenediamine (TMEDA) (⩾99%) were purchased from Alfa Aesar. Other reagents were available commercially and were used without further purification, unless otherwise indicated. All aryl tosylates were prepared according to the literature procedures.25–29 Ligands
Experimental section
General procedure A: ligand synthesis and characterization
A Schlenk tube was charged with reagents: Pd2dba3 (46 mg, 5 mol% Pd), BINAP (93 mg, 7.5 mol%), NaOtBu (231 mg, 2.4 mmol, 1.2 equiv.), 1,2-dibromobenzene
To a Schlenk tube, Pd(OAc)2 (typically 3 mol%) and DiPPF [1,1′-bis(diisopropylphosphino)ferrocene; Pd:L = 1:1.2], di(cyclohexyl)phosphine (1.2 mmol), and NaOtBu (1.44 mmol) were added, followed by the aryl halide
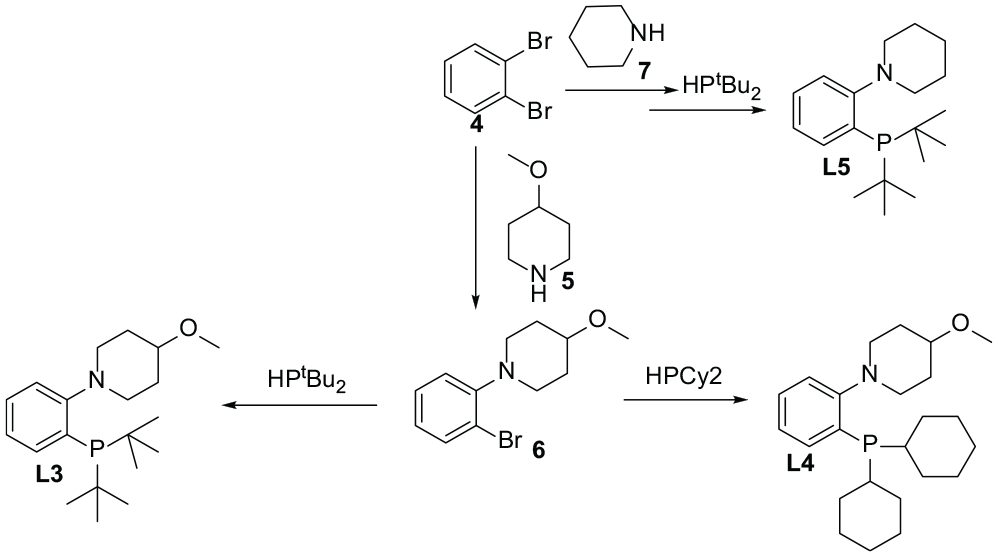
Synthesis of ligands
1-(2-(di-tert-butylphosphanyl)phenyl)-4-methoxypiperidine (
(HRMS) (EI): m/z [M]+ calcd for C20H34NOP: 335.2378; found: 335.2373. Anal. calcd for C20H34NOP: C, 71.61; H, 10.22; N, 4.18; found: C, 71.73; H, 10.29; N, 4.31.
1-(2-Dicyclohexylphosphinophenyl)-4-methoxypiperidine
(HRMS) (EI): m/z [M]+ calcd for C24H38NOP: 387.2691; found: 387.2687. Anal. calcd for C24H38NOP: C, 74.38; H, 9.88; N, 3.61; found: C, 74.43; H, 9.93; N, 3.72.
1-(2-(di-tert-butylphosphanyl)phenyl)piperidine (
(HRMS) (EI): m/z [M]+ calcd for C19H32NP: 305.2272; found: 305.2276. Anal. calcd for C19H32NP: C, 74.71; H, 10.56; N, 4.59; found: C, 74.75; H, 10.59; N, 4.62.
General procedure B: phosphination of (Het)aromatic tosylates with secondary phosphine oxides
In an argon-filled glovebox, an oven-dried Schlenk tube equipped with a Teflon stir bar was charged with Ni(cod)2/
di-tert-butyl(pyridin-3-yl)phosphine oxide
di-tert-butyl(thiophen-2-yl)phosphine oxide
(HRMS) (EI): m/z [M]+ calcd for C12H21OPS: 244.1051; found: 244.1046. Anal. calcd for C12H21OPS: C, 58.99; H, 8.66; S, 13.12; found: C, 59.01; H, 8.71; S, 13.16.
di-tert-butyl(quinolin-8-yl)phosphine oxide
(HRMS) (EI): m/z [M]+ calcd for C17H24NOP: 289.1596; found: 289.1593. Anal. calcd for C17H24NOP: C, 70.57; H, 8.36; N, 4.84; found: C, 70.63; H, 8.41; N, 4.89.
di-tert-butyl(quinolin-6-yl)phosphine oxide
(HRMS) (EI): m/z [M]+ calcd for C17H24NOP: 289.1596; found: 289.1591. Anal. calcd for C17H24NOP: C, 70.57; H, 8.36; N, 4.84; found: C, 70.64; H, 8.43; N, 4.88.
Supplemental Material
sj-pdf-1-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-1-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Supplemental Material
sj-pdf-2-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-2-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Supplemental Material
sj-pdf-3-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-3-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Supplemental Material
sj-pdf-4-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-4-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Supplemental Material
sj-pdf-5-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-5-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Supplemental Material
sj-pdf-6-chl-10.1177_1747519821994533 – Supplemental material for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides
Supplemental material, sj-pdf-6-chl-10.1177_1747519821994533 for Nickel-catalyzed C–P cross-coupling of (het)aryl tosylates with secondary phosphine oxides by Xiao-Yun He in Journal of Chemical Research
Footnotes
Acknowledgements
The author is thankful to Hebei University of Science and Technology for 13C NMR, 1H NMR, 31P NMR, HRMS(EI), and elemental analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author gratefully acknowledges the financial support from Hebei Chemical and Pharmaceutical College and the Higher Education Scientific Research Project of Hebei Province (2020) (SQ201044).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.