Abstract
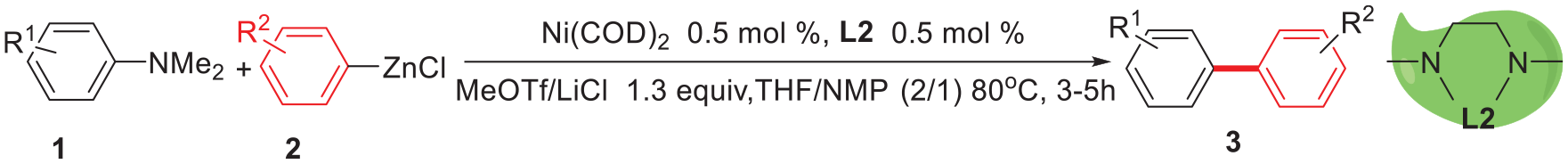
A nickel-catalyzed arylation of aromatic amines using arylzinc reagents with trimethylammonium salts formed in situ is developed. Compared with previous systems using ammonium salts as the starting materials and complexes of pre-prepared nickel catalysts, this reaction directly employed an N,N-dimethyl(het)arylamine as the coupling partner and commercial Ni(COD)2/TMEDA as the catalyst, greatly simplifying the experimental procedure, decreasing the cost, and being especially suitable for scale-up production processes.

Introduction
Transition metal–catalyzed cross-coupling reactions have become an extremely versatile tool in organic synthesis for the connection of electrophilic and organometallic fragments by the formation of either carbon-carbon or carbon-heteroatom bonds. 1 Examples include Kumada, Negishi, Suzuki, and Stille cross-couplings and, among these coupling reactions, the Negishi coupling is one of the most powerful owing to the widespread availability and high functional group tolerance of organozinc reagents.2,3 In addition to aryl iodides, bromides, and chlorides, 4 more recently, triflates and other O-based substrates, such as tosylates, mesylates, and carboxylates, are useful alternative electrophiles for cross-coupling reactions.5,6 Reactions through C–N bond cleavage of the electrophilic substrates are scarce, although Wenkert and co-workers carried out the nickel-catalyzed reaction of aryltrimethylammonium iodides with Grignard reagents in the early stage of cross-coupling studies. 7 On the other hand, nitrogen-containing compounds such as aryl- and alkyl-amines are very important and widely available in the natural world and in industry. A tactic is to convert arylamines to triazenes 8 or arylammonium salts 9 to weaken the C–N bonds before cleavage. Wang and other groups have performed some transition metal–catalyzed cross-coupling reactions using aryltrimethylammonium salts as the electrophilic partners in the past few years. 10 Recently, Wang and co-workers developed a pincer nickel complex–catalyzed Negishi coupling reaction of aryltrimethylammonium triflate with arylzinc reagents (Scheme 1). Despite the fact that long reaction times were required, this reaction provided an efficient method for using tertiary aromatic amines in aromatic couplings for the first time.

Ni-catalyzed arylation of arylammonium.
We recently became interested in using aryltrimethylammonium salts in cross-coupling reactions with organozinc reagents. As aryltrimethylammonium triflates were prepared by reactions of aromatic amines, lithium chloride, and Methyl trifluoromethanesulfonate (MeOTf), we assumed that the aromatic amines might be used directly as the starting materials, if the quaternization process would take place in situ.
11
Herein, we report such an arylation reaction in one step using aromatic amines (Scheme 1). It is worth mentioning that it was also not necessary to pre-synthesize the catalyst as the common Ni(COD)2/
Results and discussion
In this communication, we report our findings on the subsequent development of conditions which allow, for the first time, nickel-catalyzed cross-couplings using aromatic amines and arylzinc reagents. By heating a mixture of 4-(dimethylamino) toluene (
Optimization of the reaction conditions a .

NMP: N-methylpyrrolidone, THF: tetrahydrofuran.
Reaction conditions:
We next evaluated the substrate scope under the optimized conditions. The reaction of various aromatic amines with p-methoxyphenylzinc reagent was first examined (Table 2). Electron-deficient aromatic amines such as p-fluoro- and p-trifluoro-N,N-dimethylaniline exhibited high reactivity in the reactions with the p-methoxyphenylzinc reagent, affording the corresponding products in excellent yields (Table 2, entries
Coupling of various arylammonium with arylzinc reagentsab.

Reaction conditions: 1a (0.2 mmol), 2a (1.5 equiv.), MeOTf/LiCl (1.3 equiv.), catalyst 0.5 mol%, ligand

A gram scale of reaction.
Conclusion
In conclusion, the reaction reported here is applicable to simple (het)arylammonium salts prepared in situ as the coupling partners, avoiding the synthesis and isolation of these ammonium salts. In addition, commercially available Ni(COD)2 was used as the catalyst instead of pre-prepared Ni complexes, making the experimental procedure simpler, and excellent for laboratory and large-scale synthesis. Finally, the reaction provides an alternative method converting carbon-nitrogen bonds into carbon-carbon bonds.
Experimental
General information
Ni(acac)2, Ni(cod)2, NiCl2(dppf), NiCl2(dppp), and all ligands were purchased from Aldrich. NiCl2, LiCl, and Ni(Pph3)2Cl2 were purchased from Alfa Aesar. Other reagents are available commercially and were used without further purification, unless otherwise indicated. All reactions were carried out under an argon atmosphere with dry solvents under anhydrous conditions, unless otherwise noted. THF was dried over alumina under N2 using a Grubbs-type solvent purification system. All arylzinc chlorides were prepared from the corresponding arylmagnesium bromides and ZnCl2. All aromatic amines were purchased from Alfa Aesar. Spectroscopic data of the known compounds matched the published data. Reactions were monitored by Agilent GC Series 6890N and GCMS 7890A GC. All compounds were characterized by 1H NMR spectra measured on a Bruker 400 M spectrometer. 1H NMR were recorded using tetramethylsilane (TMS) in deuterated chloroform (CDCl3) using deuterium oxide as the internal standard. Chemical shifts are reported in parts per million (d). The peak patterns are indicated as follows: s, singlet; d, doublet; t, triplet; dd, doublet of doublets; dt, doublet of triplets; and m, multiplet. The coupling constant, J, is reported in Hertz (Hz). The products were purified by column chromatography on silica gel 300–400 mesh under an argon atmosphere.
Experimental procedures
An oven dried 25-mL Schlenk tube was charged with the amine
Supplemental Material
sj-doc-1-chl-10.1177_17475198211063806 – Supplemental material for One-pot nickel-catalyzed cross-coupling of (Het)arylammonium salts prepared in situ and organozinc reagents
Supplemental material, sj-doc-1-chl-10.1177_17475198211063806 for One-pot nickel-catalyzed cross-coupling of (Het)arylammonium salts prepared in situ and organozinc reagents by Yong-Mao Huang in Journal of Chemical Research
Footnotes
Acknowledgements
The author is also thankful to Hebei Chemical & Pharmaceutical College for 1H NMR.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author gratefully acknowledges funding by the Science and Technology Project of Hebei Education Department (QN2016050).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.